Abstract
Depression is commonly observed in individuals suffering from chronic pain, but the exact molecular mechanisms behind these symptoms are still not fully understood. This study highlights the important role of the TRIM14-NF-κB pathway in the anterior cingulate cortex (ACC) in regulating comorbid depressive symptoms associated with chronic pain. Our results show that the CFA model induces both chronic pain and depression-like behaviors in mice, with significant activation of the ACC brain regions. Specifically, the protein expression of TRIM14 was notably elevated in the ACC of CFA mice. Furthermore, reducing TRIM14 expression alleviated both chronic pain and depression-like behaviors in these mice. In addition, we also discovered that NF-κB may act as a downstream target of TRIM14, as silencing TRIM14 expression led to a reduction in the levels of phosphorylated NF-κB. Notably, inhibiting NF-κB produced similar improvements in chronic pain and depression-like behaviors, mirroring the effects observed with TRIM14 knockdown. In summary, our findings emphasize the critical role of the TRIM14-NF-κB pathway in regulating chronic pain and depression-like behaviors in the CFA mouse model. These insights provide a foundation for further exploration of the molecular mechanisms underlying chronic pain and depression, and may guide the development of targeted therapeutic strategies.
Introduction
Chronic pain and depressive symptoms often occur together and interact in the clinical setting, significantly complicating treatment for patients.1,2 Specifically, depressive symptoms may not only prolong pain but also exacerbate its severity,3–5 creating a vicious cycle of pain and depression. Epidemiological surveys indicate that about 50% of patients with depression also experience pain, and 85% of individuals with chronic pain have co-occurring depression.3,6 Despite this high comorbidity, the molecular mechanisms underlying the interaction between pain and depression remain poorly understood, posing a major challenge for clinical management. Therefore, it is crucial to investigate the mechanisms through which chronic pain is accompanied by depression.
Accumulating evidence suggests that the anterior cingulate cortex (ACC) plays a critical regulatory role in regulating both chronic pain and emotional disorders.7–11 The ACC acts as a relay connecting the frontal cortex, thalamus, and amygdala, integrating functions related to cognition, emotion, and autonomic regulation. 12 Several studies have reported functional and structural changes in the ACC in patients with depression, including altered glucose metabolism and reduced gray matter volume.13,14 Additionally, research has shown that stimulating ACC regions and their associated neural circuits can help alleviate symptoms of treatment-resistant depression. 15 Furthermore, damage to the ACC has been found to prevent depressive-like behaviors associated with chronic pain and aversion to spontaneous pain. 16 These findings highlight the ACC as a crucial hub for mood disorders, including the anxious-depressive symptoms often observed in chronic pain, though its precise molecular mechanisms remain unclear.
The TRIM protein family consists of over 80 members, most of which are E3 ubiquitin ligases. These proteins are characterized by the presence of RING finger, B-box, and coiled-coil domains. 17 TRIM proteins are widely recognized for their involvement in various biological processes, including tumor immunity, autophagy regulation, and inflammation.18,19 TRIM14, a member of this family, lacks the RING finger domain and instead mediates ubiquitination by interacting with USP14. It was first identified in HIV-infected humans and HIV-infected simian models of non-Hodgkin’s lymphoma. 20 Increasing evidence suggests that TRIM14 plays a significant role in cancer, autophagy, viral immunity, and other diseases, particularly through pathways such as interferon (IFN) signaling and the non-classical NF-κB pathway.21–23 Recent research has highlighted the link between dysfunction in the ubiquitin system and chronic pain, including both neuropathic pain and inflammatory pain. 24 Our previous studies also demonstrated that TRIM14 regulates CFA-induced plantar inflammatory pain in rats through the NF-κB pathway. 25 The NF-κB signaling pathway is crucial in a variety of biological processes, including immune responses, inflammation, and tumor progression.26,27 NF-KB includes RelA (p65), RelB, c-Rel, NF-kB1 (p105), and NF-kB2 (p100), 28 These subunits are activated through two major signaling pathways: the canonical and the atypical NF-κB pathways. The canonical pathway involves the proteasomal degradation of IκBα, which leads to the activation of transcription factors such as p50, p65, and c-Rel. 27 In inflammatory pain, NF-κB binds to the inhibitor IκBα, forming an inactive complex. When pro-inflammatory cytokines are present, IκBα is phosphorylated by a large multi-unit complex that includes two catalytic subunits (IKKα and IKKβ) and regulatory subunits, such as IKKγ (also known as NEMO). The development and progression of inflammatory pain also involve several other signaling pathways, including p38, ERK1/2, and JNK. However, the classical NF-κB pathway plays a particularly crucial role. Despite its importance, the precise regulatory mechanisms of the classical NF-κB pathway in pain and affective disorders remain incompletely understood. Further research into these mechanisms could lead to more effective treatments for various clinical pain conditions and their comorbidities, particularly through the use of precision medicine.
In this study, we provide enough evidence that targeting the TRIM14-NF-κB pathway markedly attenuates pain and depressive-like behavior, demonstrating that up-regulation of TRIM14 in ACC neurons plays a key role in pain and depressive behaviors. These findings deepen our understanding of the central molecular mechanisms involved in chronic pain and depression, offering potential therapeutic targets for the development of more effective treatments.
Materials and methods
Animals
The experimental animals were 6–12 weeks old SPF C57BL/6J male mice. The experimental animals were kept in strict accordance with the breeding standards, with a density of no more than five mice per cage. All mice were housed at a stable temperature of 24 ± 2°C, with humidity between 40% and 60% and a 12-h light/dark cycle. All animals had free access to food and water. All procedures were approved by the Animal Protection Committee of Soochow University, and behavioral tests were conducted in accordance with the requirements of the International Association for the Study of Pain.
Mouse model of inflammatory pain
Mice were randomly assigned to either the control or inflammatory pain group. After administering light anesthesia with isoflurane, the pain model was established while ensuring the mice continued to breathe spontaneously. Once anesthetized, the soles of the left hindpaw were disinfected with iodophor. Mice in the inflammatory pain group received a 10 μl injection of CFA (Sigma-Aldrich) into the left hindpaw sole, while mice in the control group were injected with an equal volume of saline in the same area.
Von Frey filament test
Before the behavioral test, mice were placed in a transparent Plexiglas box with a sieve-like metal mesh at the bottom and allowed to acclimatize to the environment for approximately 30 min. Once the exploratory and grooming behaviors had subsided, the left hindpaw of each mouse was stimulated with von Frey filaments (ranging from 0.008 to 4.0 g). The filaments were applied vertically to the left hindpaw until they bent, with each stimulation lasting 2–4 s. The interval between each trial was 30 s, and the test was repeated five times. A positive response was defined by behaviors such as paw withdrawal, foot licking, or running away. If no response occurred, the test continued with progressively thicker filaments until a positive reaction was observed. The pressure (in grams) at which a positive response occurred was recorded, and the average pressure was calculated after excluding the highest and lowest values
Hargreaves test
To minimize environmental disturbance, the mice are placed in a Plexiglas box with a clear glass plate at the bottom and acclimatized to the test environment for at least 30 min. During the test, the infrared light source of the infrared thermometer was pointed at the center of the left hindpaw sole of the mouse, and the time of foot contraction or licking response was recorded by pressing the start button. To avoid tissue damage, each mouse was subjected to five measurements with at least 5 min between tests. The average of the three measurements after removing the lowest and highest values was taken as the thermal pain threshold of the mice.
Open field test
Mice were individually placed in a square box (45 × 45 × 45 cm) with black walls and a white background for 30 min. They were allowed to explore freely for 10 min, during which their movement trajectory was recorded. After the recording, the mice were moved to their feeding cage, and the box was wiped with 75% alcohol to remove any odor from the previous animal. The box was then allowed to dry completely before proceeding to the next experiment. The distance traveled and the time spent in the center region of the box were analyzed using Any-maze software (Stoelting).
Forced swim test
Mice were placed in a transparent Plexiglas cylinder (25 cm high, 12 cm in diameter) filled with warm water (24 ± 1°C) to a height of 20 cm for 6 min. 29 The behavior of the mice was video recorded from the side, and the duration of immobility during the last 5 min of the trial was analyzed offline. Immobility was defined as floating or remaining motionless. At the end of the test, each mouse was dried, and the water was changed.
Sucrose preference test
The experimental procedure was as described in previous publication. 30 Briefly, to minimize potential experimenter bias, mice were housed individually, with cage numbers blinded. For the first two days, the mice were habituated to two bottles of drinking water, followed by 2 days with two bottles of 1% sucrose solution. After the habituation period, the mice were assessed for any baseline preference between the two bottles. Only mice with a preference between 25% and 75% for either bottle were included in the study, mice with a more extreme preference were excluded. During the testing phase, mice were given one bottle of water and one bottle of 1% sucrose solution for 2 h, with the positions of the bottles swapped halfway through. The volume of each liquid consumed was recorded. Sucrose preference is calculated as sucrose preference (%) = sucrose consumption volume/(sucrose consumption volume + water consumption volume) × 100%.
Western blot analysis
Mice were executed under 1.5% isoflurane anesthesia. Anterior cingulate cortical tissues were isolated on ice and stored in EP tubes at −80°C until use. For use, RIPA lysis buffer (Beyotime P0013B) and phosphatase inhibitor (PMSF, NCM Biotech P002) were add to lysate the tissues. The aliquots of proteins were separated by SDS-PAGE gel and transferred to nitrocellulose (NC) membranes. The NC membranes were incubated with TRIM14 (1: 1000, bs-16724R, Bioss), p-NF-κB (1:1000, sc-136548, Santa Cruz Biotechnology), NF-κB (1:1000, sc-8008, Santa Cruz Biotechnology), and GAPDH (1:10,000, G9545, Sigma) primary antibodies at 4°C overnight. The enhanced chemiluminescence reagents (ECL, Millipore Corporation, Billerica, MA, USA) was employed to display the bands. The band of target protein was quantified using ImageJ software (NIH, USA).
Immunofluorescence staining
Under deep anesthesia, mice were perfused via the heart with 0.9% saline followed by 4% polyformaldehyde. The dissected mouse brain tissue was placed in 4% polyformaldehyde for 2 h and then dehydrated in a gradient of 20% and 30% sucrose solution. Tissue was frozen in cryostat (CM1950, Leica, Germany) and cut into sections with a thickness of 30 µm. After rinsing three times in PBS, the sections were blocked with donkey serum for 2 h at room temperature. The sections were then incubated overnight at 4°C with primary antibody (1:200). Subsequently, the sections were rinsed three more times and incubated with secondary antibodies (Abcam, ab150129, anti-mouse 488, 1:800; Abcam, ab150074, anti-rabbit 555,1:800) at room temperature for 1 h. After three washes in PBS, the slices are mounted with the DAPI staining solution. Fluorescence images were acquired using Confocal microscopy (Leica, Germany).
Injection of TRIM14 siRNA
Mice were anesthetized under 3% isoflurane using an inhalation anesthesia machine (R500, RWD Life Science, China). The mice were fixed on a brain stereotaxic apparatus (68046, RWD Life Science, China) with incisor strips and ear strips, and then the scalp was incised with an ophthalmic instrument to expose the skull. Taking the bregma position as a reference zero point, holes were drilled at the mouse ACC (AP, +1.21 mm; ML, ±0.27 mm; DV, −1.50 mm). At the end of the injection (1 µl), the needle was stopped for ten minutes to allow the siRNA to spread. TRIM14 siRNA (siBDM0001, RIBO, China) sequence is as follows: 5′-GCATTCTGACCTTCTACGA-3′. The DEPC-treated water was employed as a vehicle for TRIM14 siRNA delivery.
Drug application
To assess the effect of NF-κB on pain behavior, bilateral stereotactic injections of DMSO (0.5%) or BAY 11-7082 (1 µl, MedChemExpress) were administered bilaterally via cannula into the ACC. Mice with inaccurate injection sites were excluded.
Data analysis
All data were presented with mean ± sem. SPSS 19.0 and GraphPad Prism 9.0 were used for statistics and graphing. Student’s t- test was used for two independent samples. Comparisons between multiple groups were analyzed using one-way ANOVA or two-way ANOVA. Statistical significance was considered at p < 0.05.
Results
CFA induced chronic inflammatory pain and depression-like behaviors
The timeline for modeling and behavioral testing in CFA mice is shown in Figure 1(a). Chronic inflammatory pain was assessed using the von Frey and Hargreaves tests, which revealed that CFA mice exhibited chronic inflammatory pain (Figure 1(b) and (c), **p < 0.01, two-way ANOVA followed by Sidak’s multiple comparison test). To assess depression-like behaviors, the forced swimming test, sucrose preference test, and open field test were conducted. The results of the forced swimming test showed significantly increased immobility time in CFA mice compared to sham mice (Figure 1(d), *p < 0.05, two-sample t-test). In the sucrose preference test, CFA mice demonstrated a lower preference for sucrose (Figure 1(e), **p < 0.01, two-sample t-test). During the open field test, CFA mice spent less time in the center zone and covered less distance compared to sham mice (Figure 1(f)–(h), *p < 0.05, **p < 0.01, two-sample t-test). However, there was no significant difference in the total distance moved between the two groups (Figure 1(i), two-sample t-test). These findings suggest that CFA mice exhibit persistent and stable chronic inflammatory pain accompanied by depression-like behaviors, without any significant impact on motor function.

CFA induced chronic pain and depression-like behaviors in mice. (a) Timeline of CFA modeling and behavioral testing. (b and c) Mechanical and thermal pain behaviors of CFA and sham mice (**p < 0.01, two-way ANOVA followed by Sidak’s multiple comparison test, n = 8 per group). (d) Time of immobility of CFA and sham mice in forced swimming test (*p < 0.05, two-sample t-test, n = 8 per group). (e) Preference for sucrose of CFA and sham mice in the sucrose preference test (**p < 0.01, two-sample t-test, n = 8 per group). (f) Representative traces of CFA and sham mice in the open field test. (g–i) Statistical analysis of CFA and sham mice for time in the center, distance in the center and total distance in the open field (*p < 0.05, **p < 0.01, two-sample t-test, n = 8 per group).
Enhanced activity and upregulated TRIM14 expression in ACC of CFA mice
To investigate the neural and molecular mechanisms underlying chronic pain in CFA mice, immunofluorescence staining was performed to assess activation of the ACC. The results revealed a significant increase in the number of c-Fos positive cells in the ACC of CFA mice compared to sham mice (Figure 2(a) and (b), **p < 0.01, two-sample t-test), indicating a dramatically increased activity in the ACC brain region. Further analysis showed that TRIM14 expression in the ACC was significantly upregulated in CFA mice (Figure 2(c), *p < 0.05, one-way ANOVA with Tukey’s multiple comparisons test). Immunofluorescence staining also revealed that TRIM14 was predominantly expressed in neurons, with minimal expression in microglia and astrocytes in the ACC (Figure 2(d) and (e)). These results suggest that TRIM14 expression is upregulated in ACC neurons of CFA mice.

Enhanced activity and upregulated TRIM14 expression in ACC of CFA mice. (a) Representative images of c-Fos with NeuN co-localization at ACC. Scalar bar = 100 µm. (b) Ratio of c-Fos with NeuN co-localization in ACC (**p < 0.01, two-sample t-test, n = 6 slices from 3 mice). (c) Representative western blot images of TRIM14 expression levels in CFA mice on days 1, 3, 7, 14, 21, 28, 35 (*p < 0.05, one-way ANOVA with Tukey’s multiple comparisons test, n = 6 per group). (d) Representative immunofluorescence images of TRIM14 with NeuN, GFAP, and Iba1. Scalar bar = 100 µm. The zoomed pictures in the right panel were to show the co-localization of TRIM14 with NeuN, Iba1, and GFAP. Scalar bar = 50 µm.
Knockdown of TRIM14 alleviates pain and depression-like behaviors in CFA mice
To assess the role of TRIM14 in modulating pain and depression-like behaviors, siRNA was used to knockdown TRIM14 expression in the ACC (Figure 3(a) and (b)). Following TRIM14 siRNA injection, TRIM14 expression was significantly reduced (Figure 3(c) and (d), **p < 0.01, one-way ANOVA with Tukey’s multiple comparisons test), confirming the effectiveness of the siRNA treatment. Compared to the vehicle group, pain behaviors in CFA mice were significantly alleviated after TRIM14 siRNA administration (Figure 3(e) and (f), **p < 0.01, two-way ANOVA followed by Sidak’s multiple comparison test). In the forced swimming test, TRIM14 siRNA injection markedly reduced immobility time in CFA mice (Figure 3(g), *p < 0.05, one-way ANOVA with Tukey’s multiple comparisons test). In the sucrose preference test, TRIM14 siRNA injection significantly increased sucrose preference in CFA mice (Figure 3(h), *p < 0.05, **p < 0.01, one-way ANOVA with Tukey’s multiple comparisons test). In the open field test, TRIM14 siRNA injection notably increased both the distance traveled and the time spent in the center region of CFA mice (Figure 3(i)–(l), *p < 0.05, **p < 0.01, ***p < 0.001, one-way ANOVA with Tukey’s multiple comparisons test). These results suggest that TRIM14 plays a critical role in regulating pain and depression-like behaviors in CFA mice.

Knockdown of TRIM14 alleviates pain and depression-like behaviors in CFA mice. (a) Timeline of CFA modeling and behavioral testing. (b) Schematic diagram of TRIM14 siRNA injection into ACC. (c) Representative western blot traces of TRIM14 after injection of TRIM14 siRNA into ACC. (d) Analysis of TRIM14 expression after injection of TRIM14 siRNA into ACC (**p < 0.01, one-way ANOVA with Tukey’s multiple comparisons test, n = 6 per group). (e and f) Mechanical and thermal pain behaviors of CFA and sham mice following injection of TRIM14 siRNA (two-way ANOVA followed by Sidak’s multiple comparison test, n = 8 per group). (g) Time of immobility of CFA and sham mice in forced swimming test following injection of TRIM14 siRNA (**p < 0.01, two-sample t-test, n = 8 per group). (h) Preference for sucrose of CFA and sham mice in the sucrose preference test following injection of TRIM14 siRNA (*p < 0.05, **p < 0.01, two-sample t-test, n = 8 per group). (i) Representative traces of CFA and sham mice in the open field test following injection of TRIM14 siRNA. (j–l) Statistical analysis of CFA and sham mice for time in the center, distance in the center and total distance in the open field following injection of TRIM14 siRNA (*p < 0.05, **p < 0.01, ***p < 0.001, two-sample t-test, n = 8 per group).
Knockdown of TRIM14 inhibited NF-κB phosphorylation
Previous study has shown that NF-κB is a downstream target of TRIM14, 25 so NF-κB expression and corresponding phosphorylation levels were assessed via western blotting. The results showed that the phosphorylation of NF-κB was significantly increased in ACC of CFA mice (Figure 4(a), **p < 0.01, one-way ANOVA with Tukey’s multiple comparisons test). Additionally, injection of TRIM14 siRNA significantly reduced the phosphorylation of NF-κB in CFA mice (Figure 4(b), *p < 0.05, **p < 0.01, one-way ANOVA with Tukey’s multiple comparisons test). Furthermore, immunofluorescence staining revealed a co-localization ratio of 89.56% for TRIM14 and NF-κB (Figure 4(c)). Altogether, these results suggest that NF-κB is a downstream target of TRIM14 and its phosphorylation level is regulated by TRIM14.

Knockdown of TRIM14 inhibited NF-κB phosphorylation. (a) Representative western blot traces and analysis of p-NF-κB/NF-κB expression of CFA and sham mice (**p < 0.01, one-way ANOVA with Tukey’s multiple comparisons test, n = 6 per group). (b) Representative western blot traces and analysis of p-NF-κB/NF-κB expression following injection of TRIM14 siRNA (*p < 0.05, **p < 0.01, one-way ANOVA with Tukey’s multiple comparisons test, n = 6 per group). (c) Representative immunofluorescence images of TRIM14 with NF-κB. Scalar bar = 50 µm.
Inhibition of NF-kB alleviates pain and depression-like behaviors in CFA mice
To further evaluate the role of NF-κB in regulating pain and depression-like behaviors, the NF-κB inhibitor BAY 11-7082 was used to block NF-κB function in the ACC (Figure 5(a) and (b)). Compared to the vehicle group, CFA mice treated with BAY 11-7082 exhibited significantly alleviated pain behaviors (Figure 5(c) and (d), **p < 0.01, two-way ANOVA followed by Sidak’s multiple comparison test). In the forced swimming test, injection of BAY 11-7082 markedly reduced immobility time in CFA mice (Figure 5(e), *p < 0.05, **p < 0.01, one-way ANOVA with Tukey’s multiple comparisons test). In the sucrose preference test, BAY 11-7082 injection significantly increased sucrose preference in CFA mice (Figure 5(f), **p < 0.01, ***p < 0.001, one-way ANOVA with Tukey’s multiple comparisons test). In the open field test, BAY 11-7082 treatment dramatically increased both the distance traveled and the time spent in the central region of CFA mice (Figure 5(g)–(j), **p < 0.01, ***p < 0.001, one-way ANOVA with Tukey’s multiple comparisons test). These results suggest that NF-κB plays a critical role in regulating pain and depression-like behaviors in CFA mice.
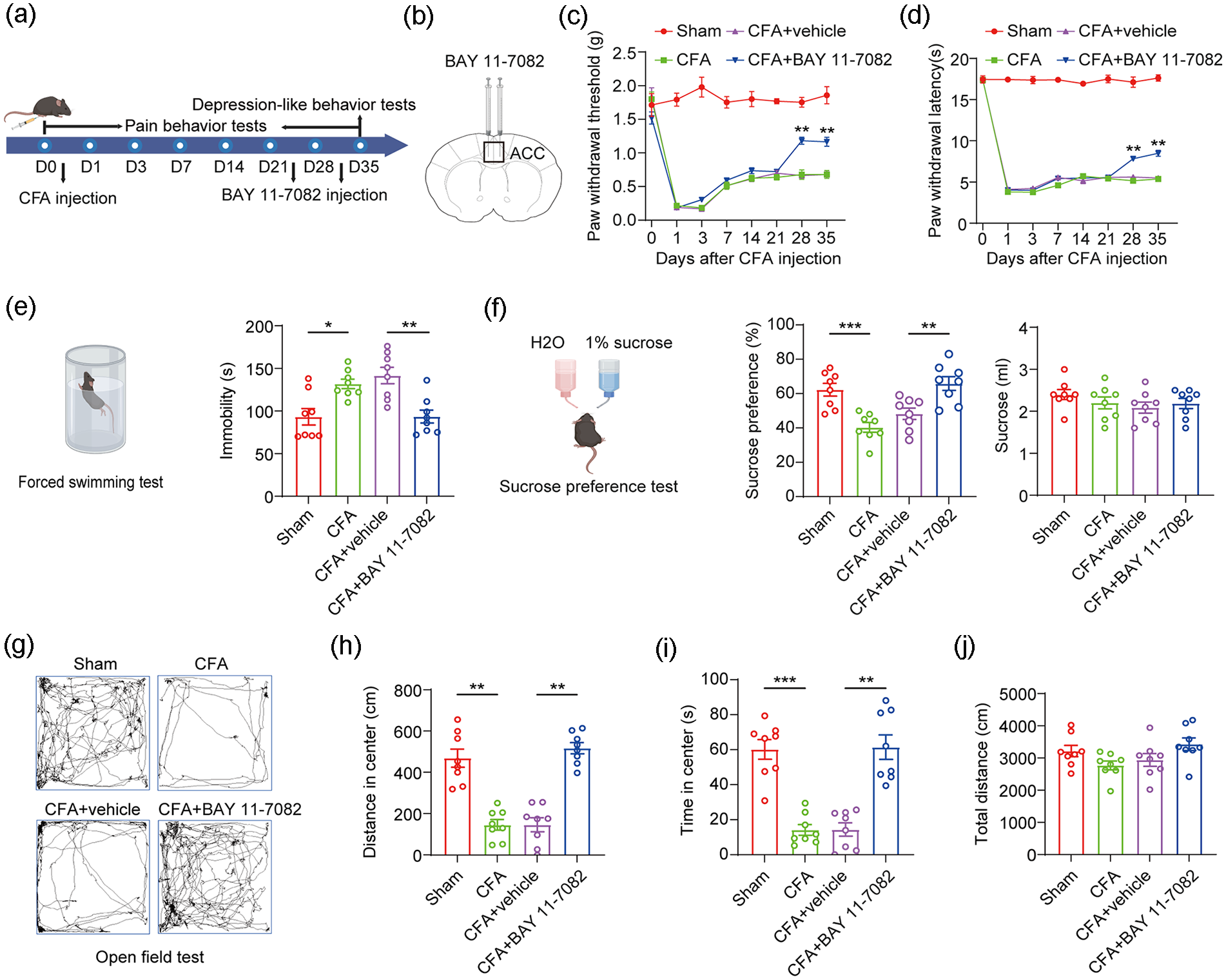
Inhibition of NF-kB alleviates pain and depression-like behaviors in CFA mice. (a) Timeline of CFA modeling and behavioral testing. (b) Diagram of injecting BAY 11-7082 into ACC. (c and d) Mechanical and thermal pain behaviors of CFA and sham mice following injection of BAY 11-7082 (two-way ANOVA followed by Sidak’s multiple comparison test, n = 8 per group). (e) Time of immobility of CFA and sham mice in forced swimming test following injection of BAY 11-7082 (*p < 0.05, **p < 0.01, two-sample t-test, n = 8 per group). (f) Preference for sucrose of CFA and sham mice in the sucrose preference test following injection of BAY 11-7082 (**p < 0.01, ***p < 0.001, two-sample t-test, n = 8 per group). (g) Representative traces of CFA and sham mice in the open field test following injection of BAY 11-7082. (h–j) Statistical analysis of CFA and sham mice for time in the center, distance in the center and total distance in the open field following injection of BAY 11-7082 (**p < 0.01, ***p < 0.001, two-sample t-test, n = 8 per group).
Discussion
Chronic pain and depressive symptoms often coexist and interact with each other, yet our understanding of the molecular mechanisms underlying both conditions remains limited, hindering the development of effective treatments. In this study, we demonstrate the critical role of the TRIM14-NF-κB pathway in regulating chronic pain and depression-like behaviors in CFA mice. These findings lay a foundation for further exploration of the molecular pathways involved in both chronic pain and depression and may offer a theoretical basis for targeted therapeutic interventions.
Depression is a common emotional disorder typically triggered by pain and chronic stress. 31 Multiple studies have indicated that pain and depression-like behaviors are closely associated with abnormal functioning of the anterior cingulate cortex (ACC), suggesting that the ACC plays a key role in modulating both pain and depressive-like behaviors.14,32–35 Changes in synaptic plasticity are a key mechanism underlying chronic pain and emotional disorders.36,37 Previous studies have identified two distinct forms of long-term potentiation (LTP) in the ACC: postsynaptic LTP and presynaptic LTP. The induction of postsynaptic LTP requires activation of NMDA receptors, while its maintenance depends on the activity of an atypical protein kinase C (PKC) isozyme in postsynaptic neurons, this mechanism sustains the affective component of chronic pain. In contrast, presynaptic LTP induction involves kainate receptor activation, with its maintenance relying on enhanced glutamate release that may contribute to pain-related anxiety. These two distinct forms of LTP give highly potent evidence for the mechanisms underlying the generation of chronic pain and emotional disorders.9,37 In this study, although conclusive evidence is lacking, we propose that the significant enhancement of postsynaptic LTP in the ACC of CFA mice may be a key factor driving the comorbidity of pain and depression-like behaviors. This hypothesis is supported by the observation that presynaptic LTP primarily mediates anxiety-related behaviors in mice, whereas its association with depression-like behavior appears less direct. Furthermore, our data demonstrate marked activation of postsynaptic neurons in the ACC of CFA mice, suggesting that synaptic plasticity alterations predominantly occur in neuronal compartments rather than presynaptic terminals. Notably, our current findings do not address potential changes in the expression or function of NMDA receptors (NMDARs) and AMPA receptors (AMPARs), this limitation precludes definitive exclusion of presynaptic LTP’s potential contribution to the observed phenotypes. These specific mechanisms will be systematically investigated in future studies.
A previous study identified KDM6B in the ACC as a key regulator of visceral pain and associated depression-like behaviors. 38 However, the molecular mechanisms underlying ACC-mediated inflammatory pain and depression remain largely unexplored. Here, we reveal that the upregulation of TRIM14 in the ACC is a critical factor in the development of inflammatory pain and concomitant depression-like behaviors. Interestingly, while there was no change in TRIM14 expression in the ACC during the acute pain phase (within 1 week), TRIM14 expression significantly increased during the stable phase of chronic pain in mice. Knockdown of TRIM14 reduced NF-κB phosphorylation and significantly alleviated both pain and depression-like behaviors. These findings suggest that the TRIM14-NF-κB pathway may represent a novel target for the treatment of chronic pain and depression-like behaviors. As a member of the E3 ubiquitin ligase family, TRIM14 has been shown to play important roles in various cancers, including gastric, cervical, and breast cancers.39–41 Additionally, the TRIM14-USP14-BRCC3 complex has been reported to alleviate inflammation by stabilizing KDM4D and preventing its degradation. 19 TRIM14-USP14 also promotes a stable response of cGAS to viral infections. However, to the best of our knowledge, the role of TRIM14 in pain and depression-like behaviors has not been well studied. Our findings therefore highlight the novel function of TRIM14 in mediating both pain and depression-like behaviors.
NF-κB is a protein that regulates DNA transcription and plays a crucial role in the immune response, mediating cellular reactions to stress, cytokines, and free radicals.42,43 The activation of NF-κB is controlled by interactions with IκB proteins. In inactive cells, NF-κB resides in the cytoplasm, bound to IκB proteins, keeping it in an inactive state. Upon external stimulation, IκB is phosphorylated, leading to the activation and translocation of NF-κB into the nucleus. NF-κB is widely expressed in the central nervous system, and its function is closely linked to its cellular location. 44 Previous studies have shown that NF-κB expression in the hippocampus plays an essential role in the development of depression-like behaviors in mice.45,46 Notably, our data reveal that NF-κB expression in the ACC also plays a pivotal role in the regulation of depression-like behaviors, providing a potential new target for therapeutic interventions in depression. Our immunofluorescence results demonstrated that NF-κB was predominantly expressed in neurons in the ACC, while in the hippocampus, it was mainly localized to glial cells. 44 This suggests that NF-κB may function through different pathways depending on its location, opening up new possibilities for drug development targeting specific brain regions. Previous research has shown that knockdown of TRIM14 significantly inhibits the activation of the atypical NF-κB pathway in vivo. 47 Additionally, TRIM14 can regulate the classical NF-κB pathway by recruiting USP14, forming the TRIM14-USP14 complex, which modulates IκBα through K63-linked ubiquitination.47,48 Our findings support this model, as we observed that TRIM14 expression in the ACC region of CFA mice closely correlated with changes in the phosphorylation of NF-κB, and knockdown of TRIM14 resulted in decreased p-NF-κB/NF-κB expression. Notably, there were no data from female mice in this study. Previous studies have established that estrogen plays a significant role in pain modulation.49,50 Furthermore, emerging evidence suggests that the menstrual cycle may influence the processing of pain signals.51,52 To control for these potential confounding factors, we exclusively used male mice in the current study. This methodological choice allows clearer isolation of pain mechanisms independent of estrogen-related variables. However, single-sex mice are a limitation of this study and systematic investigation of sex differences in pain modulation is necessary in subsequent research.
In summary, our study highlights the critical role of the TRIM14-NF-κB pathway in the development of inflammatory pain and depression-like behaviors. These findings provide new insights into the mechanisms by which TRIM14 and NF-κB modulate these conditions, offering a foundation for the development of targeted therapies for pain and depression.
Footnotes
Author contributions
J-H.D, Z-H.X, and Q-L.L performed experiments and analyzed data. J.H, Z.N, and C-H.Z analyzed data and prepared manuscript. S.H, R.S analyzed data and revised the manuscript. Y-C.L designed experiments, supervised the experiments, and finalized the manuscript. All the authors have read and approved the paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from Suzhou Technology Project (SKYD2023051), Zhangjiagang Technology Project for Youth (ZJGQNKJ202204), the Basic Frontier Innovation Cross Program of Suzhou Medical College (YXY2304059) and Postdoctoral Fellowship Program of CPSF under Grant Number GZC20231890.
