Abstract
Irritable bowel syndrome (IBS) is a common functional gastrointestinal disorder characterized primarily by chronic visceral pain, with a complex pathogenesis and limited efficacy of current therapeutic interventions. Growing evidence indicates that electroacupuncture (EA) and repetitive transcranial magnetic stimulation (rTMS) exert significant analgesic effects on visceral pain. However, the underlying neural circuit mechanisms remain poorly understood. In this study, the ventral part of the lateral septal nucleus (LSV) was identified as a critical brain region mediating the analgesic effects of EA and rTMS in a mouse model of visceral pain. Visceral nociceptive stimulation significantly increased c-Fos expression in the LSV, predominantly within glutamatergic (Glu+) neurons. Optogenetic inhibition of LSV Glu+ neurons attenuated visceral pain, whereas their activation exacerbated pain-related behaviors. Both EA and rTMS individually reduced visceral pain-induced c-Fos expression and alleviated pain behaviors, with the combined EA + rTMS treatment producing a more pronounced analgesic effect than either treatment alone. Moreover, fiber photometry recordings demonstrated that EA and rTMS decreased glutamate release and concurrently increased cannabinoid signaling in the LSV, suggesting that these interventions modulate neurotransmitter dynamics to regulate neuronal excitability. In summary, our findings highlight the pivotal role of LSV Glu+ neurons in the modulation of visceral pain. EA and rTMS exert their therapeutic effects by regulating glutamate and cannabinoid release within this circuit. These insights provide a foundation for developing targeted neuromodulatory strategies for the treatment of chronic visceral pain.
Keywords
Introduction
Irritable bowel syndrome (IBS) is a prevalent functional gastrointestinal disorder affecting approximately 7%–21% of the global population.1–5 It is characterized by recurrent abdominal pain accompanied by altered bowel habits, including diarrhea, constipation, or a mixed pattern.6,7 Among these clinical manifestations, chronic visceral pain is particularly debilitating and represents a primary reason for medical consultation among patients with IBS.8–10 Despite its high prevalence and substantial impact on quality of life, the etiology of IBS-associated visceral pain remains poorly understood, and disease-specific therapeutic targets are lacking. Consequently, current pharmacological treatments often yield suboptimal efficacy and are frequently accompanied by adverse effects. Existing therapeutic strategies largely focus on symptom management—such as dietary modification, pharmacotherapy, and psychological interventions11–15—but these approaches commonly fail to provide sustained pain relief, highlighting the urgent need for innovative therapies and a deeper understanding of the neural mechanisms underlying visceral pain in IBS.
Accumulating evidence supports a critical role for neuromodulation in pain management16–19 Techniques such as electroacupuncture (EA) and repetitive transcranial magnetic stimulation (rTMS) have demonstrated therapeutic potential in neuropathic and inflammatory pain conditions.18,20–23 However, their efficacy and mechanistic basis in chronic visceral pain remain incompletely defined. EA, a modern adaptation of traditional acupuncture, combines electrical stimulation with needle insertion at specific acupoints24–26 and has been shown to modulate endogenous opioid and neurotransmitter release, thereby inhibiting nociceptive transmission and reducing pain perception.27,28 In addition to analgesic effects, EA has been associated with improvements in bowel function, including normalization of bowel habits and reductions in diarrhea or constipation episodes, ultimately enhancing patients’ quality of life.29,30 Similarly, rTMS is a non-invasive neuromodulation technique that employs magnetic fields to stimulate cortical and subcortical brain regions.31–33 By inducing electric currents via a magnetic coil positioned over the scalp, rTMS can modulate neuronal excitability within pain-related regions, including the ventral lateral septal nucleus (LSV), anterior cingulate cortex (ACC), and insular cortex (IC)—key nodes involved in visceral sensory processing.34–38 Through modulation of these central circuits, rTMS has been shown to reduce pain perception and may represent a promising therapeutic approach for visceral pain.36,39 Nevertheless, rTMS predominantly targets central mechanisms and may exert limited effects on peripheral pathways. In addition, optimal stimulation parameters and standardized clinical protocols for IBS have yet to be established.
Both EA and rTMS therefore possess distinct advantages and limitations.40–42 EA relies on precise needle placement and individualized parameter adjustment, which may affect reproducibility and patient compliance, whereas rTMS offers precise control over stimulation parameters and selectively targets central pain circuits, but often requires repeated sessions to achieve sustained efficacy. Given their complementary mechanisms—EA primarily engaging peripheral pathways and rTMS modulating central circuits—combining these modalities may produce synergistic effects on the complex gut–brain axis implicated in IBS. Peripheral EA stimulation, particularly at acupoints along the stomach and large intestine meridians, can elicit neurochemical responses such as cannabinoid release, thereby reducing visceral hypersensitivity.43,44 In parallel, rTMS influences central pain-processing regions, including the LSV, ACC, and IC, attenuating pain perception and disrupting maladaptive central sensitization.45,46
This integrative neuromodulatory strategy has the potential not only to enhance analgesic efficacy but also to overcome the limitations of single-modality interventions. From a translational perspective, elucidating the neural circuit mechanisms underlying the combined effects of EA and rTMS may advance our understanding of IBS pathophysiology, particularly the role of gut–brain axis dysfunction in visceral pain. Clinically, such insights could facilitate the development of more effective and personalized therapeutic approaches, ultimately improving treatment outcomes, reducing side effects, and enhancing quality of life for individuals with IBS. In this study, we investigate whether LSV Glu+ neurons serve as a shared neural substrate mediating the analgesic effects of both EA and rTMS, thereby enabling more robust and sustained pain relief. Our findings provide a mechanistic foundation for the clinical development of novel combinatorial analgesic strategies.
Materials and methods
Animals
Male C57BL/6J mice were housed in the Experimental Animal Center of Soochow University under specific pathogen-free (SPF) conditions. The holding room was maintained on a 12-h light/dark cycle at 24–25°C and 40%–60% relative humidity. Animals had free access to standard chow and autoclaved water. All experimental protocols received approval from the Animal Care and Use Committee of Soochow University and were performed in compliance with the ethical standards of the International Association for the Study of Pain. To eliminate potential hormonal confounders, exclusively male subjects were utilized in this study.
Neonatal colonic inflammation (NCI) model
A model of chronic visceral pain was established in male offspring via neonatal colonic inflammation (NCI), following established methods.47,48 On postnatal day 10 (PND10), pups were randomly allocated to either the NCI or control (CON) group. Under light restraint, a flexible catheter was inserted 1 cm into the rectum. The NCI group received a 30 μL intracolonic instillation of 0.5% acetic acid in saline, which was retained for 30 s, whereas control animals received an equivalent volume of saline alone. Pups were weaned at PND21 and group-housed until behavioral testing at 6 weeks of age.
Electrode implantation and electromyography recordings
To quantitatively assess the visceral pain response, electromyography (EMG) electrodes were surgically placed in the external oblique muscles.49,50 Under deep isoflurane anesthesia and using aseptic technique, the abdominal fur was shaved and the skin disinfected. Bilateral nickel-chromium wire electrodes were implanted into the external oblique musculature. The electrode leads were tunneled subcutaneously to an externalized exit point at the nape of the neck and secured. The abdominal incision was sutured closed, and mice were allowed a 1-week recovery period before further procedures.
Visceral pain tests
Visceral hypersensitivity was evaluated by measuring the EMG response to graded colorectal distension (CRD).4,51,52 Following the recovery period, a lubricated latex balloon attached to a polyethylene catheter was inserted into the distal colorectum and taped to the base of the tail. Mice were acclimated in transparent restraint chambers for 30–60 min prior to testing. The balloon was then inflated to progressively higher pressures (20, 40, 60, and 80 mmHg), with each distension lasting 20 s followed by a 3-min rest interval. EMG signals from the external oblique muscles were recorded throughout each distension using Acknowledge software (Biopac Systems, Inc., USA). The visceromotor response was quantified by calculating the area under the curve (AUC) for the EMG signal. All CRD testing and data analysis were performed by experimenters blinded to the treatment groups.
EA treatment
Based on previous studies demonstrating the analgesic effects of electroacupuncture at Zusanli (ST36), we applied EA at this acupoint in NCI model mice.53,54 Animals were gently immobilized on a custom-built platform using a resin holder, with limbs exposed and unrestricted. Sterile acupuncture needles (0.25 mm × 13 mm; Hua Tuo, Suzhou Medical Supplies Co., Ltd., Suzhou, China) were inserted bilaterally into the ST36 acupoints to a depth of approximately 4 mm. Electrodes were connected to a nerve stimulator (Hua Tuo, Suzhou Medical Supplies Co., Ltd.), and electrical stimulation was applied at 0.5 mA, 100 Hz for 30 min per day over 7 consecutive days. After stimulation, needles were retained in place for an additional 15 min. During the entire procedure, mice remained awake but immobile, with no observable signs of distress. Mice in the sham group received the same procedure without electrical stimulation.
rTMS treatment
To alleviate visceral pain in NCI mice, rTMS was employed to intervene in the excitability of LSV neurons. Similar to the EA treatment protocol, mice were restrained on a smooth wooden board with their heads exposed. rTMS was delivered using a magnetic stimulator (YiRuiDe, Wuhan, China) targeting the LSV. The mice received rTMS treatment for 7 consecutive days, with each session lasting 5 min. Stimulation was applied at a frequency of 1 Hz and an intensity of 80% of the resting motor threshold (RMT),55,56 determined by visual inspection of bilateral forelimb movements in anesthetized mice during preliminary experiments. Throughout the procedure, mice remained awake and stationary without displaying signs of distress. In the sham group, the procedure was identical except that no magnetic stimulation was delivered.
Induced c-Fos expression
To evaluate neuronal activation in the LSV following visceral pain, c-Fos immunostaining was performed. 57 Mice were acclimated in transparent chambers for 1 h prior to stimulation to minimize stress. Visceral pain was evoked using a standardized CRD protocol: a 60-mmHg distension was applied for 20 s, repeated five times with 3-min intervals over a 15-min session to prevent tissue damage. Animals were euthanized 1.5 h post-stimulation for tissue collection.
Immunofluorescence staining
For immunofluorescence, mice were transcardially perfused with saline followed by 4% paraformaldehyde (PBS, pH 7.2–7.4). After post-fixation overnight at 4°C, brains were cryoprotected in a graded sucrose series (10%, 20%, 30%), embedded in OCT compound, and sectioned at 30 μm on a cryostat (Leica CM3050S). Following antigen retrieval in boiling sodium citrate, sections were blocked with 7% donkey serum containing 0.3% Triton X-100 and incubated overnight at 4°C with primary antibodies against c-Fos (mouse, 1:100, Santa Cruz Biotechnology) and Glutamate (rabbit, 1:200, Sigma-Aldrich). After washing, sections were incubated with Alexa Fluor-conjugated secondary antibodies (donkey anti-mouse 555 and donkey anti-rabbit 488, 1:500, Invitrogen) and mounted with DAPI. Images were acquired using Zeiss AXIO SCOPE A1 and LSM900 confocal microscopes. c-Fos-positive nuclei within the LSV were quantified from 1-2 representative slices per mouse using ImageJ software.
Stereotaxic injection
For targeted manipulation and recording, adult male mice (6–8 weeks) were anesthetized with isoflurane and secured in a stereotaxic frame (RWD, 71000-M, Shenzhen, China). Following a scalp disinfection and craniotomy, viral vectors were injected into the LSV using a micro-syringe (Gaoge) connected to a glass micropipette and an injection pump (Longer Pump, TJ-2A, Beijing, China). A volume of 200 nL was delivered at a rate of 30 nL/min, and the needle was left in place for 10–15 min post-injection to ensure adequate diffusion. For targeted manipulation of LSV Glu+ neurons, rAAV-Vglut2-ChR2-EYFP (3.0 × 1012 genome copies/mL, BrainVTA, Wuhan, China) or rAAV-Vglut2-eNpHR-EYFP (2.35 × 1012 genome copies/mL, BrainVTA, Wuhan, China) was injected into the LSV brain region (AP: +0.74 mm; ML: +0.6 mm; DV: −3.75 mm). After that, the wounds of the mice were sealed with dental cement and fixed together with skull screws. Behavioral tests were conducted 20 days later. For fiber photometry experiments, rAAV-hSyn-GRAB-eCB2.0-WPRE-hGH (2.32 × 1012 genome copies/mL, BrainVTA, Wuhan, China) or rAAV-hSyn-iGlusnFR (5.25 × 1012 genome copies/mL, BrainVTA, Wuhan, China) was injected into the LSV brain region, respectively. After injection, the wound was sealed with dental cement and skull screws. Behavioral assessments were conducted 20 days later. Animals with injection site deviations were excluded from statistical analysis.
Optogenetic and fiber photometry procedures
Optogenetic Manipulation: In mice expressing ChR2 or eNpHR in LSV Glu+ neurons, we delivered light stimuli during EMG recording. For neuronal activation, a 473 nm blue light (2–5 mW, 20-ms pulses at 10 Hz) was applied; for inhibition, a constant 594 nm yellow light (3–5 mW) was used. Light delivery was controlled via an Optogenetics-LED system (Alpha Omega Engineering).
Fiber Photometry Recording: Neural activity in the LSV was monitored using a fiber photometry system (RWD, R821). Signals from the eCB2.0 or iGlusnFR sensors were excited at 470 nm, with a 405 nm reference channel used for motion correction. Fluorescence changes (ΔF/F0) in response to graded CRD (20, 40, 60, 80 mmHg) were recorded, with F0 defined as the baseline fluorescence averaged over a 10-s pre-stimulus period. Each mouse underwent 9 trials of CRD stimulation during recording. Data were analyzed using Multichannel Fiber Photometry Software (RWD, Shenzhen, China).
Drug administration
To determine whether cannabinoid signaling mediates the analgesic effects of EA + rTMS on visceral pain in NCI mice, cannabinoid receptor 1 (CB1) signaling was selectively inhibited. The selective CB1 antagonist AM251 (2 mM, 50 μL per site) was administered via bilateral cannula injections into the LSV region.
Statistical analyses
All data are reported as mean ± SEM. The error bars in the figures indicate SEM. Statistical analysis was performed using GraphPad Prism software. Normality checks were performed on all data before conducting the statistical analysis. The data statistical significance was assessed by one-way ANOVA followed by Tukey’s post hoc test, two-way ANOVA with Bonferroni’s post hoc test and two-sample student’s t-test. p < 0.05 was considered statistically significant.
Results
NCI induces visceral pain and activates LSV Glu+ neurons in mice
As previously described, neonatal colonic inflammation (NCI) is a well-established rodent model that reliably induces visceral pain and closely mimics the clinical features of IBS.15,48 In the present study, the NCI model was employed (Figure 1(a)). Visceral pain was assessed using CRD combined with EMG. The area under the EMG curve (AUC) was significantly increased in NCI mice compared with CON mice, indicating the presence of visceral pain in the NCI group (Figure 1(b)–(d), **p < 0.01, two-way ANOVA followed by Bonferroni’s multiple comparison test). To determine whether the LSV was activated in response to visceral pain, mice were subjected to CRD stimulation (Figure 1(e)). As shown, c-Fos expression in the LSV was significantly elevated following repeated CRD, with a markedly higher expression in NCI mice compared to controls (Figure 1(f) and (g), ***p < 0.001, two-sample t-test). To further characterize the activated neuronal population, c-Fos immunostaining was combined with glutamatergic neuronal markers. Immunofluorescence analysis revealed substantial co-localization of c-Fos with Glu+ neurons in the LSV (Figure 1(h) and (i)). These findings suggest that the LSV Glu+ neurons are strongly activated in NCI-induced chronic visceral pain and may play a critical role in its modulation.

Visceral pain stimulation induces a significant increase in c-Fos expression in the LSV. (a) Experimental protocol for NCI model preparation. (b) Diagram showing EMG recording setup for assessing visceral pain in mice. (c) Representative EMG traces recorded during CRD. (d) The area under the curve of the EMG at 20, 40, 60 and 80 mmHg (**p < 0.01, two-way ANOVA followed by Bonferroni’s multiple comparison test, n = 6 per group). (e) Illustration of experimental setup for evaluating CRD-evoked c-Fos expression. (f) Representative images of c-Fos positive cells in LSV of CON and NCI mice, scale bar = 50 µm. (g) Quantification of c-Fos-positive cells in the LSV after CRD stimulation (***p < 0.001, two-sample t-test, n = 6 brain sections from 3 mice for CON group, n = 5 brain sections from 3 mice for NCI group). (h) Representative immunofluorescence images showing glutamate-positive (Glu+) and c-Fos-positive (c-Fos+) cells in the LSV, scale bar = 50 µm. (i) Quantification of co-localization between Glu+ and c-Fos+ cells in the LSV, expressed as the percentage of Glu+/c-Fos+ double-labeled cells among total Glu+ or c-Fos+ cells (n = 5 brain sections from 3 mice per group). ns indicates non-significant differences.
Optogenetic manipulation of LSV Glu+ neurons alters visceral pain
To further elucidate the functional role of LSV Glu+ neurons in chronic visceral pain, optogenetic approaches were used to selectively manipulate these neurons, and visceral nociception was assessed via CRD combined with EMG recordings (Figure 2(a) and (b)). AAV2/9-Vglut2-hChR2-EGFP or AAV2/9-Vglut2-eNpHR-EGFP was stereotactically injected into the LSV to enable light-induced activation or inhibition of Glu+ neurons, respectively (Figure 2(c)). Optogenetic inhibition of LSV Glu+ neurons significantly attenuated visceral pain responses in NCI mice (Figure 2(d) and (e), ***p < 0.001, two-way ANOVA followed by Bonferroni’s multiple comparison test). In contrast, optogenetic activation of LSV Glu+ neurons significantly enhanced visceral pain responses in CON mice (Figure 2(f) and (g), **p < 0.01, ***p < 0.001, two-way ANOVA followed by Bonferroni’s multiple comparison test). These results indicate that LSV Glu+ neurons actively contribute to the modulation of visceral nociceptive processing and are functionally involved in the maintenance or amplification of chronic visceral pain states.

Optogenetic inhibition of LSV Glu+ neurons alleviates visceral pain in NCI mice, whereas activation induces visceral pain in CON mice. (a) Experimental timeline of optogenetic modulation targeting LSV Glu+ neurons in NCI and CON mice. (b) Schematic of EMG recording setup used to evaluate pain responses following optogenetic intervention. (c) Schematic diagram of virus injection and optical fiber implantation in the LSV, with a representative image of virus expression in LSV Glu+ neurons, scale bar = 50 μm. (d) Representative EMG traces from NCI mice in response to CRD at 20, 40, 60, and 80 mmHg. (e) The AUC of the EMG in NCI mice in response to CRD at 20, 40, 60 and 80 mmHg (***p < 0.001, two-way ANOVA followed by Bonferroni’s multiple comparison test, n = 6 per group). (f) Representative EMG traces of CON mice at different levels of CRD stimulation (20, 40, 60 and 80 mmHg). (g) The AUC of the EMG in CON mice at 20, 40, 60 and 80 mmHg (**p < 0.01, ***p < 0.001, two-way ANOVA followed by Bonferroni’s multiple comparison test, n = 6 per group). ns indicates non-significant differences.
EA or rTMS intervention reduces LSV Glu+ neurons activation and alleviates visceral pain behavior in NCI mice
To investigate the therapeutic effects of EA and rTMS on visceral pain, we conducted a series of intervention experiments. A schematic of bilateral ST36 EA stimulation in NCI mice is presented in Figure 3(a). Following EA or sham-EA treatment, c-Fos immunostaining and CRD-induced EMG recordings were performed to assess neuronal activation and visceral pain behaviors, respectively (Figure 3(b)). The results showed that c-Fos expression in the LSV was significantly reduced after EA intervention, and this reduction was predominantly observed in Glu+ neurons (Figure 3(c) and (d), ***p < 0.001, two-sample t-test). Additionally, the area under the curve of EMG was significantly reduced in the EA group compared with Sham-EA, indicating that visceral pain was relieved (Figure 3(e) and (f), **p < 0.01, ***p < 0.001, two-way ANOVA followed by Bonferroni’s multiple comparison test). Similarly, rTMS intervention was applied to another group of NCI mice, followed by c-Fos staining and EMG evaluation (Figure 3(g) and (h)). CRD-induced c-Fos expression in the LSV was also significantly decreased after rTMS, particularly in Glu+ neurons (Figure 3(i) and (j), ***p < 0.001, two-sample t-test). Consistently, rTMS treatment significantly reduced the EMG AUC compared to the sham-rTMS group, suggesting an analgesic effect (Figure 3(k) and (l), ***p < 0.001, two-way ANOVA followed by Bonferroni’s multiple comparison test). These findings indicate that both EA and rTMS relieve visceral pain in NCI mice by suppressing the activity of LSV Glu+ neurons, implicating the LSV as a key target for neuromodulatory interventions in visceral pain.

EA or rTMS treatment suppresses LSV Glu+ neuronal activation and alleviates visceral pain in NCI mice. (a) Schematic illustration of bilateral EA stimulation at acupoint ST36. (b) Experimental protocol for c-Fos immunostaining and visceral pain behavioral assessment following EA or sham-EA treatment. (c) Representative images and quantification of c-Fos+ neurons in the LSV of EA and Sham-EA groups (***p < 0.001, two-sample t-test, n = 6 brain sections from 3 mice per group), scale = 100 µm. (d) Representative images and quantification of c-Fos+ neurons co-labeled with Glu+ neurons in the LSV of EA and Sham-EA group (***p < 0.001, two-sample t-test, n = 6 brain sections from 3 mice per group), scale = 100 µm. (e) Representative EMG traces of EA and Sham-EA group in response to CRD at 20, 40, 60, and 80 mmHg. (f) The AUC of the EMG for EA and sham-EA groups at each CRD pressure level (**p < 0.01, ***p < 0.001, two-way ANOVA followed by Bonferroni’s multiple comparison test, n = 6 per group). (g) Schematic diagram of rTMS stimulation targeting the LSV. (h) Experimental timeline of c-Fos staining and behavioral assessment after rTMS or sham-rTMS treatment. (i) Representative images and quantitative analysis of c-Fos+ neurons in LSV of rTMS and Sham-rTMS groups (***p < 0.001, two-sample t-test, n = 6 brain sections from 3 mice per group), scale = 100 µm. (j) Representative images and quantification of c-Fos+/Glu+ co-labeled neurons in the LSV of rTMS and Sham-rTMS groups (***p < 0.001, two-sample t-test, n = 6 brain sections from 3 mice per group), scale = 100 µm. (k) Representative EMG traces of rTMS and Sham-rTMS group at different levels of CRD stimulation (20, 40, 60 and 80 mmHg). (l) The AUC of the EMG for rTMS and Sham-rTMS groups at 20, 40, 60 and 80 mmHg (***p < 0.001, two-way ANOVA followed by Bonferroni’s multiple comparison test, n = 6 per group). ns indicates non-significant differences.
Combined EA + rTMS treatment exhibits enhanced analgesic effects compared to EA or rTMS alone
Although EA and rTMS individually attenuate visceral pain, their therapeutic efficacy may be limited when applied alone. Therefore, a combined EA + rTMS treatment strategy was employed (Figure 4(a)). c-Fos staining was performed to assess neuronal activation within the LSV following combination therapy. Results revealed that EA + rTMS treatment significantly suppressed LSV Glu+ neuron activation (Figure 4(b) and (c), ***p < 0.001, two-sample t-test). To compare the analgesic efficacy of EA, rTMS, and EA + rTMS, CRD-evoked EMG recordings were analyzed. The combination treatment group exhibited a more pronounced reduction in visceral pain responses and maintained this analgesic effect over a longer duration relative to single treatments (Figure 4(d)–(f), *p < 0.05, **p < 0.01, ***p < 0.001, two-way ANOVA followed by Bonferroni’s multiple comparison test). These findings suggest that EA + rTMS treatment mediates more potent analgesic effects by inhibiting LSV Glu+ neurons.

Combined EA + rTMS treatment exhibits more robust and sustained analgesic effects in NCI mice. (a) Schematic diagram of c-Fos staining and visceral pain behavior test following EA + rTMS or Sham-EA + Sham-rTMS treatment. (b) Representative images and quantification of c-Fos+ neurons in the LSV of EA + rTMS and Sham-EA + Sham-rTMS groups (***p < 0.001, two-sample t-test, n = 6 brain sections from 3 mice per group), scale = 100 µm. (c) Representative images and quantification of c-Fos+/Glu+ co-labeled neurons in the LSV (***p < 0.001, two-sample t-test, n = 6 brain sections from 3 mice per group), scale = 100 µm. (d) Representative EMG traces from Sham, EA, rTMS, and EA + rTMS groups during CRD stimulation at 20, 40, 60, and 80 mmHg. (e) The AUC of the EMG for Sham, EA, rTMS and EA + rTMS groups during CRD stimulation at 20, 40, 60, and 80 mmHg (*p < 0.05, **p < 0.01, ***p < 0.001, two-way ANOVA followed by Bonferroni’s multiple comparison test, n = 6 per group). (f) The visceral pain threshold of Sham, EA, rTMS and EA + rTMS treatment groups (***p < 0.001; #p < 0.05, ###p < 0.001, two-way ANOVA followed by Bonferroni’s multiple comparison test, n = 6 per group). ns indicates non-significant differences.
EA + rTMS treatment increases cannabinoid release and decreases glutamate release in the LSV
To explore the neurochemical mechanisms underlying the analgesic effects of EA and rTMS, we performed in vivo fiber photometry to monitor glutamate and endocannabinoid release in the LSV before and after the interventions. rAAV-hSyn-GRAB-eCB2.0 or rAAV-hSyn-iGlusnFR was injected into the LSV to detect cannabinoid and glutamate dynamics (Figure 5(a) and (b)). Representative image of rAAV-hSyn-iGlusnFR expression in the LSV was shown in Figure 5(c). Photometry recordings showed that all three interventions reduced glutamate release in the LSV (Figure 5(d) and (e)). Notably, the combined EA + rTMS treatment resulted in a significantly greater reduction in glutamate release than either EA or rTMS alone (Figure 5(f) and (g), *p < 0.05, **p < 0.01, ***p < 0.001, one-way ANOVA followed by Tukey’s multiple comparison test). To investigate the basis of this suppression of glutamate release, cannabinoid release was assessed. Representative image of rAAV-hSyn-GRAB-eCB2.0 expression in LSV was shown in Figure 5(h). All interventions significantly increased cannabinoid levels in the LSV (Figure 5(i) and (j)), with EA + rTMS inducing the most robust increase (Figure 5(k) and (l), **p < 0.01, ***p < 0.001, one-way ANOVA followed by Tukey’s multiple comparison test). Together, these findings suggest that the analgesic effects of EA and rTMS are mediated by enhanced cannabinoid signaling, which in turn reduces glutamate release in the LSV. The combination therapy amplifies this effect, providing a stronger neurochemical basis for its superior analgesic efficacy.
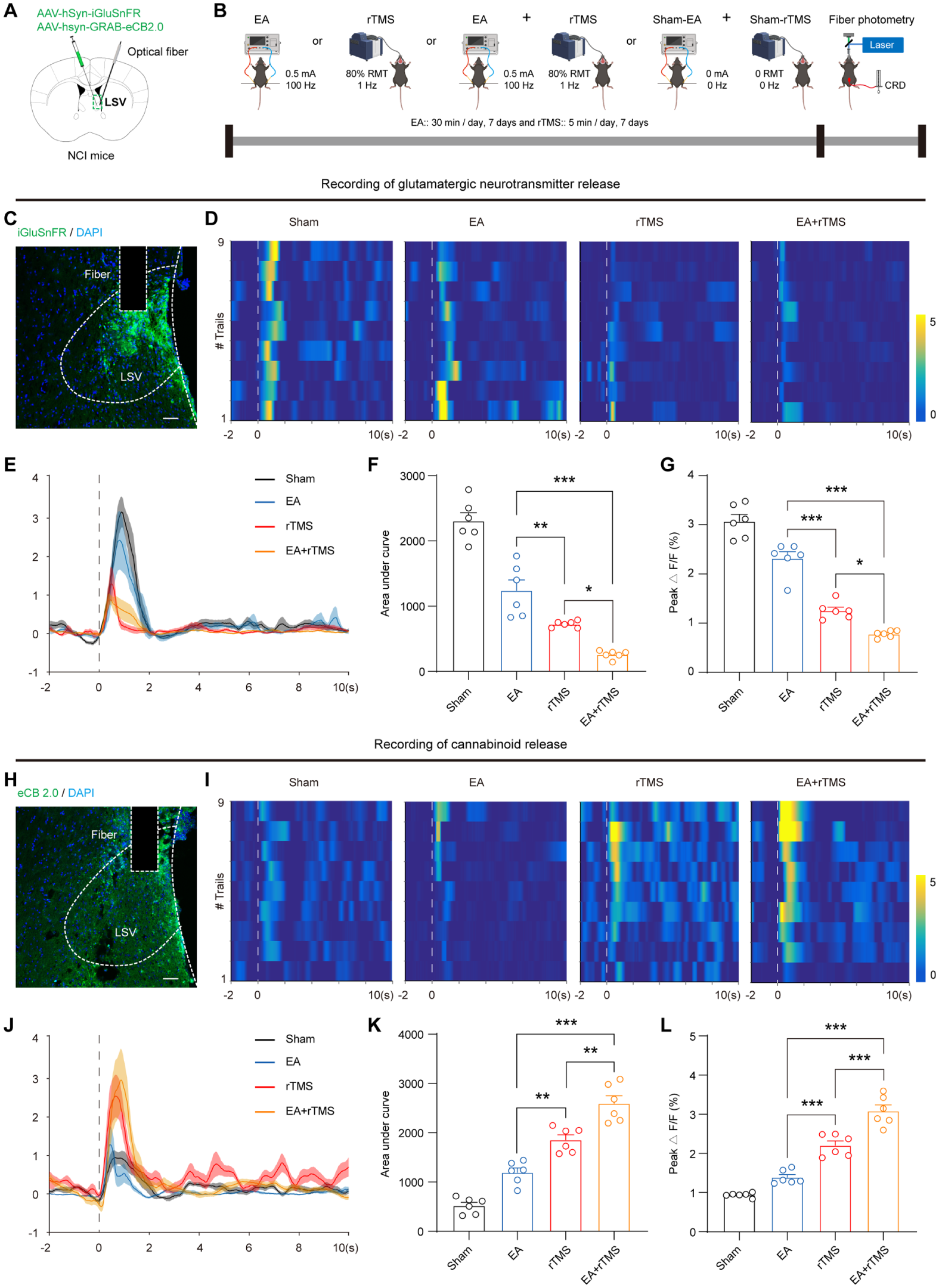
Combined EA + rTMS treatment increases cannabinoid release and decreases glutamate release in the LSV. (a) Schematic of viral injection of glutamate or cannabinoid sensors and optical fiber implantation targeting the LSV. (b) Experimental protocol for real-time assessment of glutamate or cannabinoid release following Sham, EA, rTMS and EA + rTMS treatment. (c) Representative image of glutamate sensor expression in LSV, scale = 100 µm. (d) and (e) Heatmaps and waveform plots of fiber photometry recordings in response to CRD stimulation at the 60 mmHg. (f) Quantification of AUC for glutamate release in the LSV across groups (*p < 0.05, **p < 0.01, ***p < 0.001, one-way ANOVA followed by Tukey’s multiple comparison test, n = 6 per group). (g) Average peak for glutamate release signals in LSV of Sham, EA, rTMS and EA + rTMS treatment group receiving CRD stimulation (*p < 0.05, ***p < 0.001, one-way ANOVA followed by Tukey’s multiple comparison test, n = 6 per group). (h) Representative image of cannabinoid sensor expression in LSV, scale = 100 µm. (i) and (j) Heatmaps and waveform plots of fiber photometry recordings in response to CRD stimulation at the 60 mmHg. (k) Quantification of AUC for cannabinoid release in the LSV of Sham, EA, rTMS and EA + rTMS treatment group receiving CRD stimulation (**p < 0.01, ***p < 0.001, one-way ANOVA followed by Tukey’s multiple comparison test, n = 6 per group). (l) Average peak for cannabinoid release signals in LSV of Sham, EA, rTMS and EA + rTMS treatment group receiving CRD stimulation (***p < 0.001, one-way ANOVA followed by Tukey’s multiple comparison test, n = 6 per group). ns indicates non-significant differences.
AM251 abolishes the analgesic effect of EA + rTMS in NCI mice
To determine whether the analgesic effect of combined EA + rTMS in NCI mice is mediated by cannabinoid signaling, we assessed visceral pain behaviors following administration of cannabinoid receptor antagonists. Given that CB1 receptors are predominantly expressed in the brain, the selective CB1 antagonist AM251 was delivered via bilateral cannula injections into the LSV. Compared with saline-treated controls, mice receiving AM251 failed to exhibit EA + rTMS–induced attenuation of visceral pain behaviors (Figure 6(a)–(d), **p < 0.01, ***p < 0.001, two-way ANOVA followed by Bonferroni’s multiple comparison test). These results indicate that CB1-dependent cannabinoid signaling is required for the analgesic effect of EA + rTMS in NCI mice.

AM251 significantly blocked the analgesic effect of EA + rTMS in NCI mice. (a) Schematic diagram of AM251 delivery to LSV. (b) Flowchart of EA + rTMS Intervention Following AM251 Injection. (c) Representative EMG traces from vehicle and AM251 groups during CRD stimulation at 20, 40, 60, and 80 mmHg. (d) The AUC of the EMG for vehicle and AM251 groups during CRD stimulation at 20, 40, 60, and 80 mmHg (**p < 0.01, ***p < 0.001, two-way ANOVA followed by Bonferroni’s multiple comparison test, n = 6 per group).
Discussions
This study reveals a novel neural mechanism underlying chronic visceral pain in irritable bowel syndrome and highlights the therapeutic potential of EA and rTMS, either alone or in combination. Using the NCI mouse model, we demonstrated that chronic visceral hypersensitivity is associated with increased activation of Glu+ neurons in the LSV. Optogenetic manipulation confirmed the causal role of LSV Glu+ neurons in modulating visceral pain responses. Notably, both EA and rTMS significantly suppressed LSV activity and alleviated pain behaviors, with their combination producing a more robust and sustained analgesic effect.
Emerging evidence supports a unique role of the LSV in visceral pain processing.52,57 Our findings align with previous reports showing that LSV Glu+ neurons play a key role in regulating visceral nociception. The marked upregulation of c-Fos expression in these neurons following CRD stimulation suggests that the LSV functions as an integrative hub for peripheral nociceptive input and central pain perception. Optogenetic activation of LSV Glu+ neurons exacerbated visceral pain, whereas their inhibition attenuated pain responses, providing direct evidence of their functional significance. EA has been widely employed in traditional Chinese medicine to relieve pain, and its analgesic effects have been attributed to modulation of both peripheral and central mechanisms.22,58–60 In our study, EA significantly reduced CRD-evoked visceral hypersensitivity and suppressed c-Fos expression in LSV Glu+ neurons, suggesting attenuation of central sensitization. Mechanistically, EA has been reported to enhance the release of endogenous opioids, serotonin, and endocannabinoids, which may contribute to its inhibitory effects on excitatory neurons.23,27,28 rTMS, a non-invasive neuromodulation technique, has gained traction in recent years for treating neuropathic and affective disorders.61,62 rTMS delivers repetitive magnetic pulses to modulate cortical and subcortical circuits. In this study, rTMS targeting the LSV significantly inhibited Glu+ neuronal activation and alleviated visceral pain behaviors in NCI mice, supporting its efficacy in central modulation of pain.
Crucially, the combination of EA and rTMS yielded superior analgesic outcomes compared to either treatment alone. This synergistic effect may be attributed to dual-level modulation: EA targeting peripheral input and spinal relay, while rTMS modulates central pain circuitry. Fiber photometry revealed that EA + rTMS treatment not only reduced glutamate release in the LSV more significantly but also enhanced endocannabinoid signaling. As endocannabinoids are known to inhibit glutamatergic transmission and reduce neuronal excitability,63–65 these findings suggest that cannabinoid-mediated suppression of glutamatergic activity may underlie the enhanced analgesic effects of combined treatment. The present study provides in vivo evidence that EA and rTMS synergistically inhibit LSV Glu+ neuron activity via enhanced endocannabinoid signaling, thereby relieving chronic visceral pain. These findings carry several important implications. First, they underscore the LSV as a central hub in the regulation of visceral pain. Second, they offer mechanistic insights into how neuromodulatory therapies, especially when combined, can more effectively disrupt maladaptive pain circuits. Third, they open new avenues for integrative, multimodal treatment strategies tailored to the complex pathophysiology of IBS.
Despite the promising findings, several limitations should be acknowledged. While our results support a causal role for LSV Glu+ neurons in visceral pain modulation, further studies are needed to map their downstream projections and define their precise contribution to behavioral output. In addition, although we showed that EA + rTMS increases cannabinoid release in the LSV, the expression levels and distribution of cannabinoid receptors in this region remain to be elucidated and will be addressed in future studies. Notably, while the primary focus of this study was on the combined EA + rTMS intervention, our exploratory cannabinoid release data offer preliminary insights into the relative contributions of each modality. The observed hierarchy of effect—where the combined intervention elicited the most pronounced increase in endocannabinoid levels, followed by rTMS alone, and then EA alone—suggests that both modalities contribute positively, but not equally, to the net effect. This pattern implies that rTMS may be the more potent driver of endocannabinoid release within the parameters used in this study, while EA provides a significant additive or possibly priming effect. Crucially, the supra-additive outcome of the combined treatment (EA + rTMS) compared to either intervention alone points toward a synergistic interaction rather than a simple summation of effects. The translational relevance of EA + rTMS therapy also warrants further investigation in clinical settings. Optimization of stimulation parameters such as frequency, intensity, and duration will be critical for maximizing efficacy and reproducibility in IBS patients. Furthermore, the current study included only male mice to eliminate the potential confounding effects of estrogen and estrous cycle on visceral pain sensitivity.66–69 However, given the known sex differences in pain perception and response to analgesics,70–72 the exclusion of female mice represents a potential limitation of this study and should be prioritized in future research.
In conclusion, this study identifies LSV Glu+ neurons as key regulators of chronic visceral pain in IBS and demonstrates that combined EA and rTMS treatment provides a more effective and mechanistically distinct approach to pain relief. These findings offer new strategies for neuromodulation-based therapies targeting functional gastrointestinal disorders.
Footnotes
Author contributions
R.-X.W., Y.G., and C.-H.Z. performed experiments, analyzed data and prepared the manuscript. R.-Y.Y., Q.L., and Z.-H.X. performed experiments, analyzed data, and prepared the figures. R.L., R.G., and Y.-C.L. analyzed data and prepared the manuscript. G.-Y.X. designed experiments, supervised the experiments, and finalized the manuscript. All the authors have read and approved the paper.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from National Key Research and Development Program of China (2024YFC3505200, 2024YFC3505203), National Natural Science Foundation of China (82401454, 82470573 and 82530041), Postdoctoral Fellowship Program of CPSF (GZC20231890 and 2025T180728), Jiangsu Provincial Department of Science and Technology (BE2023710) and the Priority Academic Program Development of Jiangsu Higher Education Institutions of China. The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Ethics approval
Care and handling of the animals were approved by the Institutional Animal Care and Use Committee of Soochow University and were in accordance with the guidelines of the International Association for the Study of Pain.
