Abstract
Background:
The prevalence of poor sleep is increasing. Women and men have different sleep needs, and uniquely female life transitions, such as the perinatal period and menopause, are associated with further differences in sleep patterns. In Canada, sleep disparities across socioeconomic groups are profound, but sleep research rarely differentiates between women and men.
Objectives:
To examine the current evidence on the sleep quality and quantity of Canadian women using a life course perspective, drawing comparisons to men’s sleep where possible, and to identify correlates associated with poor sleep to help determine avenues for improving women’s sleep health.
Design:
Systematic review.
Methods:
A literature search was conducted in August 2024 to identify observational studies examining the relationship between sleep and potential correlates. Community-based studies set in Canada with exclusively female participants, or those in which results were stratified by sex, were included. Risk of bias was assessed using tools developed by the CLARITY group at McMaster University. Outcomes were summarized narratively.
Results:
Fifty-five studies (including 25,952,718 total participants) were included. Among children and adolescents, short sleep was associated with poorer biopsychosocial health, particularly among girls. Sleep was affected by the menstrual cycle, and sleep difficulties were more prevalent among adult women than men. The perinatal period was fraught with sleep disturbances that influenced key aspects of motherhood. Sleep dissatisfaction was prevalent among perimenopausal and older women.
Conclusion:
Canadian women have different sleep patterns than men, and their sleep is negatively affected in uniquely female life transitions. Poor sleep is influenced by, and impacts, women’s health and behaviors. Characterizing these differences is an important step toward improving sleep health, and thus overall well-being, for women. Future research would benefit from further exploration of the role of social determinants of health and sleep.
Registration:
PROSPERO CRD42024582007.
Keywords
Introduction
Sleep is a basic human need that has a key role in neural maturation, memory consolidation, cognition, and other metabolic processes. 1 Furthermore, sleep is being increasingly recognized as a vital component of health overall, with implications for longevity, mental health, and cardiovascular health, among other elements. 2 Sleep occurs in two phases, rapid eye movement (REM) and nonrapid eye movement (NREM), which cycle approximately every 90 min. NREM is further divided into four stages, each representing a deeper level of sleep. The amount of time spent in each phase differs by age and sex,3,4 with older individuals spending less time in stages 3–4 of NREM, whereas women spend more time in these stages. 5 Further differences in the time spent in each phase and stage of sleep are observed in sleep disorders such as insomnia and obstructive sleep apnea (OSA). 6
Although sleep duration remains a primary parameter when investigating sleep health, healthy sleep encompasses sufficient quality, regularity, timing, and continuity, among other dimensions. 7 Disruption to one or more of these areas can impair the sleep cycle and cause sleep to be non-restorative, resulting in numerous health and daily functioning challenges over time. 8 Sleep can be interrupted by many factors, including clinical, behavioral, hormonal, psychosocial, and environmental. 9 The Canadian 24-Hour Movement Guidelines are an internationally recognized set of recommendations for numerous health behaviors, including physical activity, sedentary time, and sleep, across the lifespan. 10 They recommend that children and youth sleep 8–11 h per night, whereas 7–9 h of sleep per night is optimal for adults. 10 Unfortunately, many Canadians report inadequate sleep quantity (sleeping too few hours each night) as well as poor sleep quality (such as interrupted sleep or long sleep latency leading to nonrestorative sleep). 11
The link between sleep and health has been extensively studied in Canada and abroad, but there is a deficit of information relating to sex differences across the life course. While differences in sleep between young girls and boys are not consistently shown, 3 these disparities increase with age, and older women have increased risks of sleep disorders, long sleep latency, and other disturbances compared to older men, who also have more sleep troubles compared to younger groups.3,12
Such differences in sleep needs and patterns between women and men may be due to social factors such as differences in employment and at-home roles, 13 and biological factors such as adiposity and hormone profiles. 14 The circadian rhythm, which resides in the hypothalamus and is the internal sleep-wake regulator, also varies between women and men due to differences in hypothalamus-pituitary-gonadal hormone axes, body temperature, and other factors. 15
Furthermore, sleep in women is affected by fluctuations such as the menstrual cycle, 16 as well as critical life transitions (including pregnancy, postpartum, and menopause), which can negatively affect their sleep, while also increasing the need for sleep.17,18 Throughout the menstrual cycle, levels of sex hormones, melatonin, cortisol, and other pituitary hormones fluctuate, which is thought to negatively affect sleep. 3 During pregnancy, sleep is interrupted by physical changes such as abdominal distension, urinary frequency, and back pain, 3 which tend to resolve postpartum. 3 In the postpartum period, women’s sleep is fragmented by the need to respond to their infants’ needs, such as night feedings and diaper changes. 19 Finally, during menopause, women commonly experience disturbing symptoms such as hot flashes, night sweats, and insomnia as a result of hormone fluctuations. 20 Despite this, differences in sleep patterns and needs across the life course for women, and in critical female life transitions, are rarely the focus of sleep research, which has left a gap in the current picture of sleep among Canadians.
The Canadian government have defined 12 social determinants of health (SDH), which influence one’s well-being and are linked to health equity. 21 These include income, employment, education, childhood experiences, living environment, social support, healthy behaviors, access to health services, biology and genetic endowment, gender, culture, and race. 21 A recent systematic review by Etindele Sosso et al. analyzed the current research on the influence of SDH on sleep among Canadians. 22 Several critical differences in sleep quality and quantity among Canadians of different ages, education levels, income, and employment were noted in this work; however, sex differences in sleep across the life course among Canadians remains a topic to be explored. An analysis by Rodrigues et al. also investigated the role of SDH and sleep among a subset of older Canadians and found that social determinants substantially influenced various domains of sleep, with some effects differing by sex. 23 However, research is still lacking across the life course between women and men in terms of sleep and SDH, and precludes our ability to fully understand the sex differences in sleep among Canadians, and the resultant implications for overall health.
Thus, our primary objective was to identify potential correlates of sleep health and determine if they vary across the life course and in critical life transitions. By synthesizing the research on sleep among Canadian women, and making comparisons to men’s sleep where possible to glean valuable insight into the differential experiences of women and men, this review can help identify opportunities for improving women’s health and inform the ways in which sleep may be specifically targeted across different life transitions and socioeconomic groups. Furthermore, by focusing on Canadian research, we were able to include under-represented groups where available, such as Indigenous Peoples, and incorporate data from small and large studies set in remote regions and urban centers, and from coast to coast to coast. As such, this systematic review presents a comprehensive picture of sleep among Canadian women.
Methods
Literature search
Following consultation with a health sciences librarian, a comprehensive search of PubMed, Embase, and CINAHL was conducted in August 2024. A combination of keywords and controlled vocabulary (e.g., MeSH, Emtree) were used to search for three main concepts (combined with AND): (1) Canada OR (individual provinces and territories); (2) sleep OR sleep health OR sleep hygiene OR sleep quality; and (3) women OR female OR mothers OR pregnancy OR postpartum (see Supplemental Material). This systematic review follows the Preferred Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (see Supplemental Material). The protocol for this systematic review was registered with PROSPERO (registration number CRD42024582007).
Inclusion and exclusion criteria
Observational studies conducted on community-based samples in Canada in which sleep metrics were measured were included in the current systematic review. Such sleep measurements could be subjective (i.e., self-reported through interview, questionnaire, or sleep diary) or objective (i.e., actigraphy or polysomnography). See section “Characteristics of sleep metrics” for a complete breakdown of sleep variables assessed in the studies included. Studies must exclusively study women or girls, or present sex-stratified data if men were also included.
Studies were excluded for the following reasons: exclusive focus on biological males or no sex-stratification; studies conducted outside of Canada (including multinational studies that included Canada); clinical interventions; and studies conducted outside of community settings such as in secondary or tertiary care centers. Studies were excluded if they focused on individuals with sleep pathologies, though those that grouped “sleep latency” and “restless sleep” under the title “insomnia symptoms” were included. Studies that focused on individuals with other health conditions were also excluded; as many common diseases such as cancer, chronic pain, and osteoarthritis are known to cause sleep disturbances,24 –26 we elected to exclude studies that exclusively focused on individuals living with these conditions, as they would not be representative of the general population and may skew results in favor of a greater prevalence of sleep disturbances. Studies were also excluded if they were conducted during the COVID-19 pandemic, which was characterized by government-imposed changes to social structures and living and working environments. Conclusions drawn from such research are not comparable to typical non-pandemic times, which was the focus of the current systematic review.
Screening and bias assessment
Search results were imported into Covidence, for which Western University provides free access for students, which automatically removed duplicates and retrieved study information, including abstracts, author information, and full texts for Open Access articles. All stages of screening were done independently by two reviewers (SW and PN) on Covidence and are depicted in Figure 1. First, titles and abstracts were screened. This stage primarily identified studies for exclusion that were the wrong setting or population. Second, full texts were reviewed. This stage primarily identified studies for exclusion that did not stratify results by sex or had the wrong outcomes. Conflicts between reviewers were resolved by discussion and consensus. There were no studies that met all our inclusion criteria and none of our exclusion criteria that were excluded.

PRISMA flowchart of study selection process.
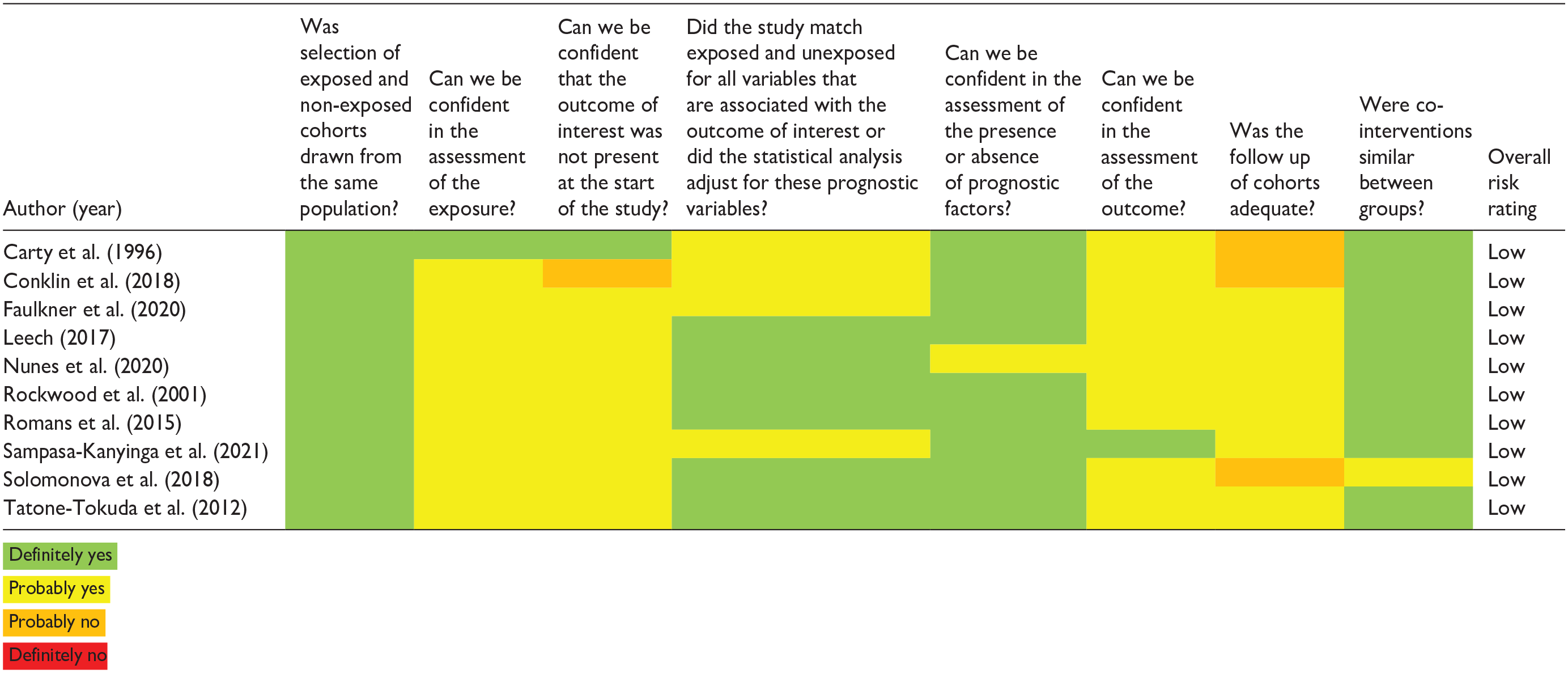
After the final list of studies to be included in the review was identified, risk of bias was assessed using the tools developed by the CLARITY Group at McMaster University. For cohort studies, eight questions were answered using the Tool to Assess Risk of Bias in Cohort Studies. For cross-sectional studies, five questions were answered using the Risk of Bias Instrument for Cross-Sectional Surveys. For case–control studies, five questions were answered using the Tool to Assess Risk of Bias in Case Control Studies. Possible answers included “definitely yes” (low risk of bias), “probably yes,” “probably no,” and “definitely no” (high risk of bias). An overall risk rating was assigned for each study based on their cumulative scores by assigning a numeric value for each answer (“definitely yes” = 1, “probably yes” = 2, “probably no” = 3, and “definitely no” = 4). Studies in the first tertile were given a low overall risk rating, studies in the second tertile a moderate overall risk rating, and studies in the third tertile a high overall risk rating. Bias assessments were done by both reviewers independently; conflicts were resolved by discussion and consensus.
Data synthesis
Data were extracted from each study by one author and then reviewed for accuracy by the other. When not explicitly described, the percentage of female participants in samples were calculated manually. The following data were extracted and summarized from each study: study setting, study design, data source, population characteristics, sample description, sleep variables, factors associated with sleep, method of variable measurement, results, and conclusions.
Results
Characteristics of included studies
Our literature search identified 2455 unique primary research articles. In total, 2232 of these texts were excluded during the abstract screening phase, leaving 223 articles for full text review. About 168 texts were excluded due to a variety of factors including wrong study design, wrong study population, wrong setting, and wrong outcomes. Ultimately, we identified 55 studies that met the inclusion criteria for this systematic review, which were published between January 1996 and August 2024. In total, 44 were cross-sectional in design, 10 were cohort studies, and 1 was a case–control study. The oldest was published in 1996, with the next in 2006, and the most recent was published in 2024. Many studies were secondary analyses of large population studies such as the Canadian Longitudinal Study on Aging (CLSA; seven studies), Canadian Health Measures Survey (CHMS; five studies), and Canadian Community Health Survey (CCHS; seven studies). Only 14 studies collected primary data; these tended to have smaller sample sizes. Sample sizes ranged from 19 to over 25 million people. Sixteen studies focused on pediatric populations, and two in older adults aged 65 years and older. The remaining 37 studies characterized sleep in adults of varying age ranges. Of these, two were set in the menstrual cycle (i.e., comparing different phases of the menstrual cycle), five were set in the perinatal period (i.e., during pregnancy and 1 year postpartum), and two collected data on menopausal status. Twenty-eight of the studies included participants from three or more provinces/territories; the remainder drew participants from smaller areas. Five studies investigated Indigenous Peoples, with four of these studies set in the same Saskatchewan First Nations community. In total, 12 studies had only female participants, whereas 43 studies contained data from both men and women and presented sex-stratified results. Results from the risk of bias assessments are shown in Tables 1–3. Studies ranged in risk of bias, with most having at least one component rated as moderate risk of bias. A summary of study characteristics is presented in Table 4.
Results of the risk of bias assessment for cross-sectional studies.

Results of the risk of bias assessment for cohort studies.

Results of the risk of bias assessment for case–control studies.

Characteristics of included studies.

CS: cross-sectional; PC: prospective cohort; RC: retrospective cohort; CC: case–control; OSA: obstructive sleep apnea; PSQI: Pittsburgh Sleep Quality Index; BMI: body mass index; SPAQ: Seasonal Pattern Assessment Questionnaire; OR: odds ratio; CI: confidence interval.
Female representation varied between questions.
Characteristics of sleep metrics
Nocturnal sleep duration was the most common sleep metric in the studies included, as it was measured in 42 studies and was the sole sleep outcome in 20 studies. Sleep metrics were mostly self-reported by participants (or by parents in some pediatric studies) in interviews, sleep diaries, or questionnaires. Most studies created their own sleep questionnaire, but some used validated questionnaires such as the Pittsburgh Sleep Quality Index (PSQI; n = 6), Mindell’s Sleep questionnaire (n = 1), Sleep Symptom Checklist (n = 1), Multidimensional Assessment of Fatigue (MAF; n = 1), Seasonal Pattern Assessment Questionnaire (SPAQ; n = 1), and the Adolescent Sleep Wake Scale (ASWS; n = 1).
Mindell’s Sleep Questionnaire was developed in 1999 to assess sleep patterns and symptoms of sleep problems over the preceding 2 weeks. Questions included weekday and weekend bedtime, weekday and weekend waketime, total sleep time, time to sleep onset, napping details, night wakings, difficulty falling and staying asleep, and other measures detailed elsewhere. 27 The PSQI also assesses sleep quality and disturbances over a 1-month period, including subjective sleep quality, sleep duration, use of sleep medication, sleep disturbances, daytime dysfunction, and other measures detailed elsewhere. 28 The Sleep Symptom Checklist assesses the severity of sleep-related symptoms over the past month, including subjective sleep quality, difficulty initiating and maintaining sleep, and other measures detailed elsewhere. 29 The MAF assesses the degree and functional impairments of fatigue the preceding week, as detailed elsewhere. 30 The SPAQ assesses seasonal effects on mood and behaviors, including the duration of sleep in each season, and other measures. 31 The ASWS is a measure of subjective sleep quality for children aged 12–18 years. It assesses 28 items relating to sleep quality, including going to bed, falling asleep, maintaining sleep, reinitiating sleep, and returning to wakefulness over the preceding month; a validated 10-item scale was used in the current study. 32
Five studies used actigraphy or polysomnography to obtain objective measurements of sleep quantity and quality. The quality of sleep assessment was reflected in the Bias Assessment (see Tables 1–3). Other sleep metrics included sleep latency (n = 10), night awakenings (n = 6), refreshment from sleep (n = 7), daytime fatigue (n = 5), daytime alertness (n = 2), restless sleep (n = 1), sleep satisfaction (n = 7), seasonal sleep patterns (n = 1), sleep efficiency (n = 4), insomnia symptoms (i.e., long sleep latency and interrupted sleep; n = 2), and problems with daytime functioning (n = 3). The full breakdown of sleep metrics is depicted in Table 4.
Childhood and adolescence
Absolute differences in average sleep duration between boys versus girls were not widely reported but were found to be comparable in several studies.33 –35 In one study, adolescent boys had the longest sleep duration when compared to adolescent girls, young girls, and young boys. 4 Two other studies found that adolescents slept less than children, irrespective of sex.35,36
Predictors of short sleep duration and sleeping difficulties included adolescent age35 –37 and low income. 36 Both young 35 and adolescent35,38 –41 girls were less likely than boys to meet recommendations for sleep, physical activity, and screen time as set by the Canadian 24-Hour Movement Guidelines. 10
One study showed that short sleep placed both boys and girls at risk for being overweight or obese compared to long sleep duration, 42 but others found this relationship true in boys but not girls.36,43 In a survey of First Nations students, sleep duration was not related to body mass index (BMI) in girls nor boys. 44 Tatone-Tokuda et al. showed that short sleep was associated with lower intake of fruit and vegetables in both boys and girls; in girls only, short sleep was further associated with greater soda consumption and decreased dairy consumption. 43 Interestingly, Vézina-Im et al. found that adolescent girls had poorer sleep quality than adolescent boys despite consuming smaller quantities of sugar-sweetened beverages, though this may have been related to increased quantities of caffeine consumed by girls. 32 Despite a positive association with body weight, Sluggett et al. did not find that short sleep was associated with metabolic derangements such as hyperinsulinemia and hypertriglyceridemia in boys or girls. 36
Finally, relationships between short sleep and psychosocial factors were noted in several studies. In girls, household chaos, 33 internalizing problems, 37 depressive symptoms,45,46 and suicidal ideation 41 were associated with short sleep duration. In both girls and boys, substance use was associated with short sleep duration. 39
Adulthood
Menstrual cycle
Menstrual cycles normally last between 24 and 38 days, beginning with 3–7 days of menstruation followed by the follicular and luteal phases.47,48 Both studies on menstrual timing found differences in sleep during the end of cycle (i.e., late luteal) compared to midcycle (i.e., follicular phase). Romans (2015) showed that luteal sleep quality was poorer than midcycle sleep quality irrespective of physical health, exercise, social support, or daily perceived stress. 49 They also found that sleep quality during menses did not differ from midcycle sleep. In contrast, Lamarche et al. found that only women with significant pre-menstrual symptoms (PMSs) had diminished alertness and greater sleepiness during the end of their cycle compared to mid-cycle, though napping habits did not differ between cycle phases. 50 Interestingly, the amount of time spent in both stage 1 and NREM sleep was reduced in all women during the late-luteal phase compared to the follicular phase.
Perinatal
Sleep disturbances during pregnancy and postpartum were widely reported. Hall et al. found that 68% of pregnant women had fragmented sleep (three or more awakenings per night) and 21% were sleep deprived (slept <6 h per night). 51 Solomonova et al. found that sleep disturbances increased from the second trimester to the third trimester, then improved postpartum. 52 Similarly, Carty et al. showed that night awakenings decreased, and sleep duration increased, postpartum compared to the prenatal period. 53 Still, these postpartum sleep disturbances contributed to tiredness and affected maternal well-being, work, and their relationship with their partner. 53 Prenatal sleep disturbances were associated with greater fatigue, 51 childbirth fear, 51 as well as postpartum delusional ideation, 52 depression, 52 and anger. 54 Prenatal night awakenings were due to using the bathroom, 53 whereas postpartum sleep disturbances were due to mothers responding to their infant crying. 53
About 51.8% of postpartum women in the study by Ou et al. had poor sleep quality, and those who perceived their sleep as poor were 2.9 times more likely to have intense anger, 54 along with poor sleep quality, intense anger was associated with having more children, depression, lower income and education levels, and more perceptions of infant sleep problems. 54 Kalogeropoulos et al. also found that while the total nocturnal sleep duration was comparable between fathers and mothers, mothers’ sleep was more fragmented and consecutive hours of sleep were fewer. 55
General studies
There was a high heterogeneity of studies that assessed sleep in the general Canadian adult population without a focus on female-specific health states (i.e., menstruation or the perinatal period). Only four of these studies were comprised of exclusively female samples.56 –59 Several studies were secondary data analyses of large community surveys, including the CHMS and CCHS, with the rest being investigations on the provincial level.
Analyses of sleep data collected in the CHMS showed various differences between men and women. Both Chaput et al. and Zandy et al. analyzed data collected between 2007 and 2013. Chaput et al. found that women between the ages of 18–64 years slept longer than their male counterparts, despite reporting greater difficulties in falling asleep. 11 Zandy et al. stratified data by urinary cotinine levels, which is a marker of tobacco smoke exposure. Sleep problems were more prevalent among those with higher cotinine levels, with women being more affected than men: they had greater odds of short or long sleep duration; poor sleep quality; and low sleep satisfaction than men for increasing cotinine levels. 60 An analysis of data from 2012 to 2015 found no difference in the prevalence of inadequate sleep between men and women, though inadequate sleep predicted high-risk alcohol consumption and inadequate physical activity among women but not men. 61 An analysis of Statistics Canada’s General Society Survey found that the average nightly sleep duration increased between 1998 and 2010 for both women and men, and women slept longer than men at both time points. 62
Several sleep trends were noted in data from the CCHS, a national cross-sectional survey. Women experienced a greater increase in the prevalence of insomnia between 2002 and 2012 compared to men. 63 Further analyses showed a higher prevalence of insomnia among women aged 40 years and older, but not men. 63 Kyeremeh et al., analyzing 2012 data, found that troubled sleep was more prevalent among women than men, except in sedentary women and immigrant women. 64 In an analysis of a female subset of the CCHS, Vézina-Im et al. found that only 55.7% of women slept the recommended amount of 7–9 h per night, though sleep quality was generally good. 59 An analysis of data from 2015 to 2017 found that a greater proportion of women reported poor sleep quality than men, but a greater proportion of men reported short sleep duration than women. 65 Several lifestyle and behavioral factors were associated with shorter sleep, the most reported being smoking and binge drinking. 65 Daily smoking was associated with a higher odd of short sleep, whereas having quit smoking over 1 year ago was associated with a higher odd of long sleep, and binge drinking was associated with a higher odd of short sleep. 65 Lastly, data from the 2021 CCHS sample showed that the prevalence of nighttime insomnia symptoms, non-refreshing sleep, and difficulty staying awake were each higher among women than men. 66
In women, sleep difficulties were associated with work injury in processing/manufacturing and professional occupations (such as teaching, medicine, and nursing) 67 ; high risk alcohol consumption, 61 and inadequate physical activity. 61 Vézina-Im et al. found that short sleep duration but not sleep quality was associated with being overweight or obese. 58 Factors associated with poor sleep duration in women of childbearing age included Asian, Arabic, or black ethnicity (versus white), living in an urban setting (versus rural), lower education level, lower intake of fruit and vegetables, alcohol consumption, having fewer children, and smoking. 59
Several provincial-level studies also investigated adults’ sleep. Both British Columbian men and women had increased risk of short sleep with nocturnal light exposure. 68 In Saskatchewan, women who engaged in more aerobic exercise had greater sleep quality and duration. 57 Shift work was associated with poor sleep latency and sleep efficiency compared to day work in Ontarian women. 56 In Quebec, both short and long sleep duration were positively associated with BMI in men and women, 69 whereas short sleep was positively associated with alcohol consumption in both sexes, though men were more affected. 70 Bassett and Moore found that restless sleep was more prevalent among women aged 25–34 years in Montreal than those 35 years and older, and divorced women and those with poorer health were more likely to report restless sleep. 71 In Atlantic Canada, frailty was associated with sleeping fewer than 4 h per night in both men and women aged 60 years and older. 72
Finally, analyses of Saskatchewan First Nations communities noted several sleep trends among study participants. Sleep deprivation was more prevalent among men than women,31,73,74 and in people over 30 years old, those who attended residential schools, had poor housing conditions, and who used tobacco non-traditionally. 73 Sleep quality was also higher among women than men and in those with increasing income. 74 Interestingly, seasonal changes in sleep patterns were reported by over two-thirds of the population. 31 Participants reported sleeping the least from May to August and the most from November to December, due to changes in wake time. 31 Compared to women, men had a higher prevalence of sleep deprivation in the springtime and a lower prevalence in the fall. 31 Sleep efficiency was not different between women and men, but people with high blood pressure, chronic pain, and those who lived in a house where people smoked inside had increased risk of poor sleep efficiency. 75
Older adulthood
Menopausal transition
The menopausal transition typically begins in the sixth decade of life 76 and lasts for approximately 4 years, but some symptoms can persist for a decade. 77 Compared to pre- or peri-menopausal women, postmenopausal women had longer sleep latency. 78 There were no differences in overall sleep satisfaction, sleep maintenance, daytime wakefulness, or other potential sleep disorders between the two groups. 78 A smaller survey of non-frail postmenopausal women found that those with long sleep durations had lower muscle mass, strength, and balance compared to those with shorter sleep durations. 79
General studies
Studies investigating sleep patterns among older adults stratified their data by sex. Several studies analyzed data from the CLSA, a longitudinal nation-wide survey of over 50,000 individuals aged 45–85 years. Sleep dissatisfaction23,80 and poor sleep efficiency 23 were more prevalent among women than men, without differences in total sleep time, 23 though both women and men slept longer with increasing age. 81 Several factors were associated with poorer sleep among women but not men, including daily smoking, 80 multimorbidity, 82 education level, 23 and South Asian ethnicity (compared to White ethnicity). 23 Factors associated with poorer sleep in men but not women include regular alcohol consumption, 80 East and Southeast Asian ethnicity (compared to White ethnicity), 23 and psychological distress. 83 Both employed women and men had poorer sleep than retired women and men. 23 In a case–control analysis of data from the CLSA, Zolfaghari et al. found that very long sleep duration (⩾10 h/day) was associated with an 83% increase in mortality in women but not men. 84
An older study using data from the Canadian Study of Health and Aging (CSHA) found that women aged 65 years and older reported more daytime sleepiness, difficulty falling asleep, and “being tired all the time” compared to men—despite men reporting more early awakenings than women—and that daytime sleepiness and feeling “tired all the time” were associated with mortality. 85 Chaput et al. noted no differences in sleep duration between women and men aged 65 years and older, though women reported more difficulties falling asleep. 11 There were no differences in sleep duration or sleep efficiency between very active older women and men. 86
Discussion
Summary of results
Our findings suggest that sleep disturbances are experienced by Canadian women of different age groups, and there are marked disruptions throughout the menstrual cycle and during critical life transitions such as pregnancy and perimenopause. Furthermore, sleep quality and quantity differ between men and women in different life stages, though many differences were minor. During childhood and adolescence, girls had shorter sleep duration than boys, though the associations with diet and metabolic factors were less consistent. Most studies were conducted during adulthood. Overall, poor sleep quality was commonly reported across studies during the prenatal and postnatal periods, permeating all aspects of maternal life, from well-being to relationships with spouses and infants, to work. Although differences in sleep between women and men were mostly inconsistent, women generally had poorer quality of sleep than men across different life stages and patterns of risk factors. Sleep was again affected during menopause, with reduced sleep satisfaction and quality during menopause and into later adulthood for women. This systematic review identified consistent links between poor sleep and SDH across age groups, with marginalized population subgroups being disproportionally affected. A visual depiction of results is presented in Figure 2.

Visual depiction of systematic review findings.
It is also important to reiterate that the above findings arise largely from subjective reports, with only five studies reporting polysomnographic or actigraphic data; however, that is not to say that these results are inaccurate. A 2004 meta-analysis investigating purely objective sleep parameters—including total sleep time, sleep latency, sleep efficiency, nocturnal awakenings, REM sleep, stage 1 sleep, stage 2 sleep, and slow wave sleep—across the lifespan noted numerous age and sex differences. 87 Increasing age was associated with shorter total sleep time, lower percentage of REM and slow wave sleep, poorer sleep efficiency, and longer sleep latency. Compared to men, women had shorter total sleep time, sleep latency, percentage REM sleep, and percentage slow wave sleep, as well as more nocturnal awakenings and stage 2 sleep. These findings are partially in keeping with those in the current review, which may in part be because the they did not investigate sex differences in different age groups, as well as the differences in populations and the inherent discrepancies between subjective and objective sleep measures. In fact, a recent study that compared subjective and objective measures of sleep duration in both good sleepers and those with insomnia found significant differences between the two measures, with good sleepers more often overestimating their sleep duration, and those with insomnia underestimating it. 88 Together, this reinforces the importance of continuing to study sleep epidemiology with robust methods, critical analyses, and in diverse populations.
Childhood and adolescence
Relationships between sleep and biological and social factors differed between girls and boys. Several studies found that girls were less likely to meet recommendations for physical activity and screen time, as well as sleep, than boys.35,38,39 This is problematic because lifestyle patterns during childhood persist into adulthood, and those with healthy habits are at lower risk of cardiometabolic disease, with the opposite being true for those with unhealthy patterns. 89 Given the increasing prevalence in Canada of diseases such as type 2 diabetes mellitus, hypertension, and obesity, in which lifestyle factors play significant roles in their pathogenesis, 90 addressing poor lifestyle behaviors among youth represents a potentially vital tool to improve the health of the Canadians. These findings are mostly in line with a previous systemic review investigating childhood sleep, albeit not differentiated by sex, which found childhood sleep to be associated with multiple health outcomes including cognition, school performance, psychological health, quality of life, cardiometabolic health, and adiposity. 91 Notably, we identified similar trends among Canadian youth across the country, illustrating the pervasiveness of these sleep deficits nationally.
The critical role of sleep for adolescent brain development is well established. 92 The prefrontal cortex, which is responsible for executive functions such as decision-making and memory, is particularly affected by poor sleep due to its late development. 92 In the present review, female sex and adolescent age were among key predictors for short sleep duration, 36 and girls were also found to have poorer sleep quality than boys. 32 These sex differences in sleep quality and quantity may affect neurodevelopment and educational attainment, and thus have lasting implications for adulthood.92 –94 Sleep education may be a viable first step for improving sleep and mitigating these problems; however, further investigation into the effectiveness of education about sleep hygiene practices as an intervention are needed. 93
Adulthood
Menstrual cycle
The cyclic menstrual cycle is dictated by a complex rise and fall of various hormones produced in the hypothalamus, pituitary, and ovaries. 95 Briefly, in the follicular phase, luteinizing hormone (LH) levels rise, resulting in an increase in estrogen. Estrogen exerts positive feedback on LH, and a surge of LH levels toward the end of the follicular stage, along with a rise in follicle stimulating hormone (FSH), triggers ovulation. 95 During the luteal phase, LH and FSH levels fall, whereas the level of progesterone increases in order to support the implantation and early survival of a fertilized ovum. Progesterone is secreted by the corpus luteum, which forms in the ovary after ovulation from some of the cells left behind after the oocyte is ovulated. 95 Although these hormones are essential to the reproductive aspect of the menstrual cycle, progesterone has also been shown to have a relaxing effect. 96 Numerous other hormones also fluctuate throughout the menstrual cycle, such as melatonin and cortisol, and exert effects on other aspects of health including sleep. 97 Previous studies have identified sleep disturbances during the luteal phase, such as increased sleep latency and decreased REM sleep, 98 which may be attributable to changes in hormone levels, though not all women are affected.
In line with this, the current review found women to have poorer sleep during the premenstrual (i.e., luteal) period.49,50 However, they differed in identified risk factors, with one study 49 finding poorer sleep irrespective of lifestyle and health risk factors, and the other 50 only finding sleep disturbances in women with significant premenstrual symptoms. Given that menstrual cycles vary in terms of duration, bleeding, and symptoms for each woman, which may be due to differences in hormone variations, anatomy, and other aspects of health, 99 these inconsistencies are not altogether surprising. Another systematic review investigating menstrual disturbances and sleep quality found that women with menstrual disturbances, including PMS, had poorer sleep quality, troubles with alertness, longer sleep latency, earlier morning awakening, and shorter sleep duration. 100
It has also been previously shown that lower socioeconomic status is associated with greater menstrual cycle disturbances. 101 As the link between SDH, sleep, and menstruation was not explored in the current review, it would be worthwhile for future studies to do so to help determine how women’s menstrual health can be best supported.
Perinatal
In the current review, perinatal sleep deficiencies were related to several negative physical and psychological outcomes,51 –53 with an eventual return to normal post-partum. 52 These outcomes follow similar patterns of sleep deficiency found to be related to multiple adverse pregnancy outcomes including risk of antepartum and postpartum depression, gestational diabetes, hypertensive disorders, preterm birth, and relationship with labor course and duration. 102 The findings of the current review are consistent with previous systematic reviews investigating sleep and pregnancy outcomes.103,104 Sleep duration was a particularly important indicator of pregnancy health, with both short and long duration being associated with increased risk of gestational diabetes mellitus,103,104 and long duration being additionally associated with cesarean section. 104 The current systematic review also brought into focus the relationship between perinatal sleep issues and SDH, which was not investigated in the aforementioned reviews. Given the small sample sizes of the perinatal studies included in this review, it would be beneficial for future studies to continue to explore these relationships, especially on a larger scale, to make better inferences for Canadian women as a whole.
Interestingly, although seemingly counterintuitive, breastfeeding may improve postpartum maternal sleep. Sleep duration is longer in breastfeeding mothers compared to those who did not, and this may be related to less ambient light and physical activity exposure which can reduce sleep. 105 Due to a lack of information about postpartum risk factors for sleep disturbances, difficulties arise in studying infant feeding types. 105
General studies
Sleep duration, quality, and insomnia symptoms differed between adult men and women. In women, shorter duration sleep was associated with multiple lifestyle and behavioral risk factors, as well as being overweight and obese, 58 reinforcing the life-course association between childhood sleep and bodyweight.36,42,43 This association was also seen in young adulthood, in a study not included in the current review, whereby poor sleep was overall associated with increased body fat; though lower sleep quality, efficiency, and inconsistent sleep patterns were more strongly associated than lower sleep duration. 106
Healthy habits are critical to maintaining health throughout the life course, as childhood habits propagate into adulthood, 89 and all aspects of sleep, from disturbances in duration and timing to poor sleep quality, can contribute to morbidity later in life. 102 Physical activity may have a positive impact on sleep health, with outdoor activity relating the positive association between daytime light exposure and sleep. 107
One aspect of sleep health among adults that was not investigated in the current review is that of sleep disorders, including insomnia, OSA, restless leg syndrome, narcolepsy, and REM behavior disorder. Although two studies investigated “insomnia” symptoms66,83 (i.e., long sleep latency and night awakenings), populations with diagnosed sleep disorders were excluded from this review. In Canada, the prevalence of insomnia is approximately 16.3%, with women and Indigenous peoples being at higher risk. 108 The proportion of individuals who report symptoms of insomnia, but lack a formal diagnosis, is much higher, 108 with children and adolescents reporting fewer symptoms than young adults, who report fewer symptoms than older adults.109,110 Although one study found that the proportion of Canadians reporting such symptoms increased between 2007 and 2015, 110 another that analyzed data from 2007 to 2021 found this to be true for women but not men. 109 Although polysomnography is not required for a formal diagnosis, several patterns are observed in those with the disorder, including a reduction in total sleep time, longer sleep latency, more nocturnal awakenings, as well as less time in slow wave sleep and REM sleep. 111 Despite this, polysomnography is not recommended for diagnosis but rather as a tool for patients who have continual sleep complaints after trying various treatments, as there may be other comorbidities contributing to their poor sleep. 111
Similarly, the prevalence of OSA in Canada is between 3%–7% for men and 2%–5% for women, but up to 22% of Canadians report symptoms of OSA. 18 Sleep apnea is also more prevalent among middle-aged adults than youth and older adults, and the prevalence is increasing in Western countries. 18 Unlike insomnia, OSA is diagnosed via a combination of subjective sleep complaints and polysomnographic findings, specifically an apnea-hypopnea index of 5 or greater. 112 Given that these disorders are diagnosed based on being chronic sleep disturbances that affect daytime functioning, we elected to exclude studies that focused solely on populations with diagnosed sleep disorders because they were not within the scope of our research question, which was focused on characterizing sleep in healthy Canadian women. By excluding these types of studies, however, we were able to see that sleep disturbances among healthy Canadian women are pervasive, and it is possible that many participants in our cited literature would meet criteria for a sleep disorder, if they were to seek a formal diagnosis. This highlights the depth of the issue in Canada and reinforces the need for further understanding as well as approaches to improve sleep.
Indigenous peoples and SDH
Sleep disturbances were more prevalent among Indigenous men than women.31,73,74 Numerous environmental, social, and biological factors were identified for their relationship with sleep deprivation, including older age, residential school enrollment, poor housing conditions, and non-traditional tobacco use.31,73 –75 It is well known that Canada’s complicated colonial history continues to permeate the lives of Indigenous peoples, 113 and the findings of this review reinforce the importance of continuing to support efforts to rectify these complex issues. One caveat to consider is that during bias assessments, all studies on Indigenous populations ranked as “Moderate” in their overall risk score. Thus, it is possible that the conclusions drawn from these studies may not be as sound as those taken from higher quality studies; therefore, it will be important to prioritize study design, including using validated assessment tools, for future work on Indigenous populations to strengthen the conclusions that are made.
Furthermore, across age groups and both Indigenous and non-Indigenous populations, some relationships were identified between poor sleep and SDH, with marginalized population subgroups being disproportionally affected. This link has been previously established, 22 but the current review showed that biological sex further affected these relationships. This is not altogether surprising, as gender itself is a social determinant of health, though it is not always concordant with biological sex. However, we demonstrated that girls and women are more susceptible to sleep disturbances than their male counterparts across social, demographic, and economic variables. These findings highlight the need to continue female-focused research to further identify, and minimize, these gaps.
However, it is important to note that the conclusions that can be drawn from this review in terms of women’s sleep and SDH are limited. Although most (n = 34) studies collected data on at least one social determinant, fewer (n = 13) studies conducted sex-stratified analyses on the relationship between these variables and sleep. As such, although many studies investigated the link between SDH and sleep in women and/or men, we were unable to incorporate the data in our review due to the lack of female-focused results. Of the few studies that did investigate the role of SDH in women’s sleep,23,33,36,37,43,49,54,56,59,64,67,68,73,74 some significant differences were shown compared to men and across different social determinants. These preliminary findings highlight the need for future consideration of sex stratification in analyses of sleep and correlates such as SDH, to ensure that an accurate understanding of the effects is being presented.
Older adulthood and menopausal transition
Older adulthood is characterized by another shift in sleep, with results seemingly contradictory at times. Findings showed a shift toward longer sleep in men and women, though women were more likely to experience sleep dissatisfaction23,80 and poor sleep efficiency. 23 This shift toward longer sleep is interesting as older adults tend to have an overall decrease in sleep duration. 114 Post-menopausal women were found to have excessively long sleep duration, which was associated with higher mortality. 78 Interestingly, one study not included in the current review found long sleep duration over the course of the day (including napping and nighttime sleep) to be protective against all-cause and cardiovascular mortality in men and women. 115 On the other hand, short and poor sleep were associated with increased risk of all-cause mortality in men and women with women having a markedly higher risk. 115
During menopause, a loss of ovarian activity leads to substantially decreased levels of estrogen and progesterone, whereas LH and FSH levels rise due to lack of negative feedback. 116 These hormone changes affect numerous aspects of health, including bone density, cardiovascular risk, and sleep. 116 Menopausal women frequently experience a variety of bothersome vasomotor symptoms, such as hot flashes, night sweats, chills, perspiration, palpitations, and anxiety; and genitourinary symptoms, such as urinary incontinence and urinary frequency. 116 The symptoms are attributable to hormone changes, with vasomotor symptoms being due to resetting of the internal thermoregulatory system due to a loss of estrogen, 117 and genitourinary symptoms being caused by estrogen deficiency in the urogenital tract. 116 Perhaps unsurprisingly, these symptoms can cause significant sleep disturbances for women, and decreases in estrogen even in the absence of these uncomfortable symptoms affect sleep. 118
A previous systematic review and meta-analysis noted some menopause-associated sleep deficiencies, finding that these effects differed between women of different ethnicities. 119 In contrast, another study found that sleep characteristics during menopause remained constant with pre-menopausal sleep. 120 In this study, poor premenopausal sleep was the strongest predictor of poor sleep during menopause, and vice versa for not having poor sleep before menopause. 120 Furthermore, sleep difficulties in menopause seemed to be more closely related to changing risk factors rather than biological changes in menopause, even if risk factors remained constant during menopause transition. 121 For example, anxiety and stress were also associated with poor sleep, though causality could not be established. 120
In the current review, we showed that menopause had a negative effect on sleep latency,78,79 but no associations were investigated between sleep, anxiety, other health markers, nor SDH. Thus, it would be worthwhile for future studies to investigate these relationships to further differentiate what might put a menopausal woman at increased risk of poor sleep.
Strengths and limitations
Strengths
This is the first review, to our knowledge, that examined sleep throughout the life course for women living in Canada. Most previous systematic reviews focused on sleep during pregnancy,103,104 or interventions for improving sleep during different life stages. The current review included studies investigating sleep duration and quality, and its associated risk factors—including SDH—and life-stage correlates, providing a critical summary across different age groups. Including only studies investigating sleep in all life stages for women is important because those conducted exclusively in men may not be fully transferable to women as sleep patterns are in part related to biological factors. 122 Additionally, this review included many diverse and under-represented groups, such as Indigenous Peoples31,44,73 –75 and pregnant women51 –54. Data from both remote regions and urban centers, and from coast to coast, were incorporated. As such, it presents a comprehensive picture of sleep health among Canadian women.
Limitations
This systematic review was limited by author bias, which can impact study inclusion and result interpretation. A bias assessment was done and demonstrated a range of bias among included studies, with most having at least one component rated as moderate risk of bias, impacting the conclusions that can be drawn due to study quality. In addition, although some longitudinal studies were included in the current review, the vast majority were cross-sectional, hence reducing the causal conclusions that can be drawn between sleep and covariate measurements. This heterogeneity in study design, combined with the high heterogeneity in sleep measurements and method of reporting and data analysis, precluded any preliminary quantitative analyses or meta-analysis. Furthermore, self-reported measures of sleep, especially those that are not previously validated, are less reliable than objective measures and different measures may introduce differences in the qualification of sleep variables. 119 Self-report sleep quality measures tend to be highly inter-correlated, either due to underlying constructs or inability of participants to distinguish between types of poor sleep. 121 Both a lack of reliability and high intercorrelations between instruments may have contributed to the heterogeneity in sleep variable definitions. Most of the included studies only measured sleep duration, restricting conclusions about sleep health in Canadian women. This is because the concept of sleep health encompasses more than just the length of time someone is asleep, but a collection of attributes encompassing attitudes toward sleep, and practicing proper sleep hygiene .91,123 Given that self-reported sleep duration can be quite inaccurate, 88 and sleep needs vary across ages and between sexes, reporting on sleep latency, refreshment, and other measures is crucial in order to get an accurate picture of an individual’s sleep health.
Furthermore, most studies included in this review focused on biological sex, rather than gender, and sleep, and analyses in relation to correlates such as SDH were limited. Consequentially, some factors affecting sleep may have been overlooked in the current review as they relate to gender identity. In our methodology, we excluded studies that investigated sleep in populations with various diseases, such as cancer and osteoarthritis. As many diseases are known to have negative effects on sleep, it would be important for future systematic reviews to synthesize data on these populations, to better understand how treatments can be improved to optimize the well-being of these individuals. Finally, this review is limited because it draws data from the Canadian populace only; as such, the findings may not be applicable to other parts of the world.
Conclusion
Sleep is an underappreciated component of health, and poor sleep health is associated with both multimorbidity and all-cause mortality.124,125 This systematic review showed that Canadian women are likely to experience sleep problems, including shorter sleep duration and poorer sleep quality, compared to men, with variations across the lifespan and SDH. Furthermore, sleep is impaired during the uniquely female life transitions of pregnancy and menopause, with profound effects on women’s lives and relationships. We also highlighted several longitudinal patterns of sleep, such as its relationship to physical activity. This systematic review has brought both the paucity of female-focused research, as well as the pervasiveness of sleep problems among Canadian women, into focus. This research also highlighted how sleep interacts with SDH and other lifestyle behaviors, and showed that, unfortunately, many of these interactions remain to be investigated. As such, it is essential to continue to invest in sleep research, without overlooking women’s health—which has been long neglected across many research disciplines 126 —and continue to identify the root causes of the widespread sleep problems affecting Canadians.
Supplemental Material
sj-docx-1-whe-10.1177_17455057251367141 – Supplemental material for Sleep patterns of Canadian women across the life course: A systematic review
Supplemental material, sj-docx-1-whe-10.1177_17455057251367141 for Sleep patterns of Canadian women across the life course: A systematic review by Siobhan E. Woods, Patricia Nistor, Kelly K. Anderson and Saverio Stranges in Women's Health
Supplemental Material
sj-docx-2-whe-10.1177_17455057251367141 – Supplemental material for Sleep patterns of Canadian women across the life course: A systematic review
Supplemental material, sj-docx-2-whe-10.1177_17455057251367141 for Sleep patterns of Canadian women across the life course: A systematic review by Siobhan E. Woods, Patricia Nistor, Kelly K. Anderson and Saverio Stranges in Women's Health
Footnotes
Acknowledgements
None.
Ethical considerations
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Author contributions
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Canadian Institutes of Health Research, Team Grant: Sleep Research Consortium awarded to SS.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
All data and materials included in the submission.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.