Abstract
Background:
Heavy menstrual bleeding (HMB) associated with iron deficiency anemia (IDA) negatively affects quality of life (QoL). Management of IDA usually begins with oral iron supplementation or, if ineffective/poorly tolerated, then intravenous iron (IVI) is given; however, no guidance exists on transitioning from oral to IVI in patients with HMB. While various IVI products exist, safety profiles and distinct properties affecting treatment logistics make product choice important.
Objectives:
Assess the IVI treatment journey for patients with HMB and IDA.
Design:
A survey was designed to assess multiple aspects of IVI treatment to evaluate patient perspectives.
Methods:
Patients (⩾18 years) from the United States with IDA currently prescribed IVI completed a survey conducted by The Harris Poll in 2023. Questions covered symptoms, time to diagnosis/treatment, IVI appointment logistics, IVI infusion experience, impact on daily activities, and patient preferences.
Results:
Of 323 respondents, 71 (22.0%) were prescribed IVI for HMB and received ⩾2 IVI infusions monthly. The mean age for these patients was 33.5 years; they experienced a mean of 2.9 years from symptom onset until IDA diagnosis, and 1.4 years between diagnosis and IVI treatment. Most patients agreed that navigating IVI treatment logistics interfered with productivity and social commitments, and felt they must schedule their life around treatment. Patients who were also diagnosed with hypophosphatemia following IVI (12/71; 16.9%) reported a mean of 8.2 additional hospital visits. Furthermore, 36.6% of patients missed an IVI dose; of these, 80.8% preferred single-dose IVI.
Conclusion:
Patients with IDA and HMB experienced substantial delays from symptom onset to subsequent IVI treatment, demonstrating a gap in management. Therefore, oral iron may not be an appropriate first-line treatment for some of these patients. Multiple-dose IVI and associated appointment logistics can negatively impact patients’ perspectives on treatment. Single-dose IVI preferences should be considered to improve patients’ adherence and QoL.
Plain language summary
Heavy menstrual bleeding (HMB) is a condition where women experience periods that are heavier and/or last longer than expected. Due to the increased bleeding, some women with HMB can develop iron deficiency anemia (IDA) if HMB is left untreated. IDA is a condition where your body does not have enough iron to, among other things, produce enough red blood cells to carry oxygen around your body. For individuals with both HMB and IDA, extra iron can be given in the form of tablets that are taken by mouth, or by intravenous infusion (IVI) directly into the bloodstream. However, healthcare providers have little guidance on when patients should be given IVI treatment. To better understand how patients with HMB and IDA are treated, a survey was carried out that asked patients about their IVI treatment experiences. A total of 323 patients with IDA completed the survey, of which 71 patients also had HMB and were having two or more IVI infusions per month. Together, these patients typically waited almost 3 years to get an IDA diagnosis after first experiencing symptoms and almost 1.5 years for treatment to be prescribed after being diagnosed with IDA. Most of these patients agreed that scheduling and attending IVI treatment appointments got in the way of their productivity and ability to attend important events. A third of patients said they had missed an IVI treatment appointment, and most also agreed they would prefer their IVI treatment to be given in a single dose rather than several doses. Therefore, healthcare providers should consider giving patients single-dose IVI treatment to make sure patients attend all their IVI appointments so that they can receive their full course of treatment, which should improve their quality of life.
Keywords
Introduction
Heavy menstrual bleeding (HMB), also known as abnormal uterine bleeding, 1 is a common gynecologic complaint with multiple and diverse etiologies.2 –4 HMB affects approximately a third of reproductive-aged women and can be defined as menstrual bleeding sufficient enough to adversely impact physical, emotional, or social quality of life (QoL). 5 Objectively, HMB is defined as a total blood loss per menstrual cycle that regularly exceeds 80 mL.6,7
Many women with HMB assume their bleeding patterns are normal and often tolerate associated symptoms for a long time, which often leads to an underdiagnosis of HMB, as well as substantial delays in diagnosis and treatment.8,9 This poses a significant challenge, especially since societal taboos and stigma around openly discussing menstruation and HMB, can hinder open dialogue about this common condition. 8 Additionally, some patients often feel that this acceptance of abnormal bleeding patterns prevents healthcare providers from taking their symptoms seriously during discussions. 8 Patients with menstrual issues tend to seek treatment only after their lives have become severely impacted by it, and afterward, patients often express regret for delaying discussions and not seeking help sooner. 10
HMB is initially diagnosed by obtaining and discussing a thorough medical history of a patient’s symptoms with a healthcare provider. Some of the necessary history may include onset of menarche, monthly duration, how many pads/tampons are used, and whether it affects the patient’s ability to participate in daily activities. Conducting a physical examination, checking a complete blood panel, evaluating for a possible underlying bleeding disorder, and/or performing a gynecologic sonogram/ultrasound may be carried out to aid in diagnosis. 5
The chronic blood loss experienced by menstruating girls and women with HMB is a major contributing factor to iron deficiency (ID) and iron deficiency anemia (IDA), which can affect their QoL. IDA is characterized by a reduction in total body iron which, among other things, affects hematological processes, such as hemoglobin synthesis and erythropoiesis. 3 While some patients with IDA will be asymptomatic, most have symptoms such as lightheadedness, fatigue, and dyspnea, 11 all of which negatively impact QoL and may be overlooked by physicians. 12 ID without anemia is also common in HMB and is associated with nonspecific symptoms, such as weakness, fatigue, restless leg syndrome, and decreased concentration, which often overlap with symptoms of IDA and may not be initially recognized by patients or healthcare providers as being a consequence of ID.13,14
Treatment for IDA in patients with HMB involves iron supplementation, either with oral iron formulations or with intravenous iron (IVI) when oral iron is ineffective or not tolerated; however, guidance on when to transition from oral iron to IVI has not been clear or adequately addressed in various related guidelines.5,6,15,16 Oral iron therapy is usually the first-line treatment but is often associated with gastrointestinal (GI) side effects, such as constipation and nausea. While appropriate in certain populations, oral iron may correct anemia slowly in patients with HMB and may fail to fully replenish iron stores. Alternatively, IVI can correct the anemia, restore iron stores more rapidly, and is also well tolerated.17 –19 While most IVI products require multiple infusions to complete a treatment course, higher doses of IVI (i.e., given as one or two infusions) have resulted in more rapid and pronounced hemoglobin responses compared to lower doses (i.e., infusions with <500 mg per infusion).20 –22 In a randomized clinical trial of two high-dose IVIs, patient-reported fatigue scores improved in both IVI groups, with a significantly faster and greater improvement with ferric derisomaltose versus ferric carboxymaltose (FCM), despite similar increases in hemoglobin; these results demonstrate that the magnitude of improvement in fatigue scores can differ between IVI products due to side effects. 23
While IVI products generally have similar safety profiles, some also have safety considerations such as hypophosphatemia, hypersensitivity, and possible cardiovascular risks. 24 With certain IV iron products, hypophosphatemia is a risk that can occur as early as after the first infusion. 25 Symptoms of hypophosphatemia can overlap with symptoms of IDA (e.g., asthenia, fatigue, myalgia), potentially leading to missed or delayed diagnoses.24,26 If worsened (e.g., by severe hypophosphatemia), it can lead to respiratory failure, osteomalacia, and bone fractures.25,27,28 While this safety consideration can potentially impact all patients, the highest incidence has been seen in populations requiring multiple treatment courses.25,27,29 –31 In most cases, the type of IVI product available to patients often depends on the products allowed by the payor, cost considerations, physician choice/comfort, and availability (e.g., on formulary). 18
The patient survey presented below evaluated the IVI treatment journey for patients with HMB and IDA with specific focus on multiple-dose infusions to better understand patients’ experiences.
Materials and methods
Between 15 February and 16 March 2023, The Harris Poll (Chicago, IL, USA) conducted an online patient-based survey in the United States on behalf of Pharmacosmos Therapeutics Inc. Eligible patients for inclusion were ⩾18 years of age with a confirmed IDA diagnosis from a healthcare provider who were currently prescribed IVI therapy; patients who did not meet these criteria were excluded. The survey was deployed to a panel of patients with IDA through two medical vendors used by The Harris Poll, which recruits patients through physicians, in-person sign-ups at disease awareness events, and referrals. These patient panels contain basic demographic information and medical diagnosis information so that certain patient populations can be targeted for such surveys. Patients identified in the panels as having IDA were invited to take part in the survey via email and, if they chose to participate, underwent opt-in and screening processes to ensure that they were not only patients with IDA but were also currently being prescribed IVI. All patients provided informed consent for study inclusion before completing the survey.
The survey for this observational cohort study consisted of 46 questions (Supplemental file 1) and covered topics such as patient demographics, appointment logistics, IVI infusion experience, impact of infusion on QoL (daily activities), reason(s) for missing any IVI doses, and ways to improve treatment adherence. The survey was conducted by The Harris Poll, which follows the American Association for Public Opinion Research’s Code of Professional Ethics and Practices. The online survey posed no risk of harm to patients, was anonymized, noninterventional, not intended to provide clinical data to support treatment decisions, and not conducted as a clinical trial for any endpoints; therefore, a separate IRB review was not deemed necessary. Raw data were not weighted and are, therefore, only representative of the individuals who completed the survey. For the purposes of this study, IVI therapies were grouped by the frequency of infusions in the treatment course rather than by brand. Descriptive statistics are reported only for the patients with IDA and HMB who were receiving ⩾2 IVI infusions per month.
Results
Patients with IDA and HMB
Of the 323 patients who completed the survey, 126 patients reported that they were prescribed IVI for HMB and 71 (56.3%) of these patients were prescribed ⩾2 IVI infusions per month. For these 71 patients, the mean (±standard deviation (SD)) age was 33.5 (±9.1) years; patients were predominantly White (54.9%), married, in a civil union, or living with a partner (52.1%), living with children (52.1%), employed full time (56.3%), and from a suburban environment (42.3% (38.0% from an urban or city area; 19.7% from a small town or rural area); Table 1). In addition to IDA, other comorbidities included rheumatoid arthritis (7.0%) and diabetes (type 1 or 2 (4.2%); Table 1).
Demographics of the 71 patients with IDA and HMB receiving ⩾2 IVI infusions per month.
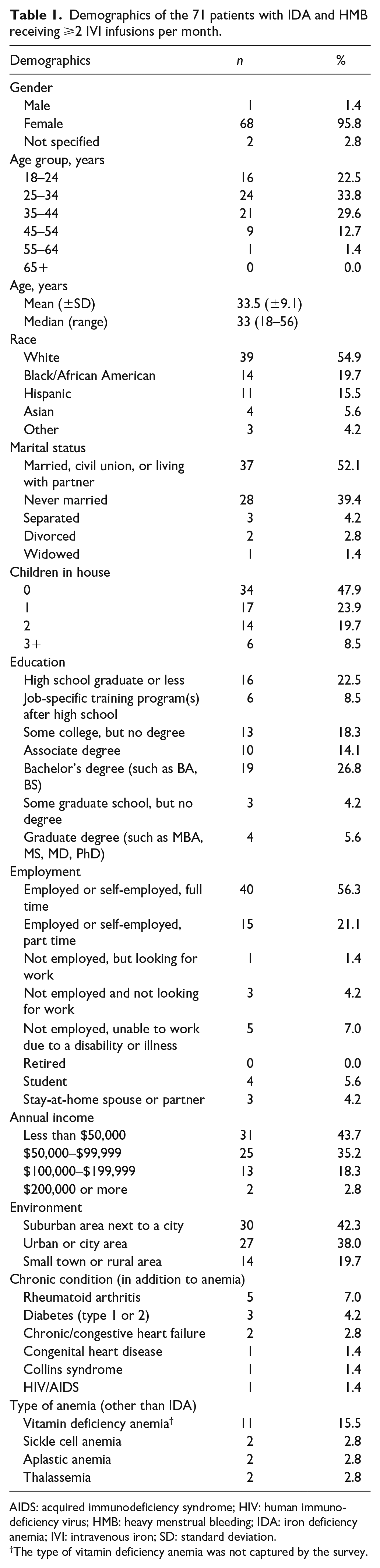
AIDS: acquired immunodeficiency syndrome; HIV: human immunodeficiency virus; HMB: heavy menstrual bleeding; IDA: iron deficiency anemia; IVI: intravenous iron; SD: standard deviation.
The type of vitamin deficiency anemia was not captured by the survey.
Most of the 71 patients (35.6%) with IDA and HMB had been experiencing symptoms associated with IDA for 1–2 years prior to diagnosis, and the mean (±SD) length of time between onset of symptoms and diagnosis was 2.9 (±3.2) years (Figure 1(a)). The mean (±SD) time since IDA diagnosis was 4.0 (±4.8) years (median (range) 2.4 (0.2–25.1) years), and the time since the first IVI was prescribed was 2.6 (±3.6) years (median (range) 1.9 (0.1–25.1) years). Together, results show that the mean length of time between IDA diagnosis and IVI being prescribed was 1.4 years and that, in total, most patients waited an average of 4.4 years from the onset of symptoms to receiving IVI therapy. The most common symptoms experienced by patients before IVI therapy were fatigue (80.3%), weakness (66.2%), and headache (63.4%), of which most improved after treatment (Figure 1(b)). The number of patients receiving treatment for HMB is unknown.

Analysis of timelines, symptoms, and appointment logistics for the 71 patients with IDA and HMB. (a) Length of time patients experienced symptoms prior to diagnosis of IDA, (b) symptoms experienced before IVI therapy and symptoms that improved after receiving IVI therapy, (c) monthly time spent scheduling IVI appointments, traveling to appointments, waiting for infusions after arriving at their appointment, and receiving IVI infusions.
Patients were mainly receiving 2 (66.2%) or ⩾3 (28.2%) IVI infusions per month, and the majority received their therapy at an infusion center (64.8%; Table 2). The mean (±SD) reported time spent on IVI appointment logistics included 38 (±83) min scheduling appointments, 67 (±56) min traveling to appointments, 47 (±53) min waiting for the infusion after arriving at the appointment, and 90 (±60) min receiving their infusions (Figure 1(c)). The mean (±SD) number of IVI infusions ever received by the patients with IDA and HMB was 9.6 (±8.3) infusions (Table 2). Regarding QoL measures for activities of daily living after IVI therapy, ⩾50% of patients reported an improvement in “doing things around the house” (59.2%) and “caring for myself” (57.7%; Table 3).
Details of iron therapy in the 71 patients with IDA and HMB.
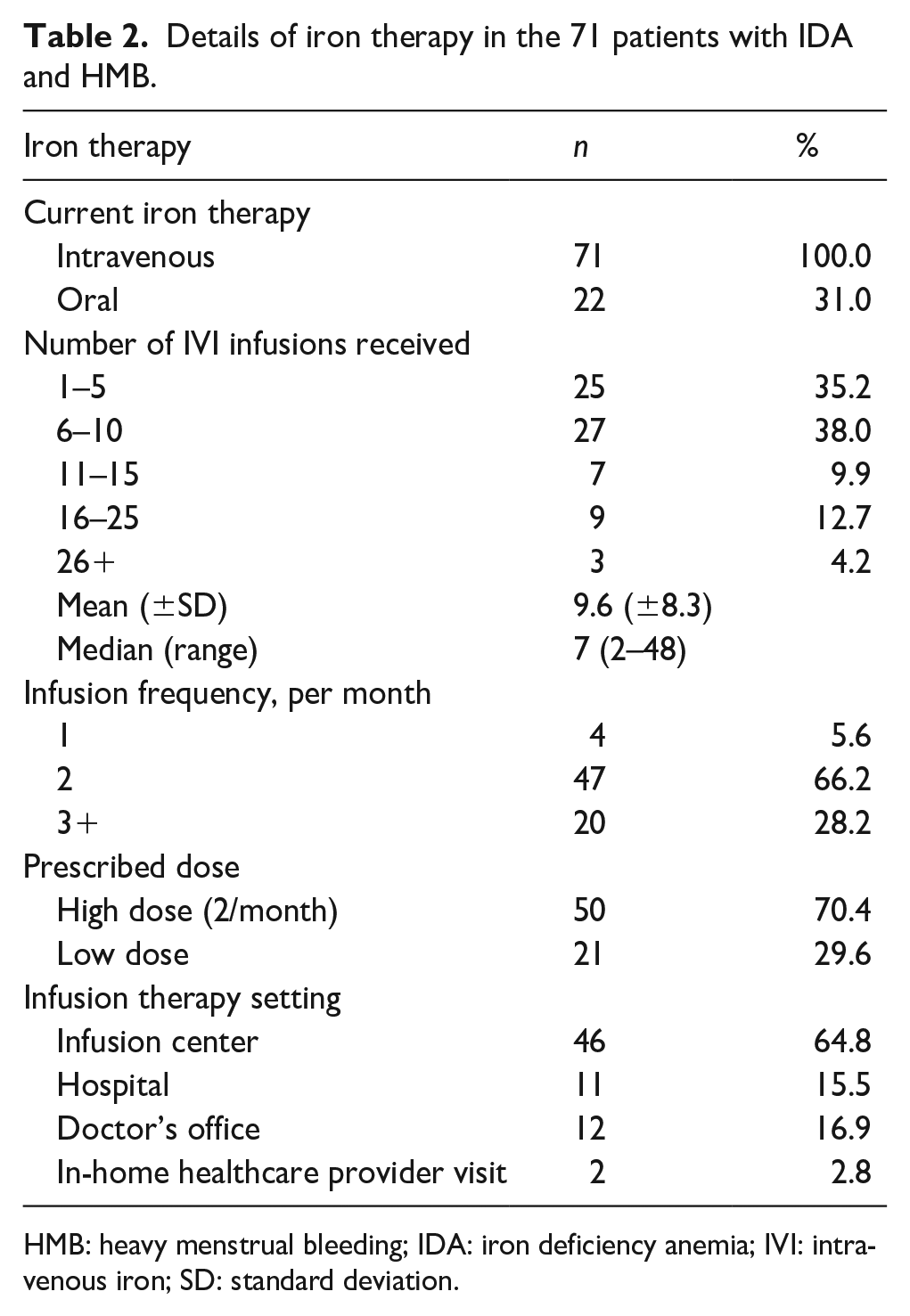
HMB: heavy menstrual bleeding; IDA: iron deficiency anemia; IVI: intravenous iron; SD: standard deviation.
Activities of daily living for the 71 patients with IDA and HMB that have improved since receiving IVI.

HMB: heavy menstrual bleeding; IDA: iron deficiency anemia; IVI: intravenous iron.
Over one-third of patients were unsatisfied with the frequency of infusions (36.6%; Figure 2(a)). Over one-half of patients also agreed that the logistics of their IVI therapy gets in the way of their productivity (53.5%) and caused them to miss important events (50.7%; Figure 2(b)). In addition, most felt that they had to schedule their life around IVI infusions (67.6%; Figure 2(b)). Consequently, 97.2% of patients indicated they would prefer to receive the IVI option that allows the most convenient therapeutic experience for IDA (Figure 2(b)), such as fewer infusions/needle sticks and fewer appointments. Most of the 71 patients with IDA and HMB also indicated that they would prefer single-dose IVI to multiple-dose IVI (83.1%).

Response rates of the 71 patients with IDA and HMB receiving ⩾2 infusions per month regarding their experiences of IVI therapy and QoL aspects. (a) Experiences of IVI therapy and (b) QoL aspects.
Patients with IDA and HMB who missed a dose of IVI
Of the 71 patients with IDA and HMB prescribed ⩾2 IVI infusions per month, over a third (

Response rates of the 26 patients with IDA and HMB who reported missing a dose of their IVI therapy. (a) Reasons provided for missing a dose of their IVI therapy, (b) considerations regarding optimal IVI therapy, and (c) agreement levels regarding optimal IVI therapy.
Patients with IDA, HMB, and hypophosphatemia
Of the 71 patients with IDA and HMB receiving ⩾2 IVI infusions per month, 12 (16.9%) reported that they were also diagnosed with hypophosphatemia following IVI infusions; 4 (5.6%) patients were uncertain. The most common hypophosphatemia symptoms reported by these 12 patients were muscle weakness (
Discussion
Results of this survey show that patients with both HMB and IDA experienced substantial delays in receiving appropriate or adequate therapy, representing a potential gap in care. These patients had to wait an average of almost 3 years from the onset of symptoms before being diagnosed with IDA and subsequently waited almost an additional 1.5 years, on average, prior to being prescribed IVI therapy. Therefore, the patients waited nearly 4.5 years from the onset of IDA symptoms to eventually receiving IVI therapy.
Approximately 30% of women worldwide are estimated to experience HMB at some point during their reproductive years, 32 and these women report that it can have a wide-ranging and debilitating impact on their lives. 8 Women also describe how symptoms of HMB are often normalized such that they often suffer silently for several years before seeking medical help. 8 This problem reflects persisting societal taboos regarding the discussion of menstruation, thereby leading to a low general awareness among healthcare providers. 8 HMB is the most common cause of IDA for girls and women of reproductive age. 33 In addition, HMB can be indicative of other health problems, such as an undiagnosed bleeding disorder or uterine mass. 33 For patients who become pregnant, IDA is associated with a variety of adverse obstetric outcomes and could affect fetal development.34,35 Therefore, it is imperative that healthcare providers openly discuss menstruation more regularly with patients. Such discussions may help raise awareness of HMB and its association with IDA. Patients with menstrual issues tend to seek treatment only after their lives have become negatively impacted by it, and afterward, patients often express regret for delaying discussions and not seeking help sooner. 10 In fact, these patients also state that the “openness” of their physician was the most important factor that encouraged them to disclose their menstrual issues.
Discussing menstrual issues and symptoms with a healthcare provider is typically the first step toward diagnosing HMB. Individuals who regularly experience >80 mL loss of blood per menstrual cycle are defined as having HMB6,7; however, there is no clear consensus on how patients are supposed to quantify this. Furthermore, patient friendly pictorial blood loss assessment charts (PBACs) are widely used for assessing menstrual blood loss in clinical trials and are primarily used in clinical practice 36 ; however, PBACs are less accurate than other methods. The alkaline hematinic method, which was once gold standard, is more accurate than PBACs, but is not practical for daily use.37,38 There are different tools that can be used to aid HMB diagnosis, but if blood tests are required (depending on prior assessments), some guidelines can be inconsistent regarding appropriate tests to assess iron levels. 6 Although anemia can be confirmed by hemoglobin levels below the lower limit of normal (<12 g/dL in women), it is important that ferritin levels are also assessed as low ferritin is the best indicator of ID in the absence of inflammation. 14 While there is no consensus on serum thresholds for the diagnosis of ID/IDA for this population, some reviews and guidelines, such as the American College of Obstetricians and Gynecologists, define ID as <30 µg/L.6,39 –42 A higher ferritin threshold such as <50 µg/L for ID has also been recommended by some experts. 43 However, this can be unreliable when there is coexisting inflammation since ferritin is an acute phase protein and levels can rise in the presence of infection, malignancy, and chronic inflammation. 44 In these scenarios, a transferrin saturation <20% can also increase the sensitivity of ID. 44
A review of international guidelines for managing HMB recently highlighted inconsistencies in the diagnosis and management of IDA. 6 Furthermore, few guidelines adequately address the need to assess and treat ID in patients with HMB; for those that do, there is a notable lack of guidance on whether iron supplementation should be given orally or intravenously. 6 Given these gaps and inconsistencies, it is likely that ID and IDA are underdiagnosed and undertreated in patients with HMB, 6 which is concerning given that HMB is also often underreported. A recent observational study in 6210 patients with HMB found that the treatment journey for these patients is associated with a large burden of suffering, delay in diagnosis, and a gap in access to therapy. 45 These results, including the current study, highlight that women with HMB-associated IDA can experience substantial delays in diagnosis and treatment. It also underscores the essential need for increased awareness of both conditions among healthcare providers, the importance of consistent guidelines for diagnosis and management, and the current gap in access to appropriate therapy for IDA due to HMB. Since guidelines for managing IDA in patients with HMB are currently inadequate, it raises the question of whether other hematology societies should introduce guidelines that can offer a consensus.
Once IDA has been diagnosed in patients with HMB, the first-line treatment is usually orally administered iron. 39 However, IVI can treat anemia and replenish iron stores more rapidly, without the GI intolerance often associated with oral iron. 19 In patients with HMB-associated IDA, IVI can achieve significantly higher increases in hemoglobin levels over a shorter period of time 46 and can provide a greater improvement in energy levels, physical function, and fatigue, than oral iron. 47 Despite the proven efficacy of IVI, some guidelines and payors coverage require a 4-week trial of oral iron without improvement or GI intolerance before approving IVI options, 6 further delaying access to effective treatment.
Most IVI products available are approved as two or more infusions per treatment course, with options often depending on payor approval, cost considerations, availability on formulary at an institution, and physician preference.18,48 Results from the current study highlight that receiving two or more IVI infusions per month can place a substantial burden on patients, not only regarding appointment logistics but also with respect to the impact on QoL and daily activities, all of which can contribute to patients missing appointments. However, further studies are warranted to fully comprehend the complexities around why patients miss appointments and the impact of these missed appointment when compared to patients who completed a full IVI treatment course; failure to receive scheduled doses of IVI can potentially result in protracted anemia and exacerbation of HMB-related blood loss. All this could potentially increase the severity of symptoms, affect the overall health of patients, and increase the overall cost of treatment. 49 A third of patients with IDA and HMB in the current study indicated they missed a dose of their IVI therapy, which may have been due to the burden associated with having multiple-dose IVI therapy. Using a single-infusion option, where clinically appropriate, may help to alleviate these burdens, improve adherence, and ultimately enhance treatment outcomes. 18
In addition to treatment logistics, product safety profiles should also have a greater factor in the selection of IVI. Side effects of IVI vary depending on the product, but most commonly include nausea or vomiting, headache, dizziness, flushing, hypertension, injection site reactions, muscle pain in the arms or legs, and infusion reactions.20,46
–48,50,51 Treatment-emergent hypophosphatemia is more recognized from the growing body of evidence; however, the association of hypophosphatemia with certain IVI products may be underrecognized in this patient population. Patients at highest risk for hypophosphatemia are those with recurrent blood loss, such as HMB, where repeat IVI infusions are required.27,31 Out of the patients receiving two or more IVI infusions per month in the current study, 17% also reported a diagnosis of hypophosphatemia, which resulted in an average of eight additional healthcare visits. This data further support the PHOSPHARE-IDA studies, which predominantly enrolled patients with HMB (pooled analysis, 68.6%; 166/242), and demonstrated that treatment with FCM resulted in a much higher incidence of hypophosphatemia than of ferric derisomaltose (pooled analysis, 74.4% vs 8.0%,
Most patients in the current study who were receiving multiple-dose IVI expressed a preference for single-dose IVI treatment and fewer infusions. Single-dose IVI has been demonstrated to have similar efficacy and safety as multiple-dose IVI20,21,54 and may be more convenient for patients and providers than multiple-dose infusions.55,56 Indeed, research has demonstrated that single-dose IVI infusions can reduce the number of infusion room visits without significantly increasing infusion reactions or compromising efficacy. 56 Therefore, it is recommended to consider patient preferences for single-dose IVI when deciding on IVI therapy.
Limitations
This observational study reports the results of an online patient survey and, as such, may be subject to self-report bias. Additionally, the survey was not validated or pilot-tested, and no power analysis for sample size calculation was performed. The reliability of the results depends on whether the patients understood the questions and answered truthfully. The causality of reported side effects, including symptoms of hypophosphatemia, cannot be definitively established in this analysis, as the results were not independently verified by a physician. The reported outcomes may therefore be influenced by underlying conditions or other confounding factors rather than a direct effect of the IVI infusions. Furthermore, the survey was completed by a small group of patients, only a small proportion of whom had both IDA and HMB and were receiving ⩾2 IVI infusions per month; therefore, results may not be generalized. The survey was also limited only to patients who were currently receiving or who had recently received IVI, so the results may not represent the perspective of all patients who have completed a full course of treatment and should be interpreted with caution.
Conclusion
Healthcare providers hold the key to improving the burden of symptoms experienced by patients with HMB-associated IDA, as well as addressing the underdiagnosis of HMB, by encouraging patients to discuss menstrual issues openly. As a treatment option, IVI rapidly treats IDA, replenishes iron stores without the GI side effects of oral iron, and provides faster hemoglobin increases and greater improvements in energy, physical function, and fatigue, especially in HMB-associated IDA.19,46,47 Current restrictions by payors requiring prolonged trials of oral iron can delay effective treatment and should be reconsidered. Clear, consistent guidelines from major medical societies can help standardize patient care, and optimize treatment decisions, such as management with IVI. The choice of IVI product is critical for patients; while multiple-infusion therapies place logistical and psychological burdens on patients, single-dose options align better with patient preferences and can improve adherence. Convenience plays an important role in adherence to IVI therapy and should be a key consideration when selecting treatment options for all patients with IDA, regardless of whether it is associated with HMB. By addressing these barriers and prioritizing patient-centered care, healthcare providers can significantly enhance the QoL and clinical outcomes for patients with HMB-associated IDA.
Supplemental Material
sj-docx-1-whe-10.1177_17455057251321221 – Supplemental material for Iron deficiency anemia in patients with heavy menstrual bleeding: The patients’ perspective from diagnosis to treatment
Supplemental material, sj-docx-1-whe-10.1177_17455057251321221 for Iron deficiency anemia in patients with heavy menstrual bleeding: The patients’ perspective from diagnosis to treatment by Imo J Akpan, Mohit Narang, Edio Zampaglione, Steve Marshall and Dawn Stefanik in Women’s Health
Supplemental Material
sj-docx-2-whe-10.1177_17455057251321221 – Supplemental material for Iron deficiency anemia in patients with heavy menstrual bleeding: The patients’ perspective from diagnosis to treatment
Supplemental material, sj-docx-2-whe-10.1177_17455057251321221 for Iron deficiency anemia in patients with heavy menstrual bleeding: The patients’ perspective from diagnosis to treatment by Imo J Akpan, Mohit Narang, Edio Zampaglione, Steve Marshall and Dawn Stefanik in Women’s Health
Footnotes
Acknowledgements
We would like to thank The Harris Poll for conducting this survey on behalf of Pharmacosmos Therapeutics Inc., and Ben McDermott of the Bioscript Group (Macclesfield, UK), who provided medical writing assistance in accordance with Good Publication Practice guidelines, which was funded by Pharmacosmos Therapeutics Inc.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
