Abstract
Keywords
Introduction
Primary dysmenorrhea (PDM), affecting 50%–90% of young women,1,2 is characterized by cyclic lower abdominal cramps during menstruation and can be associated with anxiety, depression, and irritability. 3 Brain changes observed during and outside menstrual pain episodes may be adaptive (beneficial) or maladaptive (detrimental), reflecting complex central pain processing.4–6 Long-term recurrent menstrual pain can lead to adaptive brain reorganization, shifting from emotional to cognitive pain modulation, 7 and adaptive disconnection of the default mode network to prevent central sensitization in Asian females. 4 Prolonged menstrual pain is also linked to changes in brain metabolism and connectivity in areas critical for emotional and cognitive pain processing, including the prefrontal cortex, insula, and striatum.3,5,6
The reward system (RS) includes neural structures crucial for experiencing pleasure, such as the ventral tegmental area (VTA), ventral striatum (notably the nucleus accumbens [NAc] and part of the putamen), ventral pallidum, anterior cingulate cortex, and orbital prefrontal cortex. Additional regions like the insula, amygdala, hippocampus, thalamus, and dorsal prefrontal cortex also play significant roles. 8 This system’s interaction with pain is multifaceted. 9 Pleasure can reduce pain perception by releasing neurotransmitters like endorphins and dopamine, 10 and it can modulate pain through pathways from the NAc, amygdala, VTA, and basal ganglia to the periaqueductal gray for descending pain inhibition.11–14 The RS also influences pain on emotional and cognitive levels.15,16 The VTA’s dopaminergic and the amygdala’s glutamatergic neurons trigger avoidance motivations, signaling to the NAc. 17 The putamen, along with the NAc, aids in learning reinforcement and reward processing, facilitating motivational behavior. 18 Pain relief itself may trigger a rewarding pleasure response, primarily through the NAc and VTA.10,19 Decision-making and attention, influenced by coping strategies or behaviors in response to pain, also impact pain perception.20,21 The NAc integrates nociceptive inputs from regions like the thalamus and amygdala, while the insula and prefrontal cortex, connected to the NAc, combine sensory, emotional, and cognitive aspects for pain modulation.22–24
Acute pain stimuli can activate the RS for multidimensional pain modulation, 25 while chronic pain can alter the brain’s RS, impairing the function of regions like the NAc and prefrontal cortex, and diminishing the ability to process and enjoy analgesic rewards. 26 Research on PDM during pain-free periods has shown decreased NAc functional connectivity, with the reduction’s extent linked to menstrual pain intensity and prostaglandin levels, compared to individuals without PDM. 27 Liu et al. (2023) identified changes in the mesocorticolimbic pathways during the menstrual phase of PDM, indicating both beneficial and harmful adjustments. 28 However, no studies have explored the intrinsic functional connectivity (FC) changes in the RS between menstrual pain and pain-free cycles, nor the connection between the FC of reward regions and menstrual pain experience.
Our study aims to investigate how menstrual pain affects the RS’s FC neurodynamics. Using resting-state functional magnetic resonance imaging (rsfMRI), we analyzed the RS in PDM subjects through regional homogeneity (ReHo) and amplitude of low-frequency fluctuations (ALFF) methods. These are two critical techniques for analyzing the blood-oxygen-level-dependent (BOLD) signal to deduce brain activity. ReHo evaluates the synchrony of BOLD signal fluctuations in adjacent voxels using Kendall’s coefficient,29,30 with higher ReHo hinting at tighter local synchronization. Higher ReHo values also implicate increased functional segregation from other brain areas, while lower values indicate more functional integration7,31 or impaired processing.29,32 ALFF measures the magnitude of low-frequency oscillations (0.01–0.1 Hz) within the BOLD signal, which can indicate the level of neural activity during rest, with greater ALFF values signaling more robust neural activity.33,34 These tools help identify unusual brain states associated with disorders like depression, schizophrenia, and Alzheimer’s, therefore contributing to our understanding of brain functionality and its impairments in different conditions. By combining ReHo’s coherence focus and ALFF’s activity intensity focus, one gains complementary insights into brain function. We compared ReHo and ALFF in reward-related areas across menstrual phases and linked these measurements to pain experiences, aiming to reveal the interaction between the RS and menstrual pain in PDM individuals. We hypothesized that the RS’s regional neurodynamics vary across menstrual phases, showing multidimensional pain modulation during painful periods and potential reward-like reactions during pain-free peri-ovulatory phases.
Materials and method
Subjects
Participants for our study were enrolled based on specific criteria. Women with PDM had to have a regular menstrual cycle lasting between 27-32 days, be right-handed (as determined by the Edinburgh Handedness Inventory), and experience menstrual pain for over six months with an average pain intensity greater than four on a 0-10 numerical rating scale (NRS), despite routine management for PDM. Healthy female controls (CON) met the same criteria, except they experienced no pain during menses (NRS = 0). Exclusion criteria included recent use (within six months) of any centrally acting medications, contraceptives, or hormone supplements; having a pituitary gland disorder; suffering from any organic pelvic diseases; having psychiatric or neurological disorders; suffering a head injury resulting in loss of consciousness; being pregnant, planning to become pregnant, or having a history of childbirth; and having metal implants, pacemakers, claustrophobia, or any other contraindications to MRI. Participants were required to refrain from taking painkillers 24 h before the study. All PDM subjects were diagnosed by a gynecologist and underwent a pelvic ultrasound to exclude the presence of organic pelvic diseases.
Experimental design
We conducted a comprehensive assessment of participants at two distinct phases of their menstrual cycle: during menstruation (days 1-3, MENS phase) and during the peri-ovulatory phase (days 12-16, POV phase). This evaluation included psychological assessments, blood samples for gonadal hormone analysis, and two brain MRI scans (including T1 and rsfMRI images). To measure pain experiences in PDM participants during the MENS phase, we used the short-form McGill Pain Questionnaire (MPQ). 35 Additionally, to assess negative thought patterns and pain-related emotions in all participants (both PDM and CON) during both phases, we utilized the Pain Catastrophizing Scale (PCS), Beck Anxiety Inventory, and Beck Depression Inventory.
Analysis of resting-state functional MRI data
Image acquisition
rsfMRI images for all participants were acquired using a 3.0 T MRI scanner (Magnetom Trio Tim, Siemens, Erlangen, Germany) at the National Yang Ming Chiao Tung University, equipped with a 12-channel head coil. T2*-weighted gradient echoplanar imaging (EPI) sequences were used with the following specifications: repetition time (TR) of 2500 ms, echo time (TE) of 30 ms, flip angle of 90°, field of view (FOV) of 220 mm × 220 mm, matrix size of 64 × 64, and slice thickness of 3.4 mm. Each scanning run comprised 200 volumes. High-resolution T1-weighted structural images were also obtained using the MPRAGE sequence with TR/TE = 2530 ms/3.03 ms, flip angle = 7°, FOV = 224 mm × 256 mm, matrix size of 224 × 256, and slice thickness of 1 mm. The initial three EPI scans of each rsfMRI series were discarded to allow for signal saturation and magnetic field stabilization. Participants were instructed to stay awake with their eyes open, heads still but relaxed, and not to engage in specific thoughts. Head cushions and earplugs were provided to minimize head movement and reduce scanner noise.
Image preprocessing
We preprocessed the fMRI data using the Data Processing Assistant for Resting-State fMRI (DPARSF) V5.2 advanced edition, built on the Data Processing and Analysis of Brain Imaging (DPABI) toolbox V6.0 36 and Statistical Parametrical Mapping 12 (SPM12) in MATLAB R2018b. The preprocessing steps included slice timing correction and realignment for head motion correction, with participants showing head motion exceeding 2 mm displacement or 2° rotation being excluded. T1-weighted images were co-registered with the mean functional image using intra-subject spatial alignment, followed by segmentation into gray matter, white matter, and cerebrospinal fluid. We performed nuisance regression using the Friston 24-parameter model 37 and default masks from SPM to regress out head motion parameters and signals from white matter and cerebrospinal fluid. Spatial normalization was done using a study-specific DARTEL template, 38 transformed to the Montreal Neurological Institute (MNI-152) space, with image resampling to 3 mm isotropic voxels. Temporal band-pass filtering (0.01 to 0.1 Hz) was applied to reduce high-frequency noise and low-frequency drift. The data were then smoothed using a Gaussian kernel with a 6 mm full-width at half-maximum (FWHM). Global signal regression GSR was not performed to avoid exaggerating negative correlations 39 and distorting between-group differences. 40
ReHo and ALFF exploration
Our study used DPABI software (https://rfmri.org/DPABI) 36 for ALFF and ReHo analyses. For ReHo analysis, we applied Kendall’s coefficient of concordance (KCC) to unsmoothed image data to create maps evaluating the synchrony of time series data between a voxel and its 26 closest neighbors. 29 These ReHo maps were normalized into z-score maps and smoothed with a Gaussian kernel of 6 mm full-width at half-maximum (FWHM) for further statistical analysis. 41 For ALFF analysis, we calculated each voxel’s power spectrum within the 0.01-0.1 Hz frequency range using Fast Fourier Transform (FFT) to convert the voxel time series to the frequency domain. 42 The power spectrum maps were normalized to z-score maps by dividing each voxel’s power by the global mean. Non-brain and background artifacts were filtered out, producing detailed whole-brain ALFF maps for further statistical analysis.
Statistical analysis
Demographic and psychological inventory assessment
Data analysis was conducted using GraphPad Prism 9 (version 9.1.1). The D’Agostino-Pearson omnibus normality test was used to assess data distribution. For normally distributed data, parametric tests such as two-sample and paired t-tests were applied. For non-normally distributed data, non-parametric tests like the Mann-Whitney U test for independent samples and the Wilcoxon signed-rank test for paired samples were used. Demographic differences between groups were assessed with two-sample t-tests, and psychological measures across phases were examined with paired t-tests. Results were presented as mean ± standard deviation (SD), with a p-value of less than 0.05 indicating statistical significance.
Voxel-based image statistical analysis
All image analyses were performed using a pre-defined RS mask created from a probabilistic map associated with “reward” from meta-analyses available on Neurosynth.org 43 (Supplemental Figure 1). This mask includes the VTA, NAc, amygdala, basal ganglion, insula, hippocampus, prefrontal cortex, sensory-motor cortex, and superior/inferior parietal lobe. Gonadal hormones influences were controlled by regressing them out as non-interest covariates, with detailed hormone measurement methods provided in the Supplemental material. Due to the additional pain experienced by PDM individuals beyond menstrual symptoms, we did not use the traditional 2 × 2 ANOVA for evaluating the main effect of group-condition and its interaction. Instead, we used planned contrast paired t-tests within ReHo and ALFF analyses to compare menstrual cycle phases. For comparisons between PDM subjects and control participants, planned contrast two-sample t-tests were used within ReHo and ALFF analyses via the DPABI software framework. The initial threshold for statistical significance was set at p < .005 at uncorrected voxel levels to preserve nuanced details. To protect against type I errors, we applied family-wise error (FWE) correction at the cluster level with a significance threshold of p < .05. For illustration and further correlation analyses, the averaged ReHo and ALFF values were extracted from regions of significant between-group or between-phase contrasts using the DPABI viewer and ROI signal extractor.
ROI-based image statistical analysis
Given the crucial role of the NAc and VTA in the mesolimbic circuit for pain modulation and reward processing,15,44 we conducted region of interest (ROI) analyses specifically targeting these regions. The NAc mask was created using the Harvard-Oxford Subcortical Atlas, 45 and the VTA mask was based on the 7T MRI atlas. 46 These masks were manually delineated using FSLeyes 47 (Supplemental Figure 1). We extracted mean values from the ReHo and ALFF z-score maps for these regions to perform subsequent statistical analyses between different phases. A p-value of less than 0.05 was considered statistical significance.
Correlation analysis
To explore the relationship between ReHo and ALFF measurements and pain-related assessments, we examined correlations between average ReHo and ALFF values (from significant areas) and various pain scales [pain rating index (PRI) and present pain index (PPI) subscales of the MPQ, and PCS] during the MENS and POV phases separately. Statistical significance was established at a p-value of <0.05, with a Bonferroni correction adjusting the significance level to 0.0166 due to the evaluation of three measures. We reasoned that the RS’s operation varies between menstrual phases, with intense pain highlighting more significant differences. Thus, we segmented PDM individuals into two groups based on their self-reported NRS scores: moderate (4 ≤ NRS <7) and severe (NRS ≥7). Subsequent analyses tailored to each subgroup examined phase-dependent differences in brain regions, particularly within the NAc and VTA, to investigate ReHo and ALFF patterns during various pain stages.
Results
Baseline information and demographic data
Participants were recruited via internet advertisements, resulting in 201 subjects with PDM and 169 controls. Nine PDM subjects were excluded due to secondary dysmenorrhea detected by pelvic ultrasound, and four controls were excluded for other pain experiences. Additionally, 18 PDM subjects and one control were excluded due to incidental brain findings, and 68 PDM subjects and 56 controls declined participation. Ultimately, 106 PDM subjects and 108 controls completed the MRI study, but 14 PDM subjects and 18 controls were excluded due to excessive head motion during scanning. The final cohort consisted of 92 individuals with PDM and 90 controls, all meeting stringent inclusion criteria and quality standards for neuroimaging data (Supplemental Figure 2).
Demographic data and baseline information.
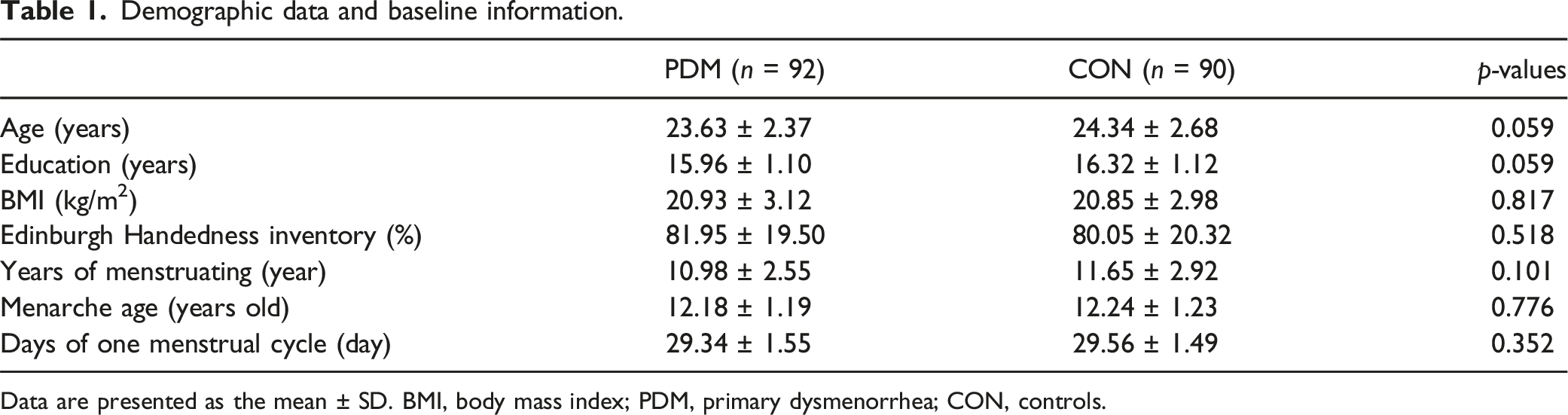
Data are presented as the mean ± SD. BMI, body mass index; PDM, primary dysmenorrhea; CON, controls.
Psychological measurements
In the PDM group, significant phase differences (MENS > POV) were observed in the PCS: total score (p = .0007), rumination (p = .0005), helplessness (p = .0068), and magnification (p = .0018). The Beck Anxiety Inventory and Beck Depression Inventory also showed significant differences between phases (both MENS > POV, p < .0001). During the MENS phase, the MPQ scales recorded a PRI total score of 30.98 ± 14.05, sensory scores averaging 17.17 ± 7.12, affective scores at 3.86 ± 2.82, evaluation scores at 2.52 ± 2.03, miscellaneous scores at 7.43 ± 4.22, and a PPI score of 2.65 ± 1.08 (Supplemental Table 1).
Voxel-based image statistical results
ReHo analysis
Between-phase comparisons
During the MENS phase, the PDM group exhibited lower ReHo values in the left putamen and right amygdala compared to the POV phase. This difference was not observed in the CON group (see Table 2, Figure 1(a)).
Result of voxel-based image statistical analysis.

Peak coordinates refer to the MNI space. Significance was set at the uncorrected voxel level p < .005 followed by the family-wise error corrected cluster level p < .05; ReHo, regional homogeneity; ALFF, amplitude of low frequency fluctuations; PDM, primary dysmenorrhea; CON, control; MENS, menstrual phase; POV, peri-ovulatory phase; R, right; L, left; NS, non-significant.
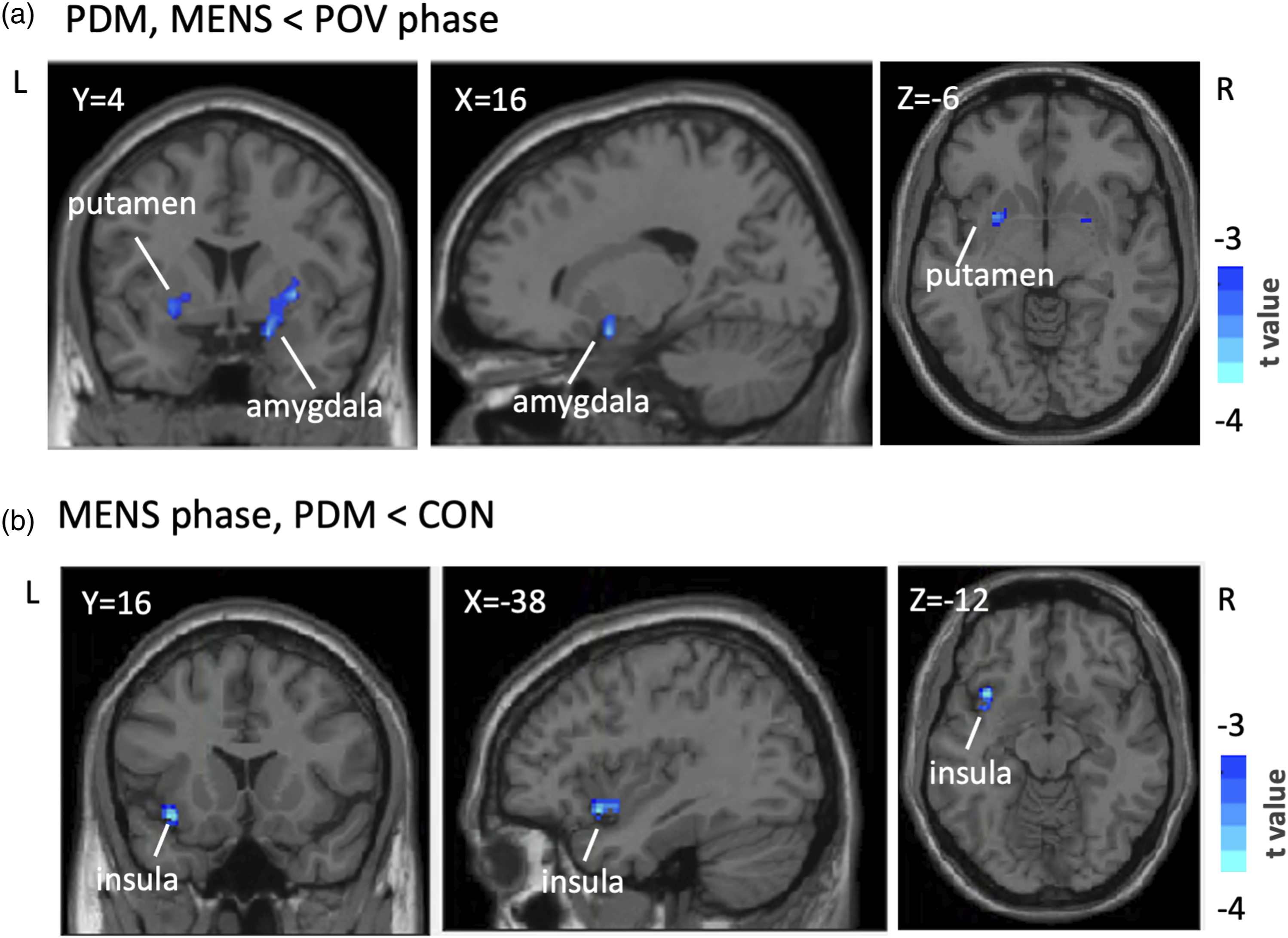
Voxel-Based ReHo Analysis in the reward network. Presented are the outcomes of voxel-based ReHo analyses, illustrating phase and group discrepancies within a reward-oriented mask defined on Neurosyn.org. (a) Identifies significant reductions in ReHo values (blue) in the left putamen and right amygdala during the MENS phase compared to the POV phase in PDM. (b) Shows the left insula’s marked decrease in ReHo values in PDM subjects compared to controls during the MENS phase. These clusters reached statistical significance at an uncorrected voxel-level threshold of p < .005, with cluster significance maintained under FWE correction at p < .05. Results are overlaid on the DPABI T1 anatomical template, with t scores represented on the accompanying color scale. Table 2 details these ReHo findings extensively. Abbreviaitons: ReHo, regional homogeneity; MENS, menstrual phase; POV, periovulatory phase; PDM, primary dysmenorrhea; CON, control; R, right; L, left; DPABI, Data Processing and Analysis of Brain Imaging.
Between-group comparisons
During the MENS phase, the PDM group showed lower ReHo values in the left insula relative to the CON group. This disparity was not present during the POV phase (See Table 2, Figure 1(b)).
ALFF analysis
Between-phase comparisons
No significant differences in ALFF values were observed between phases in either the PDM or CON groups.
Between-group comparisons
During the MENS phase, the PDM group had significantly lower ALFF values in the left putamen compared to the CON group. Lower ALFF values were also observed in the bilateral putamen of the PDM group during the POV phase (see Table 2, Figure 2). Voxel-based ALFF Analysis in the reward network. Presented are the findings from ALFF analyses indicating phase-specific differences within a reward-oriented mask established on Neurosyn.org. (a) Identifies notable ALFF value decreases in the left putamen of the PDM group in contrast to the CON group during the MENS phase, depicted in blue. (b) Depicts ALFF value decreases in both sides of the putamen in PDM subjects as opposed to CON subjects during the POV phase. These findings achieved statistical significance at an initial uncorrected voxel-level p-value of <0.005, with subsequent confirmation at a cluster level after FWE correction at p < .05. Displayed on the DPABI T1 template, the color bar signifies the range of t scores. Detailed ALFF analysis results are cataloged in Table 2. Abbreviations: ALFF, the amplitude of low-frequency fluctuations; PDM, primary dysmenorrhea; CON, control; MENS, menstrual phase; POV, periovulatory phase; R, right; L, left; DPABI, Data Processing and Analysis of Brain Imaging.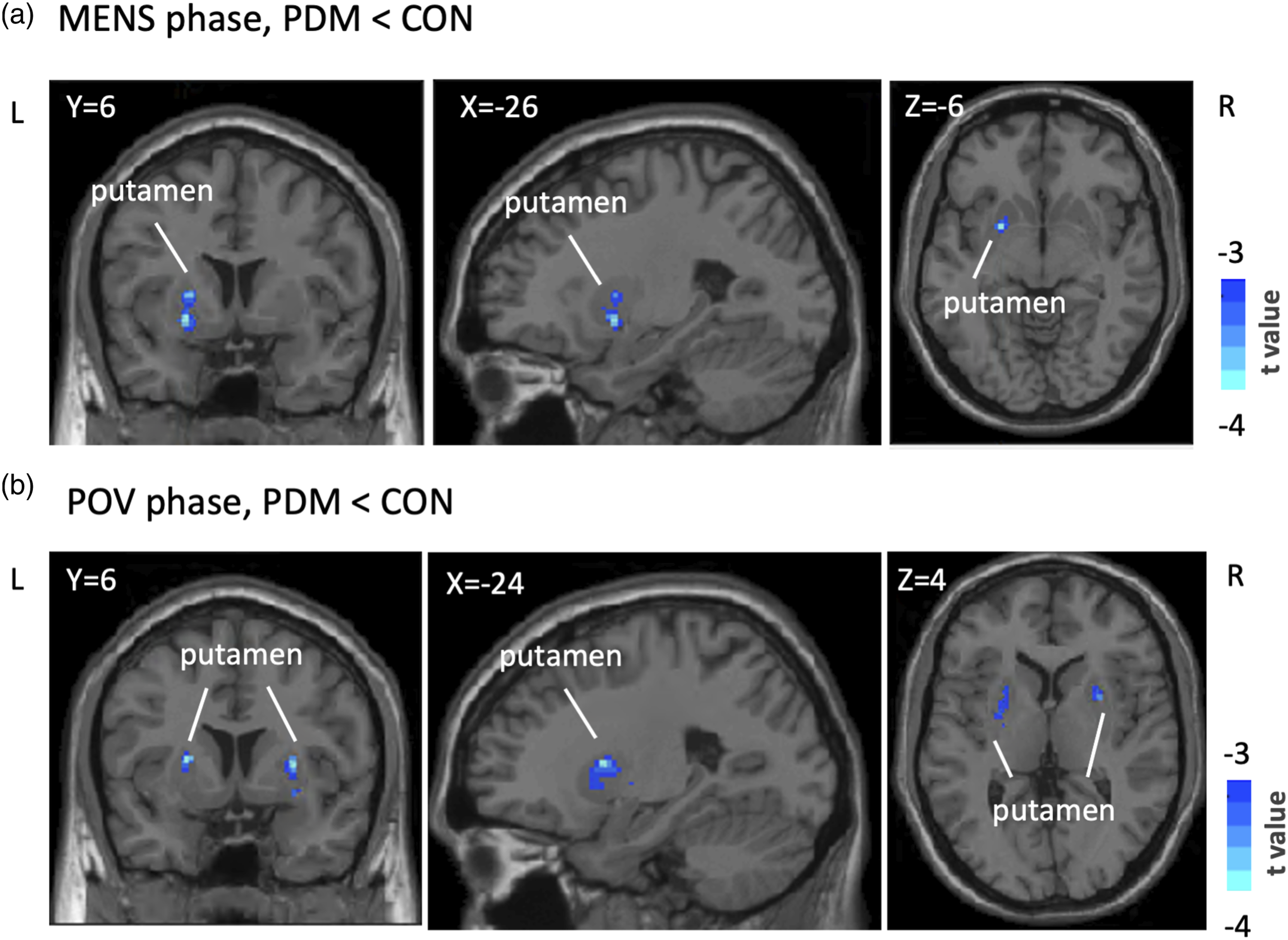
ROI-based image statistical results
In the between-phase analysis, there were no significant differences in either ALFF or ReHo values in the bilateral NAc and VTA (Supplemental Figure 3, A and B).
Correlation results
In areas with notable inter-group disparities, such as the insula in ReHo studies and the putamen in ALFF analyses, no significant correlations with pain scales were observed across the entire cohort (Supplemental Table 2).
During MENS
For the entire PDM cohort, a positive correlation was identified between ReHo values in the right amygdala and the PRI (Figure 3(a)). The NAc showed a positive correlation between both ALFF and ReHo values with the PCS, PRI, and PPI (Figures 4(a) and (b), Supplemental Table 2 and 3), while the VTA exhibited a negative correlation between ALFF values and PCS, PRI, and PPI (Figure 4(c)). Within the moderate group, the NAc displayed a positive correlation between ReHo values and PPI, and the VTA showed a negative correlation between ALFF values and PPI (Supplemental Table 3). In the severe group, a positive correlation was found between ReHo values in the left putamen and PRI (Figure 3(b)), and between ReHo or ALFF values in the right amygdala and PRI and PPI (Figures 3(a) and (c)). The NAc exhibited a positive correlation between ReHo values and both PRI and PCS, with stronger correlation coefficients in comparison to the overall cohort (Figures 4(a) and (b)). Detailed correlation analysis results are cataloged in Supplemental Tables 2 and 3. Phase-dependent correlation patterns in significant regions. This figure illustrates the correlation outcomes between ReHo and ALFF metrics and pain scales in subjects with overall and severe PDM (NRS ≥7), focusing on regions with significant inter-phase variations. (a) Shows a positive correlation between right amygdala ReHo values and PRI scores in the overall PDM cohort during the MENS phase, with a stronger correlation in the severe group. (b) Demonstrates a positive correlation between left putamen ReHo values and the PRI scores in the severe PDM group during the MENS phase. (c) and (d) shows ALFF values in the right amygdala, which positive correlate with PRI scores during the MENS phase but a negative correlation with the PCS scores during the POV phase in the severe PDM group. Supplemental Tables 2 and 3 provide detailed correlation results. PDM, primary dysmenorrhea; ReHo, regional homogeneity; ALFF, amplitude of low-frequency fluctuations; NRS, Numerical rating scale; MENS, menstrual phase; POV, periovulatory phase; R, right; L, left; PCS, pain catastrophizing scale; PRI, pain rating index. (z) Means z transformed mean value of ReHo and ALFF. Correlation Patterns of of NAc and VTA. This figure elucidates the ROI-based analysis of the NAc and VTA correlations with pain scales in the overall and severe PDM groups. (a) and (b) demonstrate a positive correlation between NAc ReHo values and PCS and PRI scores during the MENS phase, especially in the severe group. (c) Shows a negative correlation between ALFF values in the VTA and PPI during the MENS phase in the overall PDM group. Detailed correlation metrics are cataloged in Supplemental Tables 2 and 3 Abbreviations: ROI, region of interest; NAc, nucleus accumbens; VTA, ventral tegmental area; PDM, primary dysmenorrhea; ReHo, regional homogeneity; ALFF, amplitude of low-frequency fluctuations; MENS, menstrual phase; PCS, pain catastrophizing scale; PRI, pain rating index; PPI, present pain intensity. (z) Means z transformed mean values of ReHo and ALFF.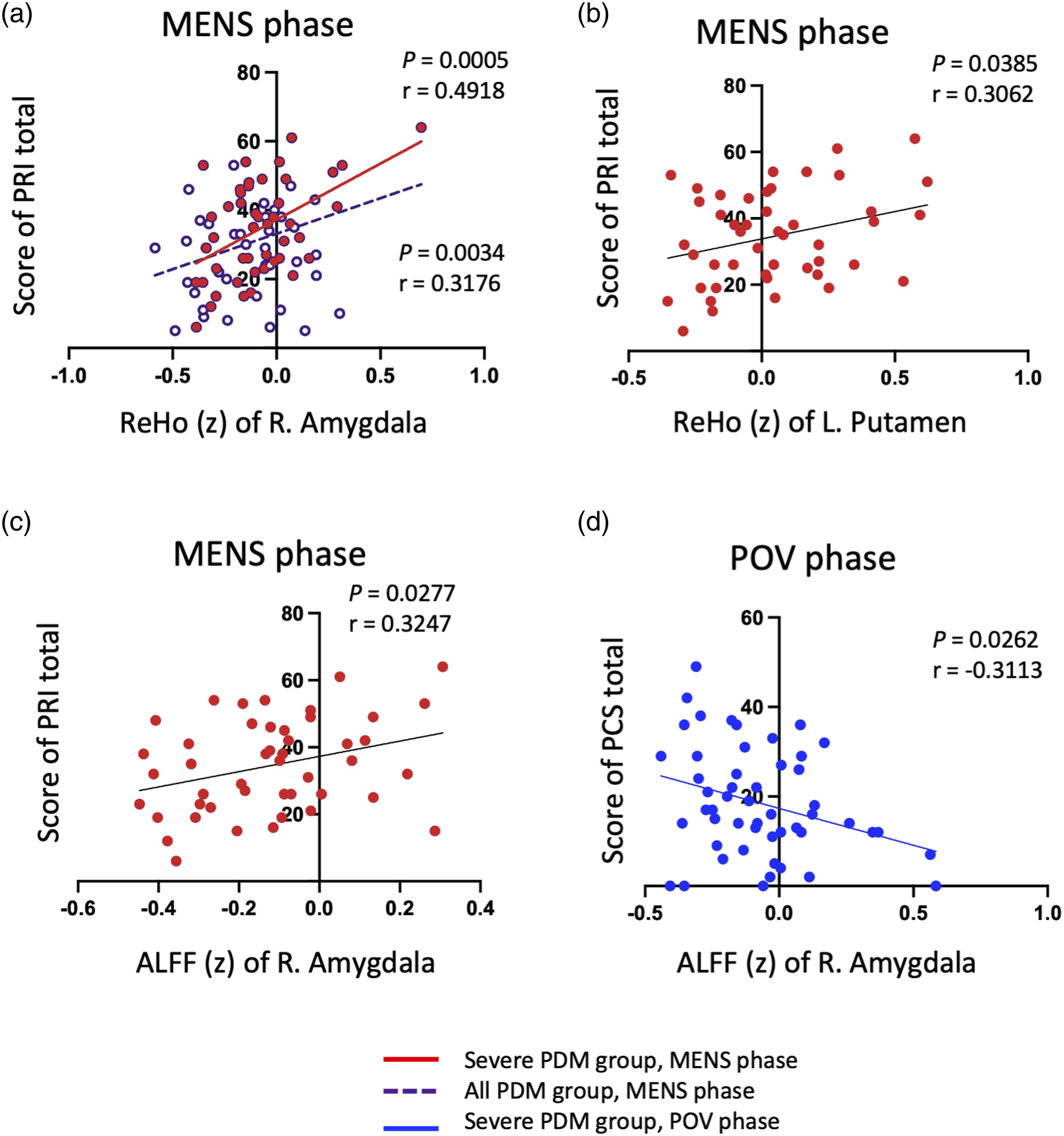

During POV phases
During the POV phase, a negative correlation was observed between ALFF values in the amygdala and PCS scores (Figure 3(d), Supplemental Table 3).
Discussion
Our findings highlight the RS’s varied responses during menstrual pain and pain-free periods in individuals with PDM, underscoring its vital role in dysmenorrhea management. This involves mechanisms for pain avoidance, appreciation of pain relief as a reward, and cognitive coping strategies throughout the menstrual cycle, demonstrating the RS’s adaptability in PDM. Our research provides a comprehensive view of how different neural substrates within the brain’s RS correlate with pain perception and modulation in individuals with PDM.
On-pain processing of the RS during MENS phase
Our research demonstrates a significant positive correlation between ReHo and ALFF levels in the NAc and amygdala with pain scales (PRI, PCS, and PPI) during MENS, particularly in subjects with severe PDM (Figures 3 and 4). The PCS reflects negative pain perceptions and thoughts, 48 while the MPQ (PRI total scores) captures the multidimensional aspects of pain, including its emotional dimension. 35 The positive correlation between ReHo and ALFF in the NAc and amygdala with pain metrics (PCS, PRI, and PPI) highlights an emotional drive to avoid pain or negative feelings during MENS. Negative (pain) and positive (relief) emotions drive behaviors, prompting avoidance or pursuit actions, which help in learning from past experiences. 49 The NAc integrates dopamine and amygdala inputs to fuel reward-seeking and motivation, assessing the value of rewards.8,50–53 The amygdala processes survival-related emotions like pain-related fear and pain-relief related pleasure, 54 playing a role in pain control and emotional processing. 55
Menstrual pain may prompt actions to alleviate discomfort. The positive link between ReHo value of the putamen and PRI scores during MEMS in those with severe menstrual pain (Figure 3(b)) suggests the putamen’s role in initiating motivational avoidance behaviors, such as quiescence or altered posture, as adaptive responses to pain. The putamen, part of the basal ganglia, is crucial for motor coordination, 56 learning, 57 and pain management. 12 Its connections with motor and reward pathways influence behaviors aimed at pain mitigation and anticipating pain relief rewards.12,58,59
ALFF values in the putamen for the PDM group are lower than those in the CON group during MENS and POV phases (Figure 2), indicating adaptive neuromodulation. After years of regular menstrual pain, the putamen might initiate motivated behavior with reduced neural activity. Pain relief acts as a motivator, using negative reinforcement to encourage actions that alleviate pain,17,60,61 leading to decreased neural activity as an adaptive mechanism. 62
In contrast to the NAc, bilateral VTA ALFF values negatively correlate with current PPI during the MENS phase (Figure 4(c)), suggesting that VTA activity may modulate the pain experience. The VTA’s dopaminergic projections influence motivational behavior, mood, and cognition by targeting brain regions like the NAc, prefrontal cortex, amygdala, and hippocampus.63–66 Additionally, VTA signals to the periaqueductal gray may help modulate pain signals.67,68 Deep brain stimulation in the VTA can alleviate pain severity and anxiety, 69 indicating its potential role in pain modulation, either indirectly through cognition and emotion or directly impacting descending pain pathways.
During the MENS phase, individuals with PDM exhibited decreased ReHo values in the left insula compared to the CON group (Table 2, Figure 1(b)), indicating reduced local synchrony, may represent an adaptive mechanism for managing dysmenorrheic pain and suppressing pain emotions. While the insula is not primarily dedicated to reward processing, it is integral to the network governing reward-related behavior. 15 The anterior insula, which includes areas connected to cognitive functions and emotional regulation,24,70,71 showed significant changes in our study. Despite the primary difference being menstrual pain, we found no correlation between insula ReHo values and pain scales (Supplemental Table 2).
Off-pain processing of the RS during POV phase
Individuals with PDM exhibit increased ReHo values in the putamen and amygdala during the pain-free POV phase compared to the MENS phase, suggesting potential engagement of the RS in response to pain relief as a reward (Table 2, Figure 1(a)). During the POV phase, the pain-free state can be considered a reward, 19 engages brain regions like the putamen and NAc involved in reward detection and processing.18,72 ReHo and ALFF values in the NAc and VTA show no significant differences between phases (Supplemental Figure 3), this may be due to their dual role in regulating pain during the MENS phase and processing pain relief during the POV phase.
In the severe PDM subgroup, elevated ALFF levels in the amygdala during the MENS phase correlate with higher PRI scores (Figure 3(c)), indicating heightened emotional processing and seeking behavior in response to increased pain intensity. Conversely, during the POV phase, higher ALFF levels in the amygdala correlate with lower PCS scores, suggesting decreased negative thinking or enhanced pleasure due to RS engagement (Figure 3(d)). These findings highlight the differing roles of RS components during pain onset (MENS phase) and offset (POV phase) in individuals with PDM, contributing to phase disparities. This aligns with the idea that the amygdala mediates between pain and pleasure, encoding both negative and positive stimuli associated with punishments and rewards 73 and playing a crucial role in descending pain control. 74
A methodological reappraisal of PDM and RS interactions
Our investigation differs in objectives from the works of Zhang et al. (2019) and Liu et al. (2023), both of which explore the interplay between PDM and the brain’s RS.27,28 While Zhang et al. focused on group differences during the POV phase and Liu et al. on the MENS phase, our study uniquely examines the RS’s response across both pain-present and pain-absent periods. We analyzed reward-related brain regions using multi-dimensional pain assessments and MRI scans during both the MENS and POV phases. Despite both our study and Liu et al.'s using ReHo analysis, our findings with a larger sample size did not replicate Liu et al.'s observation of enhanced NAc response during the MENS phase. This discrepancy may be due to our adjustment for gonadal hormonal effects on BOLD signals, brain activity, and functional connectivity.75,76 Liu et al. suggest that heightened NAc ReHo and increased functional connectivity in the supraorbital frontal region aid in pain modulation through descending inhibition, which aligns with our adaptive observations. In contrast, Zhang et al. noted reduced NAc-VTA connectivity in PDM subjects without accounting for hormonal adjustments. The link between PDM and an atypical RS warrants further investigation.
Limitations and further consideration
The study has several limitations. Firstly, offset analgesia typically refers to the immediate reward response following the cessation of pain. However, our imaging was conducted during the pain-free phase, specifically during the POV phase, which may not capture the peak reward response in participants with PDM. Secondly, we used pain-focused behavioral scales to assess brain-behavior correlations. Future research may incorporate reward-specific tasks to better understand the RS’s role in pain management, providing a more precise measure of the various reward responses. 19 Thirdly, the study involved 92 PDM and 90 CON participants, but the imaging contrast data’s significance was only at a relatively liberal threshold, indicating a subtle reward response mechanism in PDM, possibly due to its cyclic pain pattern, distinct from continuous chronic pain conditions.26,44,77 Despite the relatively weak statistical backing, we included discussions on these results to capture nuanced data that may offer key insights for future research. 78 Finally, genetic factors influence chronic pain conditions, with previous research on PDM suggesting that individual differences in pain modulation may be linked to genetics.79–84 Future neuroimaging studies should investigate how gene polymorphisms, such as Catechol-O-Methyltransferase Val158Met 64 or mu-opioid receptor A118G, 85 affect the relationship between pain and the reward experience in pain-free states.
Conclusions
Combining data from ReHo and ALFF studies, our findings suggest that the RS in individuals with PDM adapts to pain through changes in motivational, emotional, pain-relieving, and potentially cognitive processes. Various brain regions within this system modulate their activity, showing either enhanced or diminished neural responses in both painful and pain-free contexts. This suggests a potential neural coping mechanism that helps women with PDM manage recurring pain without significant psychological or social difficulties. Our research provides insights into the RS’s response to cyclic pain in young women, which may differ from its response to other acute or chronic pain conditions.
Supplemental Material
Supplemental Material -Neural adaptation of the reward system in primary dysmenorrhea
Supplemental Material for Neural adaptation of the reward system in primary dysmenorrhea by Pei-Shan Hsu, Ching-Hsiung Liu, Ching-Ju Yang, Lin-Chien Lee, Wei-Chi Li, Hsiang-Tai Chao, Li-Fen Chen and Jen-Chuen Hsieh in Molecular Pain.
Footnotes
Acknowledgements
The authors thank all participants in this study and appreciate Intan Low, Tzu-Yi Hong, and Tzu-Ling Tzeng for their technical and experimental help.
Author contributions
Pei-Shan Hsu: Writing - original draft, Conceptualization, Investigation, Formal analysis, Validation, Methodology, Data curation, Validation, Visualization; Ching-Hsiung Liu: Writing - original draft, Conceptualization, Investigation, Formal analysis, Validation, Methodology, Data curation, Visualization; Ching-Ju Yang: Writing - review & editing, Formal analysis, Validation; Lin-Chien Lee: Writing - review & editing, Investigation, Validation; Wei-Chi Li: Writing - review & editing, Investigation, Data curation; Hsiang-Tai Chao: Writing - review & editing Investigation, Resources; Li-Fen Chen: Writing - review & editing Conceptualization, Methodology, Resources, Funding acquisition; Jen-Chuen Hsieh: Writing - review & editing Conceptualization, Methodology, Resources, Funding acquisition, Project administration, Supervision.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Taipei Veterans General Hospital (V100D-001, V100D-001-1, V100D-001-2 and V101C-152), National Science and Technology Council (NSC 100-2314-B010-006-MY3, NSC 100-2629-B-010-001, NSC 102-2629-B-010-001, MOST 103-2321-B-010-020, MOST 106-2629-B-010-001-MY3, MOST 108-2314-B-010-001, MOST 109-2314-B-101-001-MY3 and MOST 109-2314-B-350-001), TVGH-NTUH joint research program (VN103-4, VN104-03, VN105-03), and the Aim for the Top University Plan of the Ministry of Education of National Yang-Ming University. The funders had no role in study design, data collection and analysis, the decision to publish, or manuscript preparation.
Ethical statement
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
