Abstract
Background:
Postpartum preeclampsia has been implicated in increasing hospital re-admissions, maternal morbidity, and mortality worldwide. The knowledge of the risk factors of postpartum preeclampsia would be helpful in formulating strategies to aid in the prevention, early diagnosis, and timely treatment of this disorder. Thus, this study aimed to identify the risk factors associated with the development of new-onset postpartum preeclampsia and persistent postpartum preeclampsia in the Ghanaian setting.
Methods:
This case–control study was conducted at the Obstetrics and Gynecology units of Komfo Anokye Teaching Hospital and the Kumasi Regional Hospital, both located in the Ashanti Region of Ghana. A total of 65 postpartum preeclamptic women (33 new-onset postpartum preeclampsia and 32 persistent postpartum preeclampsia) and 65 normotensive postpartum mothers were recruited from 48 h to 6 weeks post-delivery. Questionnaires were administered to assess the socio-demographic, lifestyle, obstetric characteristics, and past medical history of the study participants.
Results:
Physical inactivity (p < 0.0001), infrequent antenatal visits (p < 0.0001), analgesic use (p < 0.0001), and cesarean delivery (p = 0.021) were significantly associated with both the new-onset postpartum preeclampsia and persistent postpartum preeclampsia. Contraceptive use was significantly associated with the development of new-onset postpartum preeclampsia (p < 0.0001) while women with low-birthweight babies are also at high risk of developing persistent postpartum preeclampsia (p < 0.0001).
Conclusion:
Physical inactivity, infrequent antenatal visits, analgesic use, contraceptive use, and cesarean delivery are major predisposing risk factors for the development of postpartum preeclampsia. Screening using these risk factors, close monitoring and follow-up observation of women after delivery would be beneficial in identifying and managing postpartum preeclampsia.
Keywords
Background
Preeclampsia is a multisystem disorder that is characterized by new-onset hypertension (HTN) with proteinuria or multisystem dysfunction occurring after the 20th week of gestation.1,2 This disorder complicates about 2%–8% of all pregnancies globally. 3 The prevalence of preeclampsia in Ghana is estimated to be between 6.55% and 7.03%.4,5 Preeclampsia is associated with both maternal and perinatal complications,2,6 and it is currently the leading cause of maternal mortality in Ghana. 7 The etiology of preeclampsia is unclear, hence, the difficulty in formulating strategies for effective prevention or management. For a long time, it has been thought that the delivery of the fetus and placenta resolves the symptoms and effects of preeclampsia. 8 However, the onset can be sudden in the postpartum period8,9 after the fetus and placenta have been delivered.
Postpartum preeclampsia, a condition that develops after the baby is delivered, is defined as a systolic blood pressure (SBP) ⩾ 140 mmHg and/or diastolic blood pressure (DBP) ⩾ 90 mmHg on at least two occasions, and presenting more than 48 h after delivery and before 6 weeks postpartum, with or without proteinuria.8–10 Postpartum preeclampsia can occur regardless of whether a woman had preeclampsia during pregnancy or not. It could result from an antepartum preeclampsia that persists after delivery of the fetus and placenta (known as persistent postpartum preeclampsia—PPE) or a preeclampsia that develops de novo after delivery of the fetus and placenta in a woman who was normotensive during pregnancy (known as new-onset postpartum preeclampsia—NOPPE). 9
Like antepartum preeclampsia, there is evidence implicating postpartum preeclampsia in increasing hospital re-admissions, maternal morbidity, and mortality.11–14 There is a paucity of data on the risk factors of postpartum preeclampsia in sub-Saharan Africa, a setting where the incidence of hypertensive disorders of pregnancy is high and also a leading cause of maternal mortality. The early identification of women at high risk of developing postpartum preeclampsia could favorably impact maternal morbidity and mortality through prompt and effective diagnosis and management of this disorder. This study, therefore, evaluated the risk factors of postpartum preeclampsia among women presenting with the disorder. The knowledge of the risk factors would be helpful in formulating new strategies aimed at prevention, early identification of at-risk women, diagnosis, and enhancing timely treatment of this disorder.
Methods
Study design and study setting
A hospital-based case–control study was conducted at the obstetrics and gynecology (O&G) units of the Komfo Anokye Teaching Hospital (KATH) and the Kumasi Regional Hospital (KRH). Both hospitals are located in Kumasi in the Ashanti Region of Ghana. Kumasi has an average population of 4,780,380 (Ghana Statistical Service, 2012). KATH is the second largest tertiary hospital in Ghana, and with a thousand bed capacity, it serves as a major referral center offering health services to several regions across Ghana. KRH is a principal care hospital in south-central part of Kumasi and offers its services to 56 communities, which constitute about 400,000 people. The combined annual deliveries for both hospitals during the study period was about 10, 000 deliveries. This was verified through the hospital database and annual reports.15,16
Ethical approval and consent to participate
The Committee on Human Research, Publications and Ethics (CHRPE) (CHRPE/AP/037/18), School of Medical Sciences, Kwame Nkrumah University of Science & Technology (KNUST), Research and Development Unit of the KATH and O&G unit of KRH approved the study protocol. Written informed consent in the form of a signature or fingerprint was also obtained from each participant prior to enrollment in this study.
Justification of sample size
The estimated minimum sample size for this case–control study was calculated as 63 for each case and control using the formula as described by Charan and Biswas. 17
n = sample size, R = the proportion of cases to control, which was chosen as 1 based on the expected rareness of cases, Zα/2 = 1.96, is the standard normal variate for level of significance at type I error of 5%= Z0.05/2 = Z0.025 = 1.96 (from the Z statistical table), Zβ = 0.84, standard normal variate for power or type 2 error. For the purpose of this study, 80% was used = Z0.20 = 0.842 (from the Z statistical table), d = 0.25, expected mean difference between case and control, 0.25 was used for the purpose of this study, and standard deviation (SD) = 0.5, the SD. In total, we recruited 130 study participants.
Study participants
Using a simple random sampling technique, a total of 130 participants, comprising 65 cases (women with postpartum preeclampsia) and 65 controls (postpartum women who were normotensive before, during, and after pregnancy) under clinical management at the O&G department of KATH and KRH, were consecutively recruited into this study. For this case–control study, women who had elevated blood pressure within 48 h to 6 weeks after delivery were recruited as cases, and normotensive postpartum women were recruited as controls. The cases were identified and recruited by a qualified obstetrician/gynecologist using the American College of Obstetricians and Gynecologists 3 Practice Bulletin. The cases were classified into new-onset postpartum preeclampsia (NOPPE) and persistent postpartum preeclampsia (PPE). Age-matched healthy women who were normotensive before, during, and after pregnancy were enrolled as controls.
Definition of terms
NOPPE refers to women who had elevated blood pressure within 48 h to 6 weeks after delivery. PPE refers to women who had antecedent intrapartum elevated blood pressure and remained so after birth. Normotensive postpartum women refer to women who were normotensive throughout pregnancy and after delivery.
Eligibility criteria
All recruited participants were women who had delivered within the study period, irrespective of their parity, but developed HTN between 48 h and 6 weeks postpartum. Also, women who were normotensive before, during, and after delivery and had no medical history of diabetes, HIV/AIDS, and renal failure were included as controls. Women with a history of chronic HTN and those who developed HTN before 48 h and after 6 weeks post-delivery were excluded. Women with a history of diabetes, HIV/AIDS, renal failure, and those who refused to give informed written consent were also excluded from this study.
Questionnaire administration
A self-constructed, well-structured, closed-ended questionnaire was developed by a review of related literature.8,18,19 The developed questionnaire was pre-tested and administered to each participant to obtain information on socio-demographic data; maternal lifestyle factors such as smoking and alcohol consumption before and during pregnancy, contraceptive use (defined as use of combined oral pills, progesterone only pill, injectable contraceptives and barrier methods of birth control prior to conception), pain medication use, change of partner, physical activity (defined as at most 30 min of moderate-intensity aerobic activity daily), educational status, antenatal attendance, family history of HTN, residence, ethnicity, marital, and occupational status. A complete present and past obstetric history such as personal and family history of preeclampsia or eclampsia, systolic and DBP, infant birth weight, and pregnancy characteristics (gravidity, singleton/multiple gestation, gestational age at diagnosis of preeclampsia or eclampsia and at delivery, mode of delivery, antihypertensive treatment) were also obtained. All the information obtained from each subject was reviewed through the hospital database, see supplemental material.
Statistical analysis
The data obtained was entered into Microsoft Excel. Statistical analysis was performed using GraphPad Prism version 5.0 for Windows (GraphPad Software Inc., Los Angeles). The Shapiro-Wilk normality test was used to test the data for a normal distribution. Categorical variables were compared using chi-square and Fischer’s exact test. For data that assumed Gaussian distribution, comparison between two groups was performed using unpaired t-test while one-way analysis of variance (ANOVA) was used to compare the means of more than two groups. Logistic regression was used for univariable and multivariable analysis, multivariable logistic regression was performed to measure the degree at which more than one independent variable (predictors) and more than one dependent variable (responses) are linearly related. Continuous data were reported as mean ± SD while categorical data were reported as proportions and percentages. Statistical significance level was considered at p < 0.05.
Results
Demographic characteristics of the study participants are shown in Table 1. The cases, grouped into PPE and NOPPE, showed that the PPE group presented with the highest mean age (31.56 ± 8.06) followed by the NOPPE group (29.70 ± 6.62), with the controls presenting the lowest mean age (28.77 ± 6.37). However, there was no statistically significant difference in mean ages across the study groups (p > 0.05). Participants were comparable in terms of demographic characteristics (p value > 0.05; Table 1).
Demographic characteristics of the study participants.
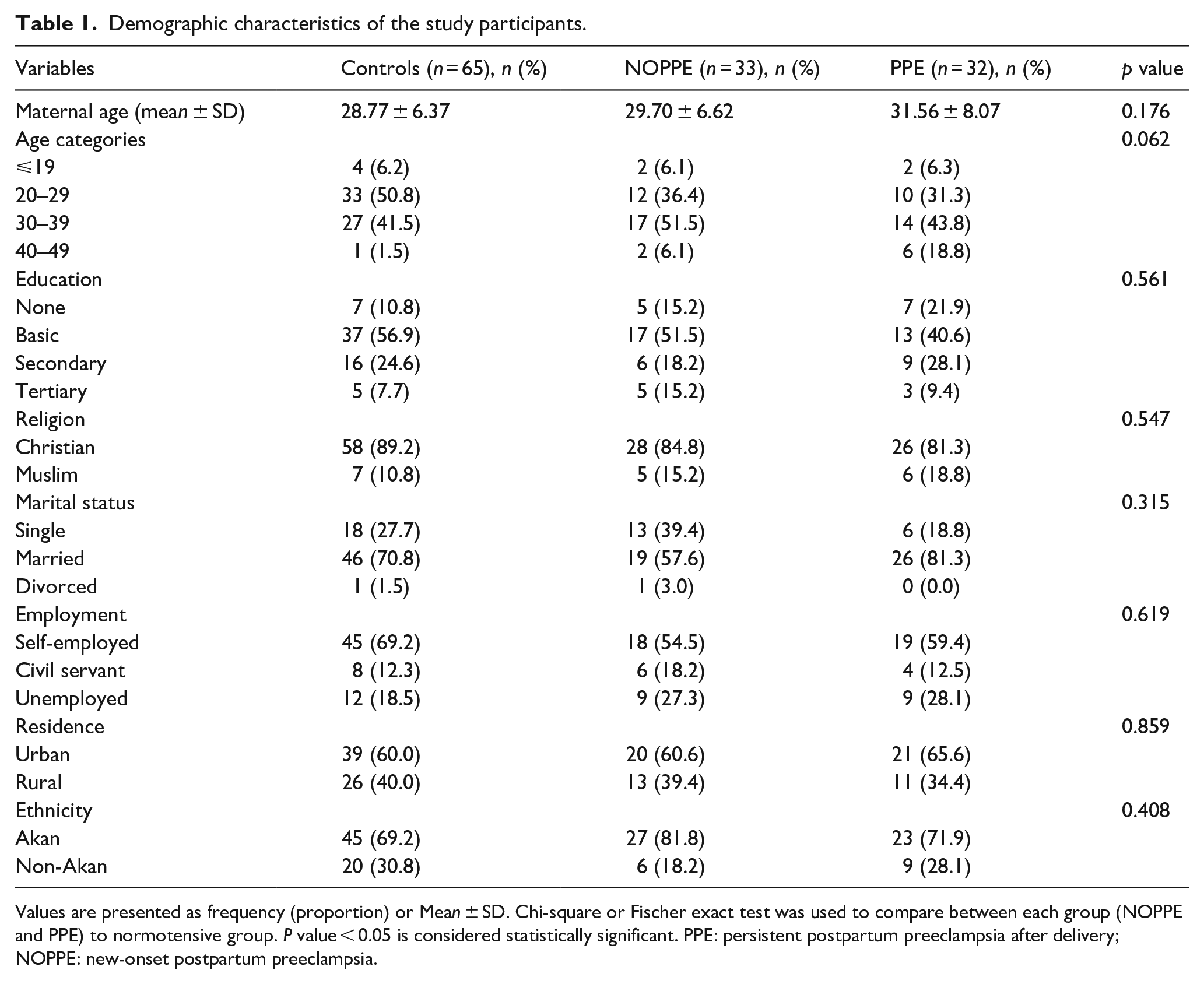
Values are presented as frequency (proportion) or Mean ± SD. Chi-square or Fischer exact test was used to compare between each group (NOPPE and PPE) to normotensive group. P value < 0.05 is considered statistically significant. PPE: persistent postpartum preeclampsia after delivery; NOPPE: new-onset postpartum preeclampsia.
The lifestyle characteristics of the study participants are shown in Table 2. There was a significant difference in the physical activity levels between the NOPPE and the PPE groups on comparison with the controls (p < 0.0001). Irregular physical activity and no exercise were significantly high among NOPPE (51.5% and 42.4%) and PPE (50.0% and 43.8%), respectively. However, the majority of controls were engaged in regular activity (69.2%). No statistically significant difference was observed among the study participants in terms of alcohol use, smoking status, parity, and change of sexual partner. Compared to the normotensive controls, family history of HTN was significantly higher among NOPPE (63.6%) and PPE (50.0%) groups (p < 0.05; Table 2).
Lifestyle characteristics of the study participants.
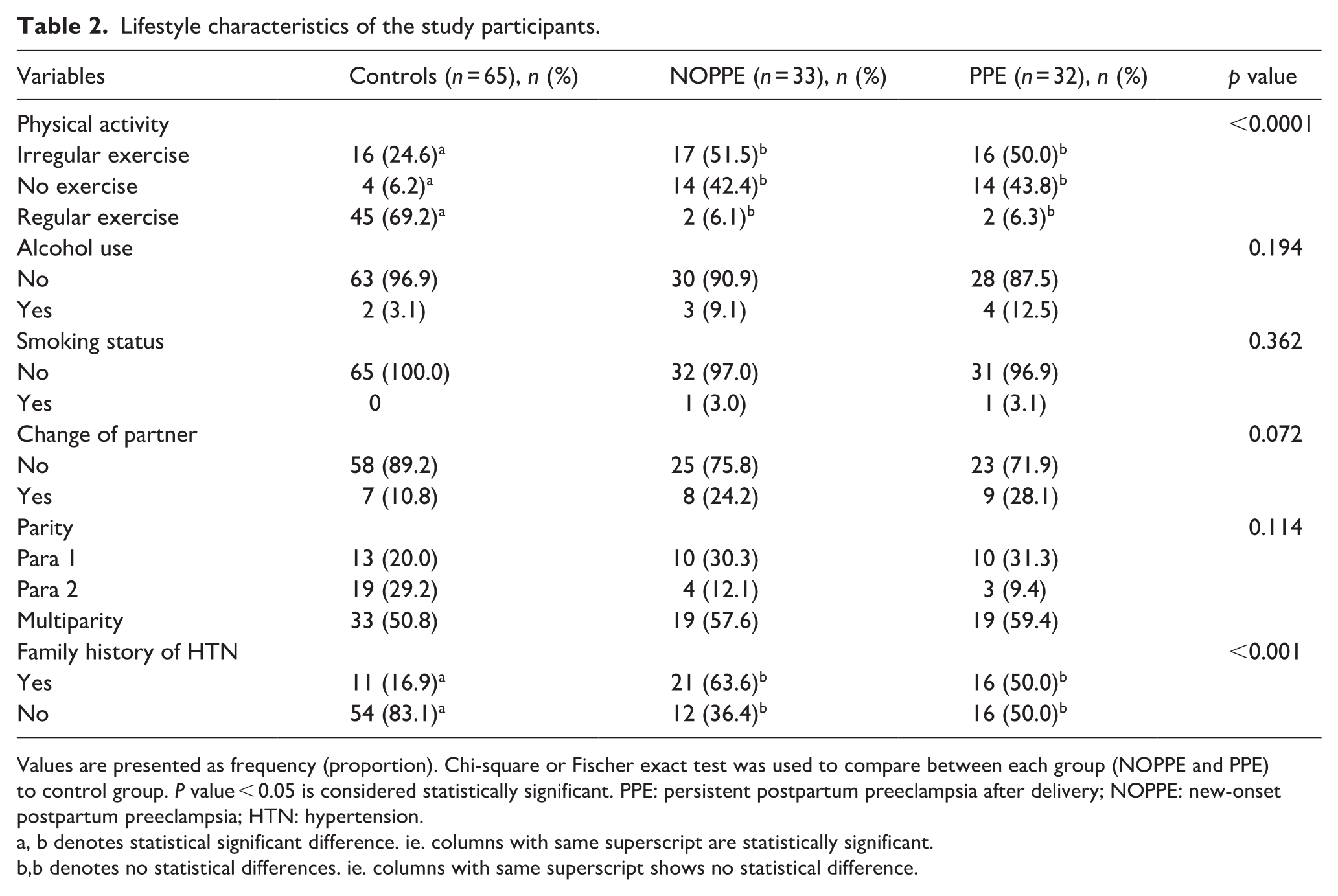
Values are presented as frequency (proportion). Chi-square or Fischer exact test was used to compare between each group (NOPPE and PPE) to control group. P value < 0.05 is considered statistically significant. PPE: persistent postpartum preeclampsia after delivery; NOPPE: new-onset postpartum preeclampsia; HTN: hypertension.
a, b denotes statistical significant difference. ie. columns with same superscript are statistically significant.
b,b denotes no statistical differences. ie. columns with same superscript shows no statistical difference.
The gestational characteristics of the study participants are summarized in Table 3. The proportions of NOPPE (39.4%) and PPE (40.6%) subjects that visited antenatal clinics less than the required number were significantly higher (p < 0.0001) compared to the control group (4.6%). Preterm delivery was higher among NOPPE (27.3%) and PPE (34.4%) compared to controls (0.0%). A larger proportion of NOPPE (45.5%) and PPE (50.0%) had cesarean delivery when compared to the control group (12.3%). Analgesic use (p < 0.0001), contraceptive use (p < 0.0001), and low birth weight (p < 0.0001) were more common among participants with NOPPE and PPE than normotensives, as shown in Table 3.
Gestational characteristics of the study participants.

Values are presented as frequency (proportion). Chi-square or Fischer exact test was used to compare between each groups (NOPPE and PPE) to control group. P value < 0.05 is considered statistically significant. PPE: persistent preeclampsia after delivery; NOPPE: new-onset postpartum preeclampsia. HTN: hypertension.
a, b denotes statistical significant difference. ie. columns with same superscript are statistically significant.
b,b denotes no statistical differences. ie. columns with same superscript shows no statistical difference.
Table 4 shows the regression analysis of factors associated with NOPPE and PPE, adjusting for family history of HTN, physical activity, and maternal age. Infrequent antenatal visits were significantly associated with the increased likelihood of NOPPE in both the univariable (odds ratio (OR) = 13.43 (3.47–51.96), p < 0.0001) and multivariable (OR = 13.16 (2.28–5.90), p = 0.004) analysis. In the univariable model, cesarean delivery increased the likelihood of NOPPE by 5.94 times (p = 0.001). However, its effect was dismissed in the multivariable model (p > 0.05). Analgesic use and contraceptive use were significantly associated with increased odds of developing NOPPE in both the univariable and multivariable models (p < 0.05). Women with low-birthweight babies were at an increased risk of developing PPE in both the univariable and multivariable models (p < 0.05).
Univariable and multivariable regression analysis of gestational factors predictive of NOPPE and PPE.
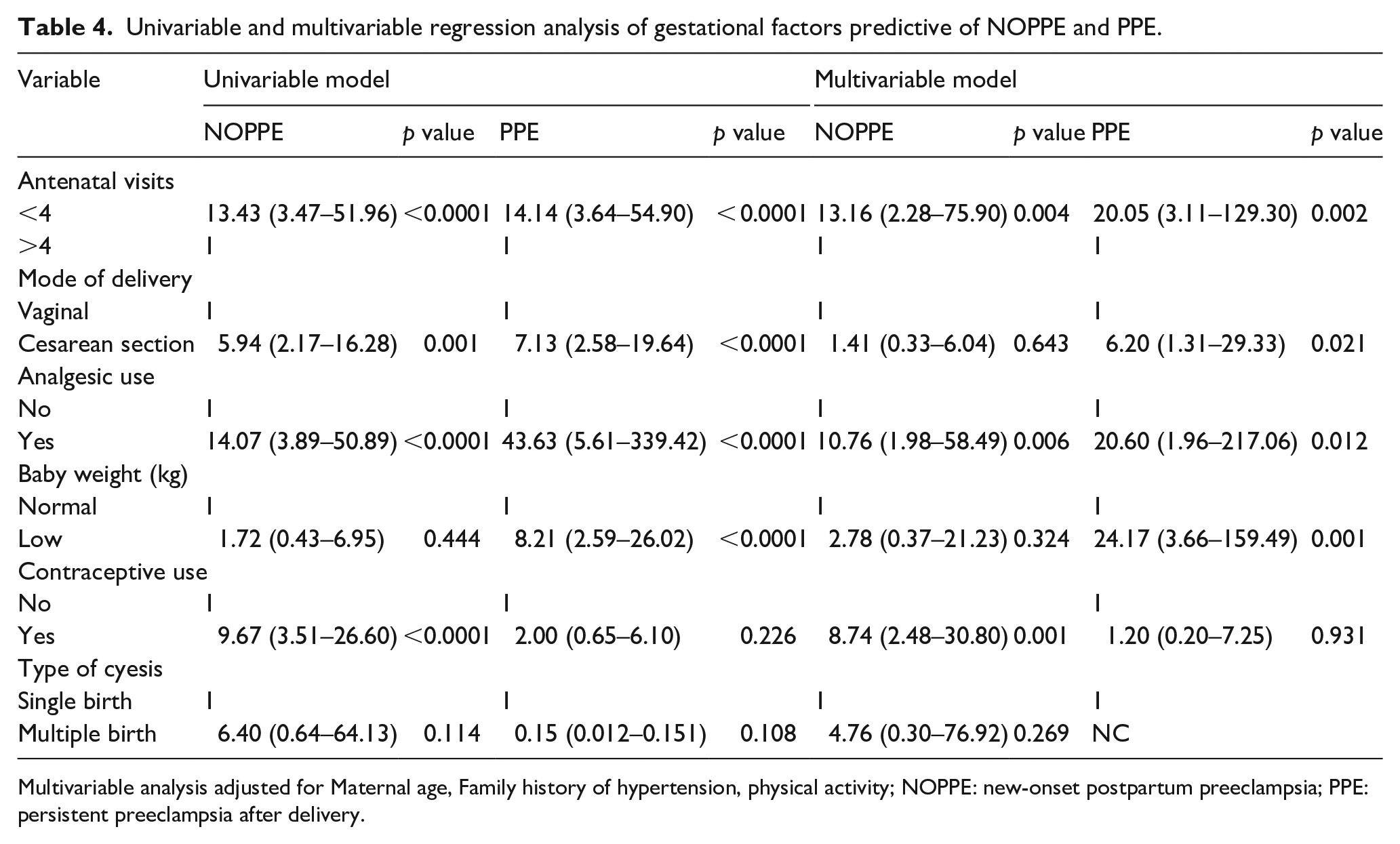
Multivariable analysis adjusted for Maternal age, Family history of hypertension, physical activity; NOPPE: new-onset postpartum preeclampsia; PPE: persistent preeclampsia after delivery.
Discussion
In this study, we observed a significant difference in gestational characteristics such as analgesic use, contraceptive use, antenatal visit, term of delivery, mode of delivery, and baby’s birth weight between the control group and women who later developed postpartum preeclampsia. Our findings identified physical inactivity, infrequent antenatal visits, analgesic use, contraceptive use, and cesarean delivery as the major risk factors for the development of postpartum preeclampsia.
This study observed that women who did not exercise at all and those who did not engage in regular physical exercise during pregnancy were at a high risk of developing both NOPPE and PPE (Table 2). Similar observations were made by Spracklen et al. 20 where they reported physical inactivity and lack of exercise as risk factors for developing preeclampsia. On the contrary, a prospective cohort study conducted among Hispanic women 21 did not observe statistically significant associations between physical activity during pregnancy and the risk of developing preeclampsia. Variation in findings may be due to differences in study design and the tools used in assessing physical activities in the various studies. That notwithstanding, physical inactivity is a modifiable cardiovascular risk factor21–23 that can be employed to minimize the effects of postpartum preeclampsia. Furthermore, in agreement with other findings,24–26 our study revealed that a family history of HTN is a risk factor for developing both NOPPE and PPE.
We identified irregular antenatal care visits as a risk factor for developing postpartum preeclampsia. Similar observations have been made in other studies26,27 where the authors identified irregular antenatal care visits as a risk factor for developing preeclampsia and other pregnancy complications. The World Health Organization (WHO) recommends antenatal care visits four or more times for every pregnant woman. However, the antenatal care visits of women who developed postpartum preeclampsia in this study were less than the recommended number of visits when compared to the controls (Table 3). Regular antenatal care visits and postnatal check-ups are necessary in the identification of high-risk pregnancies through regular monitoring during the gestational and postpartum periods, respectively.27,28 We have recently reported a high prevalence of postpartum preeclampsia in the Ghanaian population (Fondjo et al., 2022). This supports the notion that delivery of the placenta does not nullify the development of preeclampsia. Thus, attending antenatal care regularly affords healthcare providers the opportunity to identify and manage women at high risk of developing postpartum preeclampsia. Moreover, it is possible that women who presented with postpartum preeclampsia in this study might have failed to adhere to prescribed medications, which otherwise would have been noticed during regular antenatal care visits.
A combination of genetic factors and life experiences can contribute to an individual’s pain threshold. Often, inadequate family/spousal support, distress, anxiety and the positive or negative labor stories heard can increase pain and pain perception; as such, the treatment with and use of analgesics after delivery is not unexpected. Interestingly, the use of analgesics was high in the women who developed postpartum preeclampsia in comparison to the control group (Table 3). Our findings show that the use of analgesics by pregnant women predisposes them to developing NOPPE and PPE. While most of our case participants were on suppository non-steroidal anti-inflammatory drugs (NSAIDs), the exact mechanism by which NSAIDs induce HTN is unclear. However, it has been suggested that NSAIDs promote blood pressure elevation by inhibiting sodium and water loss in the postpartum period as well as through the suppression of prostaglandin synthesis. 29
Consistent with these studies,30,31 our study showed that undergoing a caesarian section was a risk factor for developing postpartum preeclampsia. However, the effects of cesarean delivery in the development of NOPPE were dismissed in a multivariate analysis. This suggests that in a group of risk factors predisposing a woman to developing NOPPE, undergoing a cesarean delivery has a redundant effect. Furthermore, both the univariate and multivariate models in this study showed that women with low-birthweight babies are at high risk of developing PPE (Table 4). It is well established that preeclampsia relates to adverse pregnancy outcomes such as having low-birthweight infants.6,32 Considering these findings, it is imperative that healthcare providers educate and regularly monitor pregnant women, especially preeclamptic women with low-birthweight infants, so as to enable early detection of postpartum PE for timely management.
In addition, this study has demonstrated that the use of contraceptives by women prior to conception is a predisposing factor to developing NOPPE (Table 4). Epidemiological studies have implicated the use of contraceptives in the pathogenesis of cardiovascular diseases such as preeclampsia.33–35 Our observation agrees with the findings of the study conducted by Asare et al. 33 which revealed that the use of contraceptives is associated with about a thirty-fold increase in the odds of developing preeclampsia. With this increasing evidence of contraceptive use in the development of preeclampsia, especially in the Ghanaian setting, larger studies are warranted to confirm these findings and provide evidence-based protocols for prevention of preeclampsia in Ghana and sub-Saharan Africa at large.
This study is not without limitations. The use of self-reporting by participants to obtain some of our data is a limitation of this study. Also, data were obtained within the Ashanti Region of Ghana, and thus the findings may not be generalized to other populations. That notwithstanding, our study, represents very robust data and an innovative study conducted to evaluate the risk factors for developing postpartum preeclampsia among Ghanaian women.
Conclusion
In summary, physical inactivity, infrequent antenatal visits, analgesic use, contraceptive use, and cesarean delivery are major predisposing risk factors for the development of postpartum preeclampsia. Intensified education, close monitoring, and follow-up observation of women after delivery would be beneficial in identifying and managing women at risk of developing postpartum preeclampsia.
Supplemental Material
sj-docx-1-whe-10.1177_17455057221109362 – Supplemental material for Risk factors for the development of new-onset and persistent postpartum preeclampsia: A case–control study in Ghana
Supplemental material, sj-docx-1-whe-10.1177_17455057221109362 for Risk factors for the development of new-onset and persistent postpartum preeclampsia: A case–control study in Ghana by Linda Ahenkorah Fondjo, Beatrice Amoah, Worlanyo Tashie and John Jude Annan in Women’s Health
Footnotes
Acknowledgements
The authors acknowledge and are grateful to all the pregnant women who voluntarily participated in the study. Special thanks to the Late Prof. CA Turpin for all for his expert advice.
Author contribution(s)
Availability of data and materials
The data sets used and/or analyzed during the current study are within the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the KNUST Research Fund (KReF).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
