Abstract
Keywords
In their prospective cohort study of apparently healthy women, Kurth and colleagues add to the growing body of literature concerning the relationship between chronic kidney disease (CKD) and cardiovascular disease (CVD) [1]. However, this and other studies that examine the relationship between mild-to-moderate kidney disease and heart disease have varying results, and indicate the need for further studies.
The relationship is well catagorized between endstage renal disease and heart disease: the National Kidney Foundation (NKF) reported in its 2002 Task Force that CVD mortality in dialysis patients is 10–30% higher than in the general population [2]. The 2006 prevalence of kidney failure was greater than 506,000 people in the USA, and is projected to rise to more than 780,000 people in 2010. Kidney failure is an automatic criteria for Medicare coverage, and resulted in the expenditure of US$ 23 billion in 2006 for kidney failure with an additional US $ 10 billion coming from private insurers [3]. This is a clear call for action on a known risk factor in a specific population.
The Task Force also states that all patients with CKD should be considered ‘highest risk’ for CVD [2]. This statement has sweeping implications for the general population as the prevalence of patients with renal insufficiency and renal failure increases. Notably, a reduction of glomerular filtration rate (GFR) with aging is expected (i.e., after 20–30 years of age the normal mean value of GFR declines by approximately 1 ml/min/1.73 m2 per year). The NKF Task Force delineates five stages of CKD. Stage 1 includes patients with a normal or increased estimated GFR with other evidence of kidney damage (pathologic abnormalities or abnormalities in blood, urine or imaging) for at least 3 months. Stage 2 includes patients with a GFR from 60 to 89 ml/min/1.73 m2 with other evidence of kidney damage (without evidence of kidney damage they can be referred to as having ‘decreased GFR’). Stage 3 represents patients with a GFR from 30 to 59 ml/min/1.73 m2 for at least 3 months, who are considered to have CKD regardless of the presence of other signs of kidney damage. The GFR of 60 ml/min/1.73 m2 is a cut-off value for CKD because it signifies a loss of more than half of the function of normal kidneys and, as will be discussed, is associated with other ramifications of decreased renal function. Stage 4 represents those patients with a GFR from 15 to 29 ml/min/1.73 m2. Finally, stage 5 represents patients who have a GFR less than 15 ml/min/1.73 m2 or who require renal replacement therapy (RRT).
The relationship between CKD and CVD is complex and presumably synergistic (Figure 1) [3]. On one level, the well-described ‘traditional’ risk factors for CVD of high blood pressure, diabetes mellitus, age and tobacco are also risk factors for CKD. Therefore, CKD in these patients may merely be a marker for the presence of these risk factors or a result of localized vascular damage to the kidneys. However, the role of nontraditional or ‘novel’ cardiovascular risk factors emerging from the milieu of Stage 3 CKD and beyond, which is marked by disorders of bone metabolism, anemia, oxidative stress, inflammation and dyslipidemia, is currently being investigated, but is not yet linked as causal [4]. Furthermore, CVD in patients with CKD may be a unique vascular phenotype as it is characterized by atherosclerotic lesions that are more often calcified and with increased media thickness compared with the fibroatheromatous plaques of the non-CKD population [5].

Cardiovascular risk factors in patients with chronic kidney disease.
While not all studies find an independent relationship between CVD and CKD [6], systematic review suggests that there is an increase in cardiovascular risk as GFR drops below 60 ml/min/1.73 m2 (when patients are considered to have CKD stage 3, based solely on GFR) (Table 1) [7]. These studies included patients with baseline CVD [8–10] and linear risk associations were also evaluated as GFR decreased below 60ml/min/1.73m2 (i.e., in patients with moderate-to-severe CKD) [11]. Interestingly, analysis of Framingham data, where participants were over 50% female, suggests a difference between men and women. The data did not show any association between mild renal insufficiency and CVD in women after adjustment for age and known risk factors. However, for men there was a significant association between mild renal insufficiency and all-cause mortality [12]. The challenge now is to better quantify the cardiovascular risk for patients with declining renal function (mild CKD) but who do not have baseline CVD or high cardiovascular risk, and to further explore the presence of gender-based differences. Currently, only one study other than that of Kurth et al. looks specifically at the risk of CVD in women with CKD. In their study of Italian postmenopausal women (mean age of 52.7 years, 31% with metabolic syndrome, followed for a mean of 73 months) Perticone et al. found that the presence of CKD as defined as a GFR less than 60 ml/min/1.73 m2 is associated with increased risk of major adverse cardiac events and death independent of known traditional risk factors, compared with women with a GFR higher than 60 ml/min/1.73 m2 [13].
Risk of cardiovascular end points in patients with chronic kidney disease in community based cohorts.
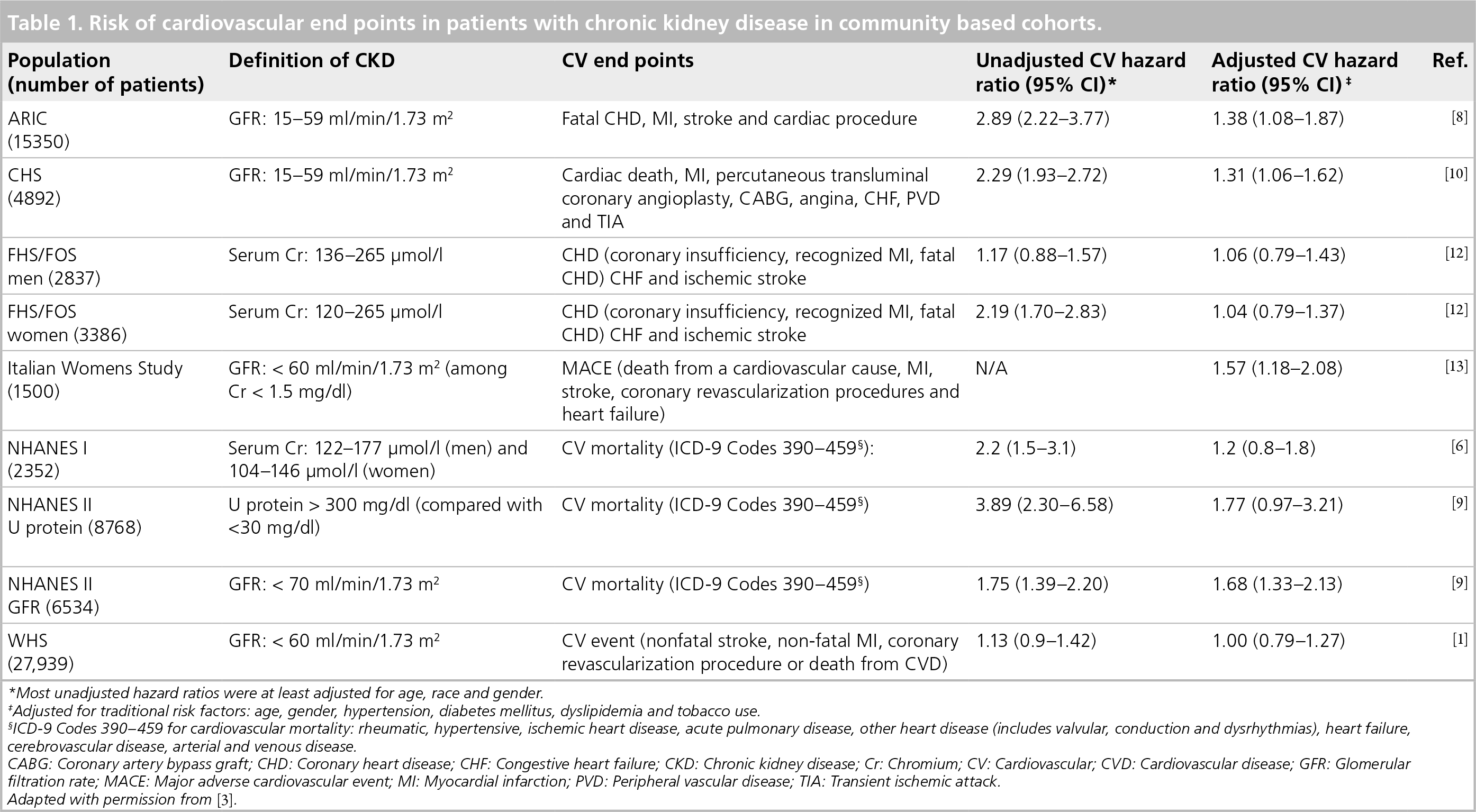
Most unadjusted hazard ratios were at least adjusted for age, race and gender.
Adjusted for traditional risk factors: age, gender, hypertension, diabetes mellitus, dyslipidemia and tobacco use.
ICD-9 Codes 390–459 for cardiovascular mortality: rheumatic, hypertensive, ischemic heart disease, acute pulmonary disease, other heart disease (includes valvular, conduction and dysrhythmias), heart failure, cerebrovascular disease, arterial and venous disease.
CABG: Coronary artery bypass graft; CHD: Coronary heart disease; CHF: Congestive heart failure; CKD: Chronic kidney disease; Cr: Chromium; CV: Cardiovascular; CVD: Cardiovascular disease; GFR: Glomerular filtration rate; MACE: Major adverse cardiovascular event; MI: Myocardial infarction; PVD: Peripheral vascular disease; TIA: Transient ischemic attack.
Adapted with permission from [3].
Results
The Women's Health Study from which Kurth and colleagues obtained their data is a large prospective cohort study of mostly Caucasian (94%) female health professionals designed to test the risks and benefits of low-dose aspirin and vitamin E for the primary prevention of CVD and cancer. This group of 39,876 US women was aged 45 years or older (baseline mean age: 55 years), was free of CVD, cancer or other major illness at entry to the study in 1992–1995 and was followed for 12 years. Kurth et al. used information from the 27,939 participants who had provided blood samples at entry to evaluate the association of kidney function with CVD and mortality among this healthy population. Blood samples were collected prior to randomization and creatinine was measured by a rate blanked method based on the Jaffe reaction. GFR was estimated by the abbreviated prediction equation developed from the Modification of Diet in Renal Disease (MDRD) study (which takes age, gender and race into account) [14]. The participants were categorized based on their GFR as less than 60, 60–74.9, 75–84 and higher than 90 ml/min/1.72 m2. Other measures of renal dysfunction (e.g., microalbuminuria) were not collected. The primary end point was the occurrence of any first cardiovascular event: nonfatal stroke, nonfatal myocardial infarction (MI), coronary revascularization procedure or death from CVD. The association between renal function and any first individual cardiovascular event, all-cause mortality and noncardiovascular mortality was also evaluated.
Of the participants, 14,979 (53.6%) had a GFR of 90 ml/min/1.73 m2 or higher, 8073 (28.9%) had a GFR of 75–89 ml/min/1.73 m2, 3572 (12.8%) had a GFR of 60–74 ml/min/1.73 m2 and only 1324 (4.7%) had a GFR less than 60 ml/min/1.73 m2. Women with a GFR less than 60 ml/min/1.73 m2 compared with women with a GFR higher than 90 ml/min/1.73 m2 were older, more likely to have elevated blood pressure and be using antihypertensive drugs, have quit smoking, be more physically active and have higher cholesterol and C-reactive protein levels. The investigators found no association between categories of GFR and risk of any cardiovascular event or all-cause mortality prior to or after multivariable adjustment. The multivariable adjusted hazard ratios for any first CVD event were 0.95 (95% CI: 0.83–1.08), 0.84 (95% CI: 0.7–1) and 1.00 (95% CI: 0.79-1.27) for women with a GFR of 75–89.9, 60–74.9 and less than 60 ml/min/1.73 m2, respectively, compared with women with a GFR of 90 ml/min/1.73m2 or higher. The all-cause mortality hazard ratios were similarly nonsignificant: 0.93 (95% CI: 0.79-1.09), 1.03 (95% CI: 0.85–1.26) and 1.09 (95% CI: 0.83–1.45), respectively. There was also no association found between GFR and MI, coronary revascularization procedures, stroke or noncardiovascular death. However, the investigators did find a significantly increased risk of cardiovascular death when comparing the women with a GFR less than 60 ml/min/1.73 m2 to those with a GFR of 90 ml/min/1.73m2 or higher where there was a multivariable adjusted hazard ratio of 1.68 (95% CI: 1.02–2.79). Further categorization of the low GFR group into women with a GFR less than 40 ml/min/1.73 m2 revealed an increased risk of all-cause mortality (HR: 2.26; 95% CI: 1.15–4.41) that was driven by increased death from CVD (HR: 3.06; 95% CI: 0.94-9.94) compared with women with a GFR of 90 ml/min/1.73m2 or higher. Analysis of renal function as measured by serum creatinine or Cockcroft-Gault yielded similar results. The lack of association between GFR and CVD was not modified by age, body mass index (BMI), high blood pressure, smoking status or total cholesterol. The lack of association between GFR and allcause mortality was not modified by age, BMI, smoking status or total cholesterol, although high blood pressure showed significant effect modification (p = 0.02) with all-cause mortality, implying increased risk of death from any cause for women with high blood pressure (HR: 1.31; 0.92-1.86).
Significance
Kurth et al. add evidence to the growing debate on the complex relationship between renal function and CVD. They conclude that there is no linear relationship as renal function declines gradually from the normal range to 60 ml/min/1.73m2, but they support current evidence that suggests that when the GFR falls below 60 ml/min/1.73 m2 (i.e., women who have CKD) there is a higher risk of death from CVD even in women who are otherwise at relatively low risk. However, in his editorial on this study, Weiner points out that this conclusion may be less robust because participants with the lowest creatinine (and thus the highest GFR) actually have a higher risk of adverse outcomes than the women with GFRs of 60–80 ml/min/1.73 m2 [15]. This J-shaped curve would not be anticipated if the participants with the lowest creatinine are presumed to be the healthiest. Weiner postulates that sick participants with decreased muscle mass could be in this high GFR group, making this group heterogeneous and impacting their cardiovascular outcomes, which could lead to underestimation of a trend between GFR and CVD. His theory is supported by surprising covariate imbalances – where the group with the highest GFR has the least physical activity and highest percentage of diabetes at baseline. Additionally, variables that were not measured in this study but may have affected the outcome include a positive family history for CVD, presence of menopause, and baseline medications (e.g., whether the enrollees were taking β-blockers or statins).
Contextualizing these findings in the larger debate is difficult since this study and the others mentioned above all have inherent differences and limitations. First and foremost, these studies are deriving an estimated GFR based on one measurement of serum creatinine. As investigators in this field are well aware, more accurate GFR calculation is not feasible in cohort studies of this magnitude, but unfortunately creatinine is not a perfect surrogate as there is significant creatinine variation by muscle mass and within and between laboratories. Given that a slight variation in creatinine measurement can cause a significant change in GFR (e.g., using the MDRD equation for GFR a change in creatinine from 1 to 1.1 can result in a 10% change in GFR from 60 to 54 ml/min/1.73 m2), this is not a trivial matter [16]. Furthermore, according to the NKF, patients should only be considered to have CKD if they have a GFR less than 60 ml/min/1.73 m2 for at least 3 months or if they have other evidence of kidney dysfunction. These criteria are not met in any of the aforementioned studies (only the NHANES II analysis includes urinary markers of dysfunction) and the investigators can only presume that by enrolling patients in the outpatient setting, none of them have acute kidney injury. In fact, it is inappropriate to definitively categorize a participant with a one-time GFR of 80 ml/min/1.73 m2 as having ‘mild CKD’ as opposed to ‘decreased GFR’ without further evidence of dysfunction. One might wonder if the present study, where the participants are relatively young and therefore less likely to have a decreased GFR secondary to aging, may be the most representative of the relationship between decreased GFR (that might be due to a pathologic process) and CVD. Since the bulk of the prior literature does not involve other markers of renal dysfunction, more recent attempts are being made to more appropriately classify renal insufficiency and to identify a specific marker that confers the additional risk when GFR is less than 60 ml/min/1.73 m2. Interestingly, analysis of the Framingham Offspring Cohort suggests that only 28% of patients with a GFR less than 59 ml/min/1.73 m2 (or stage 3 CKD) had microalbuminuria (MA) [17]. Not surprisingly, those with both reduced GFR and MA had a higher risk of all-cause and combined cardiovascular mortality than those with neither. Those with only reduced GFR and no MA were at higher risk for all-cause mortality, which those with normal GFR and presence of MA were not. This suggests that to only risk-stratify based on GFR is to miss other important aspects or markers of the interplay between renal and cardiovascular disease.
The very fact that the WHS study involves younger, healthy women begs the question – is this population at such a baseline low risk for CVD that it is too early to see meaningful results? The 12-year cardiac event rate of less than 2% calculated in the study might not be sufficient to capture true trends regarding renal insufficiency. Very few participants actually had a GFR less than 60 ml/min/1.73 m2 (1315 women – or 4.7% of enrollees), and the fact that trends did emerge is impressive, but the small numbers may potentially limit interpretation.
Kurth's study adds to the evidence that, at least in women without pre-existing CVD, a gradually declining GFR is not a risk factor for CVD per se. However, if GFR deteriorates to less than 60 ml/min/1.73 m2 (i.e., the woman has CKD based on GFR criteria with ensuing metabolic abnormalities), the study supports the theory that CKD has a causal role in cardiovascular mortality, which is not limited to men.
Future perspective
Further investigation must be carried out with more precise assessment of renal function and perhaps targeted at the acuity of decline of renal function in patients without pre-existing CVD to better quantify the interplay between cardiovascular risk and mild renal insufficiency. We await the results of the Chronic Renal Insufficiency Cohort study that utilizes radiolabeled iothalamate to measure the GFR of a third of the participants [18]. Trials comparing men and women will elucidate any sex-specific differences. As the categorization of the relationship between CVD and mild-to-moderate CKD in otherwise healthy populations improves, trials will start to examine the role for risk-factor reduction in this population. More aggressive risk reduction in this population, otherwise considered low risk, could change guidelines and improve population-wide cardiovascular health.
Executive summary
Chronic kidney disease is considered to be a risk factor for cardiovascular disease, likely due to a combination of traditional risk factors and novel risk factors.
The extent to which a gradual, natural decline in glomerular filtration rate (GFR) correlates with cardiovascular risk is not well known. Most studies suggest an increase in cardiovascular risk when the GFR drops below 60 ml/min/1.73 m2.
Analysis of data from the Women's Health Study supports a significant increase in cardiovascular mortality in otherwise healthy women when their GFR falls below 60 ml/min/1.73 m2; an increased risk of cardiovascular events was not seen in women with a GFR higher than 60 ml/min/1.73 m2.
More precise measurements of real-time renal insufficiency are needed to appropriately delineate the relationship of worsening renal function and cardiovascular disease.
Footnotes
Valentin Fuster serves as the chair of the HRP study with BG Medicine. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
