Abstract
Background:
Discontinuation of the contraceptive pill is common, but little research has explored women’s personal experiences with discontinuation symptoms and associated psychological factors.
Objectives:
These studies provide initial cross-sectional data on symptoms associated with discontinuation of the contraceptive pill, focusing on both expected symptoms in current users and experienced symptoms in former users. We examined how psychological factors (e.g., beliefs about medicine, trait anxiety) relate to these symptoms.
Design:
Two parallel online questionnaires were administered: One with current and another with former contraceptive pill users (n = 642).
Methods:
We analyzed expected discontinuation symptoms in current users and experienced symptoms in former users. Multiple linear regressions assessed the relationships between psychological factors (beliefs about and sensitivity to medicine, trait anxiety, reason for usage, time since discontinuation) and discontinuation symptoms.
Results:
In current users, expected symptoms were associated with concerns about (b = 0.32, p < 0.001, 95% CI [0.35, 0.97], f2 = 0.10) and perceived necessity of (b = 0.24, p < 0.001, 95% CI [0.22, 0.86], f2 = 0.06) the contraceptive pill, perceived overuse of medication (b = 0.17, p = 0.013, 95% CI [0.13, 1.11], f2 = 0.03), and trait anxiety (b = 0.21, p = 0.003, 95% CI [0.06, 0.29], f2 = 0.05). Former users who indicated non-contraceptive reasons for using the contraceptive pill experienced more discontinuation symptoms (b = 1.68, p < 0.001, 95% CI [0.78, 2.59], f2 = 0.03).
Conclusions:
These studies provide the first data on contraceptive pill discontinuation symptoms, showing that current users expect them, and former users experienced them. Psychological factors (e.g., trait anxiety) were linked to expected symptoms, suggesting nocebo-like mechanisms. Future research integrating nocebo theory (e.g., addressing negative expectations directly) could help empower women to make informed choices when discontinuing the contraceptive pill.
Introduction
Discontinuation symptoms of active medication are a rising topic in psychological research. An established definition and a taxonomy are still required. How are discontinuation symptoms to be separated from withdrawal symptoms and potential relapse? A growing body of research has investigated discontinuation symptoms in antidepressant medication.1 –3 Psychological factors that influence these discontinuation symptoms still need to be identified, with expectations being one of the most promising mechanisms.4,5 While most research has focused on the discontinuation of antidepressant medication, psychological factors influencing discontinuation symptoms should be found for different medications, as well. These studies focused on contraceptive pills.
The contraceptive pill is the most commonly used contraceptive method in numerous countries worldwide (e.g., in Europe, Northern America, and Oceania).6,7 While contraceptive pills are licensed to be used for contraception only, 8 studies have identified several non-contraceptive benefits (e.g., controlling the menstrual cycle; improving dysmenorrhea, menorrhagia, acne, and hirsutism).8 –14 Data on off-label use of the contraceptive pill for these non-contraceptive benefits are unclear.14,15 Research indicates that non-contraceptive benefits at least partly account for 40%–60% of the reasons for prescription of contraceptive pills.14 –18 Simultaneously, the usage of the contraceptive pill is associated with aversive side effects (e.g., weight changes, sexual side effects) during intake as well as a fear of negative long-term effects (e.g., on future fertility).19,20
Aversive side effects and a general discomfort with hormone intake consistently emerge among the most common reasons for discontinuation of the contraceptive pill.18,20 –29 Contraceptive discontinuation can be defined as the termination of episodes of use of any reversible contraceptive method due to self-initiated discontinuation, failure, or switching to other methods. 26 Studies indicate high discontinuation rates, also for women still in need of contraception.18,26,27,30,31 This enhances the relevance of discontinuation and potential discontinuation symptoms.
The research concerning discontinuation of the contraceptive pill has mainly focused on future fertility, childbirth, and the elaboration of reasons behind discontinuation.32,33 Women’s personal experiences with discontinuation and discontinuation symptoms are an understudied area of contraceptive research.34,35 To our knowledge, only a few studies exist, and these indicated cycle disturbances post-contraceptive pill intake.36 –38 Meanwhile, studies have proposed that information concerning discontinuation of the contraceptive pill is increasingly posted as personal testimonies online.39,40 Discontinuing the contraceptive pill seems to be accompanied by a variety of symptoms like weight loss, enhanced libido, mood swings, acne, or menstrual pains. Women rely on personal recommendations by their social network as well as the Internet as main sources of information on contraception.18,41 Easily accessed information online, often without link to accredited health agency sources, in combination with a perceived lack of information by clinicians could lead to a distortion of the quality of health information in users.40,42,43 Existing literature seems to have demonstrated that women have knowledge gaps and misconceptions about the contraceptive pill. 42
Information presented in the media can influence expectations. 44 As treatment seems to be more beneficial for patients with more positive expectations, a large body of research has investigated expectations in the context of medication. 45 Expectations are a key mechanism of placebo and nocebo effects.45 –49 Placebo effects are beneficial effects that occur after the administration of an inert treatment. Conversely, nocebo effects are undesired effects after the administration of an inert treatment. 48 Placebo and nocebo effects are also relevant for active pharmacological treatments.48,49 In the case of active medication, nocebo effects are undesired treatment effects that cannot or cannot be fully explained by the pharmacokinetics of the drug. 50 Research has shown that expectations and therefore nocebo mechanisms are linked to the reported appearance and intensity of side effects of active medication, with people who expect more side effects also reporting more.51,52 These expectations are formed by information patients gather about side effects, for example in medical leaflets, during medical encounters, or through information presented in the media.44,53
Expectations and therefore nocebo effects have been discussed to play a role in the unspecific side effects reported for hormonal contraception. 17 As users of the contraceptive pill have indicated concern of delayed or impaired fertility after discontinuation, 32 negative expectations and nocebo effects could likely also play a role in amplifying the intensity of discontinuation symptoms.
Women’s personal experiences with discontinuation of the contraceptive pill and associated symptoms are an understudied area of research. The overall objective of this paper is to present the first cross-sectional data on the influence of psychological factors on discontinuation symptoms. To comprehensively investigate discontinuation, we examined discontinuation symptoms expected by current users as well as discontinuation symptoms experienced by former users. We assumed that higher trait anxiety, higher negative beliefs about medication, stronger perception of one’s sensitivity to medication, and reasons for intake other than contraception (e.g., non-contraceptive benefits) would be associated with higher expected and experienced discontinuation symptoms.
Methods
We conducted two cross-sectional studies as an efficient and economic first investigation of contraceptive pills’ discontinuation symptoms, ensuring their relevance in the target population. To comprehensively investigate discontinuation, two distinct samples were recruited. We examined current (Study 1) and former (Study 2) users of the contraceptive pill. To ensure comparability, both studies were planned in parallel with respect to design, recruitment, and statistical analysis. The STROBE guidelines (Supplemental Material 1) for cross-sectional studies were followed.
Study 1: expected discontinuation symptoms by current users
Design
A cross-sectional online questionnaire was administered to current contraceptive pill users. The comprehensive questionnaire included items on sociodemographic data, intake of the contraceptive pill (e.g., age at onset of intake, duration of intake, reasons for intake, product, changes in product), and discontinuation symptoms (e.g., prior knowledge about, expectations toward, and worries about discontinuation symptoms). Psychological variables (e.g., trait anxiety, beliefs about medication) were measured as potentially associated constructs by using pre-existing questionnaires (below). The complete questionnaire can be found in Supplemental Material 2.
The online study was implemented on the platform SoSciSurvey and took approximately 20 min in total. 54 Recruitment and data acquisition took place from December 19, 2022, to January 3, 2023. There was no incentive for participation.
Recruitment
We conducted an a-priori sample size calculation with G*Power (Version 3.1.9.7). 55 Due to the lack of data on effect sizes with regard to discontinuation of the contraceptive pill, we assumed a medium effect, resulting in a necessary sample size of 114 participants (f2 = 0.15, power = 0.80, α error = 0.05, number of predictors = 9).
Inclusion criteria for participation were age ⩾16 years, competence in German language comprehension, and current usage of a contraceptive pill. There were no specific exclusion criteria except for not meeting the inclusion criteria. The study included a diverse sample of individuals who were biologically eligible to take contraceptive pills (i.e., individuals with a uterus). The link to the online study was distributed via a university’s internal research mailing list established for students and employees, via social media (e.g., X, Instagram), and via word of mouth. For recruitment purposes, individuals who were interested in participating in the study were informed that the study collected information concerning the contraceptive pill, for example on the reasons for intake or the duration of use.
Measures
Primary outcome
Following the line of prior research,56,57 we adapted the German version of the validated General Assessment of Side Effects (GASE) questionnaire 58 to measure expected discontinuation symptoms. We included potential discontinuation symptoms specifically tailored to the contraceptive pill (e.g., increased libido, mood swings, menstrual pains), as well as non-specific symptoms (e.g., headache, nausea, dizziness). Due to the lack of research on potential discontinuation symptoms of contraceptive pills, symptoms were adapted from the package inserts of contraceptive pills. Participants rated which of the 19 symptoms (see Supplemental Material 3) they expected and at what intensity on a 4-point Likert scale (1 “not present,” 2 “mild,” 3 “moderate,” 4 “severe”). Answers can be added up to an intensity-weighted sum score (range: 19–76). The reliability of the GASE is good (Cronbach’s α = 0.89). 58 Additionally, participants indicated their emotional burden concerning potential discontinuation symptoms with one item (“I worry about potential discontinuation effects of the contraceptive pill”) on a 5-point Likert scale (from 1 “strongly disagree” to 5 “strongly agree”). Comparable single-item measures have gained a good reputation in more recent papers and can be rated as very efficient in use. 59
Associated constructs
Another aim of the present study was to identify characteristics associated with the intensity of expected discontinuation symptoms.
Beliefs about medicines, in general, as well as specifically concerning the contraceptive pill, were assessed using the German translation of the 28-item Beliefs About Medicines Questionnaire (BMQ). 60 The BMQ consists of two parts: the BMQ General (BMQ-G) with the subscales overuse (e.g., “Doctors place too much trust in medicines”), harm (e.g., “Medicines do more harm than good”), and benefit (e.g., “In most cases, the benefits of medicines are greater than the risks”), and the BMQ Specific (BMQ-S) with the subscales necessity (e.g., “My life would be impossible without my pill”) and concern (e.g., “I sometimes worry about the long-term effects of my pill”). Agreement on the items was rated on a 5-point Likert scale, ranging from 1 “strongly disagree” to 5 “strongly agree.” For each subscale individually, answers can be added up to a sum score. The reliability of the BMQ varies from bad to good, depending on the subscale (Cronbach’s α = 0.51–0.86). 60
Perceived sensitivity to medicine, indicating the belief that someone is particularly sensitive to both the beneficial as well as the adverse effects of medication, was assessed with the German translation of the Perceived Sensitivity to Medicine (PSM) scale. 61 Participants rate five items (e.g., “I usually have stronger reactions to medicines than most people”) on a 5-point Likert scale (from 1 “strongly disagree” to 5 “strongly agree”). Answers can be added up to an individual item score, ranging 5–25. Higher scores represent higher PSM. Reliability of the PSM scale is acceptable (Cronbach’s α = 0.79). 61
Trait anxiety was quantified using the German version of the State-Trait Anxiety Inventory (STAI). 62 Participants answered 20 items (e.g., “I tire quickly”) on a 4-point Likert scale (from 1 “almost never” to 4 “almost always”). Seven positively framed items were inversely coded. Answers can be summed up, ranging 20–80, with higher sum scores indicating higher anxiety. The reliability of the STAI is excellent (Cronbach’s α = 0.90). 62
The initial reason for intake was quantified with the self-constructed item “Why did you initially start taking the contraceptive pill?” followed by the options contraception, skin blemishes, menstrual pains, extreme length/strength of menstruation, cycle control, mood stability, and other. Multiple answers were possible. For further statistical analyses, answers were grouped as follows: contraception only, contraception and other reasons, other reasons only.
Sociodemographic data
Sociodemographic data were collected with standard items (e.g., age, marital status, education). Additionally, information concerning the contraceptive pill in general was gathered (e.g., duration of intake, type of contraceptive pill, satisfaction with the contraceptive pill, adherence, prior knowledge about discontinuation effects, worries about discontinuation effects). All items concerning sociodemographic data can be found in Supplemental Material 2.
Statistical analyses
Our primary outcome was the intensity-weighted sum score of expected discontinuation symptoms of the contraceptive pill. We tested whether trait anxiety, beliefs about medication, and perception of one’s sensitivity to medication (operationalized as indicated above) were associated with expected discontinuation symptoms. Additionally, we tested whether reasons for intake of the contraceptive pill other than contraception increased the expected intensity of discontinuation symptoms. As this was the first investigation of constructs associated with expected discontinuation symptoms, we could not form any prior assumptions about the differential strengths of these constructs. Therefore, a forced-entry multiple linear regression analysis was conducted.
All statistical analyses were performed with IBMs SPSS Statistics (Version 23.0), 63 applying a two-tailed significance level of 0.05. Effect sizes are reported as Cohen’s f2 and categorized as small (f2 = 0.02), medium (f2 = 0.15), and large (f2 = 0.35) effects. 64 Prior to every statistical analysis, assumptions for the statistical methods (e.g., outliers) were inspected. No adjustments were necessary (e.g., transformation of data).
Results
Participants
No outliers were excluded from the analysis, as there did not seem to be systematic problems in the data acquisition, leaving these data points to represent natural variety in the data. Two hundred eighty-three participants were recruited. All participants provided informed consent. Eighteen participants were excluded for not meeting inclusion criteria (11 participants did not indicate current intake of the contraceptive pill; 2 participants did not indicate female sex; 5 participants did not indicate age ⩾16 years). Thirty-four participants were excluded for not providing full data (27 participants stopped while answering items on the contraceptive pill and on discontinuation symptoms; 11 participants stopped during measurement of potential predictors). In total, 231 current contraceptive pill users fully completed the questionnaire. We would have been interested in examining differential effects on expected discontinuation symptoms, depending on the type of contraceptive pill used. As only a minority (21.2%) of the participants indicated the usage of progestin-only pills (“mini-pill”), we further only examined participants who used a combined oral contraceptive pill (“micro-pill”). One hundred eighty-two participants who used a combined oral contraceptive pill comprised the final sample. Basic demographics and descriptive statistics of the study sample are summarized in Table 1.
Sample characteristics.

SD: standard deviation.
Refers to participants with educational degrees lower than high school.
Refers to participants with high school degrees.
Refers to participants with university degrees.
Reflects the percentage of participants agreeing to the item “Did you hear about discontinuation symptoms of the pill before?”
Reflects the answer to the item “I worry about potential discontinuation symptoms of the pill” on a 5-point Likert scale (from 1 “totally disagree” to 5 “totally agree”).
Outcome data
The multiple regression model for the intensity-weighted sum score of expected discontinuation symptoms was significant, F(9, 172) = 11.88, p < 0.001. The adjusted R2 indicated a 35.1% explained variance (high goodness-of-fit). 64 Expected discontinuation symptoms were significantly associated with the general overuse of medication (b = 0.173, p = 0.013, 95% CI [0.13, 1.11], f2 = 0.03), the necessity of the contraceptive pill (b = 0.24, p < 0.001, 95% CI [0.22, 0.86], f2 = 0.06), concerns about the contraceptive pill (b = 0.32, p < 0.001, 95% CI [0.35, 0.97], f2 = 0.10), and trait anxiety (b = 0.21, p = 0.003, 95% CI [0.06, 0.29], f2 = 0.05). An overview of all results appears in Table 2.
Multiple linear regression on expected discontinuation symptoms within the sample of current contraceptive pill users.

n = 230 (after exclusion of one outlier); adjusted R2 = 0.31 (p < 0.001).
BMQ-G: Beliefs about Medicines Questionnaire, General. 60
PSM: Perceived Sensitivity to Medicine scale. 60
BMQ-S: Beliefs about Medicines Questionnaire, Specific. 60
STAI: State-Trait Anxiety Inventory. 62
SE: standard error.
p < 0.05.
p < 0.001.
Study 2: experienced discontinuation symptoms by former users
Design
As in Study 1, a cross-sectional online questionnaire was administered to a sample of former contraceptive pill users. The comprehensive questionnaire included items on sociodemographic data, intake of the contraceptive pill (e.g., age at onset of intake, duration of intake, reasons for intake, product, changes in product), and discontinuation (e.g., reasons for discontinuation, experienced discontinuation symptoms). Time since discontinuation was measured as a construct potentially associated with discontinuation symptoms. The complete questionnaire can be found in Supplemental Material 2.
This online study was implemented on the platform SoSciSurvey and took approximately 20 min in total. 54 Recruitment and data acquisition took place in two study phases to enlarge the database, from December 19, 2022, to January 3, 2023, and November 10–23, 2023. There was no incentive for participation.
Recruitment
Similar to Study 1, we conducted an a-priori sample size calculation with G*Power (Version 3.1.9.7). 55 Assuming a medium effect due to the lack of data on effect sizes resulted in a necessary sample size of 77 participants (f2 = 0.15, Power = 0.80, α error = 0.05, number of predictors = 3).
Inclusion criteria for participation were age ⩾16 years, competence in German language comprehension, and the former usage of a contraceptive pill. There were no specific exclusion criteria except for not meeting the inclusion criteria. The study included a diverse sample of individuals who were biologically eligible to take contraceptive pills (i.e., individuals with a uterus). Participant recruitment was conducted similarly to Study 1.
Measures
Primary outcome
Comparable to Study 1, we adapted the German version of the validated GASE 58 to measure discontinuation symptoms. In contrast to Study 1, this sample was retrospectively asked for experienced discontinuation symptoms. Participants rated which of the 19 symptoms (see Supplemental Material 3) they experienced and at what intensity on a 4-point Likert scale (1 “not present,” 2 “mild,” 3 “moderate,” 4 “severe”). Answers can be added up to an intensity-weighted sum score (range: 19–76). The reliability of the GASE is good (Cronbach’s α = 0.89). 58
Associated constructs
Another aim of the present study was to identify characteristics associated with the intensity of experienced discontinuation symptoms. Time since discontinuation was quantified with the self-constructed item “When did you discontinue the intake of the contraceptive pill?”. Participants indicated the time since discontinuation (in months or years) in an open-question format.
The initial reason for intake was assessed with the self-constructed item “Why did you initially start taking the contraceptive pill?” followed by the options contraception, skin blemishes, menstrual pains, extreme length/strength of menstruation, cycle control, mood stability, and other. Multiple answers were possible. For further statistical analyses, answers were grouped as follows: contraception only, contraception and other reasons, other reasons only.
Sociodemographic data
Comparable to Study 1, standard items for sociodemographic data as well as a variety of self-constructed items were utilized. All items concerning sociodemographic data can be found in Supplemental Material 2.
Statistical analyses
Our primary outcome was the intensity-weighted sum score of experienced discontinuation symptoms of the contraceptive pill. We tested whether time since discontinuation was associated with the intensity of experienced discontinuation symptoms. Additionally, we tested whether reasons for intake of the contraceptive pill other than contraception increased the experience of discontinuation symptoms. Similar to Study 1, we could not form any prior assumptions about the differential strengths of these constructs. A forced-entry multiple regression analysis was conducted accordingly.
As in Study 1, all statistical analyses were performed with IBMs SPSS Statistics (Version 23.0), applying a two-tailed significance level of 0.05. Effect sizes are reported as Cohen’s f2 and categorized as small (f2 = 0.02), medium (f2 = 0.15), and large (f2 = 0.35) effects. 64 Prior to every statistical analysis, assumptions for the statistical methods (e.g., outliers) were inspected. No adjustments were necessary (e.g., transformation of data).
Results
Participants
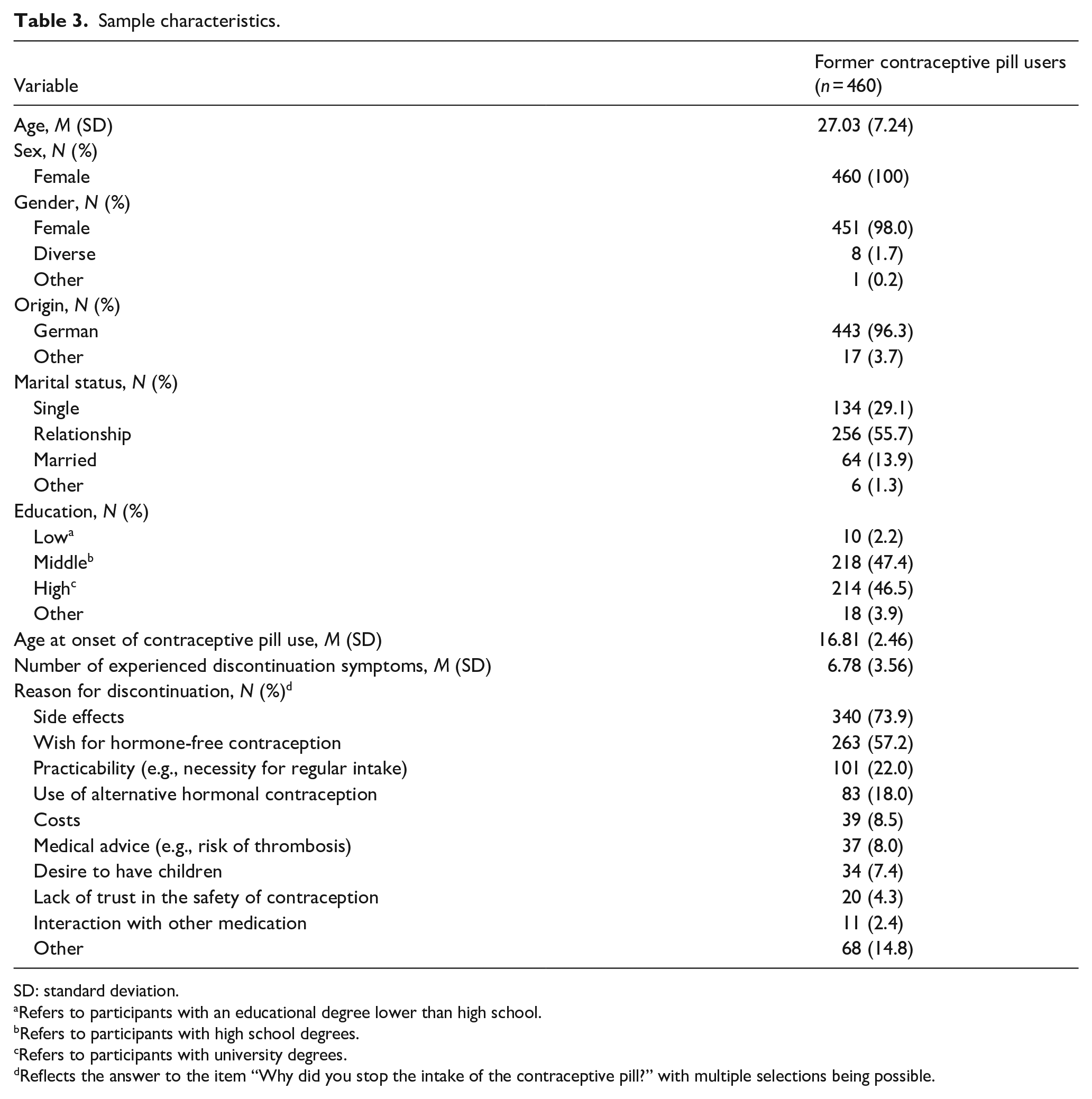
No outliers were excluded from the analysis, as there did not seem to be systematic problems in the data acquisition, leaving these data points to represent natural variety in the data. Six hundred and sixty-two participants were recruited. All participants provided informed consent. Twenty-nine participants were excluded for not meeting inclusion criteria (17 participants did not indicate former intake of the contraceptive pill; 12 participants did not indicate female sex and age ⩾16 years). Eighty-six participants were excluded for not providing full data (70 participants stopped while answering items on the contraceptive pill and on discontinuation symptoms; 16 participants stopped during the measurement of potential predictors). In total, 547 former contraceptive pill users fully completed the questionnaire. Comparable to Study 1, only a minority (15.9%) of the participants indicated the usage of progestin-only pills (“mini-pill”), so we further on only examined participants who used a combined oral contraceptive pill (“micro-pill”). Four hundred and sixty participants who had used a combined oral contraceptive pill comprised the final sample. Basic demographics and descriptive statistics of the study’s sample appear in Table 3.
Sample characteristics.
SD: standard deviation.
Refers to participants with an educational degree lower than high school.
Refers to participants with high school degrees.
Refers to participants with university degrees.
Reflects the answer to the item “Why did you stop the intake of the contraceptive pill?” with multiple selections being possible.
Outcome data
The multiple regression model for the intensity-weighted sum score of experienced discontinuation symptoms was significant, F(3, 456) = 9.91, p < 0.001. The adjusted R2 indicated a 5.5% explained variance (low goodness-of-fit). 64 The intensity-weighted sum score of experienced discontinuation symptoms was significantly predicted by the initial reason for intake. Former users who initially took the contraceptive pill for contraceptive reasons only experienced significantly less intensity-weighted discontinuation symptoms than the sample’s grand mean (b = −0.20, p = <0.001, 95% CI [−2.71, −0.99], f2 = 0.04). Former users who initially took the contraceptive pill for contraception and other reasons experienced significantly more intensity-weighted discontinuation symptoms than the sample’s grand mean (b = 1.68, p = <0.001, 95% CI [0.78, 2.59], f2 = 0.03). An overview of the results is provided in Table 4.
Multiple linear regression on experienced discontinuation symptoms within the sample of former contraceptive pill users.
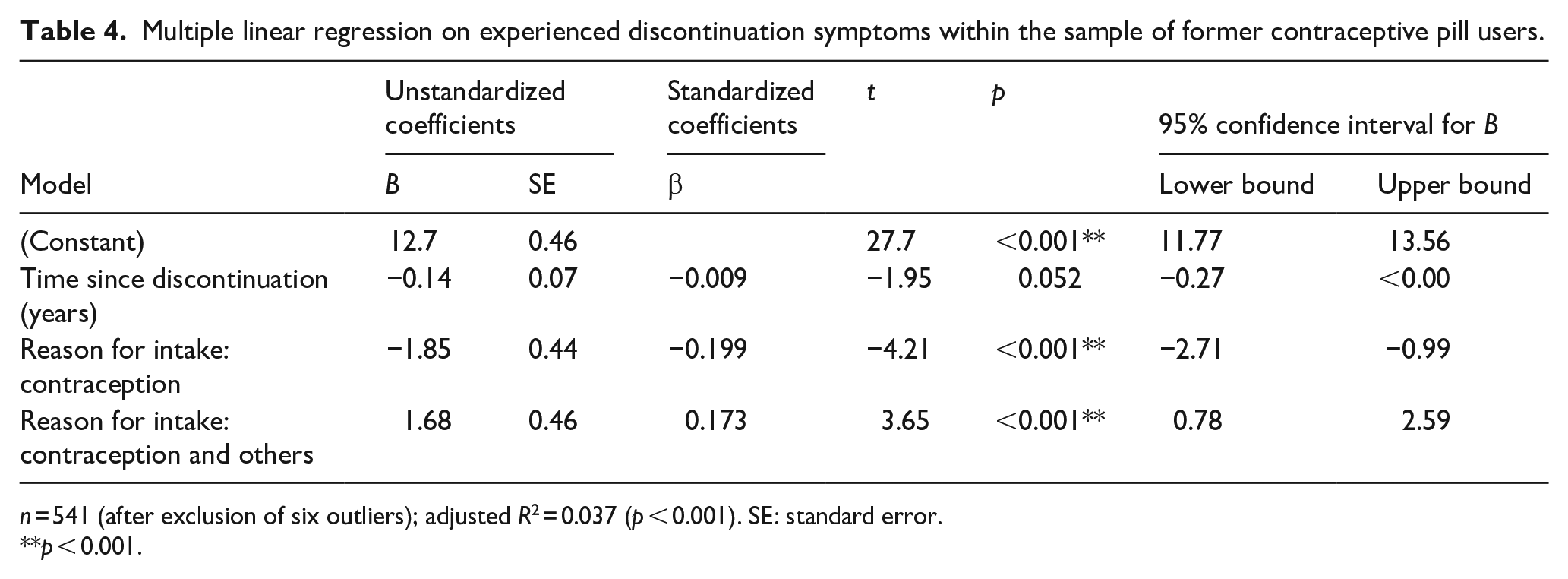
n = 541 (after exclusion of six outliers); adjusted R2 = 0.037 (p < 0.001). SE: standard error.
p < 0.001.
Descriptive comparison of samples
Here we descriptively compare the two study’s main outcomes—expected and experienced discontinuation symptoms. Figure 1 visualizes the percentage of current contraceptive pill users (participants in Study 1) who expected a specific discontinuation symptom in comparison to the percentage of former contraceptive pill users (participants from Study 2) who experienced this discontinuation symptom after discontinuation of the contraceptive pill. For further descriptive data, see Supplemental Materials 4 and 5.
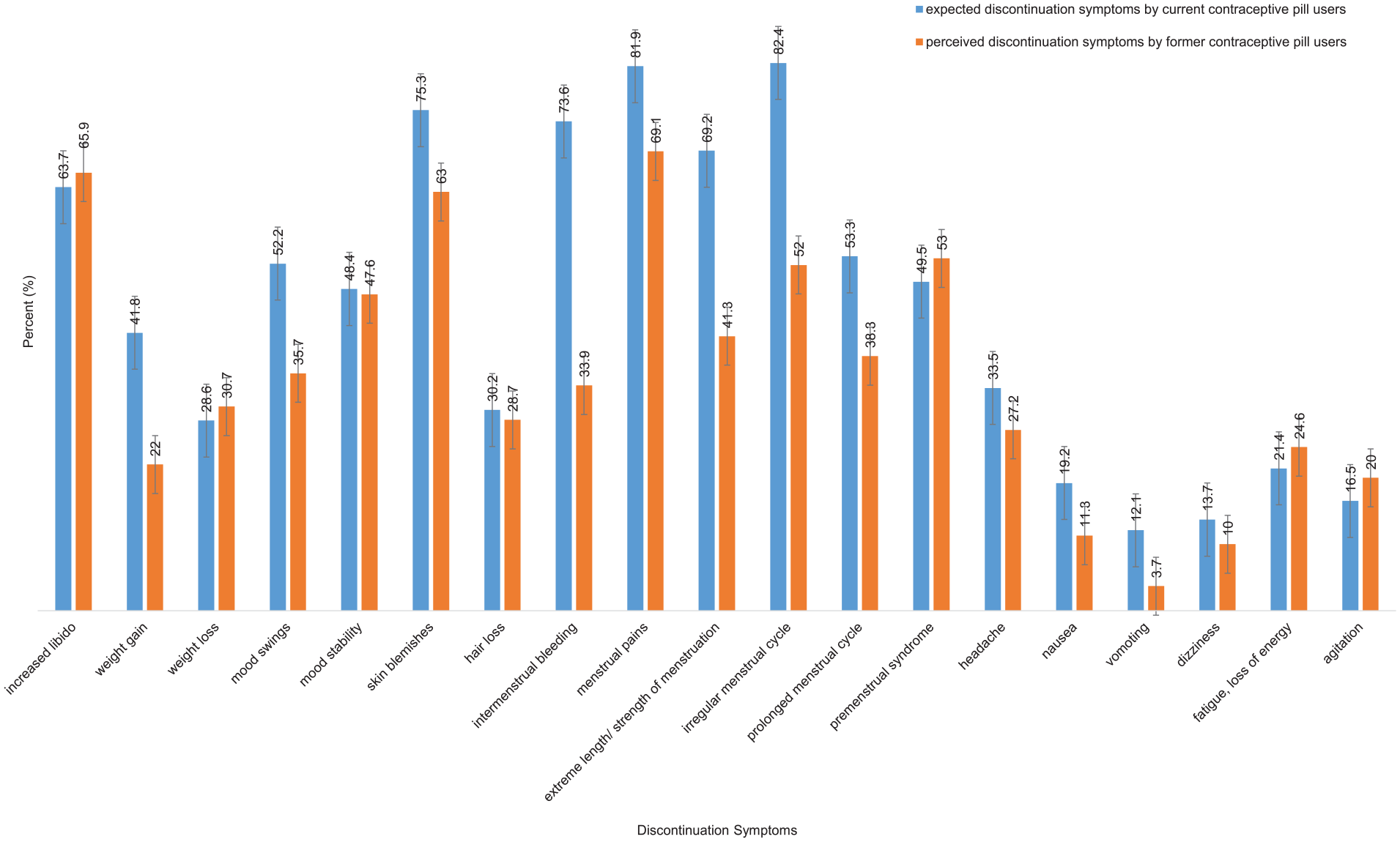
Expected and experienced discontinuation symptoms by current and former contraceptive pill users.
Discussion
The two studies’ results demonstrate the importance of symptoms associated with discontinuing the contraceptive pill: Current contraceptive pill users expected them; former contraceptive pill users experienced them. The cluster of symptoms seemed to be broad, including, for example, an increased libido, irregularities in the menstrual cycle, and menstrual pains. This was the first quantification of discontinuation symptoms, as they were neglected by previous research. Psychological constructs associated with expected discontinuation symptoms (e.g., trait anxiety) resembled those that play an important role in nocebo mechanisms.
In line with the prior hypothesis, results from the first study show that higher concerns about the contraceptive pill, as well as a higher experienced necessity to use it, were associated with a higher intensity of expected discontinuation symptoms. These findings resemble existing literature that targeted side effects, for example, by demonstrating the influence of pre-treatment beliefs about medication on side effect experience in arthritis medication. 65 Greater concerns at baseline predicted side effects 6 months later. The present research suggests the association of medication concerns and experienced necessity for intake also for discontinuation symptoms.
More general constructs such as experienced overuse of medication and trait anxiety were also associated with expected discontinuation symptoms. Participants with a higher perception of medical overuse and those indicating higher trait anxiety expected a higher intensity of discontinuation symptoms. In contrast, other general concepts like the perception of benefit or harm regarding medication did not seem to be associated with expected discontinuation symptoms. A possible explanation is that the overuse of contraceptive pills as well as negative side effects of the contraceptive pill are widely discussed online.39,40 This could influence expectation formation 4 and therefore expected discontinuation symptoms.
Contrary to the prior hypothesis, there was no significant association of expected discontinuation symptoms with experienced sensitivity to medication. This finding differs from earlier research that demonstrated more symptoms following the beginning of treatment (e.g., vaccination, antiretroviral therapy for HIV) for people indicating higher experienced sensitivity to medication.61,66 A possible explanation is the difference between the uptake and the discontinuation of medication. People perceiving themselves to be more sensitive toward symptoms when taking medication might not expect to have more symptoms after discontinuation.
In line with the prior hypothesis, the results demonstrate that the initial reason for intake of the contraceptive pill was associated with experienced symptoms at discontinuation. The use of the contraceptive pill for contraceptive reasons only was associated with a lower intensity of experienced discontinuation symptoms. Additional non-contraceptive reasons for intake (e.g., menstrual pains) were associated with a higher intensity of experienced discontinuation symptoms. The reemergence of a previously suppressed condition seems likely. However, the nature of the study’s data does not allow for a determination of whether these more intense symptoms at discontinuation stem from biological processes and an underlying condition that is no longer suppressed by the medication alone, or if they are also retraceable to the process of discontinuation. No matter the reason behind the occurrence of symptoms, there seemed to be an association with experienced symptom burden. The association of non-contraceptive reasons for intake and experienced discontinuation symptoms is highly relevant, as some literature has indicated high rates of usage of the contraceptive pill for non-contraceptive reasons.15 –18 There was no comparable association between reasons for intake of the contraceptive pill and the current users’ expectations toward discontinuation symptoms.
Regarding the overall pattern, there seemed to be a similarity between the discontinuation symptoms that current contraceptive pill users expected and those that former contraceptive pill users experienced. The literature on side effect research has demonstrated that side effects appear more frequently when they are expected by patients.52,53,57,67 A comparable mechanism is plausible for discontinuation symptoms. Perhaps negative expectations and therefore nocebo-like mechanisms amplify the experience of symptoms that occur naturally. A dominantly expectation-based occurrence of discontinuation symptoms is rather unlikely, yet the influence of nocebo-like mechanisms would suggest a window of influence that could be leveraged to help mitigate the experience of negative symptoms. At the same time, it is critical to acknowledge the reality of women’s experiences and ensure that these symptoms are not underestimated or dismissed. An example could be empathic doctor–patient communication that reduces the anticipation of negative symptoms while validating the genuine challenges women might face due to discontinuation symptoms.
For some symptoms (e.g., irregularities in the menstrual cycle, intermenstrual bleeding, extreme length or strength of the menstruation), the data indicated a more substantial gap between expectation and experience. More symptoms were expected than retrospectively experienced. It remains to be researched if these symptoms are especially prone to nocebo-like mechanisms.
The two studies’ results should be considered in light of the studies’ strengths and limitations. The studies were the first to empirically investigate discontinuation symptoms of the contraceptive pill. This research augmented the primary focus of prior studies on fertility, childbirth, and reasons for discontinuation by asking for personal experiences with discontinuation.32,34,35 A large number of participants (n = 642 in both studies combined), exceeding the required sample size, strengthens these first-derived implications. Nevertheless, the cross-sectional nature of the data must be noted as the studies’ strongest limitation. While expectations of symptoms are a relevant predictor of symptom experience (e.g., side effects),44,51 the concrete number of emerging symptoms in the case of discontinuation remains uncovered. Study 2 provides the first estimations of discontinuation symptoms, yet the goodness-of-fit of the regression model was low, and the data were likely to be biased by memory effects, as there was no limit for the time since discontinuation of the contraceptive pill (e.g., in the last year). Additionally, the generalizability of the data is limited by the participants’ relatively high educational background and the fact that the sample was composed solely of German users.
The limitations of cross-sectional data indicate the most important implication for future research. A broader, longitudinal database is necessary to ensure the validity of conclusions. Nuanced observational studies with ecological momentary assessments could be the next research step to fill the knowledge gap on the progression and course of discontinuation symptoms. Predictors different to prior reasons for intake should be incorporated to foster predictive power. A more detailed assessment could also offer the possibility to disentangle symptoms solely retraceable to the process of discontinuation from symptoms that indicate the recurrence of an underlying condition. This could also help in discriminating these different forms of symptoms and therefore aid a more sophisticated definition of discontinuation symptoms. Moreover, reappearing links to nocebo-like mechanisms (e.g., expectation of discontinuation symptoms) indicate a need for further research in this field. Experimental studies on the influence of expectations during discontinuation are necessary. 4 If nocebo-like mechanisms really do play a role in discontinuation symptoms of the contraceptive pill, they should also be susceptible to modification. Interventional trials that specifically target and possibly minimize nocebo mechanisms before and during the discontinuation process could be a promising approach. Classical nocebo research (e.g., framing) might be transferred to discontinuation symptoms. Personal characteristics (e.g., initial reasons for intake) seem to be influential and should be further elucidated.
Conclusion
This first investigation of the contraceptive pills’ discontinuation symptoms showed which discontinuation symptoms prior users report retrospectively. These symptoms seemed to be largely similar to the symptoms that current users of the contraceptive pill expected when asked about future discontinuation. Associated constructs (e.g., trait anxiety) indicated nocebo-like mechanisms that influence the formation of these expectations regarding future discontinuation. Further research that integrates existing knowledge about nocebo mechanisms (e.g., addressing negative expectations directly) could help lay the groundwork to empower women to make informed decisions when starting and discontinuing the contraceptive pill.
Supplemental Material
sj-docx-1-whe-10.1177_17455057251338401 – Supplemental material for Expectations versus reality: Two cross-sectional studies on expected and experienced discontinuation symptoms in current and former contraceptive pill users
Supplemental material, sj-docx-1-whe-10.1177_17455057251338401 for Expectations versus reality: Two cross-sectional studies on expected and experienced discontinuation symptoms in current and former contraceptive pill users by Philine Elise Wienand and Marcel Wilhelm in Women’s Health
Supplemental Material
sj-docx-2-whe-10.1177_17455057251338401 – Supplemental material for Expectations versus reality: Two cross-sectional studies on expected and experienced discontinuation symptoms in current and former contraceptive pill users
Supplemental material, sj-docx-2-whe-10.1177_17455057251338401 for Expectations versus reality: Two cross-sectional studies on expected and experienced discontinuation symptoms in current and former contraceptive pill users by Philine Elise Wienand and Marcel Wilhelm in Women’s Health
Supplemental Material
sj-docx-3-whe-10.1177_17455057251338401 – Supplemental material for Expectations versus reality: Two cross-sectional studies on expected and experienced discontinuation symptoms in current and former contraceptive pill users
Supplemental material, sj-docx-3-whe-10.1177_17455057251338401 for Expectations versus reality: Two cross-sectional studies on expected and experienced discontinuation symptoms in current and former contraceptive pill users by Philine Elise Wienand and Marcel Wilhelm in Women’s Health
Supplemental Material
sj-docx-4-whe-10.1177_17455057251338401 – Supplemental material for Expectations versus reality: Two cross-sectional studies on expected and experienced discontinuation symptoms in current and former contraceptive pill users
Supplemental material, sj-docx-4-whe-10.1177_17455057251338401 for Expectations versus reality: Two cross-sectional studies on expected and experienced discontinuation symptoms in current and former contraceptive pill users by Philine Elise Wienand and Marcel Wilhelm in Women’s Health
Supplemental Material
sj-docx-5-whe-10.1177_17455057251338401 – Supplemental material for Expectations versus reality: Two cross-sectional studies on expected and experienced discontinuation symptoms in current and former contraceptive pill users
Supplemental material, sj-docx-5-whe-10.1177_17455057251338401 for Expectations versus reality: Two cross-sectional studies on expected and experienced discontinuation symptoms in current and former contraceptive pill users by Philine Elise Wienand and Marcel Wilhelm in Women’s Health
Supplemental Material
sj-docx-6-whe-10.1177_17455057251338401 – Supplemental material for Expectations versus reality: Two cross-sectional studies on expected and experienced discontinuation symptoms in current and former contraceptive pill users
Supplemental material, sj-docx-6-whe-10.1177_17455057251338401 for Expectations versus reality: Two cross-sectional studies on expected and experienced discontinuation symptoms in current and former contraceptive pill users by Philine Elise Wienand and Marcel Wilhelm in Women’s Health
Footnotes
Ethical considerations
These studies were performed in accordance with the Declaration of Helsinki and were approved by the Ethics Committee of the Department of Psychology, chairperson Prof. Dr. Daniel Heck, Philipps-University of Marburg, Gutenbergstraße 18, 35032, Marburg, on December 14, 2022 (approval number: 2022-75k).
Consent to participate
All participants provided informed consent to participate by clicking “YES” regarding the statement “I was informed about the questionnaire. I agree to participate in this online study” prior to enrolment in the study. All participants provided informed consent for themselves. In Germany, individuals are eligible to provide informed consent for study participation starting at the age of 16 years. In accordance with the Ethics Committee of the Department of Psychology, Philipps-University Marburg, we included participants who were at least 16 years old.
Author contributions
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Open access funding provided by the Open Access Publishing Fund of Philipps-University Marburg.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets generated during and/or analyzed during the current studies are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
