Abstract
Background:
Clinically, chronic pain and depression often coexist in multiple diseases and reciprocally reinforce each other, which greatly escalates the difficulty of treatment. The alteration in neural networks underlying the chronic pain/depression comorbidity remains unclear.
Methods:
Spared nerve injury (SNI) was performed to establish a rat model of comorbidity of chronic pain and depression. Multimodal fMRI was utilized to acquire neuroimaging data of the rat brain, with BOLD and DTI data used to analyze adaptive changes in brain regions and networks associated with comorbidity.
Results:
By integrating static and dynamic ALFF analyses with DTI parameters, six key brain regions (OFR_L, CA1_L, CA3_L, PL_L, Stri_L, and Stri_R) were identified. These regions showed significant associations with both pain-related and depressive-like behaviors and formed a comorbidity-related network centered on the prefrontal cortex, hippocampus, striatum, and primary sensory cortex. Further analyses indicated that the relationship between fMRI features and depressive-like behavior was largely mediated by pain-related processes.
Conclusions:
This study reveals network-level neural alterations underlying chronic pain-depression comorbidity and supports a pain-mediated pathway linking brain activity to affective disturbances. These findings provide new insights into the neural mechanisms of this comorbidity and highlight the importance of integrated pain-depression interactions.

Keywords
Introduction
Chronic pain frequently co-occurs with depression in clinical settings, forming a bidirectional and mutually reinforcing relationship that markedly complicates treatment.1,2 Understanding the mechanisms underlying this comorbidity is therefore of critical importance.
With advancing research, brain dysfunction is increasingly recognized as a key mechanism underlying the comorbidity of chronic pain and depression.3,4 Emerging evidence from neural circuit-based studies suggests that specific brain regions and circuits contribute to its development and progression. For example, dysregulation of dopamine function in the ventral tegmental area promotes comorbidity of chronic pain with mood disorders. 5 The medial prefrontal cortex is also considered as a central hub for mental comorbidities associated with chronic pain.6,7 Additionally, a specific serotonergic neural circuit from the dorsal raphe nucleus to the central amygdala and an inhibitory pathway from the central amygdala to the parafascicular nucleus play a critical role in governing the comorbidity of chronic pain and depression.2,8 However, research investigating global changes in brain function is still lacking. Resting-state functional magnetic resonance imaging (rs-fMRI) as a non-invasive neuroimaging technique aims to macroscopically detect neuronal activity in response to the possible role of resting-state specific sustained experience in shaping functional connectivity.9,10 A large-scale meta-analysis reported common changes in patients with chronic pain, depression or anxiety, with decreased gray matter volume predominantly in the insula and medial prefrontal cortex, mainly within the brain’s default mode and salience networks. 11 Collectively, these findings highlight the necessity of leveraging fMRI-based approaches to elucidate the neural mechanisms of the comorbidity of chronic pain and depression.
Our study aims to acquire blood-oxygen-level-dependent (BOLD) and diffusion tensor imaging (DTI) signals in the rat brain using fMRI to explore the key regions and specific neural circuits, which have predictive and diagnostic power for the comorbidity of chronic pain and depression and maybe radiological biomarkers of comorbid. Our research findings will contribute to gaining a novel perspective and evidence in understanding the diseases.
Material and methods
Experimental animals
Adult male Sprague-Dawley (SD) rats, aged 6–8 weeks and weighing 150–200 g, were used in the experiment. The rats were obtained from the Institute of Experimental Animals at Sun Yat-Sen University. All animals were housed at 23 ± 3ºC, with humidity ranges between 50% and 60% under a 12-h/12-h light-dark cycle separately and with food and water available ad libitum. Efforts were made to minimize the suffering and the number of rats used. All experimental procedures and protocols used in this study were approved by the local Animal Care Committee and in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Experimental design
The specific experimental design and procedures are presented in Figure 1(a). In brief, each rat’s hind paw mechanical withdrawal threshold was pre-tested. The next day, the nerve injury group underwent a nerve injury operation, while the sham group underwent the same operation without nerve injury. Subsequently, the mechanical von Frey testing (VFT) was conducted every 2 weeks. After 6 weeks, the open field test (OFT), elevated plus maze (EPM), and forced swimming test (FST) were performed sequentially. Resting-state magnetic resonance imaging (re-fMRI) data were acquired after these tests.
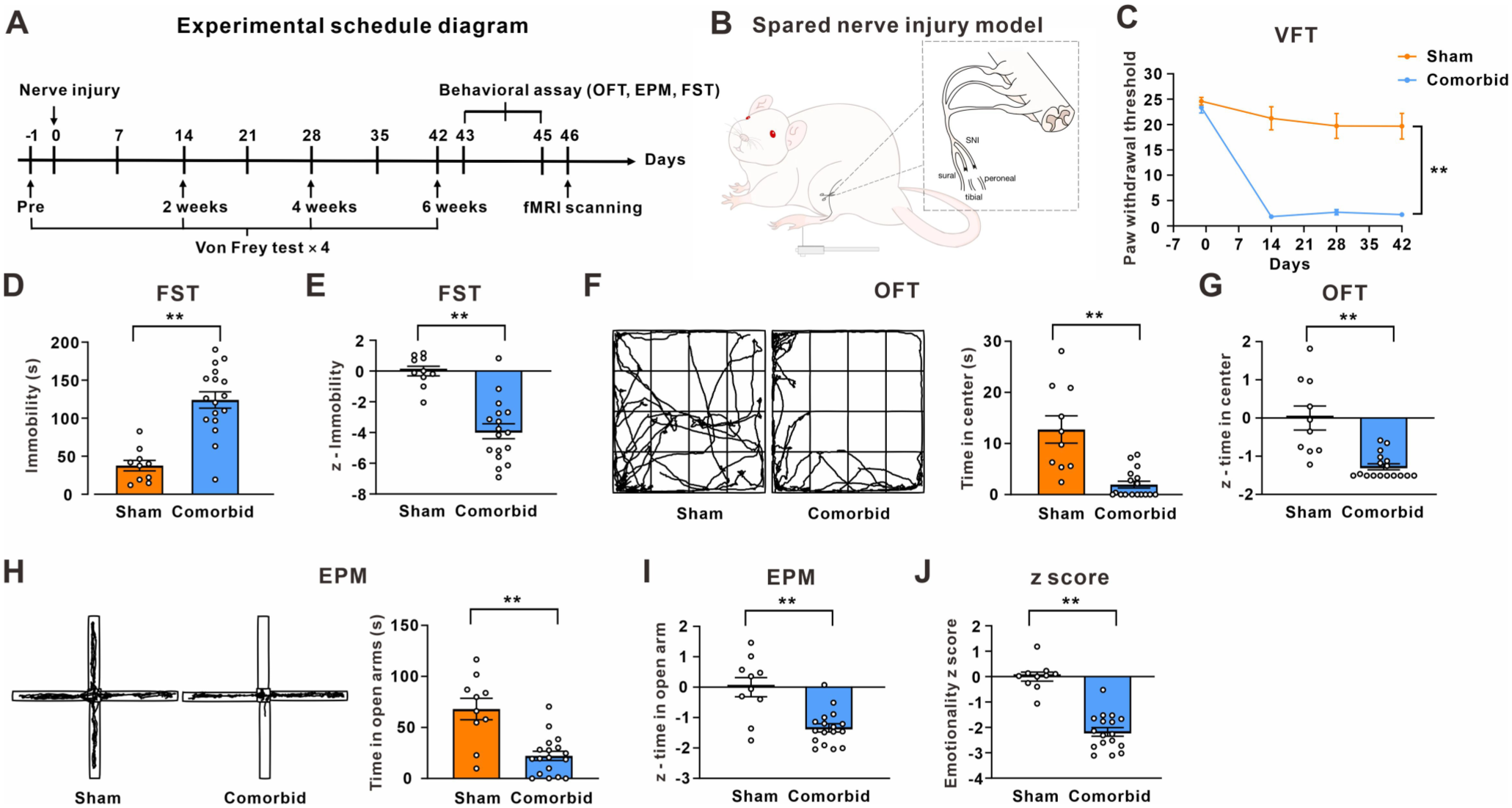
Chronic pain and depression comorbidity induced by SNI in rats: (a) a diagram depicting the experimental schedule for rats, (b) the schematic diagram of spared nerve injury model, (c) mechanical paw withdrawal threshold of sham and comorbid (SNI) rats (n = 10 in sham group and n = 17 in comorbid group; **p < 0.01 versus the sham group), (d) immobility time of rats in the FST (n = 10 in sham group and n = 17 in comorbid group; **p < 0.01 versus the sham group), (e) the z-score of the immobility time in FST in sham and comorbid rats (n = 10 in sham group and n = 17 in comorbid group; **p < 0.01 versus the sham group), (f) the representative trajectory maps and statistical analysis of the time spent of center of the sham and comorbid rats in the OFT (n = 10 in sham group and n = 17 in comorbid group; **p < 0.01 versus the sham group), (g) the z-score of the time spent of center in OFT, (h) the representative trajectory maps and statistical analysis of the time spent of open arms of the sham and comorbid rats in the EPM (n = 10 in sham group and n = 17 in comorbid group; **p < 0.01 versus the sham group), (i) the z-score of the time spent of open arm in EPM, and (j) emotional z-score for assessing anxiety and depression state in rats (n = 10 in sham group and n = 17 in comorbid group; **p < 0.01 versus the sham group).
Surgery for the chronic pain model
The spared nerve injury (SNI) model was utilized as an animal model of chronic pain. 12 The surgery of each rat was done under anesthesia induced by isoflurane (4%). Each rat’s surgery was performed under anesthesia induced by 4% isoflurane. A cut was made in the left thigh to expose the three peripheral branches of the sciatic nerve by gently dissecting the underlying muscles. The common peroneal and tibial nerves were securely tied using 5-0 silk sutures and then fully severed distal to the ligation sites (removed in 2 mm sections), while leaving the sural nerve intact. The muscles and skin layer were sutured in two layers. In the sham group, the three branches of the sciatic nerve were exposed in the same manner without any nerve injury. Following the surgery, the rats were individually housed.
Behavioral tests
Von Frey test
Von Frey test (VFT) were used to assess the mechanical sensitivity. 7 Briefly, each animal was individually placed in a transparent plastic box to adapt to the environment for three consecutive days (15 min/day) before commencing behavioral testing. Different strengths of von Frey filaments were then alternately applied to the mid-plantar surface of the hind paw to stimulate the animals. The initial pressure of the von Frey filament was selected for stimulation. If no paw withdrawal reaction occurred, a stronger filament was chosen for stimulation; if a paw withdrawal reaction occurred, a weaker stimulus was used. The “up and down” method was employed to determine the mechanical sensitivity that would produce a 50% likelihood of withdrawal. All behavioral tests were performed by a researcher who was blinded to the treatment conditions.
Open field test
The open field test (OFT) was utilized to assess the anxiety-like exploratory and locomotor behaviors.7,12 Briefly, an open-topped square box, 100 cm length × 100 cm width × 40 cm height with uniform and consistent light was used. This 1 m2 area is evenly divided into 16 small square areas, with the central 4 being the central area and the outer 12 being the outer area. Rats were placed in the testing room for 1 h to adapt to the testing environment. Then the rat was gently placed into the center of the square area and was monitored for 5 min. The traveling time and motion trajectory in the central area and outer area were captured by the camera placed directly above and recognized and calculated automatically by the software (Shanghai Jiliang Software Technology, Co., Ltd.). The chamber was cleaned with a 75% Ethanol at the end of each rat test.
Elevated plus maze test
The elevated plus maze test (EPM) was widely used to perform for the assessment of depression/anxiety-like behaviors in rodents.7,12 The apparatus was composed of a 10 cm × 10 cm central area and four 40 cm × 10 cm elevated arms, two close arms with 40 cm walls and two open arms without walls. The apparatus is elevated 55 cm above floor. At the start of the test, after habituation 1 h in the testing room, each rat was gently placed in the center area and facing the open arms. The movement of rats was tracked for 5 min by using the digital video camera in a brightly environment. The maze was cleaned with a 75% Ethanol at the end of each rat test.
Forced swimming test
The forced swimming test (FST) has been classically and widely used to perform for the assessment of depression/anxiety-like behaviors in rodents.7,12 The procedure of FST has two swimming session, including pre-test session and test session. Briefly, the pre-test session, after habituation 1 h in the testing room, each rat was taken from their home cage and gently placed in individual plexiglass cylinders (50 cm high, 18 cm in diameter) filled with water (24 ± 1°C) to a height of 40 cm for 15 min. After 24 h, test session was performed and the movement was recorded for 5 min by digital camera. At the end of both swimming sessions, each rat was removed from the cylinders, dried with towels, placed in cages for 15 min rest and recovery, and then returned to their home cages. Change the water after every test to avoid any influence on the next test. Among all behavioral tests, the FST was generally arranged as the last one.
Emotionality Z-score
Z-scores are dimensionless mathematical tools that represent the emotional aspect by calculating the emotional behavioral parameters.13–16 The Z-scores for the open field test and elevator plus maze were calculated using the following formula: Z = (X − μ)/σ while for the forced swimming test was calculated by the formula: Z = (μ − X)/σ. X represents the individual data for the observed parameter, while μ and σ represent the mean and the standard deviation for the control group, respectively. These three behavioral parameters, including time spent in the central area of the OFT, time spent in the open arms of the EPM, and time of immobility in the FST were used to calculate the Z-score for each rat representing the emotional aspect in each test. The emotionality Z-score for each rat was then calculated by averaging Z-score in the OFT, EPM, and FST.
Resting-state functional magnetic resonance imaging (rs-fMRI) acquisition
Functional MRI scans were performed for each rat of test according to the experimental schedules. All in vivo fMRI scanning took place in the morning (9:00–12:00 A.M.), when corticosteroid levels are relatively low.17–19 Imaging data were acquired using a 9.4T animal MRI scanner (Bruker Biospin GmbH, Germany) with the use of a volume coil T12054V3 (for signal excitation) and a surface coil T20011V3 (for signal reception) at the Fifth Affiliated Hospital of Sun Yat-Sen University, Zhuhai, China. Before scanning, each rat was subjected to inhalation anesthesia with 1 ~ 1.5% isoflurane before being fixed on the scanner with their heads immobilized. At the meanwhile, their respiration was monitored to maintain a respiratory rate of 70 times/min.
During imaging scan, anatomical images were first acquired using a rapid acquisition with relaxation enhancement (RARE) sequence (repetition time (TR) = 5081.564 ms, echo time (TE) = 21.59 ms, matrix size = 150 × 105, field of view (FOV) = 3.0 × 2.1 cm, slice number = 70, slice thickness = 0.4 mm, slice gap = 0, and resolution = 0.20 × 0.20 × 0.40 mm). fMRI data were obtained using a 2D multi-slice, single-shot, gradient-echo EPI sequence, for BOLD the parameters is: TR = 2000 ms, TE = 10.332 ms; flip angle = 90°, matrix size = 100 × 70, FOV = 3.0 × 2.1 cm, slice number = 45, slice thickness = 0.6 mm, slice gap = 0, and resolution = 0.30 × 0.30 × 0.60 mm; 150 BOLD images, for Diffusion tensor imaging (DTI) the parameters is: TR = 2557.136 ms, TE = 16.915 ms, flip angle = 90°, matrix size = 100 × 70, FOV = 3.0 × 2.1 cm, slice number = 90, slice thickness = 0.3 mm, slice gap = 0, number of segments = 6, symmetric diffusion gradients were applied with b = 1000 s/mm2 in 30 non-collinear directions and b = 0 s/mm2.
rs-fMRI data processing
The rs-fMRI data pre-process procedures were based on the SPM12 toolbox of the MATLAB 2013b platform (Statistical Parametric Mapping 12; http://www.fil.ion.ucl.ac.uk/spm). The first 5 volumes of each fMRI scan were removed to ensure steady-state longitudinal magnetization. The remaining volumes were processed using the following steps: voxel magnification, slice timing correction, realignment, origin correction and coregistration to echoplanar imaging (EPI) templates from SIGMA atlas before resliced at a resolution of 3 × 3 × 3 mm, spatial smoothing using Gaussian kernel with full width half-maximum (FWHM) 6 mm and linear detrending. During process, volumes of rs-fMRI with relative framewise displacement (FD) > 0.3 mm were excluded. 20 The pre-process data was performed linear detrends and temporal band-pass filtering (0.01–0.1 Hz).
Static ALFF acquisition
The static amplitude of low-frequency fluctuations (ALFF) values was calculated on detrended data by using the DPABI software (http://rfmri.org/dpabi). Transform the time series into the frequency domain using the Fast Fourier Transform (FFT) to obtain the power spectrum. The square root of the power spectrum is obtained, and then the average value is taken in the range of 0.01–0.1 Hz of each voxel, and the root mean square is taken as ALFF value. Then the individual’s ALFF values were z-transformed prior to statistical analyses. Statistical analysis is based on zALFF or each voxel, and statistically significant clusters of zALFF signal as mapped to the rat SIGMA atlas for presentation via xjview (https://www.alivelearn.net/xjview/). Brain regions of interest (ROIs) were extracted using DPABI and functional connectivity analysis of ROIs with the whole brain was performed.
Dynamic ALFF acquisition
The dynamic ALFF was performed using Temporal Dynamic Analysis (TDA) toolkits based on DPABI.21,22 The square root of the power spectrum was z-transformed using Fisher’s Z-transformation. In sliding window analysis, a smaller window size monitors potential transient signals, while a larger window can capture the lowest frequency of interest in the signal. 23 According to peer studies, we applied the window length of 30 TR (60 s) and a shifting step size of 1 TR (2 s).24,25 The remaining 145 time points after removing the first 5 time points for each individual were segmented into 116 windows in total. ALFF was calculated for each sliding window to generate a set of ALFF maps for each subject. The standard deviation (SD) divided by the global mean value of the ALFF at each voxel across each window was calculated to assess the temporal variability of the ALFF, which is defined as dALFF.
Dynamic functional connectivity acquisition
Similar to dALFF, the sliding window size was 30 TR in steps of 1 TR, which is a good trade-off between over-smoothing and sensitivity to noise. 24 The key regions from static and dynamic ALFF were used as ROIs. The Pearson correlation between the time course of each seed and the time course of other voxels were calculated for each sliding windows to obtain individual FC maps. The standard deviation (SD) divided by the global mean value of the FC at each voxel across each window was calculated to assess the temporal variability of the FC, which is defined as dFC. These maps were standardized to z-score maps using Fisher’s Z transformation.
DTI processing and calculation
For DTI data preprocessing, the first time, the data was converted to Nifti format that identified by FMRIB software Library (FSL) 4.1.9 (Oxford Centre for Functional Magnetic Resonance Imaging of the Brain Software Library, www.fmrib.ox.ac.uk/fsl/). And then, the head motion and eddy current distortions were corrected by using the FSL eddy tool. Subsequently, the diffusion tensor elements were estimated by solving the Stejskal and Tanner equation. The whole-brain voxels were segmented according to the rat SIGMA atlas, and the average of fractional anisotropy (FA), mean diffusivity (MD), axial diffusivity (AD) and radial diffusivity (RD) of all voxels within the brain region were taken as the brain region’s FA, MD, AD and RD.
Statistical analysis
SPSS 23.0 was used to analyze all of data, which were expressed as means ± SEM. Two-way ANOVA was performed by repeated measurements, including the mechanical allodynia of behavior tests. Behavioral tests including OFT, EPM, and FST that either satisfied or did not satisfy the Shapiro–Wilk normality test were analyzed using either an independent samples t-test or a Mann–Whitney–Wilcoxon test. In all cases, a p-value of less than 0.05 was deemed statistically significant for behavior analysis. rs-fMRI data were compared using two-sample t-test. The criterion of statistical significance of the static and dynamic ALFF and FC results of seed-based correlational analysis was threshold of a single voxel p < 0.001 and combined with false discovery rate (FDR) corrected of cluster level. The correlation between subjects’ comorbidity behavioral data and ALFF value of potential brain regions were analyzed using Pearson correlation (meeting Shapiro–Wilk normality test) and Spearman correlation analysis (not meeting Shapiro–Wilk normality test). Hierarchical regression analyses were performed to test whether fMRI features predict depressive-like behavior independently of pain (PWT). ALFF values were entered as predictors, with pain added as a covariate in the second block.
Results
Spared nerve injury induces the comorbidity of chronic pain and depression-like behavior in rats
The schematic diagram shows the experimental process of rats (Figure 1(a)). Consistent with previous studies,7,12 after detecting the mechanical response threshold of the rat plantar with Von Frey hairs, spared sciatic nerve injury was performed in the SNI group, while the sham group performed the same operation without nerve injury (Figure 1(b)). Von Frey test showed that the mechanical withdrawal threshold of the hind paw on the operated side in rats significantly decreased in the SNI group, compared to the sham group (Figure 1(c)). After 6 weeks, the animal’s depression status was examined using forced swimming test (FST). The results showed that the SNI group exhibited an increase in immobility time in FST, relative to the sham group (Figure 1(d)). Emotionality Z-scores, an important feature of which was to ensure consistency with behavioral data, were calculated to further confirm the occurrence of anxiety and depression-like behaviors in SNI-treated rats. 13 The z-score of FST in the SNI group was significantly lower than the Sham group (Figure 1(e)). Since patients with depression often experience anxiety-like manifestations,26,27 we further explored the anxiety-like behavior using the open field test (OFT) and the elevated plus maze (EPM). For OFT, the time spent in the center area and the z-score were obviously decreased in the SNI group (Figure 1(f) and (g)). For EPM, the time spent in the open arms and z-score decreased in the SNI group (Figure 1(h) and (i)). Importantly, the emotional Z-score, which represents the average of z-scores from the FST, OFT, and EPM was significantly lower in the SNI group, compared to the sham group (Figure 1(j)). These behavioral data collectively confirm that SNI successfully induced comorbidity of chronic pain and depression-like behavior in rats.
Static ALFF of comorbid rats
According to the experimental schedule, the fMRI data acquired after behavior assay. The representative original images of the brains from each group of animals under each collection method were presented in Supplemental Figure 1A. To explore the potential key brain region involved in comorbid rats with chronic pain and depression, we first detected the spontaneous neuronal activities by analyzing the static amplitude of low frequency fluctuation (ALFF) value in BOLD signals (Figure 2 and Table 1). The up-regulated (labeled in warm color) and down-regulated (labeled in cold color) brain regions in ALFF values compared to the sham group were shown in Supplemental Figure 1B and C. Compared with sham group, the comorbid rats showed increased ALFF signals in the left amygdalopiriform cortex (Amy_L), right entorhinal cortex (EC_R), left molecular layer of the cerebellum (MLC_L), left orbitofrontal region (OFR_L), left temporal associative cortex (TAC_L), right brainstem (BS_R) and left inferior olive (IO_L). The decreased ALFF signals were observed in the left cornu ammonis 1 (CA1_L), right cornu ammonis 1 (CA1_R), left cornu ammonis 3 (CA3_L), left dentate gyrus (DG_L), left granule cell level of the cerebellum (GCLC_L), right granule cell level of the cerebellum (GCLC_R), right molecular layer of the cerebellum (MLC_R), left olfactory bulb (OB_L), right periaqueductal gray (PAG_R), left prelimbic system (PL_L), right primary somatosensory cortex upperlips (PSCUL_R), left secondary somatosensory cortex (SSC_L), left striatum (Stri_L), right striatum (Stri_R), left brainstem (BS_L), right descending corticofugal pathways and globus pallidum (GP_R), right septal region (SR_R), left thalamus (Tha_L) and right thalamus (Tha_R) (Table 1). These results revealed that the SNI-induced chronic pain and depression comorbidity may exert impact on the activity of several brain regions.

Brain regions with significant differences in ALFF between sham and comorbid rats: (a) the ALFF value in different brains between sham and comorbid group and (b) regions showing differences in the ALFF between sham and comorbid group.
The static ALFF results of comorbid rats with chronic pain and depression.
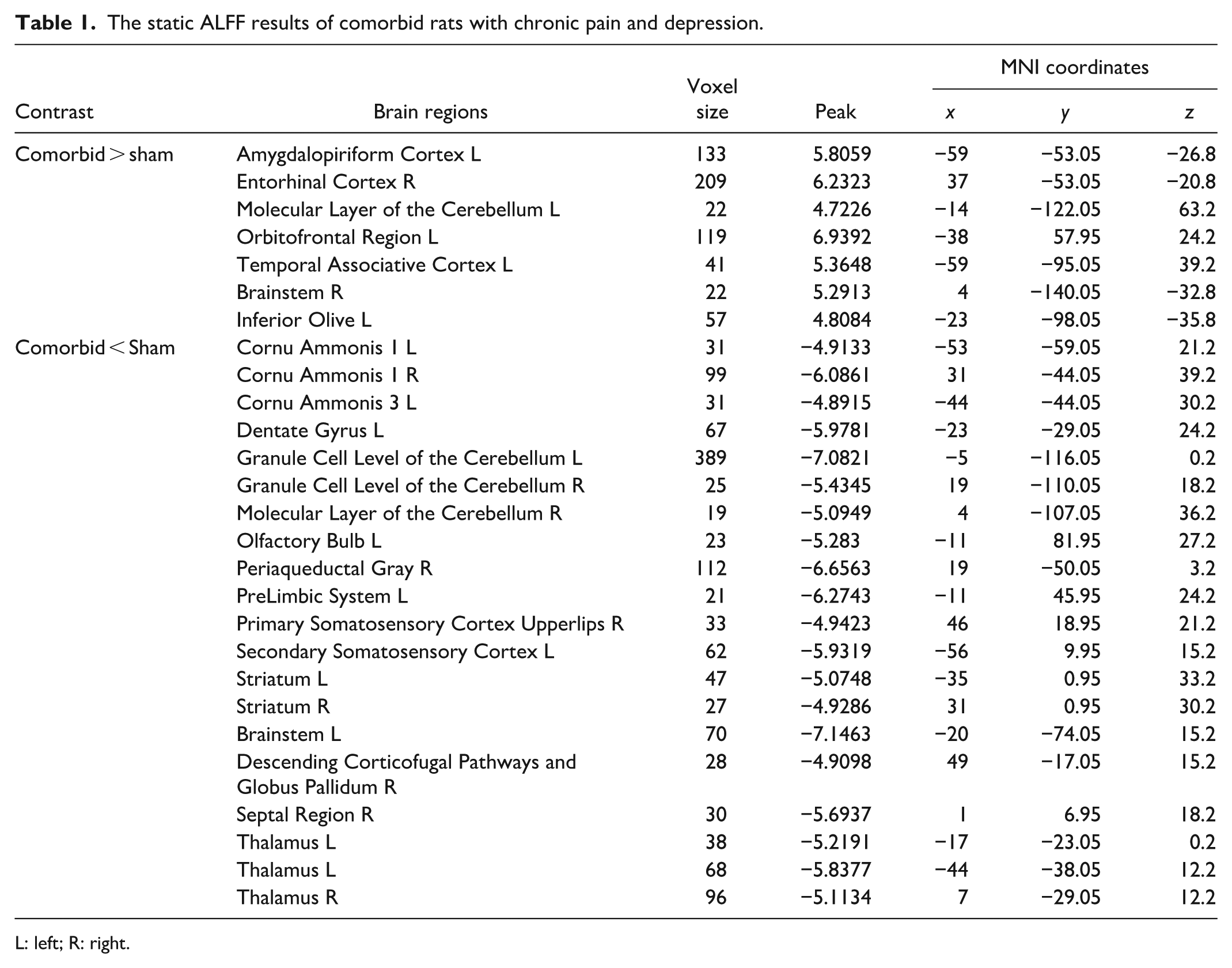
L: left; R: right.
Microstructure of comorbid rats
Due to the susceptibility of ALFF analysis to physiological noise,28,29 we further examined the microstructural changes of the brain region obtained using diffusion tensor imaging (DTI) data. We calculated the fractional anisotropy (FA), mean diffusivity (MD), axial diffusivity (AD), and radial diffusivity (RD) to compare the differences between the groups. The results showed that in the brain regions where ALFF values change, the OFR_L exhibited a significant change in four DTI indicators (FA increases while MD, AD, and RD decrease) (Figure 3(a)–(d), and Table 2). Moreover, CA1_L, CA3_L, GCLC_L, PL_L, Stri_L, and Stri_R have higher FA values, or lower MD, AD, and RD values in SNI-treated comorbid rats, compared with sham rats (Figure 3(e)–(h), and Table 2). There were no significant changes in other ALFF-change regions (Supplemental Figure 2, and Table 2). The integrated results above indicated that the brain seven regions showed concurrent changes in ALFF values and DTI data in comorbid rats (i.e. regions exhibiting both altered activity and disrupted microstructure). These regions may play a crucial role in the comorbidity of chronic pain and depression.
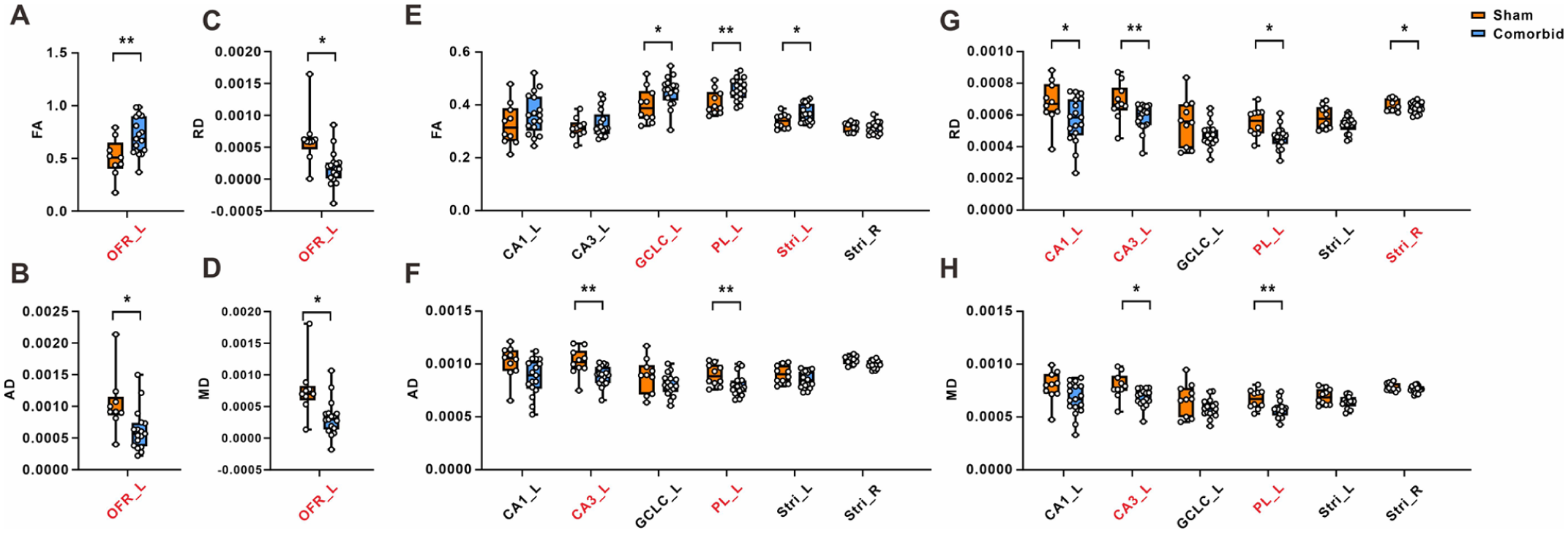
The differential regions of DTI metrics of ALFF-changed brain regions in comorbid rats. Fractional anisotropy (a, e), mean diffusivity (b, f), axial diffusivity (c, g), and radial diffusivity (d, h) diffusivities were analyzed in the ALFF-changed brain regions in chronic pain and depression comorbidity rats following with SNI treatment.
The statistics of DTI metrics from the ALFF-changing brain regions.

L: left; R: right.
The data do not meet normal distribution.
p < 0.05. **p < 0.01 versus the sham group.
Correlation analysis between ALFF values and comorbid behavior in rats
To further validate the correlation between the selected 7 regions and comorbid behaviors, we conducted the data-driven correlational analysis with the ALFF value of brain regions and the various comorbid behaviors of the subject. The results of correlation analysis implicated that the ALFF value of OFR_L was negatively correlated with the paw withdrawal threshold and the emotionality Z-score (Figure 4(a)). Conversely, the ALFF value of CA1_L, CA3_L, GCLC_L, PL_L, Stri_L, and Stri_R were all consistently exhibited positive correlation with the paw withdrawal threshold and the emotionality Z-score (Figure 4(b)–(g)). These findings indicate that the identified brain regions are closely associated with both pain-related and depression-like behaviors in the SNI-induced comorbidity model.

Correlation between ALFF value of potential region and comorbidity behavior in rats: (a-g) The ALFF value of key regions was correlated with the subjects’ paw withdrawal threshold and emotionality Z-score, respectively.
To further disentangle the relationship between fMRI features and depressive-like behavior, we conducted hierarchical multiple regression analyses by incorporating both ALFF values and PWT as predictors of emotionality Z-scores. In the initial models, several brain regions showed significant associations with depressive scores. However, after controlling for PWT, these associations were no longer statistically significant across all examined regions (Table 3). In contrast, PWT remained a significant predictor in all models. Taken together, these results suggest that while ALFF alterations in these brain regions are correlated with depressive-like behaviors, this relationship is largely mediated by pain-related processes.
Hierarchical regression analyses of fMRI features predicting emotionality z-scores.

Model 1: predictors: fMRI feature; Model 2: predictors: fMRI feature, PWT.
p < 0.05.
Dynamic ALFF of comorbid rats
We conducted the dynamic ALFF (dALFF) analysis to further identify the association between the seven selected regions and comorbid behavior. Setting the window length to 30 TR and sliding the step to 1 TR produced the findings. In comparison to the sham rats, the dALFF was decreased in 15 regions of the comorbid rats, including Tha_L, CA1_L, and CA3_L; no increases in dALFF were found (Figure 5(a), and Table 4). Combining static and dynamic ALFF and microstructure, six regions were identified, including OFR_L, CA1_L, CA3_L, PL_L, Stri_L, and Stri_R. The six selected regions were extracted from the dALFF results and showed that the dALFF signals of those regions in comorbid rats were significantly decreased (Figure 5(b)). Consistently, the dALFF signals of six key regions obviously exhibited a positive correlation with the paw withdrawal threshold and depression emotional z-scores (Figure 5(c)–(h)). These results further demonstrated that the six key regions exert a crucial role in the comorbidity of chronic pain and depression.
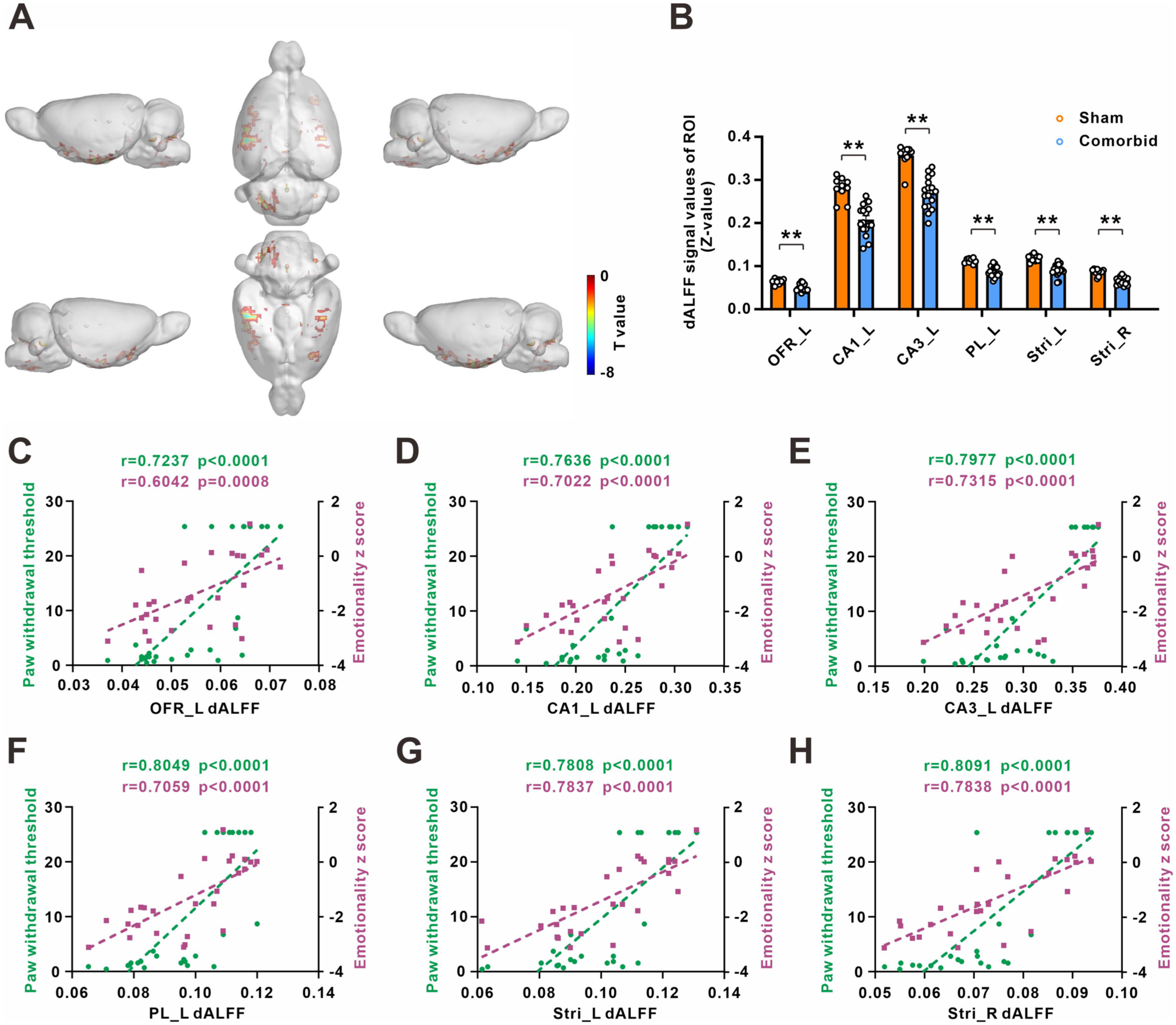
Alteration of dynamic ALFF in comorbid rats: (a) regions showing differences in the dALFF between sham and comorbid group (p < 0.001, cluster-level FDR-corrected), (b) the histogram of dALFF value in these regions in sham and comorbid rats, and (c)–(h) correlation between dALFF value of potential region and comorbidity behavior in rats.
The dynamic ALFF results of comorbid rats with chronic pain and depression.

L: left; R: right.
Dynamic functional connectivity of selected regions in comorbid rats
The key regions involved in the comorbidity of chronic pain and depression, identified through the hierarchical screening process described above, include several classic brain regions that have been confirmed to be associated with pain or depression, such as the hippocampus, prefrontal cortex, and striatum.30,31 To examine whether these selected regions reciprocally formed a functional network, we first exerted the dynamic functional connectivity analysis of regions of interest (ROI). After FDR correction, there were nineteen connections showing significantly enhanced in comorbid rats: CA1_L to the left Primary Visual Cortex (PVC_L), CA1_L to the left Primary Somatosensory Cortex (PSC_L), CA3_L to the left Primary Auditory Cortex (PAC_L), CA3_L to the left Deeper Layers of the Superior Colliculus (DLSC_L), CA3_L to the PSC_L, CA3_L to the left Retrosplenial Granular Cortex (RGC_L), OFR_L to the left Dysgranular Insular Cortex (DIC_L), OFR_L to the left Olfactory Bulb (OB_L), OFR_L to the left Primary Motor Cortex (PMC_L), OFR_L to the right Primary Somatosensory Cortex (PSC_R), OFR_L to the PSC_L, PL_L to the PSC_R, PL_L to the right Primary Cingular Cortex (PCC_R), PL_L to the PSC_L, Stri_L to the left Basal Forebrain Region (BF_L), Stri_L to the PSC_L, Stri_R to the right Basal Forebrain Region (BF_R), Stri_R to the PSC_R, and Stri_R to the PSC_L (Figure 6(a), and Table 5). The 3D visualization and schematic diagram of these 19 neural circuits were displayed and indicated that the differential network was composed of the hippocampus system, prefrontal cortex, striatum, and primary somatosensory cortex in comorbid rats (Figure 6(b) and (c)), which indicated the specific comorbid-associated network.

Dynamic functional connectivity in comorbid rats: (a) six representative dynamic functional connectivity maps, generated with CA1_L, CA3_L, OFR_L, PL_L, Stri_L and Stri_R as the seed point, respectively (p < 0.001, cluster-level FDR-corrected), (b) and (c) the three-dimensional diagram and schematic diagram of differential connection with potential regions as seed.
The dynamic functional connectivity of seed-based correlational analysis.

L: left; R: right.
Receiver operating characteristic curve of six selected regions
Given that these key regions were associated with both pain- and depression-related behaviors, receiver operating characteristic (ROC) curve analysis was performed to evaluate their classification performance in distinguishing comorbid rats from sham controls. The results showed that the AUC values of ALFF features from the selected regions were significantly elevated (Figure 7), indicating good discriminative ability.

ROC curve of ALFF value of the key region in sham and comorbid rats. (a-f) The ALFF value of key regions predicted the comorbidity of chronic pain and depression.
Importantly, as depressive-like behaviors in this paradigm arise as a consequence of prolonged chronic pain, the strong classification performance of these fMRI features likely reflects their sensitivity to pain-related neural processes that contribute to affective disturbances. Therefore, the identified regions may serve as imaging markers of pain-depression comorbidity, rather than independent predictors of depressive states.
Discussion
Chronic pain is a debilitating condition that has profound impacts on both physical and mental well-being. 32 Traditionally, the medical perspective viewed the body and mind as distinct and independent entities. However, emerging evidence suggests that the co-occurrence of chronic pain and depression can mutually exacerbate various distressing factors, including impaired functionality and increased disability rates. 11 Therefore, it is imperative to investigate the underlying neural mechanisms behind this reciprocal interaction. In the present study, we investigated the abnormal activity of brain regions by using static and dynamic frequency of ALFF methods and DTI-analysis. Next, dynamic functional connectivity was analyzed to identified the pattern of resting state networks in comorbid rats. Our findings can be summarized as follows: (1) After 6 weeks of SNI, the rats manifested remarkable mechanical allodynia as well as anxiety- and depression-like behaviors. Moreover, comorbid rats exhibited significant alternation in the activity of multiple brain regions, with the upregulation of the activity in seven regions and the downregulation in 19 regions. (2) Further analysis using DTI data revealed that among the brain regions that have significant changes in ALFF values, seven regions (OFR_L, CA1_L, CA3_L, GCLC_L, PL_L, Stri_L, and Stri_R) also exhibited alterations in microstructure. Importantly, these brain regions were significantly correlated with the comorbid behavioral phenotypes in rats. (3) Dynamic ALFF further identified the six key regions and correlated with the comorbid behavioral phenotypes. (4) Dynamic function connectivity identified the specific comorbid-associated network. (5) The ALFF value of six key regions has a good predictive value for the comorbidity of chronic pain and depression, and may be the brain imaging markers of comorbid. This study thoroughly investigated the adaptive alterations in brain neural networks resulting from the comorbidity of chronic pain and depression, highlighting an abnormal network associated with this comorbid condition. These findings offer important insights into the neural mechanisms underlying the coexistence of chronic pain and depression.
ALFF represents the magnitude of intrinsic neural activity, particularly in the gray matter regions of the brain where the results are relatively stable. 29 However, ALFF might be influenced by physiological noise unrelated to brain activity.33,34 As is well known, the structure and function of the brain are closely interconnected and inseparable. Accumulating evidence suggested that chronic pain and/or depression can significantly impact the functional activity and structural integrity of multiple brain regions.28,35–37 Recent studies have found that the cortex, striatum, parabrachial nucleus, and dorsal raphe nucleus are commonly associated with chronic pain and/or depression.38–40 Combining ALFF and DTI parameters, we found that seven key regions (OFR_L, CA1_L, CA3_L, GCLC_L, PL_L, Stri_L, and Stri_R) exhibited simultaneous significant alterations in neural activity and microstructure in comorbid rats. Importantly, the ALFF value of these key brain regions was significantly correlated with the subject’s pain- and depression-like behaviors, respectively. Notably, further hierarchical regression analyses revealed that the significant associations between ALFF alterations and depressive-like behaviors were no longer maintained after controlling for pain (PWT), whereas pain remained a robust predictor across all models. This pattern suggests that the observed correlations between neural activity and depressive phenotypes are not independent, but largely reflect shared variance with pain-related processes. Such findings are consistent with the intrinsic structure of the present model, in which depressive-like behaviors emerge as a downstream consequence of prolonged nociceptive input. Therefore, the identified ALFF alterations may primarily represent neural substrates of chronic pain, which secondarily contribute to affective disturbances. In this context, these brain regions are more likely to reflect an integrated pain-affect processing network, rather than serving as independent neural markers of depression.
The combination of static and dynamic rs-fMRI parameters could comprehensively elucidate brain function.41,42 In the present study, dynamic ALFF further identified that the six key regions significantly decreased in comorbid rats, compared with the corresponding sham group. Moreover, the dALFF value of six key regions was correlated with paw withdrawal threshold and the emotionality z-score of depression, respectively. Interestingly, we found that the results of OFR_L of dynamic measures differ from the static measures, this has also been found in other studies of diseases.23,43–45 For example, compared with mild cognitive impairment, cuneus and calcarine exhibited an increase of fraction ALFF (fALFF) and a decrease of dfALFF in patients with comorbidity of mild cognitive impairment and depression. 23 Among the six key regions, OFR_L and PL_L belong to the prefrontal cortex, CA1_L and CA3_L are the hippocampus system, and the bilateral striatum is the striatal system. Consistently, recent studies indicate that neuroinflammation and functional alterations in the prefrontal cortex, hippocampus, and striatum take place in patients with chronic pain and depression comorbidity,40,46–48 which may reflect the common radiological features of chronic pain and depression comorbidity. Importantly, the ROC analysis demonstrated that ALFF features from these regions could effectively distinguish comorbid rats from sham controls. However, in light of our hierarchical regression results, this classification performance is more appropriately interpreted as reflecting sensitivity to pain-related neural processes that contribute to depressive-like behaviors, rather than independent encoding of depression per se. Thus, these regions may serve as integrated neuroimaging markers of pain-depression comorbidity, capturing the interaction between nociceptive and affective systems.
The importance of individual brain regions in the occurrence and development of diseases is evident, but the large-scale interactions between brain regions also significantly contribute to the progression of diseases. rs-fMRI studies in the human brain have demonstrated the existence of multiple large-scale neural networks within a complex hierarchical structure of coordinated functional activity. These distributed neuroanatomical systems provide an emerging research perspective for understanding brain function and its impaired conditions in various neuropathological conditions. 49 While rs-fMRI methods have been extensively used in human connectivity research, it is only in recent years that they have been adapted for use in experimental animal studies. Although previous studies have reported that the comorbidity of chronic pain and depression is associated with marked alterations in whole-brain functional connectivity within the resting-state networks (RSNs) in rats through independent component analysis (ICA). 50 The definition of large-scale neural networks remains somewhat ambiguous, and further optimization and validation are required for the consistency of research results. 51 From a novel perspective, our study was based on the specific regions with alternation of neural activity and structure to identify the specific neural circuits correlated with the comorbid phenotype. A large and intricate brain network composed of 19 neural circuits was observed, and it was discovered that the prefrontal cortex, hippocampus, striatum, and primary sensory cortex are the most central and crucial components of this network. In the brain network, the prefrontal cortex and hippocampus belong to the default mode network (DMN), the striatum is the part of salience network (SN), and the primary somatosensory cortex is sensorimotor network (SEN). These three networks (DMN, SN, SEN) are widely recognized as important brain networks closely related to emotion regulation, emotional cognition, and pain perception.52–54 This intricate and specific comorbid-associated network integrated the DMN, SN, and SEN network into an interconnected entirety. These findings offer a fresh perspective on understanding comorbidity, and further investigations involving additional neural circuit markers and functional experiments are warranted to unravel the precise neural circuits and functions involved in this phenomenon.
Isoflurane is one of the most commonly used inhalational anesthetics in preclinical resting-state fMRI studies, as it offers rapid induction, stable maintenance, and facilitates survival studies in non-human primates and rodents.55,56 However, accumulating evidence indicates that isoflurane exerts profound effects on blood-oxygen-level-dependent (BOLD) signals and functional connectivity (FC). Previous studies have shown that isoflurane dose-dependently suppresses low-frequency BOLD fluctuations, leading to reduced signal amplitude and power. 57 Consistently, it induces a global decrease in functional connectivity strength across both local circuits and large-scale brain network. 58 Notably, despite this overall attenuation, the spatial organization of major resting-state networks is largely preserved, suggesting that intrinsic network architecture remains intact under anesthesia.58,59 In contrast, awake animal fMRI offers a more accurate reflection of intrinsic brain function, as it avoids the confounding effects of anesthetic agents on neural activity, cerebrovascular reactivity, and network dynamics. Awake preparations preserve the full integrity of functional networks, including higher-order cognitive systems such as the default mode network, and enable more reliable translation of findings to human physiology and pathology. Although awake imaging requires extensive animal training and presents challenges related to motion and stress, advances in acclimatization protocols, head fixation techniques, and motion correction strategies have increasingly rendered this approach feasible and reproducible.58,59 Therefore, future studies should prioritize awake fMRI whenever possible, as it more accurately reflects the true pathophysiological state of the animal.
It should be noted that the animals included in this study were still undergoing brain maturation during the experimental period. This developmental stage is characterized by dynamic processes, including synaptic pruning, myelination, and the refinement of large-scale functional networks, particularly within cortico-limbic and thalamocortical circuits.60,61 Previous studies have shown that adolescence represents a critical window for the integration of cortical network functions. 62 Chronic pain is a highly aversive and persistent experience that affects multiple domains, including sensory processing, endocrine regulation, brain development, and cognition. 60 Prolonged nociceptive input may exert lasting “programming” effects on the developing nervous system, leading to alterations in thalamocortical connectivity and modulation of the endogenous opioid system. These changes may ultimately result in a dual phenotype in adulthood, characterized by reduced sensitivity to acute pain but increased vulnerability to persistent pain.60,61 Notably, such early-life influences may even accelerate brain maturation, as reflected by an increased discrepancy between brain age and chronological age. 63 In addition, chronic pain itself may disrupt normal developmental trajectories, leading to reduced gray matter volume in pain-related brain regions and exacerbated abnormalities in functional connectivity. 62 These changes are thought to reflect an acceleration of pathological brain remodeling processes. 63 In the present study, although rats were approximately 6–8 weeks old at baseline, both the control and chronic pain groups followed identical experimental timelines, thereby minimizing the impact of normal developmental processes on between-group comparisons. Therefore, the observed differences are unlikely to be solely driven by maturation. Future studies should still use mature animals to eliminate the influence of development on the results.
Conclusions
In summary, by integrating static and dynamic ALFF analyses with DTI parameters, we identified six key brain regions (OFR_L, CA1_L, CA3_L, PL_L, Stri_L, and Stri_R) that are closely associated with chronic pain-depression comorbidity. These regions form a comorbidity-related network centered on the prefrontal cortex, hippocampus, striatum, and primary sensory cortex. Importantly, our findings suggest that the neural alterations in these regions primarily reflect pain-related processes that contribute to the development of depressive-like behaviors, highlighting a pain-mediated pathway underlying this comorbidity (Figure 8). Overall, this study provides new insights into the neural mechanisms linking chronic pain and depression, and underscores the importance of considering their integrated network-level interactions.

Graphic summary.
Supplemental Material
sj-docx-1-mpx-10.1177_17448069261443836 – Supplemental material for Static and dynamic perspective on brain activity and functional connectivity alternations in the comorbidity of chronic pain and depression: An fMRI study in rats
Supplemental material, sj-docx-1-mpx-10.1177_17448069261443836 for Static and dynamic perspective on brain activity and functional connectivity alternations in the comorbidity of chronic pain and depression: An fMRI study in rats by Jie Deng, Hai-Ting Fan, Yu-Ting Zhao, Yu-Juan Duan, Cui-Cui Liu, Di-Han Lu, Xiang-Zhong Zhang, Wen-Jun Xin, Xia Feng and Ting Xu in Molecular Pain
Footnotes
Abbreviations
SNI: spared nerve injury
BOLD: blood-oxygen-level-dependent
Rs-fMRI: resting-state functional magnetic resonance imaging
DTI: diffusion tensor imaging
PWT: paw withdrawal threshold
VFT: Von Frey test
OFT: open field test
EPM: elevated plus maze
FST: forced swimming test
ALFF: amplitude of low frequency fluctuation
Amy_L: left amygdalopiriform cortex
EC_R: right entorhinal cortex
MLC_L: left molecular layer of the cerebellum
OFR_L: left orbitofrontal region
TAC_L: left temporal associative cortex
BS_R: right brainstem
IO_L: left inferior olive
CA1_L: left cornu ammonis 1
CA1_R: right cornu ammonis 1
CA3_L: left cornu ammonis 3
DG_L: left dentate gyrus
GCLC_L: left granule cell level of the cerebellum
GCLC_R: right granule cell level of the cerebellum
MLC_R: right molecular layer of the cerebellum
OB_L: left olfactory bulb
PAG_R: right periaqueductal gray
PL_L: left prelimbic system
PSCUL_R: right primary somatosensory cortex upperlips
SSC_L: left secondary somatosensory cortex
Stri_L: left striatum
Stri_R: right striatum
BS_L: left brainstem
GP_R: right descending corticofugal pathways and globus pallidum
SR_R: right septal region
Tha_L: left thalamus
Tha_R: right thalamus
RSNs: resting-state networks
ICA: independent component analysis
DMN: default mode network
SN: salience network
SEN: sensorimotor network
Author’s contributions
TX, XF contributed to the conception and design of the study. JD, HTF, YTZ, YJD, acquired and analyzed the data. CCL, DHL, XZZ, WJX contributed to data quality control. TX, XF, JD were major contributors in writing the manuscript. TX, XF, JD, HTF, YTZ were responsible to the revised manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by China Postdoctoral Science Foundation (2024M763800), National Natural Science Foundation of China (82501483, 82572936, 82071224, 82371189, U25A2060, 82301354, 824B2014), Guangdong Basic and Applied Basic Research Foundation (2026A1515010860, 2023A1515030020, 2022A1515012124, 2022A1515140157), Guangdong Province Key Field R&D Plan Project (2023B0303010002), Guangzhou Clinical High tech Project (2023P-GX10).
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
