Abstract
Chronic itch is a debilitating symptom associated with many dermatological and systemic diseases. Rodent behavioral models that distinguish pain and itch responses remain limited. Our previous studies have examined behavioral responses of mice to chemicals delivered to the plantar glabrous skin and suggested that glabrous skin biting is associated with itch sensation whereas licking represents pain sensation, establishing a new mouse behavioral model to differentiate pain and itch responses in the glabrous skin. To provide further validation of this model, we here investigated behavioral responses following intraplantar injection of multiple pruritogens to examine if they can effectively evoke biting behavior. We show that most of the tested pruritogens induced dose-dependent responses. PAR2 peptide agonist SLIGRL selectively evoked licking, whereas deoxycholic acid (DCA) selectively induced biting. 5-HT triggered licking with a low concentration and both licking and biting with a higher concentration. IL-31, allergen ovalbumin, and mast cell degranulator compound 48/80 evoked both licking and biting. Importantly, morphine abolished capsaicin-induced licking but not biting induced by Bam8-22 or DCA, confirming that glabrous skin biting represents an itch-associated nocifensive behavior. Together, our results establish a robust model to differentiate between pain and itch and provide a platform for investigating mechanisms underlying glabrous skin itch.
Introduction
Chronic itch is often associated with a variety of dermatological and systemic conditions and represents a major source of discomfort for patients.1,2 Mouse models have played an important role in studying the mechanisms of both acute and chronic itch. A common approach is the intradermal delivery of pruritogens, which directly activate cutaneous sensory nerves and evoke itch-related behaviors. 3 The specific itch-related behavioral readouts vary depending on the injection site. The most widely used injection site is the nape of the neck, where mice exhibit hindpaw scratching behaviors. 3 However, this model does not allow differentiation of pain and itch behaviors since hindpaw scratching is the only behavior that mice are able to exhibit toward the nape. To address this, Shimada and LaMotte 4 established a cheek model of itch that provides a behavioral differentiation between pain and itch. Mice can exhibit both hindpaw scratching and forelimb wiping toward the cheek, while the former indicates itch responses and the latter is pain related.
Although itch sensation can originate from any area of the skin, it can be particularly debilitating when occurring on glabrous (non-hairy) skin, such as the palms and soles. These regions are known to exhibit heightened tactile and pain sensitivity.5–11 The plantar hindpaws have been frequently used in rodent behavioral assays to investigate pain sensation because of its accessibility. 12 However, itch responses from this region have been much less studied. Pioneering work from Hagiwara et al. 13 demonstrate that mice exhibit both biting and licking behaviors toward the hindpaw. Serotonin injection into the medial malleolus region of the hindpaw or dry skin treatment of the whole hindpaw induced significant biting behavior,13–16 suggesting that biting of the hindpaw is an indication of itch sensation. Recent work from our group focused on the glabrous skin area by intradermal injection of only 4 µL of tested chemicals into the plantar hindpaw. Our results demonstrated that intraplantar injection of pruritogens induced biting. 17
In the present study, we aimed to investigate mouse behavioral responses following intraplantar injection of pruritogens commonly used in itch studies to provide further validation of the behavioral model and to better understand the underlying pathways in glabrous skin itch that may be relevant to human disease.
Experimental procedure
Mice behavioral test
All experiments were approved by the Georgia Institute of Technology Animal Use and Care Committee (Protocol A100055, approved on 1/31/2025). Mice were housed in a vivarium with a 12-h light/dark cycle, in groups of up to five per cage, with food and water available ad libitum. Behavioral tests were performed with 2- to 4-months old male or female mice between 8 AM and 12 PM during the light cycle in the animal facility. To reduce stress, mice were acclimated for 30 min to the testing environment on the day before the experiment and for an additional 5 min immediately prior to behavioral assays. Each mouse received a 4 μL intraplantar injection into the center of the right hindpaw using custom-made 25 μL Hamilton syringes fitted with 28-gauge needles. The needle was inserted into the superficial layer of the glabrous skin to ensure that the injection was restricted to the plantar surface. Chemicals injected: SLIGRL (2–5 mM), deoxycholic acid (DCA, 1 mM), IL-31 (2.5–6.3 nM), 5-HT (2–10 mM), LY344864 (2–4 mM), Compound 48/80 (0.1–0.5 µg/µL), Capsaicin (16 µM), and Bam8-22 (50 µM, custom synthesized by Genscript). To test itch and pain responses after morphine treatment, mice received an i.p. injection of morphine (0.1 mg/kg). Intraplantar injections of chemicals were performed 30 min after morphine treatment.
Immediately after injection, mice were placed individually in a plastic cylinder on a clear acrylic platform. Behavioral responses were recorded with a Sony HD camcorder (FDRAX33, 4K/24P) for 10 min from a mirror underneath the platform to visualize all behaviors directed to the hindpaw. Videos were analyzed in slow motion (0.25× normal speed) using VLC media player to distinguish specific behavioral responses. Licking behavior was identified by long head movements directed toward the injected paw and visible tongue. Biting behavior was defined as the mouse placing its toes into its mouth, sometimes with a pulling motion, or scraping the paw using the incisors.
Data are represented as mean ± SEM. Statistical analysis was performed using Welch’s t test, one-way ANOVA, or two-way ANOVA due to differences in the normality and sample size of the data in GraphPad Prism 10. The specific tests used to analyze each data set are indicated within the individual figure legends.
Ovalbumin-induced allergic itch model
For the allergy itch model, mice were sensitized by intraperitoneal (i.p.) injection of 50 µg ovalbumin (OVA) dissolved in phosphate-buffered saline, mixed with 2 mg aluminum hydroxide gel. Sensitizations were given twice at 2-week interval. 10 days after the second sensitization, 2–4 µg/µL OVA was administered using intraplanar injection described earlier.
Results
SLIGRL and DCA induce distinct behavioral responses in mouse glabrous skin
We first tested peptide SLIGRL-NH2, an agonist for PAR2 and MrgprC11, 18 to assess behavioral responses toward mice glabrous skin (Figure 1(a)–(d)). Biting behavior in the control mice is rare (average 0.28 ± 0.16 s), while mice exhibit frequent grooming behavior toward their hindpaw, the majority of which is licking (2.97 ± 1.19 s background licking in 10 min). We found that intraplantar injection of 2 mM SLIGRL did not induce a noticeable response. 5 mM SLIGRL induced significant licking compared with control, which became prominent around 5 min after injection and persisted until approximately 10 min (Figure 1(c) and (d)). No biting behavior was observed at either concentration, suggesting that SLIGRL injection induced pain sensation in the plantar hindpaw. This is consistent with previous findings that plantar administration of PAR2 agonists induce thermal and mechanical hyperalgesia.18,19 Although SLIGRL can also activate MrgprC11, a key itch receptor in glabrous skin, we did not observe any biting behavior, suggesting that pain sensation induced by PAR2 activation inhibited itch transmission. This is consistent with the hypothesis that pain afferents activate spinal interneurons to suppress itch. 20

Biting and licking behaviors after intraplantar injection of SLIGRL, DCA, and IL-31. (a–b) SLIGRL (5 mM) selectively induced licking, while DCA (1 mM) selectively evoked biting. (e–f) IL-31 (3.15 nM, 6.3 nM) induced both licking and biting. Biting (c and g) and licking (d and h) responses over a 10-min period are shown as mean ± SEM for each minute.
We next tested deoxycholic acid (DCA), one of the secondary bile acids that have been implicated in cholestatic pruritus21,22 (Figure 1). DCA is elevated in the plasma of cholestatic patients and induces itch when delivered intradermally in humans. 22 Notably, bile acid signaling shows clear species difference. In mice, bile acid receptor TGR5 is expressed in the nociceptor and mediates bile acid-induce itch.23,24 However, in humans, TGR5 is not expressed in DRG sensory neurons and instead bile acids activate human MrgprX4 to trigger itch sensation.21,22 Here, we use DCA as a potent pruritogen to assess glabrous skin itch behavior in our mouse model. In contrast to SLIGRL, we found that intraplantar DCA selectively evoked biting, not licking, which began approximately 1 min after injection and was predominantly concentrated within the first 3 min.
IL-31 induced both pain and itch in glabrous skin
IL-31, produced primarily by activated Th2 cells, is a cytokine pruritogen that directly links type 2 inflammation to sensory neuron activation.25,26 Mice carrying the IL-31 transgene develop severe itching and atopic dermatitis, demonstrating the critical role of IL-31 in itch and skin inflammation. 27 Previous studies have shown that intraplantar injection of IL-31 causes paw licking, while cheek injection evokes scratching. 25 In our experiments, intraplantar administration of IL-31 at 3.15 and 6.3 nM elicited both licking and biting behaviors in a dose-dependent manner (Figure 1(e)–(h)), indicating that IL-31 evokes both pain and itch sensation in glabrous skin. The licking response emerged at about 2 min, peaked shortly after, and gradually declined by 8 min, whereas biting behavior was distributed throughout the entire observation period.
5-HT and 5-HT1F receptor agonist as mediators of glabrous skin responses
Serotonin (5-hydroxytryptamine, 5-HT) is a biogenic amine produced primarily by mast cells in peripheral tissues and signals through a diverse family of 14 receptor subtypes. 28 Since many of these receptors are expressed in cutaneous cell types such as keratinocytes, fibroblasts, and sensory nerves, 29 intradermal delivery of 5-HT into human skin evokes both pain and itch sensations. 30 Consistently, cheek injection of 5-HT in mice elicits both scratching and wiping behaviors. 31 Hagiwara et al. 13 previously reported that 5-HT injection into the medial malleolus of the hindpaw induced licking at a low concentration and both licking and biting at higher concentrations. Our findings are consistent with this pattern (Figure 2(a)–(d)). At 2 mM, 5-HT showed no noticeable response, whereas at 5 mM it significantly increased licking without biting, and at 10 mM it triggered both behaviors. Among the multiple 5-HT receptors, HT1F has been shown as one of the key itch receptors in DRG sensory neurons. 32 Therefore, we tested LY344864, a selective 5-HT1F receptor agonist (Figure 2(e)–(h)). LY344864 at 4 mM also induced both licking and biting in mice hindpaws in a dose-dependent manner, which were primarily concentrated in the early phase following injection. Together, these results highlight the dose-dependent dual role of 5-HT as a mediator of both pain and itch sensation in glabrous skin.

Biting and licking responses after intraplantar injection of 5-HT and LY344864. (a–b) 5-HT induced dose-dependent responses, with low concentration (5 mM) triggering licking and higher concentration (10 mM) inducing both licking and biting. (e–f) 5HT1F agonist LY344864 evoked both licking and biting at 4 mM. Biting (c and g) and licking (d and h) responses over a 10-min period are shown as mean ± SEM for each minute.
Mast cell-dependent itch responses in plantar glabrous skin
Ovalbumin (OVA) is a widely used allergen to generate allergic responses in mouse models. Mice are first systemically sensitized with OVA to induce a Th2-dependent immune response. Subsequent epicutaneous or intradermal challenge with OVA triggers allergic inflammation characterized by mast cell degranulation, infiltration of eosinophils, and elevated Th2 cytokines. The inflammatory mediators, released from infiltrating immune cells, can directly or indirectly activate cutaneous sensory nerves, leading to nociceptive sensation. This model has been applied to various skin locations such as the ear, cheek, and trunk. However, plantar glabrous skin application has not been reported. Here, we found that intraplantar administration of OVA after sensitization produced both licking and biting behaviors starting from 3 µg/µL (Figure 3(a)–(d)), and both responses were mainly observed during the later phase.

Biting and licking responses after intraplantar injection of OVA and compound 48/80. (a–b) Intraplantar administration of OVA after sensitization produced both licking and biting behaviors at 3 and 4 µg/µL. (e–f) Compound 48/80 evoked biting at 0.3 µg/µL, induced both licking and biting at 0.5 µg/µL. Biting (c and g) and licking (d and h) responses over a 10-min period are shown as mean ± SEM for each minute.
While OVA-induced allergic itch model involves IgE-induced mast cell activation, mast cell degranulators such as compound 48/80 can directly induce mast cell degranulation independently of IgE signaling. Here, we used compound 48/80 to test whether mast cell activation alone is sufficient to evoke itch-related behaviors in this skin region. Results showed that intraplantar injection of compound 48/80 also induced both biting and licking behaviors at 0.5 µg/µL (Figure 3(e)–(h)), suggesting that activation of mast cells is capable of eliciting itch-related behaviors in glabrous skin, independent of allergen-specific IgE signaling.
Glabrous skin biting is a nocifensive behavior associated with itch
To further confirm that glabrous skin biting is a behavioral response associated with itch rather than pain, we tested the responses of the mice to Bam8-22, DCA, and capsaicin following morphine treatment (Figure 4). As expected, capsaicin induced robust licking without biting and morphine treatment completely abolished this licking response. In contrast, both Bam8-22 and DCA induced strong biting behavior, which were not affected by morphine. The licking observed in the Bam8-22 and DCA groups are comparable to the saline controls and are part of the grooming behaviors, which was also insensitive to morphine treatment. These results confirm that glabrous skin biting is a nocifensive behavior associated with itch.

Effects of morphine on biting and licking induced by Bam8-22 (50 µM), DCA (1 mM), and capsaicin (16 µM). Morphine blocked capsaicin-induced licking (b), but did not affect biting induced by Bam8-22 or DCA (a), demonstrating that glabrous skin biting is a nocifensive response associated with itch.
Based on these behavioral results, we generated a scatter plot comparing the mean biting and licking times elicited by each pruritogen (Figure 5). Among all tested compounds, histamine, DCA, and Bam8-22 predominantly induced itch-related responses rather than pain in the glabrous skin. In contrast, capsaicin, SLIGRL, and 5-HT 5 mM mainly elicited pain-associated behaviors. IL-31, 5-HT 10 mM, LY344864, OVA, and compound 48/80 evoked both biting and licking, suggesting the activation of both itch and pain neural pathways.
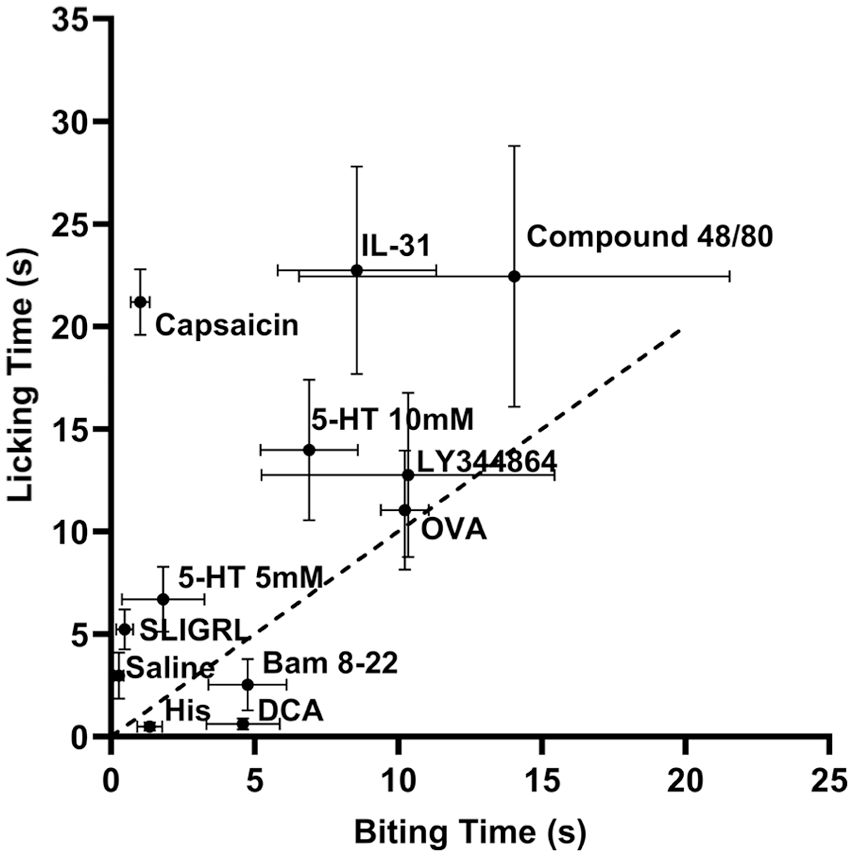
Scatter plot showing average biting and licking time induced by intraplantar injection of pruritogens.
Discussion
While the plantar glabrous skin is a well-established site for rodent pain behavioral assays, most of the previous studies have defined all recorded responses as pain-related.4,12 Our studies demonstrate that licking and biting are the two primary behaviors available to mice in response to stimuli applied to their plantar hindpaw and the dichotomy between the two naturally reflects distinct sensory experiences: licking corresponds to pain sensation, whereas biting is a nocifensive behavior associated with itch. When exposed to painful stimuli, mice typically respond with gentle licking, a behavior serves as self-comforting and protective. In contrast, pruritic stimuli elicit biting. We also often observe scraping or pressing against the glabrous skin surface using their incisors. These behaviors likely represent attempts to generate strong mechanical stimulation, analogous to human scratching, which can transiently relieve itch through activation of spinal inhibitory interneurons. 33 Mice rarely bite painful areas, as such action would worsen pain sensation. Similar behavioral distinctions have been observed and used to differentiate pain and itch behaviors following chemical injection or treatment to the whole hindpaw or the calf of hind limb.4,13–15
Opioid signaling provides a well-established pharmacological tool to differentiate between pain- and itch-related pathways. μ-opioid agonists such as morphine are potent analgesics that suppress nociceptive transmission at both spinal and supraspinal levels. 34 Paradoxically, they can evoke or exacerbate itch in humans and experimental models and do not attenuate pruritogen-evoked itch behavior in rodents. 35 This opposing modulation forms the basis for using morphine as a discriminator between pain and itch behaviors: pain-related responses are typically abolished by morphine, whereas itch-related behaviors persist. Indeed, our results show that morphine completely abolished capsaicin-induced licking, whereas biting elicited by pruritogens Bam8-22 or DCA was unaffected. These results reinforce the notion that glabrous skin licking reflects painful sensation, whereas biting indicates itch.
To further validate the feasibility of the glabrous skin itch model, we examined behavioral responses of mice following intraplantar delivery of multiple pruritogens. Our previous studies have demonstrated that both histamine and Bam8-22 specifically induced biting behaviors, consistent with itch-specific signaling.11,17 In this study, we found that DCA produced similar results, identifying it as another effective pruritogen for glabrous skin. However, SLIGRL, which activates both PAR2 and MrgprC11, induced licking without biting, consistent with pain-specific signaling. Although MrgprC11 is a well-established itch receptor, our data suggest that PAR2-mediated pain signals may override itch in this skin region, possibly via spinal inhibitory circuits. These findings support the conclusion that the glabrous skin model is a useful behavioral model to discriminate pain and itch sensation.
Using the cheek injection model, SLIGRL has been shown to induce robust scratching without wiping, consistent with itch-specific activation. 31 However, glabrous skin injection of SLIGRL specifically induced pain behavior, suggesting regional differences in somatosensory processing. We have observed similar phenomena from MrgprD+ nerves: activation of MrgprD+ nerves in hairy skin, including the cheek and nape of the neck, elicits robust scratching,17,36 whereas stimulation of MrgprD+ nerves in the plantar hindpaw evokes only innocuous tactile responses and withdrawal reflexes without nocifensive behavior.37–39 These findings support the hypothesis that identical stimuli can engage different somatosensory circuits at different skin locations.
In contrast to SLIGRL, which elicited pain without itch behavior, 5-HT, IL-31, and OVA-induced allergy model produced concurrent licking and biting behaviors. This likely reflects their ability to activate both pruriceptive and nociceptive pathways. 5-HT is known to activate mixed populations of sensory neurons in vivo, including both pruriceptors and nociceptors, therefore, both pain and itch sensation have been reported when 5-HT was delivered intradermally in mice and human.13,30,31 Consistently, our results show that 5-HT produced dose-dependent responses, with low concentrations inducing licking and higher concentrations evoking both licking and biting. Similarly, IL-31 induced both licking and biting behaviors. Receptors for both 5-HT and IL-31 are expressed not only on cutaneous nerves but also on multiple cutaneous cell types.29,40,41 Thus, these pruritogens can activate sensory nerves both directly, by stimulating the nerves, and indirectly, by triggering the release of nerve-activating mediators from cutaneous cells. In parallel, OVA-induced allergic responses rapidly increase the inflammatory mediators in the skin, which also likely stimulate both pruritic and nociceptive signaling through these same direct and indirect routes. Although nociceptive input can suppress itch transmission via spinal inhibitory interneurons, this masking is not complete, especially when pruriceptor activation is strong. Thus, itch behaviors can still emerge even when nociceptive input is present.
Taken together, our study establishes the hindpaw glabrous skin biting assay as a robust model to investigate itch mechanisms. By differentiating licking and biting behaviors, this model allows dissection of the molecular and neural pathways underlying pain and itch. These results provide a foundation for translational studies targeting itch pathways in human palms and soles.
Footnotes
Acknowledgements
We thank the Department of Animal Resources at Georgia Institute of Technology for the animal care and services.
Author contributions
Conceptualization: LH; Formal Analysis: SW, HS, KL, RS; Investigation: SW, HS, KL, RS; Writing – Original Draft Preparation: SW; Writing – Review and Editing: SW, LH; Supervision: LH; Funding Acquisition: LH.
Data availability statement
No datasets were generated or analyzed during the current study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by grants from US National Institutes of Health (HL173002) and National Science Foundation (2334697) to LH.
Declaration of generative AI and AI-assisted technologies in the writing process
During the preparation of this work the authors used ChatGPT in order to improve readability and language. After using this tool, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
