Abstract
The senescence-associated secretory phenotype (SASP) contributes to tissue degeneration and inflammation, yet its role in osteoarthritis (OA)-related pain remains poorly understood. We hypothesized that circulating SASP markers would be associated with distinct OA-pain phenotypes, defined by pain impact and radiographic OA (ROA) severity. A subset of middle-to-older-aged adults (45–85 years) from a larger multi-site study (n = 169) self-reported pain impact – defined as the extent to which pain interferes with daily functioning – and underwent knee radiography and blood collection. Hierarchical cluster analysis was used to empirically identify OA-pain phenotypes based on combined pain impact and Kellgren-Lawrence (KL) grade. Plasma levels of four SASP markers (GDF-15, activin-A, Osteopontin (OPN), and IL-15) were quantified from whole blood samples. Among 169 participants, 35.5% reported high-impact chronic knee pain and 27.8% exhibited moderate-to-severe radiographic OA. Cluster analysis identified distinct ROA-pain phenotypes. GDF-15 levels were significantly elevated in non-Hispanic White females with early ROA and high-impact pain, with race- and sex-dependent differences. Activin-A levels were higher in non-Hispanic Black participants without pain or ROA and varied by sex in early ROA/low-impact pain phenotypes. Osteopontin levels were elevated in males compared to females within the same phenotype group. IL-15 levels showed no association with ROA-pain phenotypes but were higher in males and positively correlated with age. SASP factors, particularly GDF-15, Activin-A, and Osteopontin, demonstrated race- and sex-dependent associations with OA-pain phenotypes. These findings underscore the importance of demographic context in OA pathophysiology and support further investigation into SASP factors as potential biomarkers and therapeutic targets for OA-related pain.
Introduction
Osteoarthritis (OA) is a degenerative joint disease predominantly affecting the knees and hips, characterized by the breakdown of cartilage, leading to pain, stiffness, and reduced mobility. 1 It is the most common age-related disease and a leading cause of chronic pain and disability. 2 The condition predominantly affects older adults, with around 75% of those diagnosed being over the age of 55. 3 Chronic pain associated with OA is often debilitating, contributing to limitations in daily activities and function, also known as high-impact chronic pain. 4 Despite the high prevalence of OA, no disease-modifying osteoarthritis drugs are currently approved to halt or reverse the disease. Similarly, current pain management approaches are inadequate, particularly in older adults, in whom the use of opioids adds to concerns of polypharmacy. 5 Studying the mechanisms of aging may provide insights into the pathologies of OA and its associated pain, as aging and OA share common biological pathways, including inflammation, mitochondrial dysfunction, and epigenetic alterations. 6 Some have postulated that OA contributes to biological aging, in part due to pain and disability. 7 Indeed, our work and others’ have shown that chronic pain, driven largely by high-impact chronic pain, is associated with markers of accelerated biological aging, including epigenetic aging,8,9 and brain aging. 10
Cellular senescence is involved in many age-related diseases, including OA.11–14 It is characterized by a permanent proliferative arrest and apoptotic resistance. 15 Senescent cells secrete pro-inflammatory cytokines, chemokines, growth factors, and proteases, collectively known as the senescence-associated secretory phenotypes (SASP). 15 These SASP factors alter the surrounding cells and tissues, contributing to chronic inflammation, tissue degradation, and extracellular matrix remodeling, which are OA hallmarks and drive disease progression and pain.15–18
Additionally, the transplantation of senescent cells into the knee joint area of mice induces pain-like behaviors and structural joint remodeling consistent with OA.11,19 Studies have also shown that neuronal senescence in the dorsal root ganglion (DRG) plays a key role in pain modulation, with increased senescence markers (i.e. p16INK4a, p21) and SASP factors such as IL-6 observed after injury. 20 Aged DRG neurons exhibit these senescence markers, which are further exacerbated after peripheral nerve injury and correlate with nociception, suggesting that senescent DRG neurons contribute significantly to pain in aging and injury models. 20 Despite promising pre-clinical findings, the relationship between senescence markers and OA-related pain has not been explored in humans.
Therefore, in this study, we evaluated SASP factors in relation to clinical (i.e. self-reported) pain impact and radiographic OA (ROA) severity in middle-to-older aged adults. We selected the following SASP factors because of their strong relevance to senescent cells (SnCs) and the progression of OA. SnCs, through SASP, mediate cellular cross-talk and drive inflammation, extracellular matrix degradation and joint degeneration.15,21 The factors we focused on are growth differentiation factor-15 (GDF-15), Activin-A, osteopontin (OPN), and interleukin-15 (IL-15); these are key mediators of these processes. GDF-15 is among the most upregulated genes in early OA and may drive chondrocyte senescence through MAPK14 activation. 22 It also promotes angiogenesis, which contributes to inflammation and tissue degradation, indicating its role in synovial inflammation. 23 Activin-A is highly expressed in OA-affected cartilage compared to unaffected cartilage. 24 OPN induces the production of pro-inflammatory cytokines in human OA chondrocytes. 25 IL-15 has been identified as one of four circulating proteins associated with the onset of both knee and hand OA. 26
Because the discordance between clinical pain severity and OA severity has been well documented,27–29 we employed cluster analysis to identify empirically derived radiographic OA-pain phenotypes. We hypothesized that SASP factors would be associated with radiographic OA-pain phenotypes, where individuals with the highest impact pain and greater radiographic OA severity would have the greatest circulating levels of SASP factors. Importantly, since cellular senescence levels are strongly associated with obesity and older age,30–33 these were adjusted in our analysis along with additional biobehavioral factor (e.g. sex).
Methods
The present study consists of a cross-sectional analysis of data from middle-to-older age adults (45–85 years) with and without symptomatic knee OA who took part in a larger, multi-site study conducted at the University of Florida (UF; Gainesville, FL, USA) and the University of Alabama-Birmingham (UAB; Birmingham, AL, USA). Participants were recruited from the surrounding areas at both study sites using community- and clinic-based recruitment methods. Exclusion criteria consisted of conditions that could confound knee OA-related outcomes, preclude consent, or prevent completion of study procedures, such as rheumatic disease (e.g. rheumatoid arthritis, fibromyalgia), history of surgery to the index (i.e. most painful) knee, uncontrolled hypertension, neurological disease, heart diseases, peripheral neuropathy, pregnant or nursing, had more severe pain in body sites other than the index knee, chronic opioid use, hospitalization for psychiatric disorders in the preceding year, and diminished cognitive function. For this investigation, we included a random subset of participants in whom an array of senescence biomarkers was completed. The study was approved by the Institutional Review Boards at both sites, and all participants provided written informed consent.
Assessments
Statistical analysis
Hierarchical cluster analysis was employed to identify empirical clusters based on the combined impact of self-reported pain and KL grade, excluding controls with no pain/no ROA. After confirming that GCPS grade and KL scores were not highly correlated (r ≤ 0.51), we used Ward’s clustering method with squared Euclidean distances as the similarity measure. The optimal cluster solution was established through visual inspection of the dendrogram and changes to the agglomeration coefficients. We then examined the cluster group differences across the clustering variables to determine the appropriateness and internal validity of the cluster solution, identifying empirically derived ROA-pain phenotypes, summarized in Figure 1.

Phenotype clusters based on radiographic osteoarthritis (OA) severity and graded-chronic pain scale pain-impact.
Sample characteristics are presented as means and standard deviations or counts and percentages where appropriate. The ROA-pain clusters were compared across demographic and pain characteristics using chi-square tests for categorical variables and analysis of variance (ANOVA) for continuous variables. We examined the independent associations between SASP factors and ROA-pain group classifications using one-way analysis of covariance (ANCOVA), adjusting for age, sex, race, BMI, and study site as covariates. Given the established relationships between pain and demographic factors, we also tested two-way interactions between ROA-pain group and both sex and race. Statistical significance was defined as an alpha level of ≤0.05, with Bonferroni corrections applied for post hoc comparisons.
Results
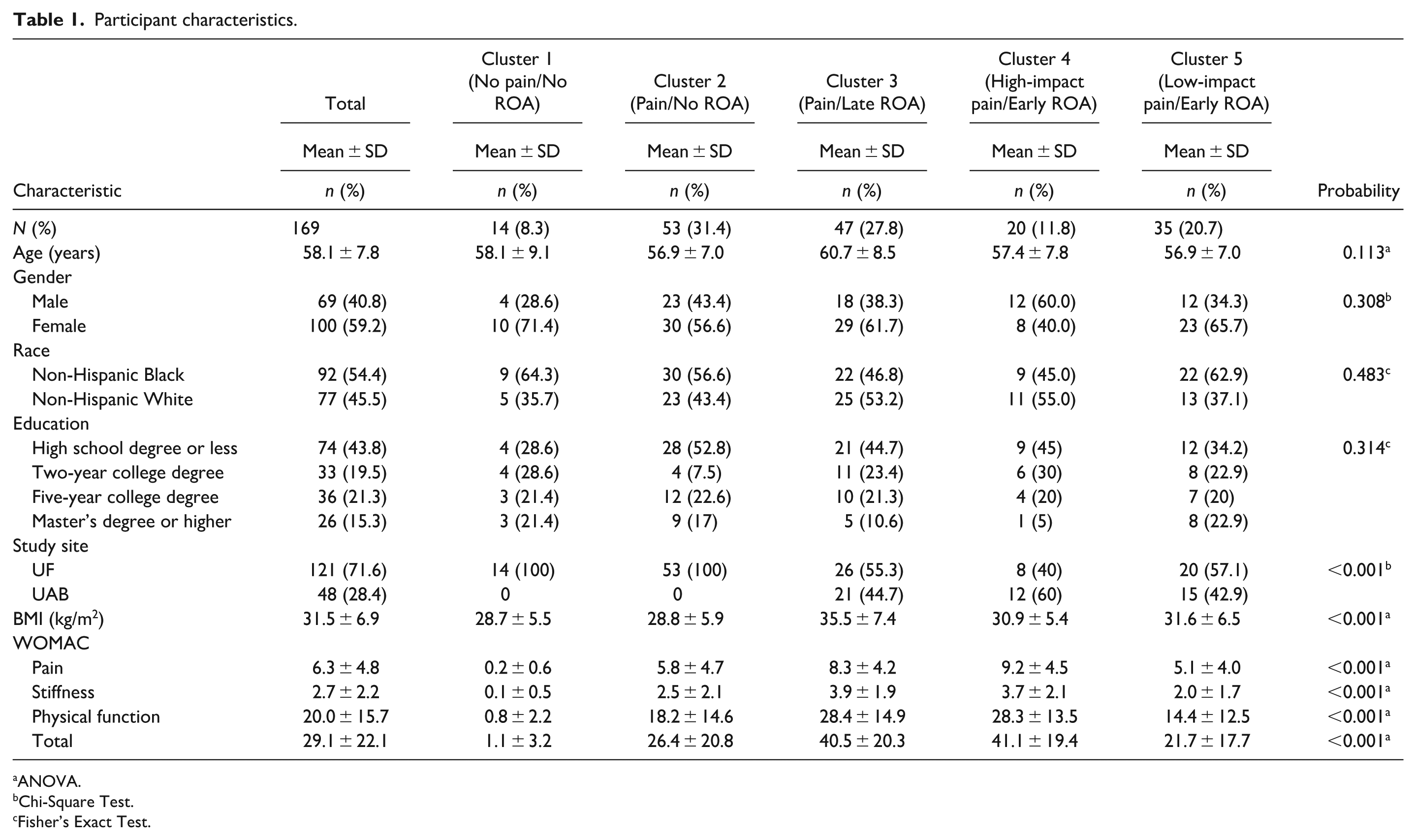
Participant characteristics are shown in Table 1. In total, 35.5% of participants (n = 60) reported high-impact chronic knee pain, and 56.2% reported low-impact chronic pain (n = 95). Forty-seven (27.8%) participants showed moderate-to-severe radiographic OA (ROA) with KL grades 3–4. Participants had a mean age of 58.1 ± 7.8 (mean ± SD), 100 (59.2%) were female, and 92 (54.4%) identified as non-Hispanic Black. The mean BMI was 31.5 ± 6.9 kg/m2.
Participant characteristics.
ANOVA.
Chi-Square Test.
Fisher’s Exact Test.
Table 1 and Figure 1 depict the ROA-pain groups represented by individuals in cluster 1 (no pain/no ROA), cluster 2 (pain/no ROA), cluster 3 (pain/late ROA), cluster 4 (high-impact pain/early ROA), and cluster 5 (low-impact pain/early ROA). The clusters did not significantly differ in most sociodemographic characteristics. Notably, cluster 1 (no pain/no ROA) did not include male participants because all individuals meeting these criteria were female. BMI was significantly higher in individuals in cluster 3 (pain/late ROA) compared to all other clusters (Bonferroni-corrected p < 0.05), except for cluster 4 (high-impact pain/early ROA). Additionally, WOMAC scores were highest among individuals in cluster 3 (pain/late ROA), and cluster 4 (high-impact pain/early ROA), whose ratings were significantly higher than those in cluster 1 (no pain/no ROA); (Bonferroni-corrected p < 0.05 for both) and cluster 5 (low-impact pain/early ROA) (Bonferroni-corrected p < 0.01 for both).
GDF-15
ROA-pain phenotypes were associated with GDF-15 levels in a race- and/or sex-dependent manner; shown in Figure 2. A significant three-way interaction was found between OA-pain phenotypes, race, and sex on GDF-15 levels (F (3, 149) = 2.92, p = 0.036). Among non-Hispanic White females, cluster 4 (early ROA/high-impact pain) had significantly higher GDF-15 levels compared to cluster 1 (no pain/no ROA, Bonferroni-corrected p = 0.022), cluster 2 (pain/no ROA, Bonferroni-corrected p = 0.024), cluster 3 (pain/late ROA, Bonferroni-corrected p = 0.006), and cluster 5 (low-impact pain/early ROA, Bonferroni-corrected p = 0.005). No significant associations between ROA-pain clusters and GDF-15 levels were measured among non-Hispanic Black participants (all p’s > 0.05). In cluster 4 (early ROA/high-impact pain), Non-Hispanic White females had significantly higher circulating GDF-15 levels compared to non-Hispanic Black females (Bonferroni-corrected p = 0.006) while non-Hispanic White females had significantly higher GDF-15 levels than non-Hispanic White males (Bonferroni-corrected p = 0.044). On the other hand, non-Hispanic White females in cluster 5 (early ROA/low-impact pain) had significantly lower GDF-15 levels than non-Hispanic White males (Bonferroni-corrected p = 0.010). Finally, GDF-15 levels were also positively correlated with age (F (1, 149) = 6.17, p = 0.014), and BMI (F (1, 147) = 5.86, p = 0.017.
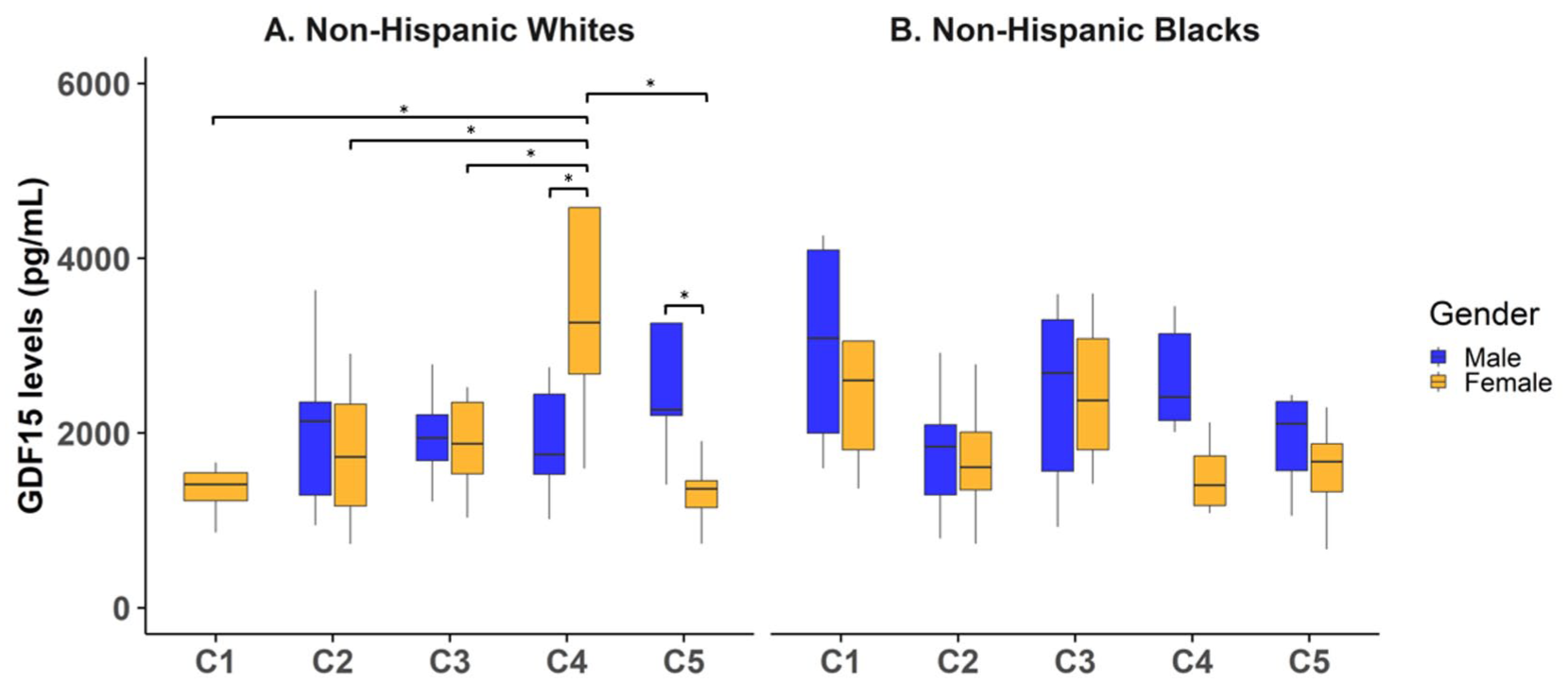
Association between OA-pain phenotypes with GDF-15 levels. The x-axis includes individuals in cluster 1 (no pain/no ROA), cluster 2 (pain/no ROA), cluster 3 (pain/late ROA), cluster 4 (high-impact pain/early ROA), and cluster 5 (low-impact pain/early ROA). The y-axis indicates the corresponding plasma GDF-15 levels. (a) Among non-Hispanic White females, individuals with high-impact pain and early OA exhibited significantly elevated GDF-15 levels compared to all other phenotype clusters (p = 0.006). (b) No significant associations between OA-pain phenotypes and GDF-15 were observed among non-Hispanic Black participants.
Activin-A
ROA-pain phenotypes were associated with circulating Activin-A levels in a race- and/or sex-dependent manner. There was a significant two-way interaction between ROA-pain phenotype by race (F (4, 147) = 2.53, p = 0.043), where circulating Activin-A levels were significantly higher in non-Hispanic Black participants compared to non-Hispanic White participants within cluster 1 (no pain/no ROA, Bonferroni corrected p = 0.034, Figure 3). Also, there was a significant two-way interaction between ROA-pain phenotype by sex (F (4, 147) = 2.62, p = 0.037). While non-Hispanic White females in cluster 5 (early ROA/low-impact pain) had lower Activin-A levels compared to non-Hispanic White males (Bonferroni-corrected p = 0.05). Finally, Activin-A levels were also positively correlated with age (F (1, 149) = 9.44, p = 0.003) and BMI (F (1, 147) = 8.04, p = 0.005).
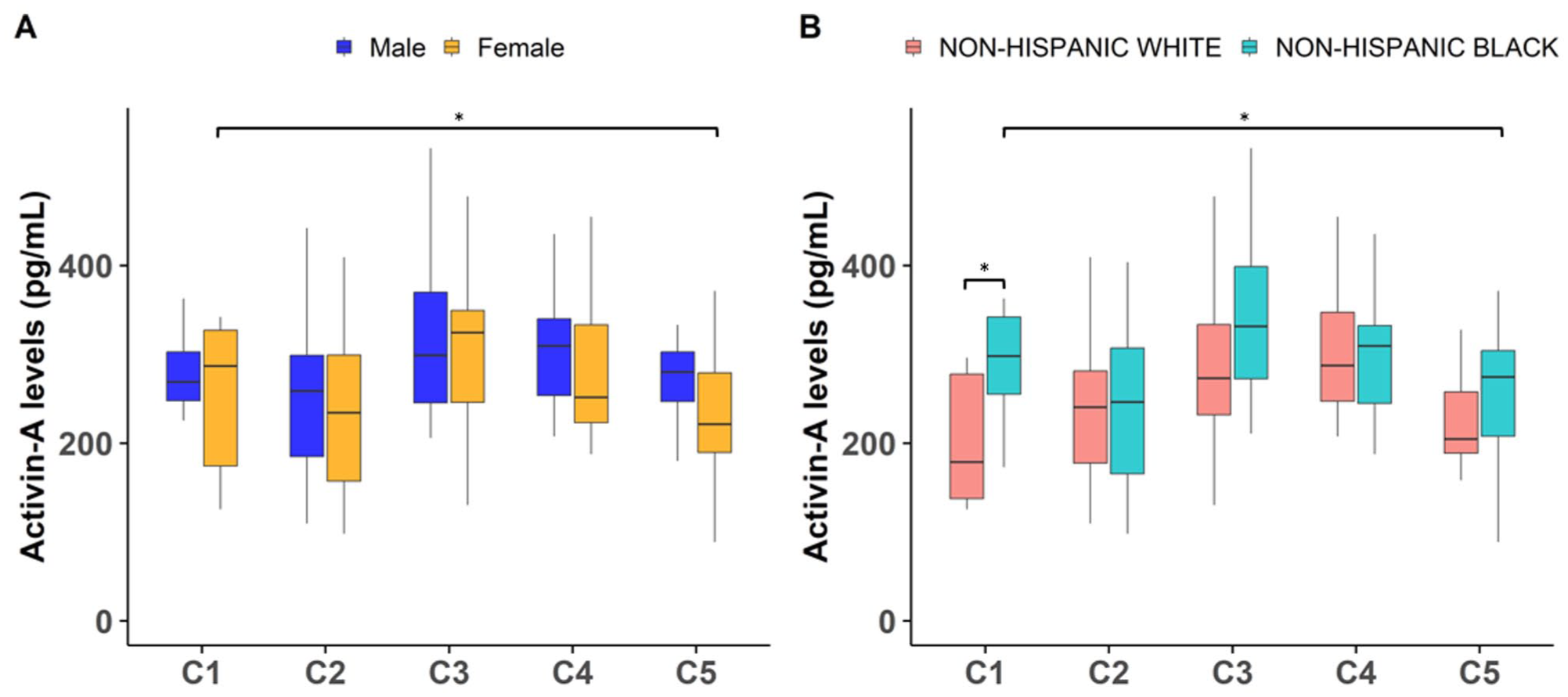
Association between OA-pain phenotypes with Activin-A levels. The x-axis includes individuals in cluster 1 (no pain/no ROA), cluster 2 (pain/no ROA), cluster 3 (pain/late ROA), cluster 4 (high-impact pain/early ROA), and cluster 5 (low-impact pain/early ROA). The y-axis indicates the corresponding plasma activin-A levels. (a) Activin-A levels were significantly lower among individuals with low-impact pain and early OA compared to those with no pain and no OA, but only within female (p = 0.026), and (b) non-Hispanic Black (p = 0.037) subgroups.
Osteopontin
ROA-pain phenotypes were associated with circulating Osteopontin levels in a sex-dependent manner. There was a significant two-way interaction between ROA-pain phenotype by sex (F (4, 147) = 4.28, p = 0.003), where females in cluster 5 (early ROA/low-impact pain) had lower Osteopontin levels than males in the same cluster 5 (Bonferroni-corrected p = 0.003). Finally, Osteopontin levels were significantly associated with BMI (F(1, 147) = 4.36, p = 0.038; Figure 4).

Association between OA-pain phenotypes with Osteopontin levels. The x-axis includes individuals in cluster 1 (no pain/no ROA), cluster 2 (pain/no ROA), cluster 3 (pain/late ROA), cluster 4 (high-impact pain/early ROA), and cluster 5 (low-impact pain/early ROA). The y-axis indicates the corresponding plasma osteopontin levels. A significant interaction between OA-pain phenotype and sex was also observed for Osteopontin levels (p = 0.003).
Il-15
There were no associations between ROA-pain phenotypes and circulating IL-15 levels (Supplemental Figure 1). However, there was a main effect of sex on IL-15 levels where males had significantly higher levels than females (F(1, 147) = 10.25, p = 0.002). Further, age was positively correlated with circulating IL-15 levels (F(1, 149) = 7.66, p = 0.006).
Discussion
Cellular senescence is linked to various age-related diseases, and senescent cells, along with their senescence-associated secretory phenotype (SASP) factors, significantly contribute to the progression of OA. 36 However, the relationship between senescence and OA-related pain has not been thoroughly investigated in humans. In this study, we examined whether circulating SASP factors were associated with OA-related pain by evaluating both clinical pain impact and radiographic knee OA severity in middle-aged and older adults with or at risk of knee OA. We identified distinct OA-pain phenotypes that differentiated individuals based on the level of pain impact (low- vs high-impact chronic pain) and the structural severity of knee OA, ranging from early to advanced OA disease, as determined by radiographs. These empirically derived phenotypes align with the well-documented discordance between clinical pain severity and radiographic OA findings.27–29
Circulating levels of Growth Differentiation Factor-15 (GDF-15) varied among OA-pain phenotypes in a sex- and race-dependent manner, with the most striking finding being elevated levels in Non-Hispanic women with high-impact pain but early radiographic OA compared to all other groups, including controls and individuals with advanced radiographic disease. GDF-15, a stress-responsive cytokine member of the transforming growth factor-β (TGF-β) superfamily, is a key SASP protein strongly associated with aging and serves as a potent predictor of all-cause mortality,37–46 including in persons with OA, 47 supporting its crucial role in biological aging processes. This sex-specific elevation is particularly relevant given that higher plasma GDF-15 levels predict accelerated decline in bone parameters in women but not men48,49 and aligns with the well-established association between post-menopausal status and OA development, where hormonal changes may accelerate cellular senescence and inflammatory aging within joint tissues.49,50 Mechanistically, GDF-15 drives OA progression through multiple pathways. Weng et al. 22 demonstrated that GDF-15 is among the most upregulated genes in early OA and may drive chondrocyte senescence through MAPK14 activation. Beyond cartilage effects, GDF-15 facilitates angiogenesis, contributing to inflammation and degradation, 23 and is found in elevated concentrations in synovial fluid and membranes of OA patients, 22 indicating its role in synovial inflammation. Studies have also shown that GDF-15 modulates nociception and neuronal excitability of dorsal root ganglia neurons in arthritic rats by regulating pain-related ion channels like Nav1.8, 51 while elevated serum and muscle levels of GDF-15 are associated with chronic muscular pain, decreased skeletal muscle mass and function, and show inverse relationships with muscle strength in aging populations – all known risk factors for chronic muscle pain. 52
Further, elevated GDF-15 in women experiencing severe pain despite minimal radiographic changes may also reflect the limitations of X-ray assessments, which primarily visualize bony structures and fail to capture early cartilage degradation, synovial inflammation, or other soft tissue changes contributing to cellular stress. This finding underscores the critical importance of considering self-reported pain impact as an early alarm system indicating significant underlying pathological processes that require therapeutic intervention to prevent future joint damage and OA progression. Importantly, this GDF-15 elevation was not observed in groups with late radiographic OA, suggesting that in advanced disease stages cartilage involvement may differ, or that the senescence signal is obscured when high- and low-impact pain groups are combined, highlighting high-impact pain as a key differentiator of underlying senescence burden occurring before structural damage becomes radiographically apparent. Moreover, since inflammation of the synovium (i.e. synovitis) can be missed with radiographic assessments, the association between GDF-15 and pain observed here may reflect a more inflammatory phenotype of OA rather than structural disease severity. Ultimately, additional work incorporating more sensitive imaging and inflammatory measures will be necessary to clarify the underlying pathophysiologic drivers.
There were significant differences in Activin-A levels between ethnic/racial groups among the control participants, as well as sex differences observed in non-Hispanic White participants with early radiographic osteoarthritis (ROA) and low-impact pain. Previous studies have consistently reported that activin-A exerts detrimental effects on knee joints.24,53 Zou et al. 24 found elevated serum levels of activin-A in OA patients compared to controls. Activin-A levels in both serum and synovial fluid were significantly associated with OA severity based on KL grading. Additionally, Alexander et al. 54 reported that activin-A is highly expressed in OA-affected cartilage compared to unaffected cartilage. However, previous studies have not considered a combination of radiographic OA and pain impact as in our study. Thus, it is possible that the relationship between activin-A and OA-pain phenotypes is not uniform across populations. Future larger studies are needed for clarification.
Males in the low-impact pain and late-stage osteoarthritis (OA) group exhibited higher levels of Osteopontin (OPN) compared to females. OPN promotes the production of pro-inflammatory cytokines, such as IL-6 and IL-8, in human OA chondrocytes, which may contribute to OA development.25,55 Additionally, elevated OPN levels in both plasma and synovial fluid have been linked to greater radiographic severity and increased pain intensity in knee OA.31,55–57 Further research is needed to explore how OPN levels vary between sexes during OA progression, particularly in relation to OA-related pain.
Although our study revealed new insights into OA-pain phenotypes linked to SASP factors, we did not find a significant association between IL-15 levels and OA-pain phenotypes. In contrast, Sun et al. 32 reported that serum IL-15 levels were independently and positively associated with increased pain severity in knee OA patients, though no link was found with radiographic OA severity. Additionally, a proteomic case-control study identified IL-15 as one of four circulating proteins connected to both established OA and the onset of knee and hand OA. 26 These conflicting findings highlight the need for further research to clarify the role of IL-15 in OA-related pain.
As expected, we found significant associations between SASP factors and chronological age, although most of these associations lost significance after adjusting for other covariates. Our results also support the well-established link between SASP factors and body mass index (BMI). Obesity is known to increase certain SASP factors, potentially exacerbating osteoarthritis (OA) and related pain. 58 A multicenter study further showed that chronically obese patients face a higher risk of medial knee cartilage damage, which accelerates disease progression. 59
This study has several strengths. Firstly, we measured established SASP factors in a well-characterized cohort,60,61 enabling meaningful comparisons across different studies and diverse populations. Another key strength is the use of cluster analysis to identify OA-pain phenotypes, which provided an empirical basis for differentiating individuals according to both pain impact and OA radiographic severity – reflecting the known disconnect between pain experience and radiographic findings. On the other hand, the cellular source of circulating SASP factors cannot be determined from our study, as these were measured in blood. Several factors, including obesity and biological aging, and comorbidities, may have overlapping systemic influences that share identical proteins to the reported SASP factors30,62–64; as such, our models were adjusted for age and BMI. In addition, non-senescent cells, such as tissue resident immune cells, can also secrete overlapping SASP factors.65,66 The direction of the relationship between circulating SASP factors and knee OA pain cannot be determined within our cross-sectional study design. Future research with larger cohorts is necessary to validate these findings and investigate the potential overlap between circulating and local (i.e. knee synovial fluid) SASP factors. Additionally, controlled preclinical models are essential to better understand how inflammatory SASP profiles arising from underlying conditions like age-related comorbidities intersect with specific tissues undergoing cellular senescence.
Conclusion
Joint pain is a hallmark of osteoarthritis (OA), though it does not always reflect the extent of tissue remodeling. While the mechanisms driving joint pain remain unclear, local inflammation is likely a critical contributor. Senescent cells may play a key role in OA pain, with evidence showing that clearing these cells before structural tissue changes can lead to pain relief.67,68 Pain-inducing cytokines from synovial and other cell types may also contribute to this process. Understanding how senescence-associated secretory phenotype (SASP) factors cause pain and the involvement of synovial, immune, and other cells will deepen our knowledge of OA’s clinical features and support the development of targeted therapeutics. 16 Our findings highlight that SASP factors, especially GDF-15, may be indicative of females experiencing high-impact pain in early OA, but not males, with these effects also varying by race. This underscores the importance of considering demographic factors in OA pathophysiology. Continued research is essential to validate GDF-15 as a potential biomarker and therapeutic target for OA-related pain.
Supplemental Material
sj-docx-1-mpx-10.1177_17448069261422994 – Supplemental material for Circulating cellular senescence biomarkers in persons with chronic knee osteoarthritis pain: An exploratory study
Supplemental material, sj-docx-1-mpx-10.1177_17448069261422994 for Circulating cellular senescence biomarkers in persons with chronic knee osteoarthritis pain: An exploratory study by Muhammad Abbas, Javier A Tamargo, Carlos J Cruz, Yutao Zhang, Stephanie Wohlgemuth, Kevin Wu, Li Chen, Roland Staud, Roger B Fillingim, Christiaan Leeuwenburgh and Yenisel Cruz-Almeida in Molecular Pain
Footnotes
Acknowledgements
We want to thank our research teams and participants for their efforts to this research.
Author contributions
Conceptualization: YCA, MA; Writing – original draft: YCA, MA, JAT; Investigation: MA, SW, KW, CL, RS; Formal analysis: YCA, MA, YZ, LC; Writing – Review & Editing: all authors; Supervision: YCA, CL.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health under award number R90AR085802, the National Institute on Aging T32AG049673 and L32AG096730, and the University of Florida Pain Research & Intervention Center of Excellence.
Availability of data and materials
The data supporting this study’s findings are available from the corresponding author, YCA, upon reasonable request.*
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
