Abstract
Background:
Treatment protocol for patients with cervical histologic low-grade squamous intraepithelial lesion (LSIL) such as that preceded by serious cytology and repeated diagnosis for at least 2 years is unclear.
Objective:
This study investigates the follow-up results of patients with cervical histologic LSIL and aims to provide evidence support for treatment and follow-up strategy.
Design:
A retrospective observational study design was used.
Methods:
The retrospective study included 4263 patients with cervical histologic LSIL diagnosed between August 2014 and February 2021. The follow-up period ended in August 2023.
Results:
During the followed-up of 6–101 months, 3246 (76.1%), 628 (14.7%), and 389 (9.1%) of the 4263 patients had lesion regression, persistence, and progression. Multiple gravidities, high-risk human papillomavirus (HPV) positive, HPV 16 positive, and cytologic examination (⩾atypical squamous cells cannot exclude high grade squamous intraepithelial lesion (ASC-H)) were independent risk factors for histologic LSIL progression. The annual cumulative cervical intraepithelial neoplasia (CIN)3+ rate of patients with histologic LSIL preceded by cytologic ASC-H and gravidity >2 was 6.3% (1-year), 22.4% (2-year), 28.2% (3-year), 28.2% (4-year), and 28.2% (5-year). The cumulative CIN2+ and CIN3+ rates in patients with histologic LSIL under risk factors and repeated diagnosis for at least 2 years were significantly higher than patients preceded by cytologic negative for intraepithelial lesion or malignancy, atypical squamous cells of undetermined significance, and LSIL.
Conclusion:
Cervical histologic LSIL had a high natural regression rate and a low progression rate. Multiple gravidities, high-risk HPV positivity, HPV 16 positivity, and cytological examination ⩾ASC-H were risk factors for histologic LSIL progression. For patients with histologic LSIL preceded by cytologic ASC-H, stratified management based on the number of gravidities might be an option.
Keywords
Introduction
The persistent infection of high-risk human papillomavirus (HPV) is the main cause of most cervical cancers and precancerous lesions.1–3 Histologic low-grade squamous intraepithelial lesion (LSIL) occurs when HPV infection becomes productive in cells that have initiated maturation. 4 Since 2014, the World Health Organization has declared that 2-tier terminology (histologic LSIL/high-grade squamous intraepithelial lesion (HSIL)) should be used to report the histopathology of squamous lesions of the lower anogenital tract. 4 Cervical histologic LSIL includes cervical intraepithelial neoplasia (CIN)1, suspicious CIN2 with p16 negative, condyloma, koilocytes, and previously named mild atypical hyperplasia. 4 Previous studies have shown that a large number of cervical histologic LSIL will spontaneously regress, and some lesions will further progress.5,6
The management of cervical lesions has shifted from result-based management to risk-based management, according to the 2019 ASCCP guidelines. 7 However, certain aspects of managing histologic LSIL remain controversial and merit further investigation. Specifically, for persistent LSIL (lasting >2 years) or LSIL preceding HSIL cytology, the immediate estimated risk of CIN3 or higher (CIN3+) may fall below the recommended treatment threshold (>25%). Nevertheless, the guidelines recognize these cases as exceptions, permitting therapeutic intervention as an acceptable option. 7 This apparent discrepancy highlights the need for additional research to refine clinical decision-making.
Several studies were devoted to exploring the influencing factors that can be used to predict the progression of lesion. High viral load (⩾100 relatively light units (RLU)/positive control), pre-biopsy cytology of atypical squamous cells cannot exclude high-grade squamous intraepithelial lesion (ASC-H) and HSIL, positive p16/Ki67 double staining and many other factors were found to predict the progression of cervical histologic LSIL, but some other studies suggest that the results are variable.8–11
Therefore, we retrospectively collected the basic clinical epidemiological information of cervical histologic LSIL patients to analysis the follow-up outcomes and explore the risk of lesion progression caused by different influencing factors. The aim of the study is to provide more clarity based on evidence support for the treatment and optimize the follow-up strategy of patients with cervical histologic LSIL.
Methods
Study design and participants
This retrospective study reviewed patients diagnosed with cervical histologic LSIL through colposcopy directed biopsy between August 2014 and February 2021 with follow-up ending in August 2023. Patients were included if they were followed up for more than 6 months. Patients were excluded if they met the following exclusion criteria: (1) incomplete cervix and previously underwent partial or total cervical resection; (2) previous cervical squamous intraepithelial lesions or cervical cancer without surgical treatment recorded as recorded in the medical records; and (3) infected with human immunodeficiency virus. During the follow-up, surgical treatment, lesion progression, and recurrence after lesion regression were selected to terminate the follow-up. The minimum sample size required was calculated to be 593 using PASS software (Version 15; NCSS, LLC, Kaysville, UT, United States), based on an assumed LSIL progression rate of 10%, 5 a 5% margin of error, and a 95% confidence interval (95% CI). A total of 4263 patients diagnosed with cervical histologic LSIL through colposcopy directed biopsy for the first time were determined to be eligible for study. The follow-up period ranged from 6 to 101 months. This study was reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 12
Clinical data of patients, including age, gravidity, parity, menopause status, mode of delivery, condom use, HPV testing, cytologic examination, and histologic results, were collected from clinical medical records. Referring to the studies of Bansal et al. 13 and Rong et al., 14 the follow-up results were determined by cytologic examination (with/without HPV testing) and histologic results. If both cytologic and histologic results were present, the histologic results were used to determine the follow-up results. Regression was defined as (1) histology confirmed negative and (2) cytologic examination of negative for intraepithelial lesion or malignancy (NILM). Persistence was defined as (1) histology confirmed LSIL and (2) cytologic examination of atypical squamous cells of undetermined significance (ASC-US) or LSIL. Progression was defined as (1) histology confirmed CIN2+ (including CIN2, CIN3, adenocarcinoma in situ (AIS), squamous cell carcinoma, and adeno-carcinoma of the cervix) and (2) cytologic examination of ASC-H or HSIL. During follow-up, patients with new indications for colposcopy-guided biopsy (according to the current cervical screening management guidelines) underwent new histologic analysis. Patients with progressive lesion were recommended to undergo treatment.
HPV testing, cytologic examination, and histologic results
HPV testing was performed using the Aptima HPV test (Hologic, Inc., San Diego, CA, United States), Digene Hybrid Capture 2 test (HC2; Qiagen, Gaithersburg, MD, United States), and Kaipu HPV 21 typing test (Kaipu Biochemistry Co., Ltd., Chaozhou, China). The result of Aptima HPV test included 3 categories (HPV 16, HPV 18/45, and the other 11 high-risk HPV genotypes (31, 33, 35, 39, 51, 52, 56, 58, 59, 66 and 68)). The Digene Hybrid Capture 2 test detects 13 oncogenic genotypes (16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, and 68). The results were classified as positive at a RLU/cut-off value of ⩾1 pg/mL. The Kaipu HPV 21 typing test included 13 high-risk HPV genotypes (HPV 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, and 68), 2 possible high-risk HPV genotypes (HPV 53 and 66) and 6 low-risk types (HPV 6, 11, 42, 43, 44, and 81).
Cytologic examination was categorized as follows based on the 2001 Bethesda classification system: NILM, ASC-US, LSIL, ASC-H, HSIL, and atypical glandular cells (AGC; including subcategories of AGC). 15 Cytologic examination ⩾ASC-H includes ASC-H, HSIL, and AGC.
Histologic results were determined based on the worst result obtained of biopsies or cervical conization. Normal saline, 5% acetic acid, and 5% iodine were administered to act on the cervix in order to identify the abnormal colposcopy findings and to perform colposcopy directed biopsy. Endocervical curettage (ECC) and cervical conization was performed when necessary. For statistical analysis, histologic results were categorized as (1) negative, (2) histologic LSIL, and (3) CIN2+ (including CIN2, CIN3, AIS and cervical cancer) and CIN3+ (including CIN3, AIS and cervical cancer).
Statistical analysis
All statistical analyses were performed using the SPSS (Version 21.0; SPSS Inc., Chicago, IL, United States), MedCalc (Version 8.0.0.1; MedCalc Software, Mariakerke, Germany), and PASS software (Version 15; NCSS, LLC, Kaysville, UT, United States). Continuous variables were presented as mean ± standard deviation if normally distributed, or as median (range) if non-normally distributed. Categorical variables are presented as frequencies and percentages. Data were analyzed using the Mann–Whitney U-test, chi-square test, or Fisher’s exact test where appropriate. The cumulative progression rate was analyzed by the Kaplan–Meyer survival curve, COX regression analysis and Log-rank test. Receiver-operating characteristic (ROC) curves were performed and the area under the curve (AUC) was evaluated to compare the prediction capability of the features that have been found to be significant in multivariate analysis. All statistical tests were two-sided, and a p-value <0.05 was considered statistically significant.
Ethics approval and consent to participate
This retrospective study was approved by the ethics committee of the Women’s Hospital, Zhejiang University School of Medicine (IRB-20220025-R) on November 17, 2021. Patients received colposcopy-guided biopsy after obtaining informed consent. The written broad consent was obtained from all participants.
Results
Follow-up outcomes and the baseline clinical characteristics
From 2014 to 2022, 3 (0.1%), 127 (3.0%), 168 (3.9%), 848 (19.9%), 1046 (24.5%), 1085 (25.5%), 880 (20.6%), and 106 (2.5%) cases were included in the study, respectively. All 4263 patients with cervical histologic LSIL confirmed by colposcopy directed biopsy for the first time were followed up for 6–101 months, with a mean follow-up period of 25.5 months. During the follow-up, 3246 (76.1%), 628 (14.7%) and 389 (9.1%) patients with cervical histologic LSIL had lesion regression (611 (18.8%) cases with histology confirmed negative and cytologic examination of NILM, 733 (22.6%) cases with histology confirmed negative, and 1902 (58.6%) cases with cytologic examination of NILM), persistence (469 (74.7%) cases with histology confirmed LSIL and 159 (25.3%) cases with cytologic examination of ASC-US or LSIL), and progression (372 (95.6%) cases with histology confirmed CIN2+ and 17 (4.4%) cases with cytologic examination of ASC-H or HSIL), respectively. Among 389 cases of histology confirmed CIN2+, 162 (41.6%) cases were histologic CIN3+ and 9 (2.3%) cases progressed to cervical cancer (including 7 cases of AIS and 2 cases of early-stage cervical squamous cell carcinoma). The baseline clinical characteristics of different follow-up outcomes were presented in Table 1.
Baseline clinical characteristics of different follow-up outcomes.
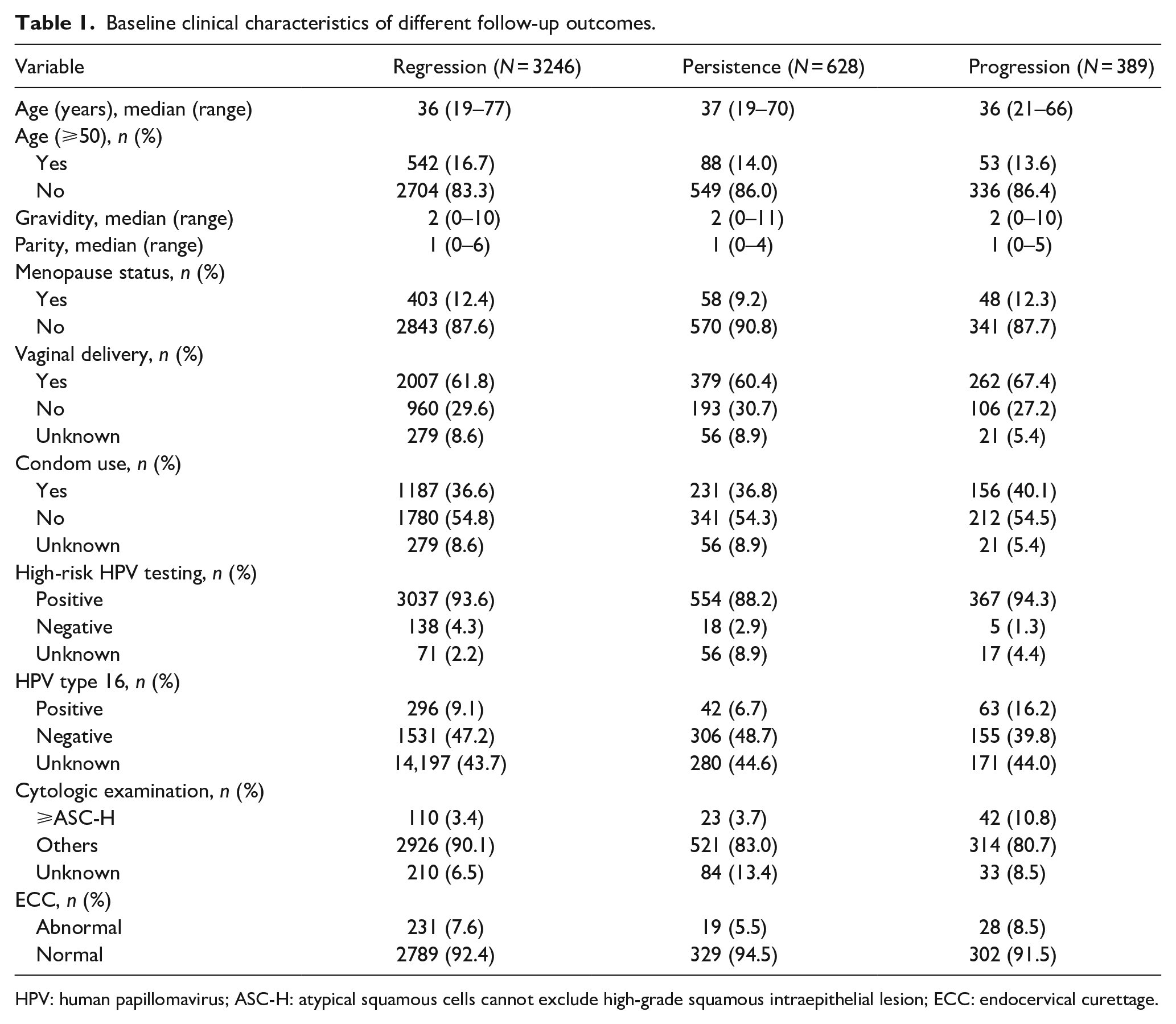
HPV: human papillomavirus; ASC-H: atypical squamous cells cannot exclude high-grade squamous intraepithelial lesion; ECC: endocervical curettage.
COX regression and ROC curve analysis for lesion progression
Univariate COX regression analyses showed that the gravidity, high-risk HPV positive, HPV 16 positive, and cytologic examination (⩾ASC-H) were significantly correlated with occurrence of LSIL progression. No significant correlation was found between LSIL progression and other baseline clinical characteristics. Multivariate COX regression analysis by backward elimination of nonsignificant associated factors in the univariate analysis showed that gravidity (hazard ratio (HR) = 1.097, 95% CI: 1.008–1.193, p < 0.05), high-risk HPV positive (HR = 3.686, 95% CI: 1.361–9.978, p < 0.05), HPV type 16 positive (HR = 2.058, 95% CI: 1.516–2.795, p < 0.05), and cytologic examination (⩾ASC-H; HR = 4.316, 95% CI: 2.811–6.627, p < 0.05) were independent risk factors for lesion progression (Table 2). Table 3 shows the AUC, optimal cut-off values, and corresponding sensitivity and specificity of each independent risk for predicting lesion progression through ROC curve analysis.
Univariate and multivariate COX regression analysis of histologic LSIL progression during follow-up.

LSIL: low-grade squamous intraepithelial lesion; HR: hazard ratio; 95% CI: 95% confidence interval; HPV: human papillomavirus; ASC-H: atypical squamous cells cannot exclude high-grade squamous intraepithelial lesion; ECC: endocervical curettage.
p < 0.05.
Sensitivity, specificity, and Youden index using cut-off points to predict histologic LSIL progression.

LSIL: low-grade squamous intraepithelial lesion; AUC: area under the curve; 95% CI: 95% confidence interval; Sens: sensitivity; Spec: specificity; HPV: human papillomavirus; ASC-H: atypical squamous cells cannot exclude high-grade squamous intraepithelial lesion.
Cumulative progression rate of patients with histologic LSIL under different risk factors
During the first 5 years of follow-up, the annual cumulative CIN2+ rate of patients with histologic LSIL preceded by cytologic NILM, ASC-US, and LSIL was 3.5% (1-year), 8.7% (2-year), 11.9% (3-year), 14.0% (4-year), and 17.2% (5-year), whereas the annual cumulative CIN3+ rate of these patients was 1.5% (1-year), 3.7% (2-year), 5.2% (3-year), 6.0% (4-year), and 7.5% (5-year), respectively (Figure 1). In patients with histologic LSIL preceded by cytologic ASC-H, the annual cumulative CIN2+ rate was 12.6% (1-year), 25.3% (2-year), 29.4% (3-year), 29.4% (4-year), and 29.4% (5-year), whereas the annual cumulative CIN3+ rate was 7.4% (1-year), 14.4% (2-year), 16.8% (3-year), 16.8% (4-year), and 16.8% (5-year; Figure 1(a) and (b)). The annual cumulative CIN2+ rate of patients with histologic LSIL preceded by cytologic ASC-H and gravidity >2 was 17.6% (1-year), 34.4% (2-year), 43.8% (3-year), 43.8% (4-year), and 43.8% (5-year), whereas the annual cumulative CIN3+ rate of these patients was 6.3% (1-year), 22.4% (2-year), 28.2% (3-year), 28.2% (4-year), and 28.2% (5-year), respectively (Figure 1(a) and (b)). The annual cumulative CIN2+ rate of patients with histologic LSIL preceded by cytologic HSIL was 20.3% (1-year), 43.1% (2-year), 58.3% (3-year), 68.7% (4-year), and 68.7% (5-year), whereas the annual cumulative CIN3+ rate of these patients was 7.3% (1-year), 18.9% (2-year), 40.5% (3-year), 40.5% (4-year), and 40.5% (5-year), respectively (Figure 1(c) and (d)). The cumulative CIN2+ and CIN3+ rate of patients with histologic LSIL under risk factors was significantly higher than patients preceded by cytologic NILM, ASC-US, and LSIL. There is no statistical difference between patients with histologic LSIL under different risk factors.

The cumulative CIN2+ and CIN3+ rate of patients with histologic LSIL under different risk factors. (a, b) Comparion of the cumulative (a) CIN2+ and (b) CIN3+ rate of patients with histologic LSIL preceded by cytologic ASC-H (with or without other risk factors) and those preceded by cytologic NILM, ASC-US, and LSIL. (c, d) Comparion of the cumulative (c) CIN2+ and (d) CIN3+ rate of patients with histologic LSIL preceded by cytologic HSIL (with or without other risk factors) and those preceded by cytologic NILM, ASC-US, and LSIL.
Cumulative progression rate of patients with histologic LSIL diagnosed repeatedly for at least 2 years
There were 357 cases of patients with histologic LSIL diagnosed repeatedly for at least 2 years. The annual cumulative CIN2+ and CIN3+ rate of patients with histologic LSIL diagnosed repeatedly for at least 2 years was 15.1% (1-year), 26.7% (2-year), 38.6% (3-year), 46.5% (4-year), 46.5% (5-year), and 6.8% (1-year), 10.5% (2-year), 18.8% (3-year), 25.8% (4-year), 25.8% (5-year), respectively (Figure 2(a) and (b)). The cumulative CIN2+ and CIN3+ rate of those patients was significantly higher than that of patients preceded by cytologic NILM, ASC-US, and LSIL.

The cumulative (a) CIN2+ and (b) CIN3+ rate of patients with histologic LSIL diagnosed repeatedly for at least 2 years.
Discussion
In our study, patients with cervical histologic LSIL were found to have a lower risk of progression during follow-up. For patients with progressive risk factors, such as multiple gravidities, high-risk HPV positivity, HPV 16 positivity, or cytological examination ⩾ASC-H, strengthening follow-up strategies or active treatment might optimize disease management. Due to the higher cumulative CIN3+ rate, histologic LSIL after cytologic HSIL required more aggressively intervention than that after cytologic ASC-H. In addition, when the gravidity was more than 2, the long-term cumulative CIN2+ and CIN3+ rate of patients with histological LSIL after ASC-H increased by about 1.5 times, suggesting that active treatment might be more suitable for those patients.
Previous studies have indicated that most of cervical histologic LSIL will spontaneously regress, but some will further progress.13,16,17 An early study reviewed the natural history of cervical lesion from the viewpoint of regression, persistence, and progression, the composite data indicate the approximate likelihood of regression of LSIL (CIN1) is 60%, persistence 30%, progression to HSIL (CIN3) 10%, and progression to invasion cancer 1%. 5 Our study confirms the similar findings in a large group of 4263 patients who were diagnosed with cervical histologic LSIL for the first time. During the longest follow-up period of 101 months, 3246 (76.1%) patients had lesion regression. However, 389 patients (9.1%) still experienced disease progression, with 9 of them progressing to cervical cancer. The 1- and 5-year cumulative CIN3+ rate of patients with histologic LSIL preceded by cytologic NILM, ASC-US, LSIL was 1.5% and 7.5%. Though cervical histologic LSIL has a high regression rate, identifying cases with progression risk is essential to optimize clinical follow-up strategies.
Numerous studies had concluded that cytological result ⩾ASC-H was associated with the progression of histologic LSI.7,9 As reported in the KPNC database, the 1- and 5-year CIN3+ risk in patients diagnosed with histologic LSIL preceded by cytologic ASC-H was 1.4% (under the 4.0% risk threshold) and 5.6% (above the 0.55% risk threshold). 7 This study found the 1- and 5-year cumulative CIN3+ rate of those patients was 6.3% and 28.3%, respectively. Due to regional and ethnic differences, as well as varying HPV vaccine coverage rates, the cumulative CIN3+ rate obtained is higher than previously reported and above the 4% risk threshold. In addition, several studies also had reported other influencing factors that increase the risk of histologic LSIL progression. A meta-analysis that included 38 studies showed that in patient with cervical histologic LSIL, the risk of progression (relative risk (RR): 3.04; 95% CI: 2.41–3.83; p < 0.001) and persistence (RR: 1.48; 95% CI: 1.17–1.87; p = 0.001) was higher in the HPV-positive group than HPV-negative group. 18 Furthermore, HPV16 positive is considered the major risk factor for CIN1 disease progression.19,20 Although ECC can improve the detection rate of histologic HSIL+, its effectiveness and application indications in evaluating endocervical lesions are still controversial.21,22 There is also no definite conclusion on whether high gravidity, marriage, sexual relationships, and condom use influence the risk of histologic LSIL progression.8,23–25 This study identified multiple gravidities, high-risk HPV positive, HPV 16 positive, and cytologic examination ⩾ASC-H influence the lesion progression. Combine cytologic results with other risk factors to further analyze the cumulative risk of progression. Especially for patients with histologic LSIL preceded by cytologic ASC-H, the long-term cumulative CIN2+ and CIN3+ rates increased by approximately 1.5 times when the gravidity is more than 2, suggesting that active treatment might be more suitable for those patients. According to the latest guidelines, treatment was acceptable in some special situation for patients with histologic LSIL preceded by cytologic ASC-H and HSIL, even if the immediate estimated CIN3+ risk did not reach the 25% treatment threshold. 7 Therefore, according to the different incidence rates in different regions, adjusting the management strategy of patients with histologic LSIL in some controversial situations is more in line with the significance of risk management.
Persistent cervical histologic LSIL is also common in clinical practice, which may easily cause anxiety in patients. A study of 126 women who underwent conization for CIN1 diagnosed at least 2 years found that 87% postoperative pathology was CIN1 or negative, whereas 13% was histologic HSIL (CIN2+). 7 In our study, the 1- and 5-year cumulative CIN3+ rate of the 357 patients with histologic LSIL diagnosed repeatedly for at least 2 years was 6.8% and 25.8%. Based on the 1-year cumulative CIN3+ rate not exceeding the 25% treatment threshold and considering the potential harms of treatment, it was appropriate to continue follow-up. But considering the long-term cumulative CIN2+ and CIN3+ rate of patients with histologic LSIL diagnosed repeatedly for at least 2 years was significantly higher than that of patients preceded by cytologic NILM, ASC-US, and LSIL. We draw the same conclusion that treatment is an acceptable option to relieve patients’ anxiety.
Limitations
The major limitation of our study is that it is a single center retrospective study. Meanwhile, the inadequate follow-up period among a subset of patients compromised the analytical accuracy of our study findings. However, the follow-up period of 6–101 months might reflect the natural history of cervical histologic LSIL, which provides evidence for optimizing the screening and triage. Second, the number of histologic LSIL patients with multiple high-risk factors and repeated diagnosis for at least 2 years was small, which prevented us from drawing a more definitive conclusion. Additionally, nine cases of cervical cancer were found during the follow-up. Among them, there were seven cases of AIS and two cases of early-stage cervical squamous cell carcinoma with minimal lesions. These cases were concealment and difficult to diagnose, requiring vigilance during follow-up.
Conclusion
Histologic LSIL had a high natural regression rate and a low progression rate. Multiple gravidities, high-risk HPV positivity, HPV 16 positivity, and cytological examination ⩾ASC-H were the potential risk factors for histologic LSIL progression during follow-up. Based on the number of gravidities might be a stratified management option for patients with histologic LSIL preceded by cytologic ASC-H.
Footnotes
Acknowledgements
The authors acknowledge all the staff who participated in this work at the Women’s Hospital, Zhejiang University School of Medicine, Hangzhou 310006, China.
Ethical considerations
This retrospective study was approved by the ethics committee of the Women’s Hospital, Zhejiang University School of Medicine (IRB-20220025-R) on November 17, 2021. Clinical trial number: not applicable. We confirm that all methods were performed in accordance with the 1964 Declaration of Helsinki and its later amendments.
Consent to participate
Patients received colposcopy-guided biopsy after obtaining informed consent. The written broad consent was obtained from all participants, permitting utilization of their data and anonymous publication, provided such use receives proper ethical review and approval. This article does not contain any studies with animals performed by any of the authors.
Consent for publication
The written broad consent was obtained from all participants, permitting utilization of their data and anonymous publication, provided such use receives proper ethical review and approval.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Natural Science Foundation of China (grant number 32300460) and Natural Science Foundation of Zhejiang Province (grant number LQ23H160031).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All relevant data are within the article. Requests for additional information should be addressed to the corresponding author and data may be provided on reasonable request.
