Abstract
Although combined oral contraceptives (COCs) are commonly used and highly effective in preventing pregnancy, they may not be suitable for some women. COC use is associated with increased rates of cardiovascular events and is not recommended in nonbreastfeeding women in the immediate postpartum period or in breastfeeding women during the initial 6 months of breastfeeding. Moreover, estrogen-related adverse effects, such as headache, are common. Estrogen-free progestin-only pills (POPs) are a valuable option in women who prefer to take an oral hormonal contraceptive, but are ineligible for, or choose not to use, COCs. Although some POPs have been associated with lower contraceptive effectiveness than COCs, the POP containing desogestrel has shown similar contraceptive effectiveness to COCs. The most commonly reported complaints in women using all POPs are bleeding problems. Counseling women interested in using POPs about the variable bleeding patterns associated with this method may improve compliance and acceptance.
Keywords
Since the introduction of the first hormonal contraceptive in 1960, ‘the pill’ has become the most widely established method of reversible hormonal contraception around the world. The pill is currently used by 17% of women in the world's more developed regions and 7% of those in less developed regions [201]. Following decades of manipulation of the specific types and dosages of estrogens and progestins used in oral contraceptives (with modifications aimed at minimizing adverse effects without compromising efficacy) dozens of different formulations of combined oral contraceptives (COCs) and progestin-only pills (POPs) are currently available for women who favor an oral form of hormonal contraception.
Over the past 50 years, COCs have demonstrated an excellent overall contraceptive efficacy and safety record. The progestin component of COCs provides the primary contraceptive mechanisms of action, whereas the estrogen component provides cycle control and adds to the ovulation inhibitory action of progestin [1]. Recent reports indicate that the overall incidence of ovulation with COCs is 2.0% [2], and only 0.3% of women experience an unintended pregnancy during the first year of perfect COC use [3].
Beyond effective contraception, COCs also offer other important health benefits, including improvements in menstrual bleeding and related symptoms, and decreased risk of iron-deficiency anemia [4]. In the Royal College of General Practitioners' Oral Contraception Study, conducted in a cohort of 46,112 women observed for up to 39 years, COC users had significantly lower rates of death from any cause, as well as death from all cancers and several specific cancers, including uterine and ovarian cancers, compared with never-users [5]. However, use of COCs may constitute an unacceptable health risk in some women, such as in those with cardiovascular risk factors, and may result in intolerable adverse effects in others. Indeed, according to findings of the most recent US National Survey of Family Growth, nearly a third of oral contraceptive users discontinue therapy due to dissatisfaction, which is attributed to adverse effects in most women [6].
Women seeking oral contraception who are not suitable candidates for COCs may find estrogen-free POPs to be a viable alternative. The early POPs contain 19-nortestosterone progestins such as norethindrone (norethisterone) and levonorgestrel. To minimize androgenic adverse effects, the progestin dose used in these pills is lower than in COCs. As a result, their contraceptive action is derived primarily from effects on mucosal thickening and thinning of the endometrial lining, rather than ovulation inhibition [1]. Compared with COCs, traditional POPs (i.e., those containing norethisterone or levonorgestrel) are associated with a higher incidence of ovulation (42.6%) [2]. Pregnancy rates with POPs may approximate those with COCs [7], ranging from 1 to 13% within the first year of use [3,8], but these rates may differ between traditional and newer POP formulations [9]. Newer progestins, including desogestrel and norgestimate, were developed from the levonorgestrel prototype to reduce androgenic adverse effects and increase progestational activity [10]. The POP containing desogestrel 75 μg has been shown to inhibit ovulation (incidence: 1.25%) to a similar extent as COCs [2]. Furthermore, the desogestrel 75 μg POP is associated with a lower Pearl Index than levonorgestrel 30 μg (0.14 vs 1.17 pregnancies per 100 woman-years) [9].
The aim of this article is to explore major obstacles to the use of COCs and describe the strengths and limitations of POPs as a valuable alternative method of family planning.
Estrogen-containing oral contraceptives: potential barriers to use
Healthcare providers can help women choose from the wide range of available contraceptive methods by providing complete product information. For women who express a personal preference for oral hormonal contraception, details on COCs and POPs should be offered. Ideally, this should include a description of their mechanisms of action, as well as an overview of their product failure rates, safety issues, common adverse effects, costs and requirements for services and supplies. Both types of oral contraception have advantages and disadvantages that should be weighed along with the woman's individual medical history, lifestyle, attitudes and perceptions. The specific safety and tolerability issues related to COC use that women and healthcare professionals should consider during the decision-making process are summarized in the following sections. Because the synthetic estrogen ethinyl estradiol has been used almost exclusively in COC formulations since the pill's introduction, the term ‘estrogen’ appearing in these sections and elsewhere in this article generally refers to this molecule.
Cardiovascular risks
Since their introduction in the 1960s, important associations have been identified between use of COCs and cardiovascular disease. As a result, the US FDA mandates that COC product labeling contain contraindications for women with current or past history of thromboembolic disorders, cerebrovascular or coronary artery disease, and diabetes with vascular disease. In addition, warnings regarding the risks of thromboembolic disorders and other vascular problems associated with use of COCs are clearly stated in COC labeling.
Venous thromboembolism
Estrogen-related effects on the risk of venous thromboembolism (VTE) are among the main concerns. Epidemiological evidence suggests that COCs are associated with an increased risk of VTE, although the absolute risk is relatively low (i.e., 3.0 per 10,000 women per year in COC users vs 0.8 per 10,000 women per year in premenopausal COC nonusers) [11]. Estimates of the relative VTE risk associated with COCs vary considerably among studies; one review of findings from case–control studies suggested that the risk was approximately four- to eight-times greater in current COC users compared with nonusers [12].
In the European Active Surveillance Study (EURAS), which followed 58,674 women for 142,475 woman-years, the VTE risk ranged from approximately eight to ten per 10,000 woman-years in current COC users compared with approximately five per 100,000 woman-years in non-COC-using women of reproductive age
Incidence of venous thromboembolism among 58,674 women who participated in the European Active Surveillance Study trial on oral contraceptive use.

COC: Combined oral contraceptive.
Adapted with permission from [13].
Venous thromboembolism risk in women with prothrombotic conditions is increased further with use of COCs. Hereditary or acquired thrombophilias are therefore considered contraindications for COC use. In an observational study in which researchers compared 155 premenopausal women with deep-vein thrombosis with 169 population controls, the relative risk (RR) for VTE was increased nearly fourfold in COC users compared with controls (RR: 3.8; 95% CI: 2.4–6.0) [11]. The risk of thrombosis among women who used COCs and were carriers of the factor V Leiden mutation (hetero- and homozygous) was nearly 35-times higher than that among non-COC users who were not carriers of this mutation (RR: 34.7; 95% CI: 7.8–154); this risk was increased by nearly 100-fold in COC users who were homozygous carriers of the mutation [11]. Results from a subsequent population-based, case–control study confirmed that users of COCs and factor V Leiden carriers have elevated RR of idiopathic VTE, although the risk was somewhat lower than previously reported (odds ratio [OR]: 10.2; 95% CI: 3.8–27.6) [30]. In a well-conducted screening study, COC use was shown to significantly increase the risk of VTE in women with thrombophilias, including deficiencies of antithrombin (OR: 12.60; 95% CI: 1.37–115.62) and protein C (OR: 6.33; 95% CI: 1.68–23.87), and elevation of factor VIIIc levels (OR: 8.80; 95% CI: 4.13–18.75) [31]. In addition, as described in COC prescribing guidelines, COC use may pose an undesirable health risk in women who do not have a current or past history of thrombosis or prothrombotic conditions, but who have other VTE risk factors (e.g., hypertension, dyslipidemia, diabetes, smoking and/or obesity)
Overview of oral hormonal contraceptive prescribing guidelines for women with cardiovascular risk factors.

BP: Blood pressure; COC: Combined oral contraceptive; CV: Cardiovascular; LDL-C: Low-density lipoprotein cholesterol; POP: Progestin-only pill.
Adapted with permission from [34].
Myocardial infarction
Results of studies examining the risk of myocardial infarction (MI) in current COC users have been inconsistent. In a pooled analysis of two population-based case–control studies, researchers evaluated the relationship between low-dose COC use and MI risk in women aged 18–44 years who had no prior history of ischemic heart disease or cerebrovascular disease (271 MI cases; 993 controls) [35]. After adjustment for major risk and sociodemographic factors, current COC users had a pooled OR for MI of 0.94 (95% CI: 0.44–2.20) compared with noncurrent users, and 0.56 (95% CI: 0.21–1.49) compared with never-users. By contrast, a meta-analysis of the association between oral contraceptive use and MI risk in 19 case–control studies and four cohort studies showed an overall adjusted OR for MI of 2.48 (95% CI: 1.91–3.22) in current COC users compared with COC never-users; MI risk was not significantly different for past COC users (OR: 1.15; 95% CI: 0.98–1.35) versus never-users [36]. Researchers conducting a more recent meta-analysis, including 14 independent peer-reviewed studies of the cardiovascular risk associated with low-dose COCs, reported an OR for MI of 1.84 (95% CI: 1.38–2.44) among current COC users compared with controls
Associations between current combined oral contraceptive use and cardiovascular disease outcomes.

COCs: Combined oral contraception.
Data taken from [32].
Inconclusive findings have been reported in five studies that directly compared the effects of second-generation COCs (i.e., low-dose COCs containing levonorgestrel or a progestogen other than gestodene or desogestrel) and third-generation COCs (i.e., low-dose COCs containing gestodene or desogestrel) on MI risk [37–43]. Only two of these studies were designed to detect a difference in the effect of second- and third-generation COCs on MI risk and included an adequate number of women taking third-generation COCs to allow a meaningful comparison [41,42]. In a study that included 448 cases (women aged 16–44 years who had an MI between 1993 and 1995) and 1728 matched controls, Dunn et al. reported that MI risk was higher for women receiving third-generation COCs than for those taking second-generation COCs (OR: 1.8; 95% CI: 0.7–4.8), although the difference observed was not statistically significant [41]. However, Tanis et al. observed a lower MI risk with third-generation compared with second-generation oral contraceptives (OR: 0.5; 95% CI: 0.2–1.1) in a similar study conducted in 248 cases (women aged 18–49 years who had an MI between 1990 and 1995) and 925 controls [42]; however, the CI was wide for the latter finding, which may preclude a definite conclusion. The risk for MI was increased relative to nonusers in women receiving second-generation COCs (OR: 2.5; 95% CI: 1.5–4.1), but not in women receiving third-generation COCs (OR: 1.3; 95% CI: 0.7–2.5).
Stroke
According to the results of three meta-analyses, the risk of stroke appears to be increased in women currently exposed to COCs, including preparations containing low doses of ethinyl estradiol [32,44,45]. Gillum et al. analyzed data from 16 studies and found an ischemic stroke RR of 2.75 (95% CI: 2.24–3.38) in current COC users [44]. Although lower ethinyl estradiol dosages were associated with lower risk, all ethinyl estradiol dosages were associated with a significantly elevated stroke risk. Chan et al. reported a significant association between COC use and stroke risk based on pooled results from 16 case–control studies (OR: 2.13; 95% CI: 1.59–2.86); however, the pooled OR from four cohort studies showed no increase in stroke risk among COC users (OR: 0.95; 95% CI: 0.51–1.78) [45]. In a recent meta-analysis, which included 14 independent cohort or case–control studies, current use of low-dose, second- and third-generation COCs significantly increased the risk for ischemic stroke (overall OR: 2.12; 95% CI: 1.56–2.86)
Multiple cardiovascular risk factors
Use of COCs may be discouraged in women who have additional cardiovascular risk factors. COC use has been found to further compound the risk of MI and stroke associated with migraine, especially migraine with aura [46–49]. Hypertensive COC users also appear to have unacceptably high MI and stroke risks, as do COC users who are more than 35 years of age, smoke or have hypercholesterolemia or diabetes [40,42,50–53].
A woman's age is a particularly important consideration when determining COC-associated risks. Mortality from cardiovascular disease is very low in women of reproductive age, but the mortality risk rises with increasing age and presence of other risk factors. The risk of VTE, MI and stroke associated with COC use in women less than 35 years of age is small, even in smokers, but the risk increases substantially in older women who are COC users and smoke [53]. The adverse effects of COC use on cardiovascular disease incidence and mortality can be reduced by screening women for risk factors, both before and during COC use. The opportunity for risk reduction via appropriate screening is particularly great in older women.
Carbohydrate & lipid metabolism
Use of COCs appears to influence carbohydrate and lipid metabolism, although the effects may be modest and vary depending on the dose of ethinyl estradiol and the dose and type of progestin used. In large nonrandomized studies, worsening of glucose tolerance was observed in COC users overall [54–56], but this effect was deemed minimal when only low-dose COC users were evaluated [57]. Based on a systematic review of findings from randomized clinical trials, Lopez et al. concluded that combined hormonal contraceptives, including COCs, have limited clinical impact on carbohydrate metabolism in women without documented diabetes [58]. Inconsistent findings, ranging from dyslipidemias [59] to eulipidemia, to beneficial effects on lipid parameters [60,61], have been reported in randomized trials evaluating the effects of various formulations of COCs on serum lipid levels in general populations of women.
In a systematic review that included three randomized clinical trials of women with Type I or II diabetes mellitus [62–64], Visser et al. reported conflicting outcomes related to carbohydrate and lipid metabolism with COC use [65]. High-dose COCs and one low-dose COC (i.e., 30 μg ethinyl estradiol plus 75 μg gestodene) were found to slightly impair glucose homeostasis, whereas other low-dose COCs had no such effect [65]. In a study in women with diabetes, COCs were associated with significant increases from baseline in serum cholesterol and triglyceride levels, although these levels remained within the normal range [64]. Investigators for the two other studies reported either no significant changes in lipid metabolism or favorable changes before and after COC use [62,63], but some significant differences were observed between COC regimens [63]. The metabolic evidence surrounding use of COCs to date is difficult to interpret. However, because even relatively small changes in glucose tolerance and insulin resistance may increase the risk of Type II diabetes and cardiovascular disease, caution may be warranted when considering COCs as a contraceptive option in women at risk for diabetes or vascular disease.
Breast cancer risk
Although COCs have been shown to protect women from certain reproductive cancers (e.g., endometrial and epithelial ovarian cancers [5,66–71]), the pills are contraindicated in women who currently have, or have had in the past, breast cancer or other estrogen- or progestin-sensitive cancer. Warnings about COC-related breast cancer risk are primarily derived from the findings of a pooled analysis of 54 epidemiological studies conducted by the 9th Collaborative Group on Hormonal Factors in Breast Cancer, which included more than 53,000 women with breast cancer and 100,000 controls [72]. Small increases were observed in the risk of localized breast cancer in women less than 35 years of age who were current COC users (RR: 1.24; 95% CI: 1.15–1.33), and who had used COCs in the previous 1–4 years (RR: 1.16; 95% CI: 1.08–1.23) and in the previous 5–9 years (RR: 1.07; 95% CI: 1.02–1.13) compared with controls

Relative risk (shown with 95% CI) of breast cancer in combined oral contraceptive users with respect to time since last use compared with combined oral contraceptive never-users.
Research results from a historical cohort study suggest that women with a family history of breast or ovarian cancer (specifically, a mother or sister) and who used early, high-dose formulations of COCs have a threefold higher risk of breast cancer (RR: 3.3; 95% CI: 1.5–7.2) than women without a family history [73]. In a meta-analysis of 34 case–control studies, prior COC use was associated with a 19% overall increase in premenopausal breast cancer risk [74]. A stronger association was found in women who used COCs before their first full-term pregnancy (OR: 1.44; 95% CI: 1.28–1.62) compared with those using COCs after their first pregnancy (OR: 1.15; 95% CI: 1.06–1.26). The magnitude of this association was greater in those women using COCs for 4 years or more before their first pregnancy (OR: 1.52; 95% CI: 1.26–1.82). However, conflicting reports about breast cancer risk in COC users have been published. The Centers for Disease Control and Prevention, which conducted a population-based, case–control study including 4575 women with breast cancer and 4682 controls, found no significant increase in breast cancer risk among current or former COC users and no increase in risk associated with family history of breast cancer or initiation of COC use at a young age [75].
Postpartum/breastfeeding
Owing to the increased risk of thromboembolism, oral contraceptives containing estrogen are not recommended for use in nonbreastfeeding women in the immediate postpartum period. The WHO advises that nonlactating women avoid COCs in the first 3 weeks postpartum Table 4) [76]. Class labeling for COCs in the USA recommends that breastfeeding mothers use other forms of contraception until they have weaned their children because estrogen-containing COCs can reduce milk production [202]. Combined oral contraceptives are usually not recommended for use in breastfeeding women because estrogen appears to have a detrimental effect on the quantity and quality of breast milk produced. According to WHO guidelines, the risks of COC use during the first 6 months of breastfeeding generally do not outweigh the benefits [76]. Indeed, this contraceptive method is considered to be the least preferred choice during any state of lactation [76,203]. The WHO recommendations are consistent with other practice guidelines, including those of the International Planned Parenthood Federation [77] and the Royal College of Obstetricians and Gynaecologists [204]. However, according to a 2003 systematic review of the literature, evidence from randomized controlled studies evaluating the effects of hormonal contraceptives, including COCs, on lactation is limited and of poor quality [78].
WHO medical eligibility criteria for the initiation and continuation of combined oral contraceptive and progestin-only pill use in postpartum and breastfeeding women.
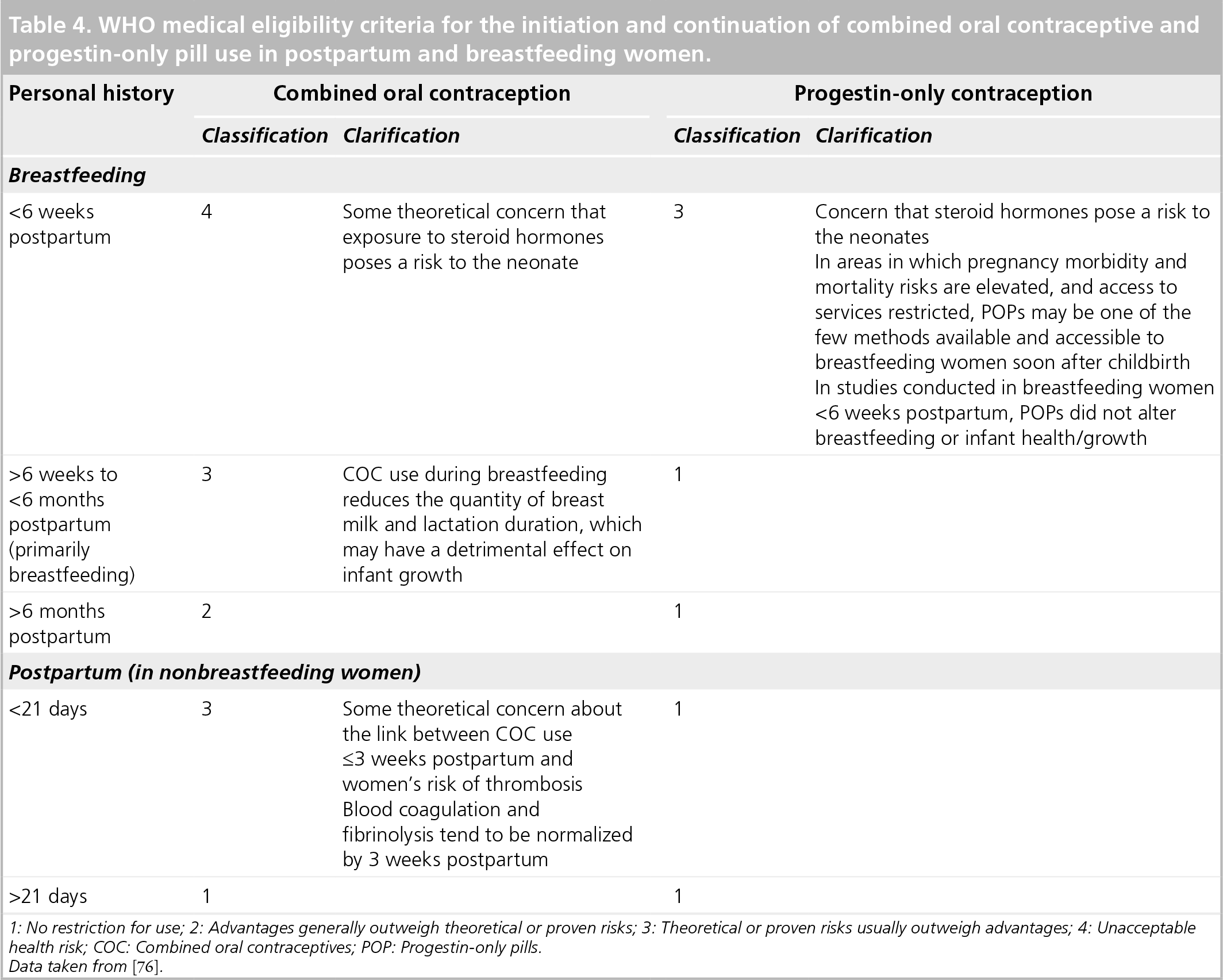
1: No restriction for use; 2: Advantages generally outweigh theoretical or proven risks; 3: Theoretical or proven risks usually outweigh advantages; 4: Unacceptable health risk; COC: Combined oral contraceptives; POP: Progestin-only pills.
Data taken from [76].
Adverse effects
The ethinyl estradiol contained in COCs can cause undesirable adverse effects that may have a detrimental effect on quality of life and result in discontinuation of this contraceptive method. Although the incidence and severity of adverse effects associated with the early COCs has improved as the ethinyl estradiol dose has been progressively reduced from as high as 100 μg to as low as 15 μg, the low-dose COCs are more likely to provide inadequate cycle control. In a systematic review of trials comparing the effects of COCs that contained no more than 20 μg ethinyl estradiol with those containing more than 20 μg, women receiving the lower dose COCs showed higher rates of early discontinuation due to irregular bleeding patterns, higher rates of bleeding disturbances and higher rates of some adverse effects (including headache, dizziness, mood changes and weight gain) than women receiving the higher-dose COCs [79].
Hormone-dependent headache and menstrual migraine are among the most common adverse effects reported with COCs containing ethinyl estradiol [80,81]. Headaches associated with COCs may be partly related to the periodic discontinuation of the oral hormones during pill-free intervals [81,82]. In an open-label pilot study (n = 20), most COC-induced migraines appeared within the first 5 days after pill cessation, were rated as moderate to severe, and lasted for at least 1–3 days [83]. Other unwanted effects related to ethinyl estradiol that can lead to COC discontinuation include breast tenderness, mood changes, bloating and nausea [84]. In a study of 1716 young (aged <25 years) women who initiated COCs in a family-planning clinic, women who reported an increased frequency of headaches or moodiness after starting this method of contraception were more likely to discontinue use [85]. According to the most recent report from the US National Survey of Family Growth, 29% of pill users discontinued because of dissatisfaction, with 65% of these women reporting adverse effects as the reason for discontinuation [6].
Dysmenorrhea
As many as half of menstruating women experience primary dysmenorrhea, which can negatively affect their daily function, quality of life and workplace attendance [86]. This condition is associated with prostaglandin- and eicosanoid-induced uterine contractions that result in uterine ischemia and subsequent pain fiber sensitization [86–89]. COCs are generally considered to be effective in alleviating symptoms of primary dysmenorrhea [86–88,90]. However, this condition may continue to be problematic in a substantial proportion of women receiving cyclical COCs, potentially because these pills promote withdrawal bleeding and therefore may not completely assuage uterine cramping. In a meta-analysis of six randomized, controlled trials, COCs taken in an extended-cycle regimen were more effective in mitigating signs and symptoms of primary dysmenorrhea than those taken in a 21-day cycle [91].
POPs: benefits & limitations
Estrogen-free POPs are considered contraceptives of choice in women who prefer to use an oral hormonal contraceptive method, but who may not be good candidates for COCs. The progestin-only class of oral contraceptives has received renewed attention due to the introduction of a POP containing desogestrel in Europe in the past decade [92,93]. Women interested in using this method will require detailed information on the advantages and disadvantages of POPs as a class and on the different commercially available formulations.
Contraceptive efficacy
The most commonly used POPs contain low doses of norethindrone, ethynodiol diacetate, levonorgestrel or desogestrel. Traditional POPs containing norethindrone, ethynodiol diacetate and levonorgestrel do not consistently inhibit ovulation, relying primarily on progestogenic effects on the cervical mucus to prevent pregnancy [1,2]. The effectiveness of POPs overall has not been well established, but reported Pearl Indices range from 1 to 13% [3,8]. Traditional POPs appear to be less effective than COCs in suppressing ovulation; ovulation appears to be inhibited in 50–57% of women taking these POPs [2,94]. The contraceptive efficacy of traditional POPs may therefore be more dependent on secondary mechanisms, such as increased cervical mucus viscosity, which is more sensitive to changes in serum progesterone levels [2]. The POP containing 75 μg of desogestrel, a highly selective progestogen with low androgenic activity, is a more reliable estrogen-free option [95]. Desogestrel 75 μg has been shown to inhibit ovulation in up to 99% of cycles [93,96], an ovulation rate similar to that reported with COCs [2].
The desogestrel 75-μg pill is associated with greater contraceptive efficacy and similar overall acceptability compared with the levonorgestrel 30-μg pill [9].
Timing of pill-taking
Because ovulation is suppressed in only approximately half of women using traditional POPs, it is important to consider the clinical implications of secondary mechanisms. Changes in cervical mucus are time-dependent; conditions for sperm penetration may return to pretreatment levels within 24–27 h of taking traditional POPs, requiring that these pills be taken consistently at the same time every day within a 3-h window to achieve maximum contraceptive efficacy [2,94,97]. Women who are more than 3 h late in taking POPs are instructed to take the missed pill when remembered and use additional contraception for 48 h to improve protection against pregnancy.
Evidence from a 2005 pharmacodynamic study suggests that the contraceptive efficacy of desogestrel is not jeopardized in women who delay taking tablets for up to 12 h [93]. Among 103 women who took the POP containing desogestrel for 56 days, with three tablets scheduled to be taken 12 h late, ovulation was observed in one woman (ovulation incidence: 1.0%; 95% CI: 0.02–5.29). These data suggest that women may not need to take this POP at exactly the same time each day to ensure effectiveness. However, if the desogestrel POP is taken more than 12 h late, the missed pill should be taken as soon as it is remembered and a backup contraceptive method used for 7 days.
The requirement for taking POPs at the same time each day, particularly with traditional POPs for which the ‘missed pill window’ is 3 h, and the demands of managing late or missed pills are important compliance issues for this contraceptive method. As strict adherence to scheduled pill-taking may be more challenging for young women, POP use is more often recommended for older women. Women who initiate POP use require counseling and education about the need to take pills at the same time each day and the importance of selecting the optimum time of day for pill-taking based on the likelihood of remembering and their individual pattern of sexual behavior.
Safety & tolerability
Although data are less robust than for COCs, progestin-only contraceptives, including POPs, do not appear to increase cardiovascular risk [94,98,99]. In a 1998 WHO case–control study of cardiovascular disease and progestin-only contraceptives, no significant increase was found in the risk of VTE (RR: 1.8; 95% CI: 0.8–4.2), acute MI (RR: 1.0; 95% CI: 0.2–6.0) or stroke (RR: 1.1; 95% CI: 0.6–1.9) among women using continuous POPs compared with nonusers [99].
According to WHO medical guidelines, POPs are preferred over COCs in women with multiple cardiovascular risk factors (i.e., older age, hypertension, smoking and/or diabetes), hypertension, thromboses and thrombogenic mutations, hyperlipidemias, migraine headaches, and complications of diabetes [76]. Findings of several studies reviewed by McCann et al. indicated that POPs had no effects on total cholesterol, low-density lipoprotein (LDL) cholesterol or very LDL cholesterol [94]. In some studies, a small decrease in high-density liproprotein (HDL) cholesterol was observed. In a randomized, crossover study comparing the effects of a POP (lynestrenol 0.5 mg) and a COC (lynestrenol 2.5 mg plus ethinyl estradiol 50 μg) on carbohydrate and lipid metabolism in women with Type I diabetes, investigators found that the POP did not change insulin requirements or blood glucose levels, whereas the COC significantly increased insulin requirements (p < 0.01) without affecting blood glucose levels [64]. In addition, POP users showed significant reductions in serum levels of cholesterol (p < 0.001) and triglycerides (p < 0.001), with no effect on levels of HDL cholesterol; COC users had a significant increase in serum triglyceride levels (p < 0.01), but no impact on serum HDL cholesterol levels. Results of a study of desogestrel suggest that this progestin may have less marked effects on measures of carbohydrate metabolism compared with COCs [100].
In contrast to COCs, which are not recommended in the immediate postpartum period due to concerns about the risk of thrombosis, POPs are the treatment of choice in nonbreastfeeding women immediately after childbirth
Progestin-only pills lack the estrogen-related adverse effects associated with COCs and can therefore reduce estrogen-related symptoms in women switching from COCs. In an observational study (ERS study) evaluating the effects of the POP containing desogestrel 75 μg administered daily for three 28-day cycles in women reporting common estrogen-related symptoms with prior COC use (n = 403), approximately three-quarters or more of women who received desogestrel reported resolution or significant improvement in estrogen-related symptoms
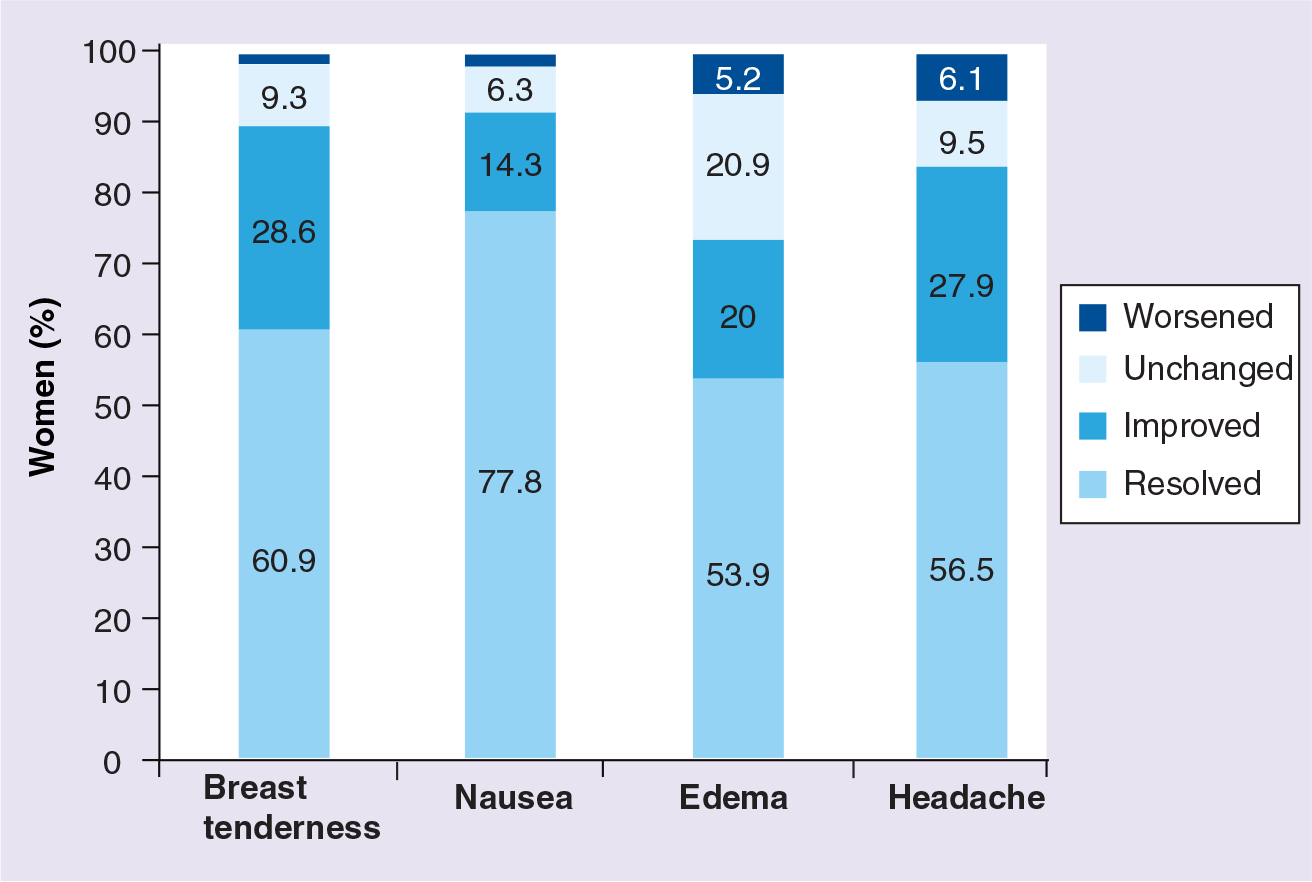
Baseline to study end changes in estrogen-related adverse effects among 403 women who switched from a combined oral contraceptive to 3 months of desogestrel 75 μg/day (ERS study).
Limited information regarding the effects of POPs on dysmenorrhea is available. However, positive findings from a prospective, noncomparative, observational study have been reported for a 3-month continuous regimen of the POP containing desogestrel [102]. Continuous administration of desogestrel 75 μg/day over three cycles reduced the incidence of moderate-to-severe primary dysmenorrhea from 84% at baseline to 7% at study end

Severity of dysmenorrhea in 406 women before and after 3 months of desogestrel 75 μg/day (Dysmenorrhea study).
Bleeding patterns
An important advantage of COCs is their protection against menstrual cycle irregularities, menorrhagia and iron-deficiency anemia [4]. Although POPs avoid the estrogen-related risks and adverse effects reported with COCs, POPs often do not provide good control of menstrual cycle regularity. The most frequent complaints reported by women using POPs are related to variable bleeding patterns or bleeding problems, such as amenorrhea, infrequent bleeding or prolonged bleeding. Compared with COCs, POPs are associated with a greater overall number of bleeding and spotting days [8]. In a randomized controlled study [103], irregular cycles were reported more frequently with a POP containing ethynodiol diacetate 0.25 mg than with a COC containing ethynodiol diacetate 1.0 mg plus mestranol 0.1 mg (OR: 135.96; 95% CI: 7.63–2421.02), as was intercycle bleeding (OR: 6.20; 95% CI: 2.11–18.22) [104].
Bleeding variability is the main reason cited by women who discontinue use of POPs [9,81,102,105]. In a WHO randomized trial comparing two POP formulations (norethisterone 350 μg vs levonorgestrel 30 μg) and two COC formulations (norethisterone 1 mg plus mestranol 50 μg vs levonorgestrel 150 μg plus ethinyl estradiol 30 μg), discontinuation because of bleeding was more common with both POP formulations than with the levonorgestrel/mestranol-containing COC [106]. In a 12-month comparative trial, 45% of women taking desogestrel 75 μg and 39% of women taking levonorgestrel 30 μg discontinued pill use [9]. In half of all cases, irregular vaginal bleeding was the reason for discontinuation [9]. In the first 3 months of the study, amenorrhea and frequent or prolonged bleeding were more common among desogestrel users than levonorgestrel users [9]. During the final 3 months of the study, approximately half of the women receiving desogestrel were amenorrheic or had infrequent bleeding or spotting episodes compared with 10% of women still receiving levonorgestrel [9]. Based on these findings, it was concluded that desogestrel, which provides the more pronounced ovarian inhibition, was associated with wider bleeding variability, but less bleeding overall than levonorgestrel. In the observational studies of desogestrel, women taking the POP containing desogestrel experienced up to a 35% increase from baseline in irregular/noncyclical bleeding pattern and up to a 43% decrease in normal/cyclical bleeding pattern [102]. However, only 5–7% of women discontinued POP use because of variable bleeding patterns or bleeding problems, and up to 87% indicated that they would continue taking this contraceptive after completing the study. These data suggest that the altered bleeding pattern was not necessarily considered a disadvantage of the contraceptive method.
To improve women's adherence to and acceptance of POPs, it is important that healthcare providers offer adequate counsel regarding the possible changes that may occur in bleeding patterns before initiating this contraceptive method [105]. Several studies have shown that contraceptive counseling significantly improves user satisfaction and adherence with selected contraceptive methods [107–110].
Conclusion
Combined estrogen/progestin contraceptives are currently the most commonly used form of hormonal contraception worldwide. However, COCs may be unsuitable for use in some women due to estrogen-related health risks or adverse effects. Estrogen-free POPs that effectively inhibit ovulation, such as the POP containing desogestrel, is a valuable contraceptive alternative to COCs, particularly for women who have contraindications to or cannot tolerate the adverse effects of COCs, but prefer a daily oral contraceptive method. The POP containing desogestrel has contraceptive efficacy comparable to that of COCs, is without estrogen-related risks and adverse effects, and has a missed pill window of up to 12 h, which allows greater flexibility in dosing. POPs that do not effectively inhibit ovulation have several disadvantages that may limit their usefulness, including potentially lower contraceptive efficacy and a missed pill window of 3 h, which may be challenging, especially in young women. Patient counseling regarding the more variable bleeding pattern associated with POP use is recommended to improve adherence and acceptance of this method.
Future perspective
Although advances in contraceptive technology and family-planning programs have been achieved over the past several decades, the contraceptive needs of a high percentage of women remain unmet and the rate of unintended pregnancy continues to increase in low- and high-income regions around the world. Strategies aimed at reversing these trends in the future include increasing the use of contraceptives overall and of specific contraceptive methods, such as the ovulation-inhibiting POP containing desogestrel, that have demonstrated high efficacy and acceptance rates, and developing new contraceptive methods to expand contraceptive choices for women and men with an unmet need for safe and effective contraceptives. These approaches will focus on providing better access to existing contraceptive methods, improving understanding of the benefits and risks of these methods and identifying means of maximizing their correct and consistent use, and pursuing new areas of basic research, including genomics and proteomics, that may result in the discovery of new contraceptives that are convenient, affordable, reversible, safe and effective. With more than 100 trials of contraceptives currently registered on the clinical trials database, the future promises many new advances in this field of research and therapeutics.
Executive summary
Oral hormonal contraceptives are currently the most widely established method of reversible contraception worldwide. For women who favor this form of contraception, various formulations of combined oral contraceptives (COCs) and progestin-only pills (POPs) are currently available.
Although COCs are highly effective in preventing pregnancy and offer health benefits beyond contraception, with an excellent long-term safety profile, their use may be unsuitable for some women due to undesirable health risks or intolerable adverse effects. For these women, POPs may be a valuable contraceptive option.
When considering use of a COC, women and healthcare professionals will review the potential benefits of this method as well as the specific safety and tolerability issues related to its use.
As stated in product prescribing guidelines, COC use may pose an undesirable health risk in women who have current or a past history of thromboembolic, cerebrovascular or coronary artery disease, diabetes with vascular disease, or prothrombotic conditions, as well as those who have other risk factors (e.g., hypertension, dyslipidemia, diabetes, smoking and obesity).
COCs are contraindicated in women who currently have breast cancer or other hormone-sensitive cancer, or who have a history of these conditions.
COCs are not recommended for use in nonbreastfeeding women in the immediate postpartum period or in breastfeeding mothers for the first 6 months or until they have weaned their children.
Women taking COCs may experience estrogen-related adverse effects, including headache, which may adversely affect quality of life and/or result in discontinuation of this method.
Although COCs are generally considered effective in improving symptoms of primary dysmenorrhea, cyclical COCs promote withdrawal bleeding and therefore may not completely alleviate symptoms of dysmenorrhea in some women.
Estrogen-free POPs are frequently the contraceptive method of choice in women who prefer to use an oral contraceptive, but who are not suitable candidates for COCs.
Traditional POPs that do not consistently suppress ovulation may be less effective than COCs and must be taken consistently at the same time each day, within a 3-h window, to obtain maximum contraceptive efficacy. However, the POP containing 75 μg desogestrel more effectively suppresses ovulation and has demonstrated greater contraceptive efficacy than 30 μg levonorgestrel; in addition, its efficacy does not appear to be jeopardized by delays in pill-taking of up to 12 h.
POPs do not appear to increase cardiovascular risk and are a preferred contraceptive method in nonbreastfeeding women immediately after childbirth and in breastfeeding women beyond 6 weeks postpartum. POPs do not cause the estrogen-related adverse effects associated with COCs and continuous use of the POP containing desogestrel has been shown to substantially reduce the incidence of moderate-to-severe primary dysmenorrhea.
Variable bleeding patterns or bleeding problems are a common complaint in women using POPs and are the most common reason for discontinuation of this method. Counseling about the bleeding pattern changes associated with POPs may improve women's satisfaction and adherence.
In women who prefer a daily oral contraceptive method but are not suitable candidates for COC use due to estrogen-related health risks or adverse effects, the estrogen-free POP is a valuable contraceptive alternative.
The POP containing desogestrel is at least as effective as COCs, without estrogen-related risks and adverse effects, and has been demonstrated to substantially reduce the signs and symptoms of primary dysmenorrhea. Counseling women on variable bleeding patterns associated with POP use may improve adherence and acceptance.
Footnotes
Dr de Melo is a consultant for Bayer Schering, Wyeth, Organon, Sanofi-Aventis, Jansen Cilag, and Johnson & Johnson. He has served as a clinical investigator on studies of lasofoxifene, ethinyl estradiol plus gestodene, ethinyl estradiol plus desogestrel, tibolone 17b, transdermal estradiol, and 17b-estradiol plus nortestosterone. In addition, Dr de Melo has been a keynote speaker at meetings sponsored by Bayer Schering, Organon, Wyeth, Sanofi-Aventis, Jansen Cilag, Roche, and Merck Sharp & Dohme. The author has no other relevant af filiations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
Editorial support was provided by Donna McGuire of Evidence Scientific Solutions and was funded by Schering Corp., a Division of Merck & Co.
