Abstract
In estrogen-receptor-positive tumors, adjuvant endocrine therapy has been shown to be highly beneficial for both overall and disease-free survival. Estradiol is key in regulating bone and mineral physiology, and several studies found a strong correlation between these therapies and the risk of fractures. Since these therapies are often given for 5 through 10 years, the timing for bisphosphonates or denosumab initiation seems essential to managing bone metabolism. However, gray zones and discrepancies between guidelines remain as to the best threshold when to start antiresorptive treatment, or whether antiresorptive treatment should be administered to every woman undergoing adjuvant endocrine therapy, independent of their risk factors for fractures. Treatment options and strategies should be discussed at the start of hormone adjuvant therapy to come to a shared decision with the patient, with the final aim of reducing the risk of future fractures as much as possible. This review will cover present guidelines and literature on antiresorptive treatment in this setting, to provide clinicians with useful clues for managing these patients.
Introduction
The spread of adjuvant endocrine therapy has been an extraordinary tool to improve overall survival and disease-free survival in women with early breast cancer (EBC), although it has led to some challenges for specialists in bone metabolism. There is no unanimous consensus in the guidelines on thresholds when to start or to delay anti-osteoporotic treatment because although several studies have shown a positive impact of antiresorptive medications on bone mineral density (BMD) in these patients, there has only been one fracture endpoint trial that showed reduction in fracture rates in patients treated with denosumab. The goal of this review is to support clinicians in the management of these patients, providing them with evidence-based data and graphic illustrations to easily familiarize with the main national and international guidelines.
Breast cancer epidemiology
Breast cancer accounts for 30% of female cancers and in North America its incidence appears to be greater than in other countries (97 in 100,000 person/year versus 27 in 100,000 person/year in Africa and east Asia), possibly reflecting the association between breast cancer and lifestyle factors associated with economic development. 1 In the United States, through an entire life span, up to one in eight women will receive a diagnosis of breast cancer, with the 5-year overall survival estimated to be 80% or higher in high-income countries. The significant improvement in breast cancer survival is likely related to considerable advancement in early diagnosis and implementation of effective and tailored therapies. Most of these tumors express the estrogen receptor (ER) and are therefore eligible for endocrine therapies with aromatase inhibitors (AIs), GnRH analogs (GnRHa), or tamoxifen (TAM). However, all these medications have consequences on bone metabolism because they interfere with estradiol production or signaling.
Hormone adjuvant therapy in breast cancer
Management of ER-positive breast cancer is different according to menopausal status. In premenopausal women, the main sources of estradiol are the ovaries, and the goal of therapy is to suppress estradiol production by inhibiting the hypothalamic-pituitary-gonadal axis. GnRHa are the main pharmacologic tool but are often not sufficient to reach a complete estradiol suppression because these medications are not able to inhibit sex hormone production by non-ovarian tissues like fat, liver, or adrenal glands, therefore, adding a concomitant treatment with AIs or TAM is also necessary.
In postmenopausal women, the goal of adjuvant cancer therapy is to suppress estradiol production from sources other than the ovaries. In this setting, the first-line options are AIs, or when these medications are not tolerated, TAM is used as a second-line option to reduce the risk of recurrence.
Bone physiology and annual bone loss
Following the acquisition of peak bone mass, the health of the bone mostly depends on remodeling, a complex dynamic process where the bone is constantly resorbed by osteoclasts and formed by osteoblasts. 2 When the coupling between these two cellular components is lost, the integrity of the bone tissue can be compromised. The main actor in this process is the BMU (basic multicellular unit), composed of osteoblasts, osteocytes, osteoclasts, and a capillary blood supply. 3 The remodeling process is more pronounced in the trabecular skeleton (e.g. spine, calcaneus, and proximal femur), which is the most metabolically active compartment of bone and therefore more vulnerable to perturbations by local or systemic factors that can cause significant imbalances in bone turnover. The main mechanisms involved in bone remodeling are the RANK ligand (RANKL)/RANK/Osteoprotegerin (OPG) pathway for osteoclast production and the Wnt signaling for osteoblast production.
The interactions between sex hormones and bone metabolism are complex and not completely understood. During the menopausal transition, estradiol levels decrease by 85–90% relative to premenopausal levels, and the rate of bone remodeling increases by 2-fold to 4-fold with a significant increase in bone resorption mediated by osteoclasts, resulting in a negative balance of the remodeling cycle. In the first 5–8 years after the cessation of menses, about 20–30% of the trabecular bone and 5–10% of the cortical bone are lost with resultant trabecular deterioration and cortical porosity.4–6 Furthermore, compared with postmenopausal osteoporosis, complete suppression of estradiol levels during AIs treatment could lead to additional pathophysiologic mechanisms, for example, the inhibition of insulin-like growth factor 1 7 and vitamin D bioavailability (by interfering with the protein carrier), 8 thereby causing several bone-active hormones disruptions. 9
Bone health in cancer patients
Women starting hormone adjuvant therapy for breast cancer could have additional risk factors for developing osteoporosis compared with the general population, beyond low estradiol levels. These risk factors are usually exposure to corticosteroids given along with chemotherapy, vitamin D deficiency and advanced age (mean age at onset of breast cancer is 60 years).10,11 A higher remodeling rate promotes bone loss thereby increasing fracture risk. In this setting, circulating tumor cells might also be more prone to seed within the bone tissue, thereby giving rise to the pre-metastatic niche in selected secondary organs. 12 By reducing the rate of bone remodeling with therapeutics, that is antiresorptive drugs, the spread of malignant disease might also be contrasted. The effects of adjuvant therapy on BMD have been quantified by several studies that have reported an annual bone loss in healthy postmenopausal women of 1–2% per year, while AIs therapy alone causes 2–3% BMD loss per year, greater during the first year, and progressively lower in the following years. In premenopausal women bone loss is as high as −7% per year for GnRH agonists with concomitant AIs, and up to −7.7% for ovarian failure secondary to chemotherapy.13–15
Aromatase inhibitors
AIs inhibit aromatase (CYP19A1), a key enzyme responsible for the conversion of testosterone into estradiol and androstenedione into estrone, especially in breast cancer cells and adipose tissue, therefore estradiol levels fall by over 80–90% in postmenopausal or menopause-induced women while receiving treatment with AIs. 16 There are two classes of AIs: steroidal (exemestane) and non-steroidal (anastrozole and letrozole). Exemestane is a steroidal analog of androstenedione and binds irreversibly to aromatase 17 while anastrozole and letrozole are triazole agents which bind reversibly to the heme group of the enzyme aromatase. Exemestane, anastrozole, and letrozole cause about 98% aromatase inhibition or greater in postmenopausal women, and the estrogen deficiency induced by AIs and GnRHa leads to a significant increase in bone resorption and accelerated bone loss, especially at the trabecular bone-rich sites. In vivo animal studies have suggested that exemestane, due to its androgenic structure, may be more bone-sparing as compared with non-steroidal counterparts, 18 but this hypothesis has not been confirmed in human studies. The MA-27 trial compared exemestane and anastrozole in postmenopausal women, 19 and although self-reported new diagnoses of osteoporosis were significantly less frequent on exemestane compared with anastrozole, there were no differences in clinical fragility fractures between the two groups, and a complementary analysis could not confirm significant BMD variations between the two medications. 20
Tamoxifen
TAM is a selective-ER modulator (SERM), with antagonist properties on breast and cancer tissue. In premenopausal women, it has been associated with higher bone loss, while in postmenopausal women, or in association with GnRHa, TAM progressively increases lumbar BMD, thereby benefiting bone metabolism. 21 This paradoxical effect could be explained by the competition of SERMs and endogenous estradiol for the binding with the ER: premenopausal women show high estradiol levels and the occupations of ERs by SERMs could lead to a blunted receptor response, whereas in postmenopausal women, with much lower endogenous estradiol levels, the occupation of the receptor by SERMs can lead to greater signaling activation.
Ovarian suppression and GnRH analogs
Ovarian suppression is generally indicated in young women (⩽35 years) or in women with high-recurrence risks factor (e.g. pathologically involved lymph nodes, tumor ⩾ 5 cm, involvement, genetic factors, etc.). There are three available strategies to suppress ovarian function:
GnRHa: administration of goserelin, leuprolide, or triptorelin, typically in monthly formulations, produces an early receptor activation with subsequent downregulation and desensitization in gonadotropic cells. As a result, luteinizing hormone (LH) and follicle-stimulating hormone (FSH) are reduced, with consequent estradiol deficiency. 22
Oophorectomy: causes a sudden drop in circulating estradiol levels, although it involves the risks associated with anesthesia and surgery. 23
Ovarian irradiation: requires cumulative doses over 12 Gy and is rarely used because of the side effects of radiation therapy.
AIs compared with TAM
Several randomized trials have shown the superiority of AIs to TAM with significant improvement in disease-free, bone metastases-free, and overall survival.24,25 On the other hand, AIs cause a greater decline in BMD and a higher fracture risk as shown in some trials that compared TAM versus anastrozole,21,26 TAM versus letrozole,27–29 or TAM versus exemestane.30–32 However, it is important to consider that most data on bone loss related to adjuvant endocrine therapy derive from several randomized studies where the bone loss was reported as an adverse event or from sub-analyses conducted on the same trials to describe BMD changes and the risk of bone fractures. 33 Furthermore, AIs have mostly been studied against TAM amplifying their perceived negative effect in terms of BMD loss or fractures (Table 1).
Clinical trials with aromatase inhibitors in postmenopausal women diagnosed with early breast cancer.

BMD: bone mineral density; DFS: disease-free survival; LS: lumbar spine; TH: total hip; ANA: anastrozole; LET: letrozole; EXE: exemestane; y: years; m: months; d: day; NA: non-available.
Non-significant difference of clinical fractures between the letrozole group and the placebo group.
Non-significant difference of BMD changes at the total hip after 12 months.
A recent metanalysis 33 has confirmed a 35% increase in fracture risk in patients during AIs treatment in comparison to TAM, with similar results confirmed also in a large-scale real-world cohort study. 35 Moreover, several breast cancer trials compared extended treatment schedules of AIs versus placebo or no further treatment, and even then, a higher fractures risk was reported for prolonged AIs therapy, with an odds ratio for fractures at 1.34 [1.16–1.55] 95% confidence interval (CI). 36
Comparative studies between anastrozole, letrozole, and exemestane have shown that none of the three AIs approved for breast cancer treatment is superior to the others in terms of cancer outcomes or their effects on bone metabolism.37,38
Vertebral fractures and AIs
High variability of fracture rates has been reported in trials investigating AIs, ranging from 5% to 10% at 5 years of treatment. With prolonged treatment, fracture rates are expected to increase. The high variability of fracture rates reported in trials investigating AIs, ranging from 5 to 10% at 5 years of treatment, is likely due to the fact that fracture was not a pre-specified outcome of these studies and there may be differences in the reporting practices of patients when they sustain a fracture across the different trials.
Prevalence of vertebral fractures (VFs) in women receiving AIs therapy in EBC has been described by a cross-sectional Italian study in which 263 consecutive women underwent a dual-energy X-ray absorptiometry (DXA) and a morphometric X-ray of the spine. Prevalence of VFs was 18.9% in AI-naïve patients, and up to 31.2% in those assessed during AI therapy (odds ratio (OR) 1.90, p = 0.03). Mean age was similar between the two groups (64 years in AI-naïve patients and 65 years in AI-treated patients, p = 0.07), and in AI-naïve patients, VFs were associated with older age and lower BMD values at femoral neck and total hip. Oddly, in the AI-treated group, the prevalence of VFs was not significantly different between patients with osteoporosis and those with normal BMD. 34
Treatment options
Non-pharmacological interventions
A healthy lifestyle is crucial for bone health, even more so in people who have increased fracture risk. It is recommended physical activity be increased, including balance training, weight-bearing, flexibility, or stretching exercises, to reduce the risk of fractures caused by falls.14,39
Clinicians should also actively encourage patients to stop smoking and limit alcohol consumption.14,39
If an adequate intake of calcium and vitamin D is not being consumed, then supplements to reach those levels are generally suggested. The optimal intakes in this population are uncertain, and different guidelines expressed their views on this point. The Joint statement of 2017, according to the IOF, mentions a daily calcium intake of 1200 mg/day, whereas ASCO 2019 guidelines and European Society for Medical Oncology (ESMO) 2020 guidelines recommend an intake of 1000–1200 mg/day. The optimal vitamin D3 intake is at least 800–1000 IU/day according to ASCO 2019 guidelines, 1000–2000 IU/day conforming to ESMO 2020 guidelines, and 800–2000 in agreement with the Joint position statement of 2017.14,39,40
Pharmacological interventions
The main treatments available for the prevention of AI-induced bone loss are bisphosphonates and denosumab, on the contrary, parathyroid hormone (PTH) analogs like teriparatide and abaloparatide, are generally not used in breast cancer because of their potential risk for the development of osteogenic sarcoma in patients undergoing radiation therapy. Also, romosozumab is currently not recommended in women with EBC due to a lack of evidence, although no contraindications have emerged so far. 40
Bisphosphonates
Bisphosphonates are analogs of inorganic pyrophosphate, which is one of the main constituents of the bone mineral matrix. Osteoclasts take up nitrogen-containing bisphosphonates, which then bind to and inhibit the activity of farnesyl pyrophosphate synthase (FPPS), a key enzyme in the mevalonic acid pathway, also critical to the production of cholesterol and other lipids. The inhibition of FPPS results in a reduced synthesis of proteins (e.g. Rab, Rac, and Rho) which play key roles in the regulation of core osteoclast cellular activities including stress fiber assembly, membrane ruffling, and survival. FPPS inhibition ultimately leads to osteoclast apoptosis. 41
In several randomized clinical trials, bisphosphonates prevented or reduced BMD loss in women receiving AIs, but so far, no randomized trials have shown a consistent reduction in fracture risk in patients treated with bisphosphonates. However, data on fractures have been reported in a large-scale observational cohort study, with a mean follow-up of 10 years, in which fracture incidence was 30% lower in the subgroup of high-risk patients treated with oral BP compared with untreated patients (hazard ratio (HR) 0.69 (0.48–0.98). 42 Although this study has some limitations, such as the lack of randomization, the uncertain severity of breast cancer at the diagnosis and the impossibility to discern between traumatic fractures and osteoporotic fractures, its large population database and the real-life design provide important messages about the effectiveness of bisphosphonates in these patients. Also, the Early Breast Cancer Trialists’ Collaborative Group showed that, by administering bisphosphonates, the 5-year fracture risk was reduced from 6.3% to 5.1%, with little effect in years 0–1 and most of the benefit in years 2–4. 43
Alendronate
The pivotal study published on alendronate during AIs therapy was the BATMAN trial, 44 in which 303 postmenopausal women with EBC were enrolled. At baseline, all osteoporotic women (N = 25) received weekly 70 mg alendronate, while of the 146 patients classified as osteopenic at baseline, only 11 commenced alendronate therapy. The 126 patients with normal BMD did not receive any anti-osteoporotic treatment. In osteoporotic patients receiving alendronate BMD significantly increased at 3 years (LS + 15.6%, TH + 5.6%), and only one non-traumatic fracture occurred during that period (3.85%) Osteopenic patients who started alendronate at baseline (N = 11) showed a significant increase in LS BMD at 3 years (+6.3%) with no significant changes at the hip and no osteoporotic fractures reported, while osteopenic women without anti-fracture therapy (N = 124) had a non-significant decrease of −1% at the LS and –1.7% at TH and a total of 10 fragility fractures (8.06%) occurred over 3 years. Patients with normal BMD showed a significant BMD decrease at 3 years (LS −5.4%, TH −4.5%) and 2 atraumatic fractures (1.59%) were documented.
In the Pineda-Moncusí cohort study 42 , women at high risk of fractures during AIs therapy receiving alendronic acid or alendronic acid plus cholecalciferol showed a non-significant reduction in fractures risk compared to those who did not receive anti-osteoporotic treatment (alendronic acid HR 0.84 (0.58–1.23) 95% CI, alendronic acid plus cholecalciferol HR 0.35 (0.10–1.21) 95% CI (Table 2).
Risedronate during aromatase inhibitors therapy.

BMD: bone mineral density; DFS: disease-free survival; LS: lumbar spine; TH: total hip; AI: aromatase inhibitor; Wk: week; Py: person-year; HR: hazard ratio; y: years; m: months; d: day.
Sestak study had a cohort of postmenopausal women at high risk of developing breast cancer, but without previous diagnosis of breast cancer.
Risedronate
In the SABER trial 45 postmenopausal women receiving anastrozole were divided by their baseline T-scores into low risk (T-score ⩾−1.0), moderate risk (T-score ranging from −1.0 and −2.0), and high risk (T-score <−2.0) for future fractures. Women belonging to the moderate risk group were randomized to receive oral risedronate 35 mg/week orally or placebo. After 2 years of therapy, there was a significant difference in BMD between those who received risedronate and the placebo group (at the LS + 2.2% versus −1.8% and at the total hip + 1.8% versus −1.1%, respectively). Furthermore, women in the low-risk group, who did not receive any anti-osteoporotic treatment, had a significant decrease in BMD at LS (−2.1%), as opposed to women in the high-risk group who received risedronate (LS + 3%, TH + 2%).
In 2019, Sestak et al. 46 studied a cohort of postmenopausal women at high risk of developing breast cancer and the difference in BMD change between the anastrozole/risedronate group versus the anastrozole/placebo group was significant at the LS (−0.1% versus –4.2%, p < 0.0001) and non-significant at the TH (−2.5% versus –3.8%, p = 0.2).
A recent meta-analysis of 16 randomized trials showed that zoledronate and denosumab resulted in significantly higher BMD of LS and TH at 1 and 2 years compared to risedronate. However, risedronate, contrary to zoledronate, was associated with lower fracture risk. 47 This is quite unexpected, considering previous studies about bone turnover markers suppression, in which risedronate seemed to be less effective than other bisphosphonates (i.e. alendronate and ibandronate) in reducing bone resorption markers. 48
In the 2019 study by Pineda-Moncusí et al., 42 the subgroup of patients treated with risedronic acid showed the lowest fracture risk (HR 0.47, (0.25–0.86) 95% CI) (Table 3).
Alendronate during aromatase inhibitors therapy.

BMD: bone mineral density; LS: lumbar spine; TH: total hip; AI: aromatase inhibitor; Wk: week; Py: person-year; HR: hazard ratio.
Ibandronate
Ibandronate is a bisphosphonate available in both oral and intravenous formulations. The BONADIUV trial 49 evaluated the impact of ibandronate treatment on osteopenic breast cancer patients receiving adjuvant AIs, with 171 osteopenic patients randomized in a 1:1 ratio to receive either monthly 150 mg oral ibandronate or placebo. At the 2-year follow-up, the mean change was statistically significant in favor of ibandronate at both sites (LS + 6.09% versus −4.22%, TH + 4.64% versus −1.51%). The ARIBON 50 trial had a similar design with 50 osteopenic patients randomized to receive either treatment with ibandronate 150 mg orally every month or a placebo. After 2 years, osteopenic patients treated with ibandronate gained + 2.98% and + 0.6% at the LS and TH, respectively, while patients treated with a placebo lost −3.22% at the LS and −3.9% at TH (Table 4).
Ibandronate during aromatase inhibitors therapy.

BMD, bone mineral density; LS, lumbar spine; TH, total hip; AI, aromatase inhibitor; Py, person-year; HR, hazard ratio; d, days.
Zoledronate
Zoledronic acid is an intravenous amino-bisphosphonate with a high binding affinity for mineralized bone matrix and for the FPPS active site, which allows for a long therapeutic duration. Several dosing schedules of zoledronic acid have been studied based on different indications, including anti-osteoporotic dosing (5 mg/12 months), conventional dosing (4 mg/3–4 weeks), maintenance dosing (4 mg/3–6 months), and metronomic dosing (1 mg/week). Most of the studies on zoledronate in early BC considered the administration of 4 mg doses every 6 months. No data are available on the 5 mg/12 months dosing schedule.
Two large, randomized trials, ZO-FAST 51 and Z-FAST,52,53 evaluated zoledronic acid in postmenopausal women, with ER + EBC receiving letrozole. Patients were randomized to receive either immediate or delayed treatment with zoledronate 4 mg q6m. In the “immediate” arm, patients initiated zoledronate within 1 month of randomization and continued for 5 years. In the “delayed” arm, patients received zoledronate after initiating letrozole, when their BMD T-score fell below −2 or if they sustained any fractures (non-traumatic clinical fracture or asymptomatic fracture detected at the 36-month assessment). In the ZO-FAST study, at 60 months, the immediate treatment group showed an increase in BMD at LS (+ 4.3%) and at TH (+ 1.6%), compared with significant BMD loss in the delayed group (LS −5.4%, TH −4.2%). Comparable results were presented in the Z-FAST trial, with average BMD differences between the two groups at 61 months of 8.9% at LS and 6.7% at TH.
The N03CC trial 54 confirmed similar results in a cohort of postmenopausal women of 551 with breast cancer who had completed TAM treatment and were undergoing daily letrozole treatment (difference in mean percent change between the upfront arm and delayed arm was 5.3%, 7.34%, and 9.42% at 1 year, 2 years, and 5 years, respectively).
The E-ZO-FAST trial 55 showed a significant BMD difference between the two groups (delayed and immediate treatment) already at 12 months both for LS and TH.
Lee et al. 56 analyzed BMD changes at 1, 2, and 3 years, in a cohort of 107 breast cancer patients treated with an AI either alone or in combination with zoledronic acid and the results showed a significant difference between the two groups at every endpoint (Table 5).
Zoledronate during aromatase inhibitors therapy.
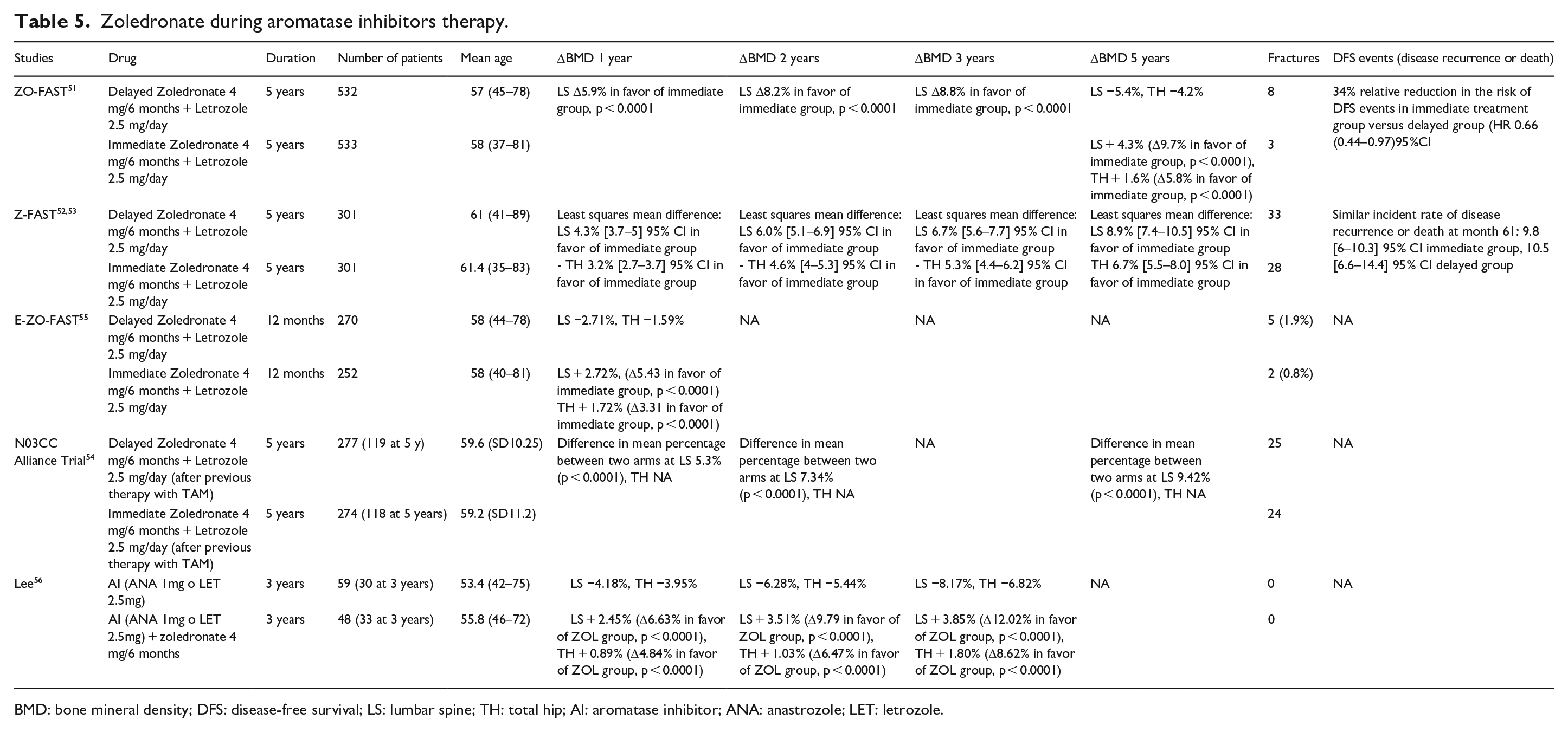
BMD: bone mineral density; DFS: disease-free survival; LS: lumbar spine; TH: total hip; AI: aromatase inhibitor; ANA: anastrozole; LET: letrozole.
Adjuvant effect of bisphosphonates
Bisphosphonates in breast cancer have been associated with a reduction in bone recurrences and an improvement in survival. 43 This effect might be explained by the “pre-metastatic niche hypothesis”: circulating tumor cells can be attracted to surfaces within the bone, where they can displace hemopoietic stem cells and bind to the osteoblastic niche. These disseminated malignant cells can remain quiescent for years and subsequently, they can exit this dormant state, start to proliferate, and progress to macrometastases in the bone or elsewhere. 43 Bisphosphonate’s effect on osteoclasts and T-cell function seems to be key in preventing or delaying bone recurrences. 57 Interestingly, this oncological benefit has been demonstrated only in postmenopausal women or in older women, and, possibly, it might require low concentrations of reproductive hormones. The recent American Society of Clinical Oncology-Ontario Health Cancer Care Ontario (ASCO-OH CCO) guidelines 58 recommend discussing adjuvant bisphosphonate therapy with all postmenopausal patients with primary breast cancer. The benefit of adjuvant bisphosphonate therapy will depend on the underlying risk of recurrence and the NHS PREDICT tool 59 provides an estimate of these benefits and may aid in shared decision-making.
Denosumab
Denosumab is a fully human IgG2 monoclonal antibody that binds with high affinity and specificity to RANKL, a member of the tumor necrosis factor superfamily of ligands and receptors. The drug blocks the binding of RANKL to RANK, thereby reducing osteoclasts’ formation, function, and survival. Denosumab is approved for the treatment of osteoporosis (60 mg sc. every 6 months) and bone metastases (120 mg sc. monthly) at different dosages.
In a 2-year randomized trial from Ellis et al., 60 250 osteopenic postmenopausal women receiving AIs were randomly assigned to receive a placebo (n = 125) or subcutaneous denosumab 60 mg (n = 127) every 6 months. At 24 months, BMD increased not only at LS (+ 7.6%) and TH (+ 4.7%) but also in the femoral neck (+ 3.6%) and one-third radius (+ 6.1%) where the cortical bone is predominant.
In ABCSG-18 61 trial, 3240 women with EBC ER/PR + receiving adjuvant AIs were randomly assigned in a 1:1 ratio to receive either denosumab 60 mg or placebo, administered subcutaneously every 6 months, in 58 trial centers in Austria and Sweden. This is the only randomized trial that found a significant reduction in the incidence of clinical fractures between the treated group (92/1711) and the placebo group (176/1709) with HR 0.50 (95% CI 0.39–0.65, p < 0.001). Subgroup analysis confirmed that the recorded reduction in clinical fractures was similar in all patient subgroups, including in the 1872 patients with normal BMD at baseline and the vertebral fracture analysis set at 36 months (N = 1634) showed a significant reduction in vertebral fracture incidence, with 27 events every 835 patients in the denosumab group compared with 49 every 809 patients in the placebo group (OR 0.53 (0.33–0.85) p = 0.009). (Table 6)
Denosumab during aromatase inhibitors therapy.

BMD: bone mineral density; DFS: disease-free survival; LS: lumbar spine; TH: total hip; FN: femoral neck; 1/3 radius: one-third distal radius; AI: aromatase inhibitor; Dmab: denosumab; HR: hazard ratio.
The impressive results of ABCSG-18 were extremely debated by the scientific community and several limitations were mentioned about this study, starting with the fact that the primary endpoint was not the number of fractures in the two groups, but the time from randomization to the first clinical fracture. Thirty-six months after randomization, 9.6% [8.0–11.2] 95% CI of patients in the placebo group reported a fracture and this proportion is somewhat higher than that reported in other trials, which may be indicative of more complete ascertainment of clinical fractures in studies in which fracture incidence was the primary endpoint. 62 In addition, the clinical and pathological characteristics of participants with underlying breast cancer were associated with quite a good prognosis, with only 25% of patients needing neoadjuvant or adjuvant chemotherapy, and patients who had osteoporosis (T-score <–2.5) at study entry were not specifically excluded. 1548 study participants (45%) had a T-score lower than –1.0, but the prevalence of other risk factors for fracture in the study population was not reported.
Finally, Coleman and Hadji 62 remarked on the survival benefit associated with the use of adjuvant bisphosphonates in EBC treatment. Denosumab’s adjuvant efficacy instead, had no robust evidence on disease-related outcomes for women with high-risk EBC. Indeed, there is an important discrepancy in data from the ABCSG-18 trial 63 that showed a significant improvement in disease-free survival in women treated with denosumab and data from the D-CARE trial that did not show efficacy on the onset of bone metastasis or disease recurrence in the patients with EBC. 64 This discrepancy is likely due to differences in the study populations, dosing schedules, or concomitant anticancer treatments. In particular, it should be noted that in the D-CARE trial, patients with high-risk cancer were given a higher dose of Denosumab (120 mg every 4 weeks for approximately 6 months, followed by 120 mg every 3 months for a total duration of treatment of 5 years) and, even then, it did not confirm the promising results from ABCSG-18, in which patients with low-risk breast cancer were given 60 mg every 6 months. In addition, in the ABCSG18 trial cancer outcomes were a post hoc analysis and not a primary endpoint, so the trial may not have been adequately powered for this outcome. Hopefully, future studies will clarify the adjuvant efficacy of denosumab.
When comparing these medications, we should also consider that bisphosphonates are considerably less expensive than denosumab and they can be discontinued without worrying about the “rebound effect,” which is characterized by rapid bone loss after denosumab cessation. On the other hand, severe renal function impairment constitutes a contraindication to the use of bisphosphonates, as opposed to denosumab. 65
Risk assessment
Although BMD measurement by DXA identifies those patients at the greatest individual risk of fracture in the population, a great number of fractures occur in individuals with osteopenia or normal BMD values.66,67 To improve the identification of patients at high fracture risk, different algorithms have been devised to integrate the risks associated with clinical risk factors as well as BMD. The most widely used tool is the Fracture Risk Assessment Tool (FRAX®) in which AIs exposure is not a direct input but can be captured under “secondary osteoporosis.” This input affects FRAX calculations when BMD is not entered, but not when BMD is included, since the risk is assumed to be mediated through BMD. Contrary to the common view that FRAX would underestimate the risk of fracture in women during AIs therapy, in a Canadian large registry-based cohort study,, 68 it was observed that fracture incidence was lower than predicted if secondary osteoporosis was included in the baseline fracture risk estimation without BMD. However, when BMD was included in the fracture probability, there was no significant difference between observed and predicted fracture risk.
Another novel assessment that enhances the detection of patients with elevated fracture risk is the Trabecular Bone Score (TBS), a gray-level texture measurement that uses lumbar spine DXA images to discriminate changes in bone microarchitecture. The combination of FRAX, TBS, and BMD maximized the identification of patients who may benefit from an anti-osteoporotic treatment in a cohort of 100 patients with EBC hormone-positive treated with AIs. 69 Recently, FRAX has been modified to return fracture risk-adjusted for TBS values.
Clinical guidelines
Several guidelines have been produced regarding the management of women taking AIs, nonetheless, a unanimous agreement has not been reached about thresholds for the initiation of anti-osteoporotic treatment. A few guidelines are summarized below.
The Belgian Bone Club (BBC) in 2007 recommended starting bisphosphonates in women with a T-score <−2.5 or a history of fragility fracture. In addition, the panel suggested considering treatment for osteopenic patients based on the severity of osteopenia and the presence of other risk factors. Regular BMD assessment in untreated patients and treatment initiation if significant bone loss is detected in osteopenic patients was also recommended. 70 The first-line choice was zoledronate 4 mg every 6 months, based on the Z-FAST trial’s results. Other regimens of bisphosphonate treatment were under evaluation at the time of writing these recommendations (Figure 1).
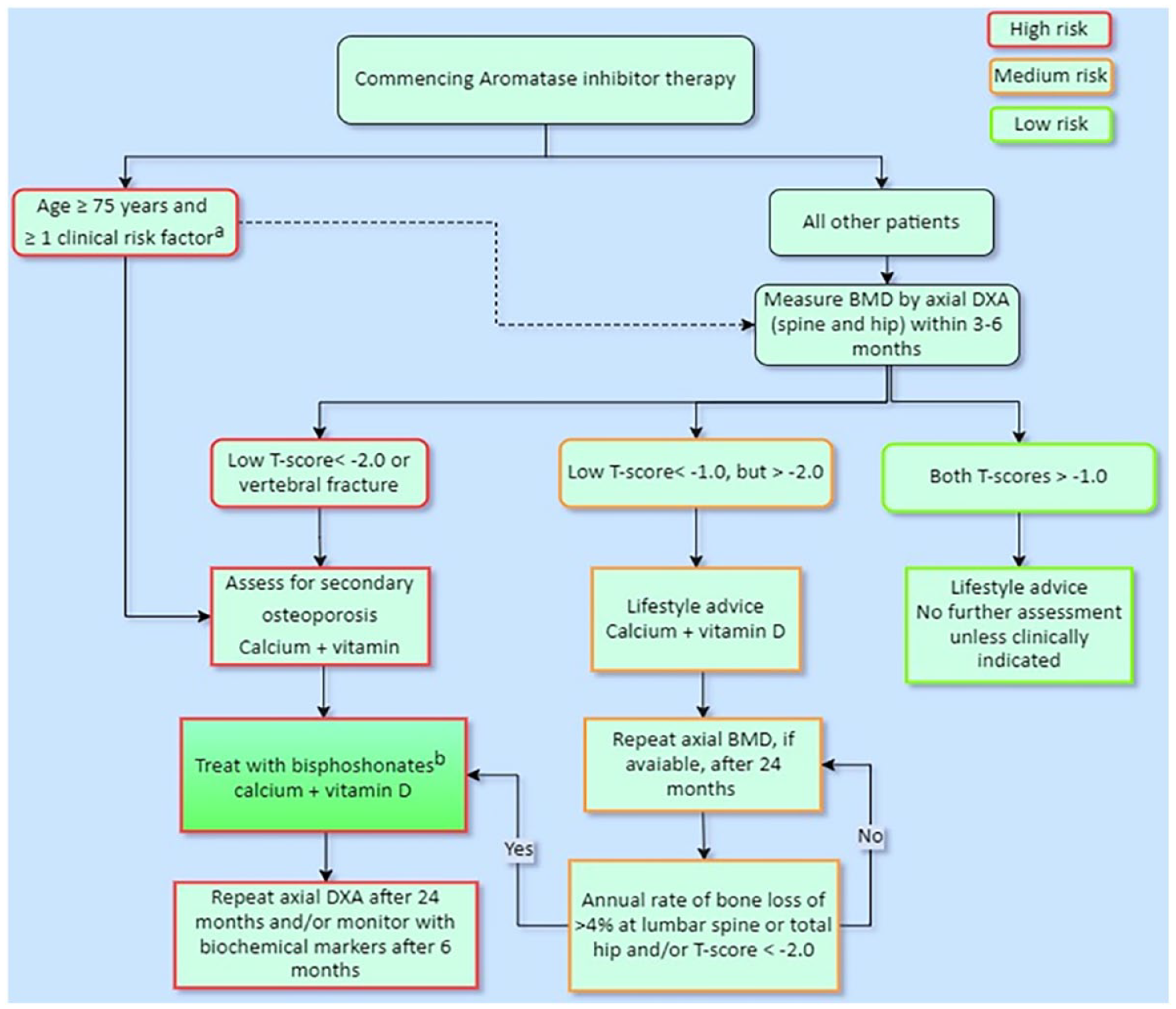
A 2008 Consensus position statement from a UK Expert Group recommended bisphosphonates therapy for women older than 75 years who had one or more risk factors for osteoporotic fracture, regardless of their baseline BMD. In addition, the panel recommended bisphosphonates for postmenopausal women younger than 75 years with T-scores <−2.0 or with bone loss ⩾ 4% per year in pre-existing osteopenia. Follow-up DEXA at 24 months was recommended in women who were not candidates for bisphosphonate therapy to reevaluate the necessity to start therapy. 71 This algorithm was not concise, but, interestingly, it suggested starting therapy regardless of DXA BMD in women older than 75 age with at least one clinical factor risk. On the other hand, for women with normal BMD, no further evaluation was suggested (Figure 2).
The National Comprehensive Cancer Network incorporated FRAX in its guidelines considering AIs treatment as secondary osteoporosis and suggested checking the box entitled “glucocorticoids” while using the algorithm. Treatment was recommended when FRAX 10-year fracture risk is >20% for major fracture or >3% for hip fracture, when T-score was <−2.0, or when T-score is <−1.5 with significant BMD loss due to cancer therapy. 72 The merit of these guidelines is the introduction of FRAX, however, as we have already discussed in this review, this useful tool was not designed to evaluate women receiving hormone adjuvant therapy. Furthermore, in these guidelines of 2013 denosumab appears among available therapies (Figure 3).
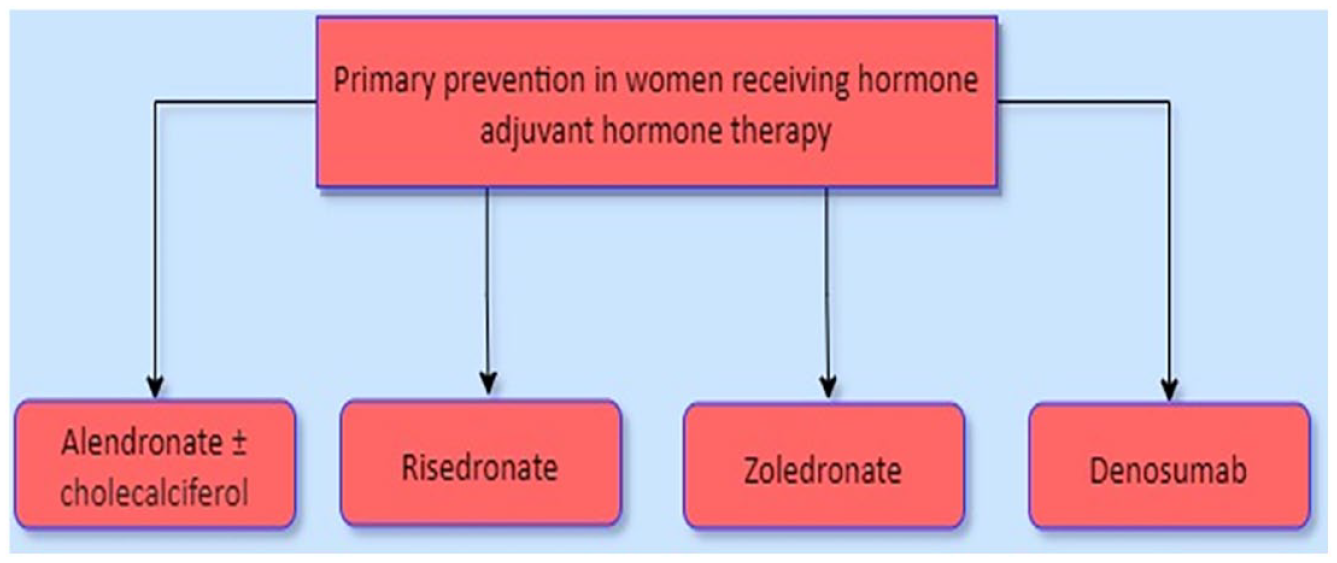
The Joint position statement of the International Osteoporosis Foundation, Cancer and Bone Society, European Calcified Tissue Society, International Expert Group for AIBL, European Society for Clinical and Economics Aspects of Osteoporosis, Osteoarthritis, and Musculoskeletal Diseases, International Menopause Society and International Society for Geriatric Oncology (2017) 40 recommended starting anti-osteoporotic therapy when T-score is <−2.0 or with ⩾ 2 risk factors (including T-score <−1.5). This consensus updated the previous 2008 practical guidance for the management of AI-associated bone loss 73 (Figure 4) and included denosumab and other bisphosphonates among treatment options (Figure 5).
The ASCO (2019) recommended starting treatment with bisphosphonates or denosumab in osteoporotic patients, in presence of risk factors for osteoporotic fractures, and when FRAX 10-year fracture risk is >20% for major fractures or >3% for hip fractures. 14 These guidelines are probably the easiest to read and to remember, however, women with normal bone density tend to be undertreated and might develop osteopenia/osteoporosis during treatment (Figure 6).
The ESMO guidelines (2020) agree with the 2017 Joint statement and suggest starting anti-osteoporotic treatment when T-score is lower than −2.0 or in the presence of 2 or more risk factors. 39 These guidelines, therefore, tend not to treat patients with mild osteopenia and normal bone density (Figure 7).
The Italian association of Medical Oncology (2021) strongly recommends carrying out a bone metabolism evaluation and considering anti-osteoporotic therapy during the entire course of AIs therapy for EBC. 74 The Italian National Health System fully reimburses both bisphosphonates and denosumab as first-line treatments in primary prevention in all patients receiving hormone adjuvant therapy for breast cancer or prostate cancer, regardless of their BMD T-scores at DXA 75 (Figure 8).

Consensus paper from the Belgian Bone Club (2007).
Guidance for the management of breast cancer treatment-induced bone loss: a consensus position statement from a UK Expert Group (2008).

Journal of the National Comprehensive Cancer Network (2013).

Practical guidance for the management of aromatase inhibitor-associated bone loss (2008).

Joint position statement of the IOF, CABS, ECTS, IEG, ESCEO, IMS, and SIOG (2017).

American Society of Clinical Oncology (ASCO) Clinical Practice Guideline (2019).

European Society for Medical Oncology (ESMO) guidelines (2020).
Nota 79 from AIFA (Italian medicines agency) (2015). 75
Limitations
This is a narrative review that summarizes the current international guidelines about the use of antiresorptive treatments during adjuvant endocrine therapy for ER + breast cancer and its goal is to offer a quick reference tool to clinicians to make therapeutic choices based on these guidelines. It is not a systematic review, and specific protocols about article selection were not followed, thus leading to potential selection bias. However, major clinical trials and their outcomes were summarized in detail, to offer comprehensive evidence to the reader.
Conclusion
Adjuvant endocrine therapy has dramatically improved cancer survival in women with EBC. Fragility fractures are an untoward side effect of this therapy, although many pharmacologic options are now available to contrast cancer treatment-induced bone loss. Bisphosphonates and denosumab should be discussed with the patient initiating adjuvant endocrine therapy for the first time, depending on age, BMD T-score, clinical risk factors for fractures, and patient preference. The local health system or insurance coverage of DXA and drug reimbursement should also be considered, given the usual 5–10-year treatment period of AIs. Uncertainties remain as to the duration of the pharmacologic therapy with bisphosphonates or denosumab, and if bisphosphonates holidays or less frequent dosing of zoledronic acid should be considered for a patient with low fracture risk with AIs prolonged for up to 10 years.
