Abstract
Background:
After menopause, reductions in ovarian hormones increase the risk of cardiovascular disease. Aerobic exercise training has been shown to reduce cardiovascular risk in older adults, but its effectiveness in postmenopausal females is less definitive.
Objectives:
The objectives of this study were to examine the: (1) effects of aerobic training, and (2) association between aerobic training intensity and cardiometabolic health outcomes in postmenopausal females.
Design:
Systematic review and meta-analysis of randomized controlled trials.
Data Sources and Methods:
Six electronic databases were searched from inception to July 21, 2023 for aerobic training interventions reporting cardiometabolic outcomes in postmenopausal females. Data were synthesized qualitatively and random-effects meta-analyses and subgroup analyses (light, moderate, and vigorous intensity) were performed. Grading of Recommendations, Assessment, Development and Evaluation was used to assess the certainty of evidence.
Results:
Fifty-nine studies (
Conclusion:
Aerobic training may improve cardiometabolic health outcomes in postmenopausal females. There may be differential effects of exercise intensity on BMI, blood triglycerides, and blood glucose; however, this warrants further investigation.
Registration:
PROSPERO—CRD42022313350
Introduction
Menopause is the permanent cessation of menstruation due to ovarian follicular depletion, with females considered postmenopausal after 12 consecutive months of amenorrhoea.1,2 Following menopause, there is a reduction in estrogen and its cardioprotective effects, 3 which can lead to worsening cardiovascular health, characterized by increased blood pressure, body fat, dyslipidemia, and reduced glucose tolerance. 4 Postmenopausal females are at a two-fold higher risk for cardiometabolic disease compared to premenopausal females 5 and the risk of stroke nearly doubles in the 10 years following menopause. 6
Although pharmacological treatments may be used to mitigate cardiometabolic decline, their effectiveness attenuates with duration of use; thus, alternative strategies to sustain long-term cardiometabolic health are needed.
7
Aerobic exercise training is an increasingly popular,
8
and effective9,10 strategy to promote cardiometabolic health. A systematic review of 93 trials with 5,223 males and females >18 years of age with normal blood pressure, prehypertension, and hypertension found that exercise, including aerobic exercise, decreases systolic blood pressure (SBP) and diastolic blood pressure (DBP).
11
In other reviews of individuals who are overweight or obese,
12
living with metabolic syndrome
13
and type 2 diabetes,
14
aerobic exercise training is effective for improving SBP,
14
DBP,
13
body composition,12
–14 high-density lipoprotein cholesterol (HDL-C),
13
serum triglycerides,13,14 blood glucose,
13
and cardiorespiratory fitness (CRF).
13
Importantly, the American College of Sports Medicine suggests that
To date, four systematic reviews have focused specifically on postmenopausal females and reported that aerobic or resistance training interventions were effective in improving SBP,19,20 DBP,19,20 CRF (
Thus, the primary objective of this systematic review was to examine the effects of aerobic exercise training on traditional cardiometabolic health measures in a broad population of postmenopausal females. Based on the importance of exercise intensity, the secondary objective was to examine the association between different aerobic exercise intensities and traditional cardiometabolic health measures.
Methods
This systematic review and meta-analysis was conducted following Cochrane guidelines for Systematic Reviews of Interventions, 30 and reported based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. 31 The protocol is registered in the PROSPERO International Prospective Register of Systematic Reviews (CRD42022313350).
Eligibility criteria
This review was restricted to peer-reviewed, published randomized controlled clinical trials in English. Non-randomized trials, conference abstracts, cross-sectional studies, study protocols, and ongoing trials were excluded.
In this review, we considered the study sample to represent postmenopausal females if they met at least one of the following three criteria: (1) explicitly confirmed participants to be postmenopausal (e.g., via self-report); (2) if postmenopausal status was not explicitly reported but age range was, a conservative lower bound of age ⩾55 years old, or (3) if age range was not reported, mean age ⩾63 years old. The latter two criteria were based on data from large population-based studies that report the average age of menopause to be 50–52 years old,32 –36 with onset of menopause varying by 1 or 2 years due to factors such as race, body composition, physical activity levels, diet, socioeconomic status, and smoking. 37 Furthermore, a mean age of 63 with an estimated standard deviation (SD) of 4 years yields a 95% confidence interval (95% CI) with a lower limit age of 55 years. Lastly, to increase the generalizability of our findings, we included studies of participants with hypertension, diabetes mellitus, obesity, and dyslipidemia, as these metabolic conditions are common postmenopause.23 –29 Participants with a history of overt cardiovascular events were excluded (e.g., stroke and myocardial infarction).
Included trials were aerobic exercise training interventions (i.e., activities that use large muscle groups that are maintained continuously and in a rhythmic nature) 38 that reported at least two of the following training parameters: frequency, intensity, type, and duration. There was no restriction to the frequency, intensity, type, and duration of aerobic interventions. To better isolate the effect of aerobic exercise training, co-interventions, and non-aerobic exercise interventions, such as resistance training, yoga, tai chi, virtual reality, pharmacological, or nutritional interventions were excluded. Control groups included non-exercise, non-diet, or non-pharmacological interventions, such as health education or stretching.
We included trials that examined the most prominent modifiable cardiometabolic health outcomes in females, (1) Resting SBP and DBP39,40; (2) resting heart rate (HR)41,42; (3) body composition and anthropometric measures43,44 (body mass index (BMI), 45 body fat, 46 waist-to-hip ratio (WHR) and WC 47 ); (4) blood lipids (low-density lipoprotein cholesterol (LDL-C), 48 HDL-C, 49 and blood triglycerides 50 ); (5) blood glucose51,52 and (6) CRF.53 –55
Studies not reporting at least one of these traditional cardiometabolic outcomes were excluded.
Information sources
The initial search of six electronic databases (OVID MEDLINE, EMBASE, CINAHL, CENTRAL, SPORTDiscus, and Web of Science) was conducted from inception to April 25, 2022 and updated on July 21, 2023. The search strategy was developed in consultation with a research librarian and included terms related to “cardiometabolic health,” “aerobic exercise,” “older adults,” and “menopause.” The final search strategies are displayed in Supplemental File 1.
Data extraction
Two sets of independent reviewers (EH and EW, EH and KSN) assessed study eligibility based on titles and abstracts followed by full-text review. All reviewers (EH, EW, KSN) piloted title and abstract screening by screening two sets of abstracts independently (
Data was extracted into Covidence systematic review manager (Covidence systematic review software; Veritas Health Innovation, Melbourne, Australia) by three sets of independent reviewers (EH and EW, EH and KSN, EH and HF) and included lead author name, year of publication, summary of participant eligibility criteria, population characteristics, control group characteristics, exercise intervention parameters (i.e., frequency, intensity, type, and duration), qualitative summary of effect on cardiometabolic health outcome, sample size and change in outcome (pre- to post-intervention) in intervention and control groups, respectively.
Statistical analysis
To assess the effects of aerobic exercise training on measures of cardiometabolic health (objective 1), random-effects meta-analyses and inverse variance-weighted estimation were used. Mean difference (MD) or standardized mean difference (SMD) in change scores from pre- to post-intervention with 95%CIs were used to determine the effect of aerobic exercise training on outcomes of interest. 30 Pooled SDs were calculated. Studies that contained two or more intervention groups were combined, outcome measures reported in median (interquartile range), mean (standard error) or mean (95%CI) were transformed to mean and SDs, and SDs for change scores were calculated per Cochrane guidelines. 30 Corresponding authors were contacted for studies with missing pre- and post-intervention means and SDs; otherwise, these studies were excluded. Forest plots were created to visually display the overall effects of aerobic exercise on outcomes of interest.
A priori subgroup analyses were conducted to compare the individual effects of different intensities (light, moderate, and vigorous) of aerobic exercise on cardiometabolic health (objective 2). Subgroups were categorized according to the American College of Sport Medicine criteria. 56 To preserve number of studies in each intensity category, categories of very-light and light, and vigorous and near-maximal to maximal were combined. For studies that progressed exercise intensity throughout the intervention period, studies were categorized based on the intensity performed at ⩾50% of the total intervention duration.
The robustness of the findings was tested with four a priori sensitivity analyses excluding studies of (1) older age females without confirmed postmenopausal status; (2) females with reported chronic conditions (i.e., reported hypertension, diabetes mellitus, dyslipidemia, and obesity); (3) outliers where high heterogeneity was observed; and (4) comparator groups of stretching, health education, relaxation, and placebo drug interventions.
Heterogeneity was assessed using Higgins
Quality appraisal
The methodological quality of included studies was evaluated by three sets of two independent reviewers (EH and EW, EH and KSN, EH and HF) using the Cochrane Risk of Bias 2 tool 57 (low, high, some concerns) for domains of random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data and selective reporting. Certainty of evidence was evaluated using the Grading of Recommendation, Assessment, Development and Evaluations (GRADE) tool for high, moderate, low, or very low certainty, based on assessments of risk of bias, imprecision, inconsistency, indirectness, and publication bias. 58 Publication bias was assessed using visual inspection of funnel plots and the Egger’s test conducted using Stata statistical software (Version 16.1; Stata CorpLLC, College Station, TX, USA). Disagreements were resolved by discussion and if necessary, resolution with a third reviewer (AT).
Results
Study flow diagram is displayed in Figure 1. Fifty-nine articles59 –117 from 57 trials were included in this review. Of these 59 articles, 6 were excluded from the quantitative synthesis due to incomplete reporting of means and/or SDs.59 –63,118 Thus, 53 articles65 –117 from 51 trials were included in the meta-analysis. Thirteen articles119 –131 were also excluded from the meta-analysis as the data were already represented by the primary trial article which had the largest sample for the reported outcomes of interest.

Study flow diagram.
Qualitative synthesis
Details of all included studies are summarized in Supplemental Table S1.
Participant characteristics
A total of 4,225 participants were included in this review, with study sample sizes ranging from 14 68 to 464. 105 Participants’ ages ranged from 45 years old 73 with confirmed postmenopausal status to 78 years old. 93 Forty-eight studies (81%) reported menopausal status, and 11 studies were included based on age criteria.62,66,68,71,79,93,101,104,108,116,117 Forty studies (68%) included sedentary postmenopausal females without reported chronic disease, and 19 studies included postmenopausal females living with 1 or more cardiometabolic conditions.59,60,67,71,72,76,86 –89,94,95,100,104,105,108,110,113,117
Aerobic exercise intervention
Aerobic exercise intervention durations ranged from 3 96 to 52 weeks,86,90,98 –100,106,107 with 8 to 60 min per session, except four studies87,105,111,112 that had a calorie or step goal. Interventions took place 3 to 21 times per week (3 times/day × 7 days). Aerobic interventions consisted of treadmill or cycle ergometer training, aquatic aerobic training, outdoor walking, and bench stepping. Forty-nine studies (83%) contained 1 aerobic intervention group, whereas 10 studies59,78,87,90,92,103,105,111 –113 contained multiple intervention groups that varied in intensity, duration, or type.
Comparator
Fifty-one studies compared aerobic exercise with no intervention (maintain usual diet and activity), and eight studies used stretching,77,111,112,116 health education,60,117 relaxation,104,116 or placebo drug interventions 65 as the control group.
Outcomes
SBP and DBP were investigated in 26 of 59 studies (44%), and 16 studies (27%) examined HR. Among body composition and anthropometric measures, BMI was the most frequently investigated outcome (
Quality assessment
From the quality assessment using the Cochrane Risk of Bias 2 tool, most studies were determined to be at high risk of bias due to failure to report participant attrition data (drop-out, reasons for drop-out) or had missing outcome data with failure to estimate intervention effects (intention-to-treat analyses) (Supplemental Table S2).
Adverse events
Thirty (51%) of the 59 studies did not report monitoring of adverse events.59,61 –63,65,66,68,70,73 –76,78 –80,84 –86,88,91,93 –96,101,102,110,113,117,118 Of the studies that monitored adverse events, 19 (32%)67,69,71,72,77,81 –83,89,92,97 –100,103,104,107 –109 reported no occurrence of adverse events, 4 (7%)87,105,112,116 studies reported adverse events unrelated to the exercise intervention, 3 (5%)60,114,115 studies reported adverse events but did not specify its relation to the intervention, and 3 (5%)90,106,111 studies reported adverse events due to the intervention.
Quantitative synthesis
Fifty-three studies (
Table 1 provides a summary of findings from the meta-analyses to examine the exercise-related effects on cardiometabolic outcomes of interest with the GRADE certainty of evidence. There were beneficial effects of aerobic exercise on all measures of cardiometabolic health except DBP, WHR, blood triglycerides, or blood glucose. Forest plots for beneficial effects are displayed in Figures 2 to 9; non-significant findings are displayed in Supplemental Figures S1–S4. All outcomes were downgraded from high to very low certainty of evidence due to multiple factors, including “very serious” or “serious” risk of bias, heterogeneity, indirectness, imprecision, and the suspicion of publication bias (Table 2).
Effect of aerobic exercise on traditional cardiometabolic health measures in postmenopausal females.
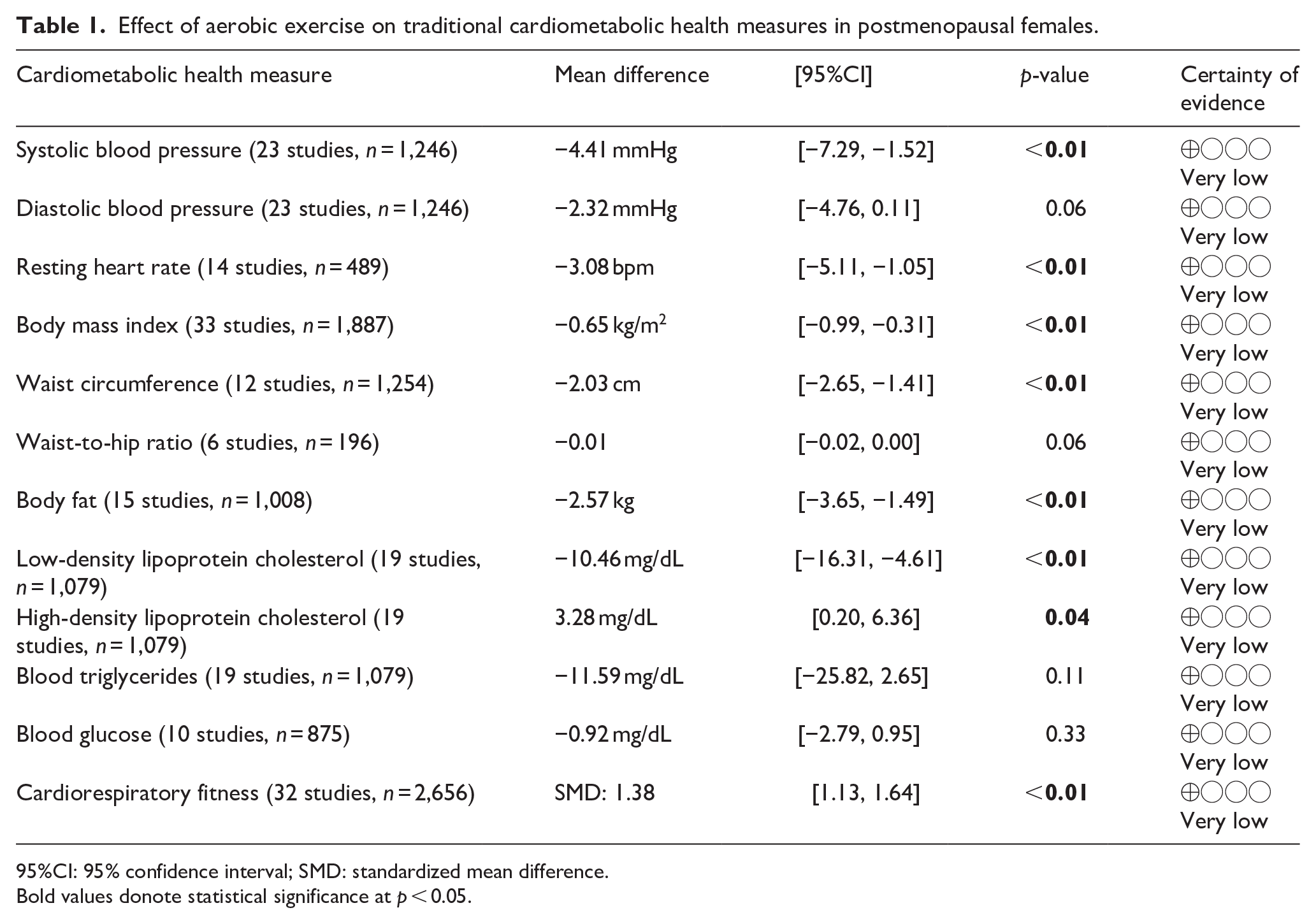
95%CI: 95% confidence interval; SMD: standardized mean difference.
Bold values donote statistical significance at

Forest plot of the effect of aerobic exercise on systolic blood pressure.
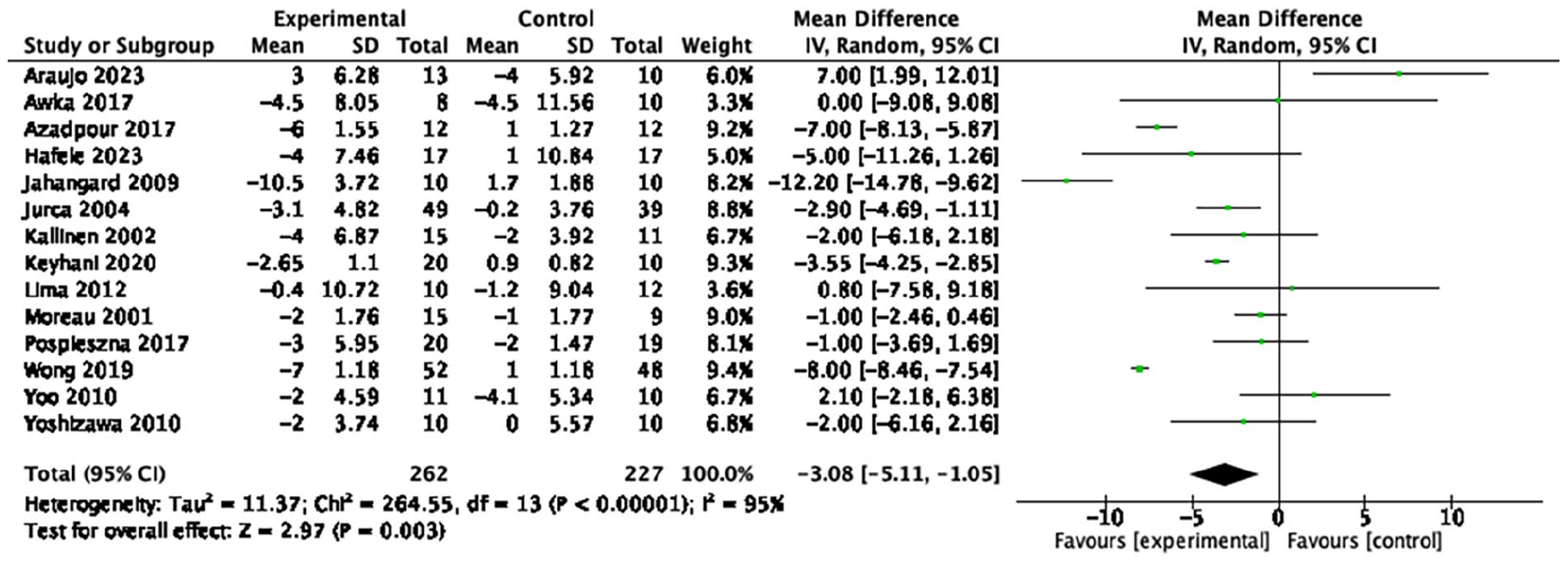
Forest plot of the effect of aerobic exercise on resting heart rate.
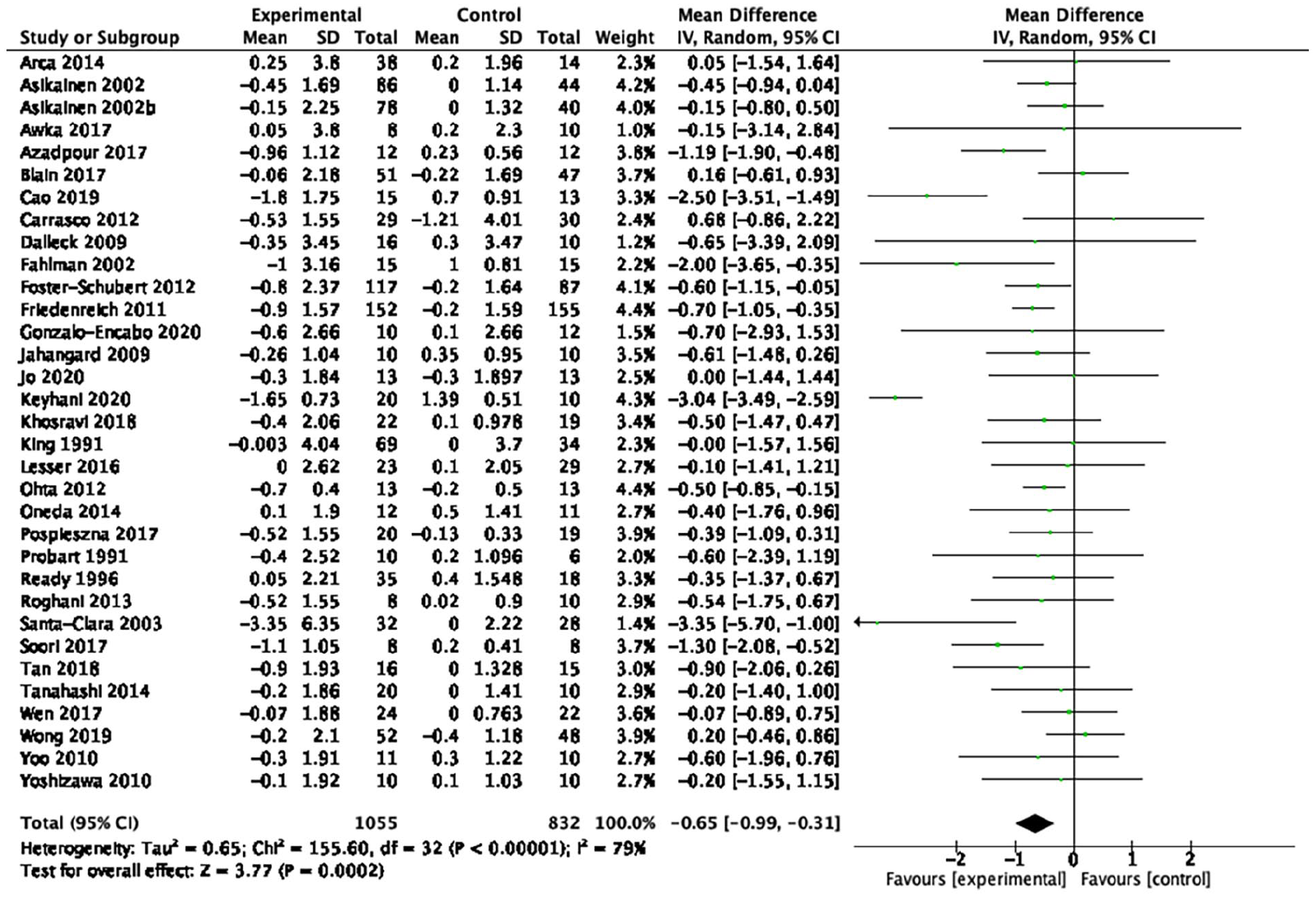
Forest plot of the effect of aerobic exercise on body mass index.

Forest plot of the effect of aerobic exercise on waist circumference.

Forest plot of the effect of aerobic exercise on body fat.
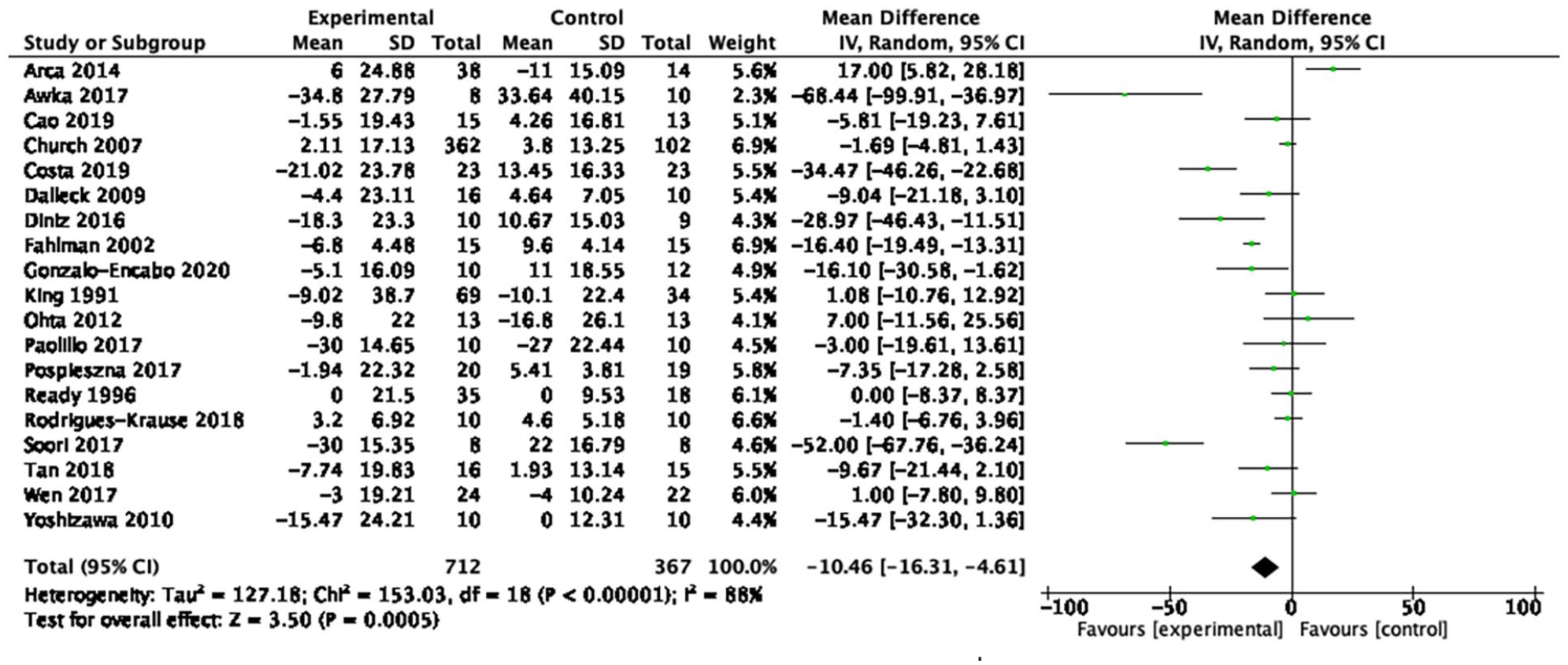
Forest plot of the effect of aerobic exercise on low-density lipoprotein cholesterol.
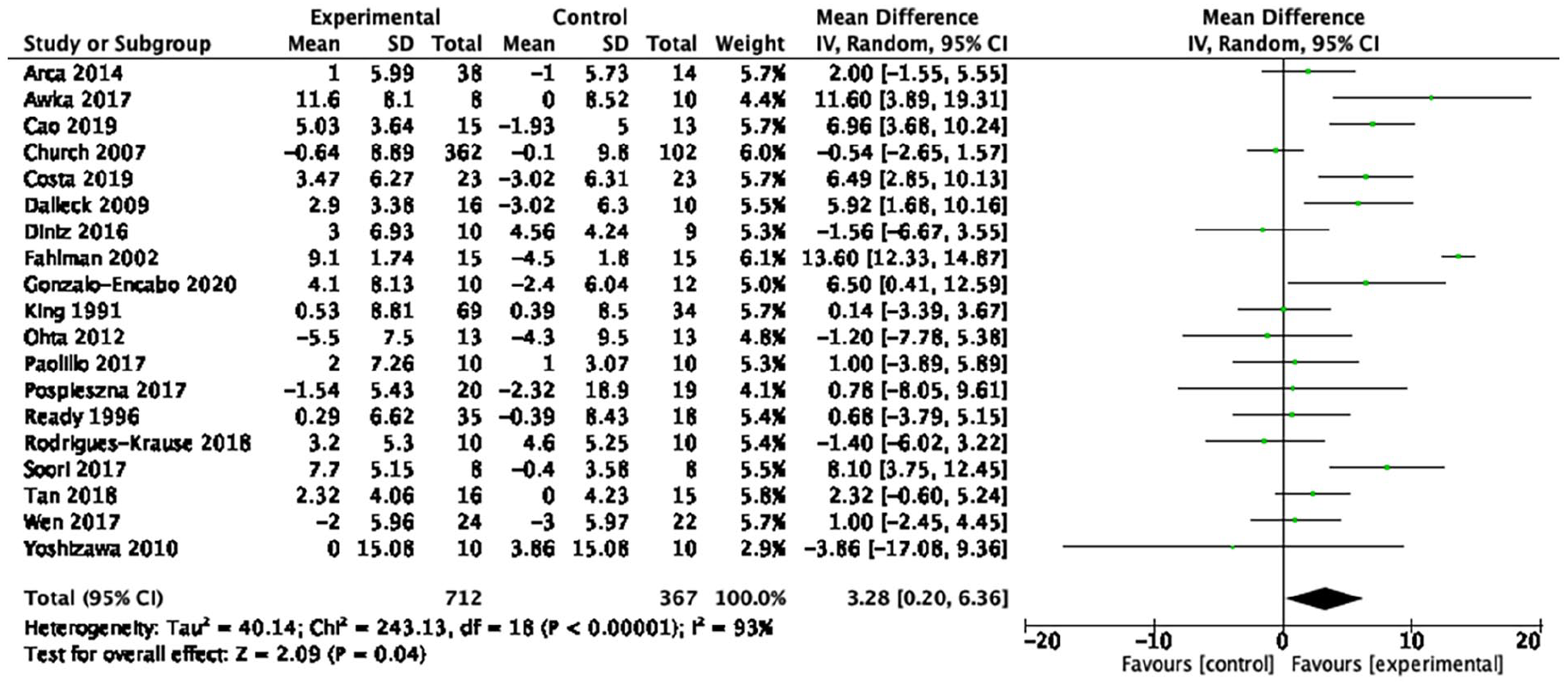
Forest plot of the effect of aerobic exercise on high-density lipoprotein cholesterol.
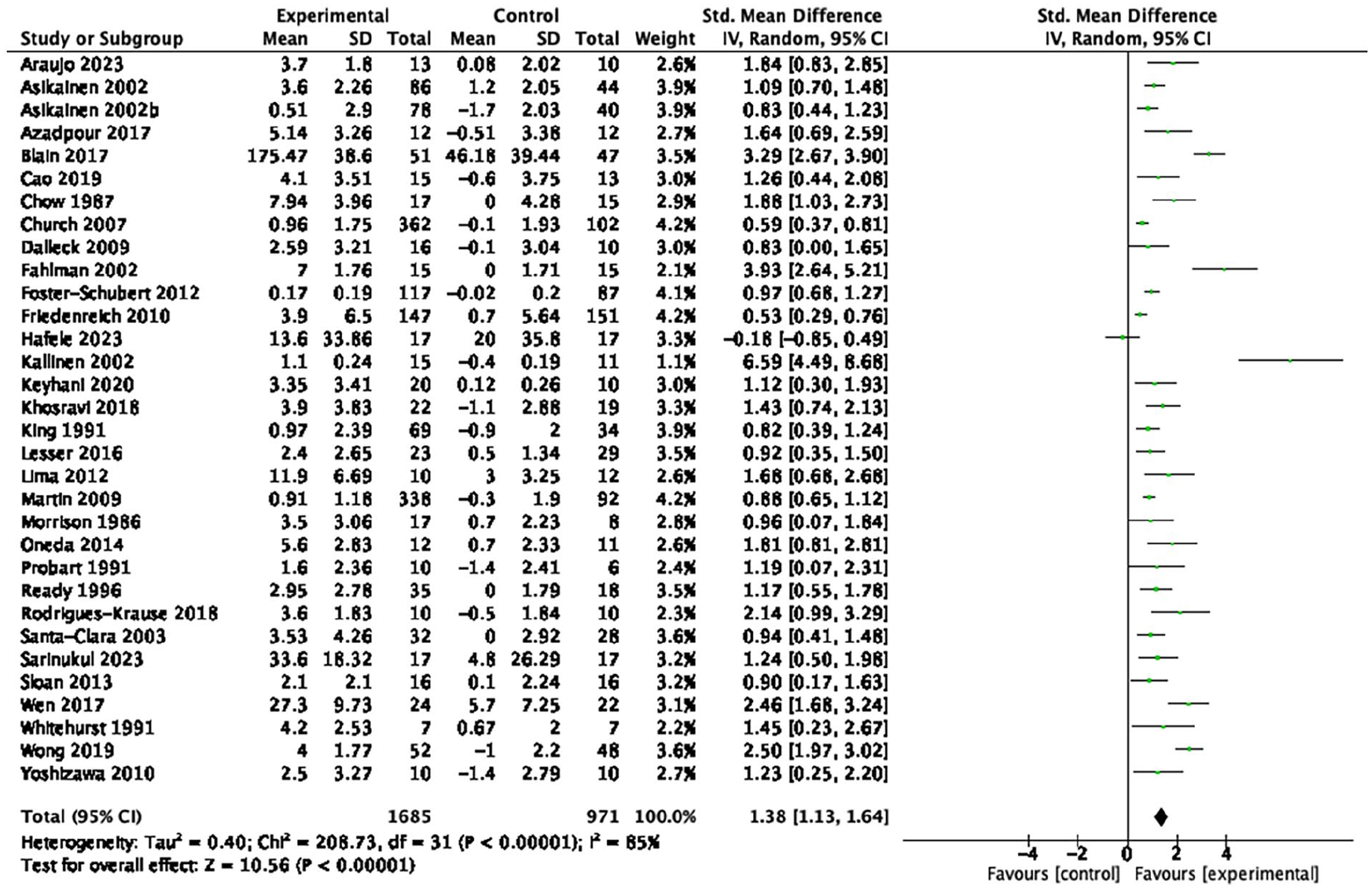
Forest plot of the effect of aerobic exercise on cardiorespiratory fitness.
Certainty of evidence of included studies using GRADE tool.

95% CI: 95% confidence interval; MD: mean difference; CRF: cardiorespiratory fitness; SMD: standardized mean difference; GRADE, Grading of Recommendation, Assessment, Development and Evaluations.
Downgraded for high risk of bias in majority of RoB-2 domains including attrition bias.
Downgraded for high statistical heterogeneity with overlapping CIs.
Downgraded due to heterogeneity in population and heterogeneity in comparison groups.
Downgraded for large 95%CI and crosses line of no effect.
Downgraded for large 95%CI, but sample >400 participants.
Downgraded for evidence of publication bias from Egger test and visual funnel plot assessment.
Downgraded for “some concerns” in majority of RoB-2 domains.
Downgraded for high statistical heterogeneity with minimal overlapping CIs.
Downgraded for crossing line of no effect but narrow 95%CI and sample >400 participants.
Positive effects in all outcomes were maintained following sensitivity analyses removing studies of (1) older age females without confirmed postmenopausal status; (2) females with reported chronic conditions; (3) outliers where high heterogeneity was observed; and (4) comparator groups of stretching, health education, relaxation, and placebo drug interventions.
Subgroup analyses
To conduct subgroup analyses based on intensity, 5 (10%)71,73,91,94,108 studies were categorized as light intensity, 25 (51%)65,67,68,72,74,76
–80,87
–89,93,96,103
–105,107,109,111
–115 were moderate intensity, and 19 (39%)66,69,75,81,82,84,86,90,92,95,97
–102,106,110,116 were vigorous intensity. Forest plots for the subgroup analyses are displayed in Supplemental Figures S5–S7. There was significant subgroup effect for BMI (χ2 = 9.79, df = 2,
Comparison between aerobic exercise intensity subgroups on cardiometabolic health measures.

95%CI: 95% confidence interval.
Denotes
No significant differences were found in the effects of exercise intensities on BMI and blood triglycerides when studies involving females with comorbidities were excluded. The results of the subgroup analyses remained after excluding studies that (1) did not explicitly report menopausal status; (2) studies with high heterogeneity; and (3) studies with comparator groups of stretching, health education, relaxation, and placebo drug interventions.
Discussion
This systematic review is the first to examine the effects of aerobic exercise in older postmenopausal females and those with common metabolic conditions. It is also the first to complete a comprehensive assessment of the certainty of evidence using the GRADE assessment, allowing clinicians and guideline developers to consider this information in their knowledge utilization and translation.
The positive effects of aerobic exercise on body fat, WC, LDL-C, HDL-C, and CRF found in this review are aligned with previous reviews,21,22 and is the first to report improvements in HR, and BMI. Moreover, these beneficial effects are consistent with reviews in adults with obesity and type 2 diabetes, which included combined interventions of aerobic training and diet, 132 and aerobic and resistance training. 133 There was little to no effect observed for DBP, WHR, blood triglycerides, and blood glucose, although the evidence is very uncertain. We note that many outcomes that demonstrated positive effects had a large proportion of studies with baseline values outside of desired levels and thus had greater potential for improvement. For example, most studies had participants who began the interventions in prehypertensive or hypertensive ranges (⩾120 mmHg) for SBP (68%), 134 or with high resting HR (50%), 135 BMI ⩾25 kg/m2 (84%), 136 WC ⩾90 cm (91%), 136 LDL-C ⩾100 mg/dL (89%) 137 and HDL-C ⩽50 mg/dL (44%). 138 This is likely a product of the broader range of studies eligible for this review, which included participants with common metabolic conditions and may explain, in part, the expanded positive effects observed in comparison to prior reviews. This approach, nonetheless, enables a more comprehensive representation of postmenopausal females, thereby increasing the generalizability of findings to underscore the benefit of aerobic exercise for individuals at higher risk for cardiovascular disease.
The null effect of aerobic exercise on DBP may be attributed to low baseline levels making it challenging to induce further reductions. The age of participants may play a factor; we note that >70% of studies in our review had a mean age of greater than 58 years old, and previous research has reported that DBP increases with age until approximately 58 years for females, after which it tends to decline,
139
possibly contributing to the normotensive DBP observed. We also did not observe an effect of aerobic exercise on WHR, although this analysis may have been underpowered due to the small number of participants included in the analysis (
The physiological mechanisms that underlie the positive effects of aerobic exercise training on cardiometabolic health are well supported. Aerobic exercise training decreases sympathetic nerve activity and increases parasympathetic activity leading to long-term peripheral vasodilation. 143 Exercise may also improve SBP by attenuating age-related decreases in arterial elasticity and compliance through increased pulse pressures and mechanical distension of collagen fibers. 144 Moreover, exercise-induced enhancements in stroke volume, leading to increased cardiac output during rest, can potentially contribute to a reduction in HR. 145 Aerobic exercise can improve body composition through increased total energy expenditure, 146 leading the body to rely on adipose tissue for energy. 147 Additionally, the upregulation of Proprotein Convertase Subtilisin/Kexin Type 9 due to aerobic exercise may accelerate the absorption and excretion of LDL-C, and upregulation of the liver X receptor gene has a strong effect on the formation of HDL-C. 148 CRF may be increased through changes to cardiac output by improved stroke and blood volumes,145,149 and arteriovenous oxygen content difference. 149
The studies included in our review found either beneficial or null effects, and the 95%CIs encompassed clinically important values for most outcomes, but we acknowledge that the positive effects observed are very uncertain based on the GRADE assessment. Exercise intervention trials are often poorly reported across a range of health conditions, 150 and the overall quality is inferior to pharmacological trials. 151 Nonetheless, we propose that clinicians should consider recommending aerobic exercise to improve cardiometabolic health and mitigate menopause-related cardiovascular risk. This conservative proposition also considered in part that postmenopausal females have shown interest in physical activity including aerobic exercise to improve body composition 152 and subsequently cardiometabolic health, and that some aerobic exercise training interventions such as walking are both feasible and low in cost making them a practical option. 153
In our secondary analyses, we observed that both light and vigorous intensities were effective in reducing BMI compared to moderate intensity, and light-intensity exercise for improving blood triglycerides and blood glucose levels. The positive benefits of light-intensity exercise but not moderate-intensity exercise were unexpected but may be a product of exercise volume,154 –156 which was not examined in this review. The small number of studies in the low-intensity category may have contributed to type I error, although we note that the baseline characteristics of the study samples were elevated in terms of cardiometabolic disease risk factors and thus may have been more responsive to change. For example, the light-intensity subgroup showed the largest effect on BMI but comprised a single study of overweight or obese females, two out of the three studies on blood triglycerides focused on females with hyperlipidemia, and the only study in the light-intensity subgroup for blood glucose specifically targeted females with type 2 diabetes mellitus. In sensitivity analyses with studies of overweight and obese females and females with hyperlipidemia removed, subgroup differences in exercise intensity on BMI and blood triglycerides were no longer observed. Hence, the positive effects of light aerobic exercise should be interpreted with caution.
Limitations
We acknowledge the limitations of this review. We only included studies published in English; there may have been potentially eligible studies from the 31 papers published in other languages. Moreover, while we used conservative age criteria to ensure with a degree of confidence that the females included were postmenopausal, there is a small possibility of including premenopausal females and excluding postmenopausal females who did not meet our age criteria. Next, although we conducted a sensitivity analysis excluding studies of older females to confirm our results, we were underpowered to perform a split analysis to observe the true effects of the influence of age. Finally, recent evidence suggests that while females with lifelong exercise behaviors accrue the greatest benefit to vascular health, 157 exercises initiated early in the menopause transition may mitigate age-related declines in vascular health more so than later in life. 158 Our review however was not designed to assess the influence of lifetime exercise patterns. Future work should incorporate data such as age and duration since the final menstrual period, and consider performing a split analysis or meta-regression to better understand the influence of these factors on the effects of aerobic training.
Conclusion
Aerobic exercise training may be an important strategy to mitigate the increased cardiometabolic risk that occurs following menopause. Aerobic exercise should be considered by clinicians and clinical practice guidelines for postmenopausal females as a non-pharmacological and non-diet primary preventative approach for cardiometabolic disease.
Supplemental Material
sj-docx-1-whe-10.1177_17455057241290889 – Supplemental material for The effects of aerobic exercise on cardiometabolic health in postmenopausal females: A systematic review and meta-analysis of randomized controlled trials
Supplemental material, sj-docx-1-whe-10.1177_17455057241290889 for The effects of aerobic exercise on cardiometabolic health in postmenopausal females: A systematic review and meta-analysis of randomized controlled trials by Eric Huynh, Elise Wiley, Kenneth S Noguchi, Hanna Fang, Marla K Beauchamp, Maureen J MacDonald and Ada Tang in Women’s Health
Supplemental Material
sj-docx-2-whe-10.1177_17455057241290889 – Supplemental material for The effects of aerobic exercise on cardiometabolic health in postmenopausal females: A systematic review and meta-analysis of randomized controlled trials
Supplemental material, sj-docx-2-whe-10.1177_17455057241290889 for The effects of aerobic exercise on cardiometabolic health in postmenopausal females: A systematic review and meta-analysis of randomized controlled trials by Eric Huynh, Elise Wiley, Kenneth S Noguchi, Hanna Fang, Marla K Beauchamp, Maureen J MacDonald and Ada Tang in Women’s Health
Supplemental Material
sj-docx-3-whe-10.1177_17455057241290889 – Supplemental material for The effects of aerobic exercise on cardiometabolic health in postmenopausal females: A systematic review and meta-analysis of randomized controlled trials
Supplemental material, sj-docx-3-whe-10.1177_17455057241290889 for The effects of aerobic exercise on cardiometabolic health in postmenopausal females: A systematic review and meta-analysis of randomized controlled trials by Eric Huynh, Elise Wiley, Kenneth S Noguchi, Hanna Fang, Marla K Beauchamp, Maureen J MacDonald and Ada Tang in Women’s Health
Supplemental Material
sj-docx-4-whe-10.1177_17455057241290889 – Supplemental material for The effects of aerobic exercise on cardiometabolic health in postmenopausal females: A systematic review and meta-analysis of randomized controlled trials
Supplemental material, sj-docx-4-whe-10.1177_17455057241290889 for The effects of aerobic exercise on cardiometabolic health in postmenopausal females: A systematic review and meta-analysis of randomized controlled trials by Eric Huynh, Elise Wiley, Kenneth S Noguchi, Hanna Fang, Marla K Beauchamp, Maureen J MacDonald and Ada Tang in Women’s Health
Footnotes
Acknowledgements
EH was supported by a Canadian Institute for Health Research-Masters Canada Graduate Scholarship. EW and KSN are supported by the Ontario Graduate Scholarship. MB supported by a tier 2 Canada Research Chair in Mobility, Aging, and Chronic Disease.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
