Abstract
Itch is a somatosensory sensation to remove potential harmful stimulation with a scratching desire, which could be divided into mechanical and chemical itch according to diverse stimuli, such as wool fiber and insect biting. It has been reported that neuropeptide Y (NPY) neurons, a population of spinal inhibitory interneurons, could gate the transmission of mechanical itch, with no effect on chemical itch. In our study, we verified that chemogenetic activation of NPY neurons could inhibit the mechanical itch as well as the chemical itch, which also attenuated the alloknesis phenomenon in the chronic dry skin model. Afterwards, intrathecal administration of NPY1R agonist, [Leu31, Pro34]-NPY (LP-NPY), showed the similar inhibition effect on mechanical itch, chemical itch and alloknesis as chemo-activation of NPY neurons. Whereas, intrathecal administration of NPY1R antagonist BIBO 3304 enhanced mechanical itch and reversed the alloknesis phenomenon inhibited by LP-NPY treatment. Moreover, selectively knocking down NPY1R by intrathecal injection of Npy1r siRNA enhanced mechanical and chemical itch behavior as well. These results indicate that NPY neurons in spinal cord regulate mechanical and chemical itch, and alloknesis in dry skin model through NPY1 receptors.
Introduction
The sensation of itch elicits an important protective scratching response to harmful stimuli, such as irritants and parasites, which is a defensive mechanism for animal survive.1,2 According to different sensory elements, itch evolves two forms: mechanical and chemical itch. The former one is activated by light tactile stimuli, 3 while the latter one is evoked by noxious painful stimuli. 4 It has been proved that selectively ablation or silencing of NPY-lineage interneurons expressing NPY transiently during development or persistently into adulthood in spinal cord induced a histamine-independent chronic itch state, which presented as an increase in sensitivity to mechanical itch, without affecting chemical itch evoked by compound 40/80 and chloroquine or pain behaviors. 3 This limited effect of NPY neurons is surprising, causing NPY neurons have been proved to modulate acute and chronic pain.5,6
Neuropeptide Y neurons are widely located in spinal cord dorsal horn laminae I-IV, mainly in laminae III–IV (70.4 ± 0.3%), and receive Aβ, Aδ and C fiber inputs, 3 including areas innervated from glabrous skin, while mechanical itch is restricted to hairy skin.7,8 They also account for one-third of all inhibitory interneurons in superficial dorsal horn (SDH: laminae I and II), which participating in modulating somatosensory information. 7 Several studies also indicated that NPY neurons innervated a population of nociceptive projection cells in the anterolateral system (ALS) underlie the perception of pain, skin temperature and itch.9–12 To sum up, Bourane et al have demonstrated NPY neurons’ limited role in modulating mechanical itch perhaps due to targeting a broad population of inhibitory interneurons that express NPY during development.
It has been indicated that NPY-NPY1R signaling is essential for the central transmission of light punctate touch and mechanical but not chemical itch. 13 They draw the conclusion mainly depending on pruritogen-evoked scratching behaviors unaffected by ablation of Y1Cre neurons. 13 Same with the strategy in ablating NPY interneurons, 3 they also ablated a broad population of Y1-expressing neurons during development transiently and persistently into adulthood. Furthermore, Y1 receptor is widely expressed in mouse spinal cord and dorsal root ganglion, containing at least seven different kinds of subtypes. 14 Y1 receptor is a Gi-protein coupled receptor that exerts potent effect on neuronal function via inhibition of intracellular calcium signaling.15,16 Also, Npy1r, coding the Y1 receptor, is abundantly expressed in glutamatergic excitatory neurons in the superficial dorsal horn of the spinal cord.17,18 It has been reported that LP-NPY intrathecal injection could attenuate both mechanical and cold allodynia in chronic constructive injury neuropathic pain model, 19 indicating that NPY-NPY1R signaling also has an analgesic effect in pain regulation. Recently, it has been demonstrated that activation of inhibitory NPY interneurons could reduce chloroquine-evoked chemical itch behavior via a circuit involving GABAergic input to excitatory interneurons expressing the gastrin-releasing peptide receptor (GRPR). 5 Given that the controversial role of NPY-NPY1R signaling in somatosensory information transmission, we set out to investigate the relationship between NPY-NPY1R signaling network with mechanical, chemical itch and alloknesis using chemogenetic, targeted pharmacological and small interfering RNA treatment approaches.
Materials and methods
Animals
Experiments were carried out on C57BL/6J obtained from Shanghai SLAC Laboratory Animals (Shanghai, China), Npycre mice (Stock no.027851, Jax mice), and their wild-type littermates unless indicated otherwise. All mice were housed in clear plastic cages with no more than five mice per cage in a controlled environment at a constant temperature of ∼23°C and humidity of 50 ± 10% with a light-dark cycle of 12 h–12 h. The animals had food and water available ad libitum. All experiments conform to guidelines set by the Institutional Animal Care and Use Committee of Shanghai Jiao Tong University School of Medicine and implemented in accordance with the relevant regulations of the Experimental Animal Center of Shanghai Jiao Tong University School of Medicine. All the efforts were made to minimize animal suffering and to reduce the number of animals used.
Intraspinal virus injection and chemogenetics
NpyCre mice were anesthetized with sodium pentobarbital (50 mg/kg) intraperitoneally (i.p.) for analgesia. Cervical vertebrae were exposed at C2-C6, and mounted onto a stereotaxic frame with spinal adaptors. After exposing spinal cord, the dura was incised with a sharp needle to expose the spinal cord surface. AAV8-Syn-DIO-hM3Dq-mCherry (2.0 × 1013 vg/mL) was injected into the both sides of the spinal cord at six sites between successive vertebrae at C3-C4-C5 with a Hamilton Neuros-syringe with beveled needle (catalog number: 65458-02, 34 gauge, 20° angle). The syringe needle was inserted into the dorsal spinal cord vertically at a depth of ∼500 μm to target the lamina II-III. 500 nL AAV per site was injected at a rate of 50 nL/min with a Stoelting Quintessential Injector (QSI, catalog number: 53311). After injection completed 10 min, the needle was slowly removed. The skin was sutured and painted with triple antibiotic ointment and lidocaine. After surgery 3 weeks, mice were ready for behavior tests. For chemogenetic experiments, behavioral tests were performed 2 h after clozapine (0.1 mg/kg, Sigma, Cat. No. C6305) i.p. injections.
Dry skin model
The dry skin model was implemented as described. 20 Briefly, the nape of mice at 8∼12 weeks of age was shaved and a mixture of acetone (Sigma, Cat. No. 179124) and diethylether (Sigma, Cat. No. 309966) (1:1) was painted on the neck skin for 15 s, followed immediately by a 30 s of distilled water application (AEW). This regiment was administrated twice daily for 5∼7 days. Littermate control mice received water only for 45 s on the same schedule. Spontaneous scratches were recorded for 30 min on the morning following the 5th day of AEW treatment. Pharmacological interventions were carried out after the 6th or 7th day of AEW treatment.
Behavior tests
Mechanical itch or alloknesis test
The fur on the nape was shaved at least 3 days before experiments. Mice were acclimated in a plastic chamber (20 × 10 × 12.5 cm) for 3 days. Mechanical stimuli on the nape were delivered with von Frey hairs ranging from 0.008 g to 1.0 g and held for up to 1 s or until the mice responded. Positive responses were counted as hindlimb scratching towards the site of mechanical stimulation. Each von Frey filament was tested 10 times on different random points of the nape with an interval of 10 s. For mice with AEW treatment, mechanical stimuli were delivered on the border of the AEW-treated area. NPY1R agonist (LP-NPY, 1 nmol/10 µl, Tocris, Cat. No.1176) or NPY1R antagonist (BIBO 3304, 1 µg/10 µl, Tocris, Cat. No. 2412) was intrathecally (i.t.) injected 10 min before mechanical itch test. The number of scratching episodes for each von Frey hair were plotted for comparisons.
Chemical itch behavior
Scratching behaviors induced by chloroquine (CQ, Sigma, Cat. No. C6628), histamine (His, Sigma, Cat. No. H7125), Compound 48/80 (Sigma, Cat. No. C2313) were performed as previously described. 21 Briefly, the injection area was shaved at least 3 days before experiments. Prior to the experiments, each mouse was placed in a plastic arena (10 × 11 × 15 cm) for 30 min to acclimate. Mice were briefly removed from the chamber and intradermally (i.d.) injected in the right side of nape with CQ at the dose of 200 µg in 50 μL saline, while for Npy1r siRNA experiment, which was at 50 µg. For the LP-NPY test, mice were i.d. injected in the right side of nape with CQ 200 µg, histamine 200 µg or Compound 48/80 100 µg. For the BIBO 3304 test, mice were i.d. injected in the right side of nape with CQ 50 µg, histamine 50 µg or Compound 48/80 25 µg. LP-NPY (1 nmol/10 µl) or BIBO 3304 (1 µg/10 µl) was i.t. injected 10 min before reagents injection. Hind limb scratching behavior towards the injected area was observed and counted by observers blinded to the group or genotype of the mice.
C-Fos induction
C-Fos induction was performed as previously described. 22 Briefly, for chemogenetic activation, NpyCre mice with AAV8-Syn-DIO-hM3Dq-mCherry virus in cervical were i.p. injected with clozapine (0.1 mg/kg) without other stimulation. After 2 h, mice were perfused for c-Fos immunohistochemistry. For mechanical itch stimulus, the right nape was stimulated with 0.07 g von Frey hair 20 times with 10-s intervals. For chemical itch stimulus, mice were i. d. injected with CQ (200 μg in 50 μL saline) at the right nape after anesthetized by isoflurane. For sham group, mice were not given any stimulations. After 90 min, mice were perfused for c-Fos immunohistochemistry.
Immunohistochemistry
Immunohistochemistry (IHC) was performed as previously described. 23 After c-Fos induction, mice were anesthetized (pentobarbital, 50 mg/kg, i.p.) and perfused intracardially with 0.01 M PBS pH 7.4 followed by 4% paraformaldehyde (PFA) in PBS. Spinal cord tissues were dissected, post-fixed for 6-8 h, and cryoprotected in 20% sucrose in PBS overnight at 4°C. Subsequently, tissues were embedded in optimum cutting temperature medium (Tissue-Plus O.C.T. compound, Fisher Healthcare) and were cut at 30 µm by a Leica CM1950 cryostat. Free-floating frozen sections were blocked for 1 h in a 0.01 M PBS solution containing 2% donkey serum and 0.1% Triton X-100 followed by incubation with primary antibody (rabbit anti-c-Fos, 1:4000, Abcam, ab190289; goat anti-NPY, 1:1000, Novus, NBP1-46535) overnight at 4°C, washed three times with PBS, secondary antibody (488-conjugated donkey anti-rabbit, 1:500, Jackson ImmunoResearch, 711-545-152; 594-conjugated donkey anti-goat, 1:500, Jackson ImmunoResearch 705-585-147) for 2 h at room temperature and washed again for three times. Sections were mounted on slides and ∼200 μL FluoromountG (Southern Biotech) was placed on the slide with a coverslip. Fluorescent Images were taken using a Nikon C2 + confocal microscope system (Nikon Instruments, Inc.).
Small interfering RNA (siRNA) treatment
Negative control siRNA (SIC001) and selective duplex siRNA for mouse Npy1r mRNA (NM_010934) were purchased from Sigma. RNA was dissolved in diethyl pyrocarbonate-treated PBS and prepared immediately prior to administration by mixing the RNA solution with RVG-9R peptide (Bachem, Cat. No. H-7502.0500), in a molar ratio of 1:10 (siRNA:RVG).24,25 The final concentration of RNA was 2 µg/10 µl and was intrathecally injected in spinal cord. Mice were injected once daily for six consecutive days as described previously.26–28 Behavioral testing was carried out 24 h after the last injection. The spinal cord and dorsal root ganglion tissues were collected for RT-PCR after one more day of siRNA injections.
Real-time RT-PCR
The spinal cord and DRG samples were dissected from control siRNA and Npy1r siRNA-treated mice. Real-time RT-PCR was performed as previously described with Fast-Start Universal SYBR Green Master (Roche Applied Science).26,27 All samples were assayed in duplicates (heating at 95°C for 10 s and at 60°C for 30 s). Data was analyzed using the Comparative CT Method (StepOne Software version 2.2.2.), and the expression of target mRNA was normalized to the expression of Gapdh. The primers used are Gapdh: foward 5′-CCCAGCAAGGACACTGAGCAA-3’; reverse 5′-TTATGGGGGTCTGGGATGGAAA-3’; Npy1r: forward 5′- CTCGTCCCGCTTCAACAGAG-3’; reverse 5′- TCAAAACGGATCAAATCTTCAGCA-3’; Npy2r: forward 5′- AAATCGGACCTGCTTTGGGT-3’; reverse 5′- ACCAGTTCACTCTCACTTGGC-3’.
Statistical analysis
The mice were distributed into different groups randomly. The number of mice is no less than six per group for behavior experiments. All the data were presented by means ± standard error of the mean. Statistical tests are indicated in figure legends when performed, including two-tailed, paired Student’s t test for two groups comparison, two-way ANOVA with Tukey post hoc for more than two-group comparison for results from behavior tests and IHC images. Statistical analyses were performed using Prism 7 (v7.0c, GraphPad, San Diego, CA). Normality and equal variance tests were performed for all statistical analyses. p < .05 was considered statistically significant.
Results
The effect of activation of NPY neurons on mechanical and chemical itch
Conditional ablation of NPY inhibitory neurons has been shown to increase mechanical itch.
3
The effect of activation of NPY neurons on itch, however, was unclear. Firstly, we used c-Fos, a neuronal activity marker, as a surrogate to determine whether NPY neurons are activated in mechanical and chemical itch stimuli. According to previous study, NPY neurons were localized in laminae III-IV mainly and laminae I-II lessly.
3
We compared the c-Fos expression pattern between the laminae after mechanical and chemical itch stimuli. The sham group mice without any stimuli showed little c-Fos expression in laminae I-II and III-IV (Figure 1(a), (d) and (e)). The IHC results showed that both mechanical itch, evoked by a von Frey filament (0.07 g) to the hairy skin of the nape, and chemical itch, evoked by i.d. injection of chloroquine (CQ), could induce c-Fos activity in NPY neurons (Figure 1(b)–(d)). For chemical itch, significant amounts of c-Fos were found in NPY neurons, same with mechanical itch (30.4 ± 2.73% vs 22.69 ± 2.89%) in laminae I-II, while less than mechanical itch (9.26 ± 1.52% vs 23.25 ± 2.26%) in laminae III-IV (Figure 1(d)). The percentage of c-Fos/NPY double-positive neurons in c-Fos neurons were similar between mechanical and chemical itch (laminae I-II: 31.72 ± 3.68% vs 39.44 ± 1.96%; laminae III-IV: 38.06 ± 2.98% vs 30.78 ± 4.57%) (Figure 1(e)). To further examine the role of NPY inhibitory neurons in itch, chemogenetic activation of NpyCre neurons was performed after injection of AAV8-hSyn-DIO-hM3Dq (Gq)-mCherry virus into the spinal cord of NpyCre mice. Compared to the control mice, NpyCre mice treated with clozapine showed pronounced deficits in mechanical and chemical itch (Figure 1(f) and (g)). IHC image showed that the c-Fos expression in NpyCre neurons infected with mCherry virus after clozapine 0.1 mg/kg i.p. injection 2 h (Figure 1(h) and (i)), confirming the activation of NpyCre neurons. These results make sure that activation of NPY neurons could inhibit both mechanical and chemical itch. Activation of NPY neurons in cervical spinal cord could inhibit both mechanical and chemical itch. (a)–(c) IHC images of NPY+ (red) and c-Fos+ (green) neurons in the cervical spinal cord of C57BL/6J mice among sham, MI and CQ group. Arrowheads indicated c-Fos/NPY double-positive neurons. Scale bar, 50 μm. (d, e) Comparison of the percentage of c-Fos/NPY double positive neurons in NPY positive neurons (d) and c-Fos positive neurons (e) in laminae I-II, III-IV under different conditions. n = 6 sections from three mice per group. Two-way ANOVA with Tukey post hoc test, ns: p > .05, *p < .05, **p < .01, ***p < .001. (f) The scratching number induced by mechanical stimulation (von Frey 0.04 g, 0.07 g, 0.16 g and 0.4 g) decreased after chemogenetic activation of NPY neurons in cervical spinal cord. N = 9 mice per group. **p < .01, ***p < .001 versus the WT group by two-way ANOVA with Tukey post hoc test. (g) The scratching number induced by chemical itch stimulation CQ 200 μg decreased after chemogenetic activation of NPY neurons in cervical spinal cord. N = 9 mice per group. *p < .05 versus the WT group by two-tailed unpaired Student’s t test. (h) IHC image of mCherry+ (red) and c-Fos+ (green) neurons in the cervical spinal cord of NpyCre mice infected with AAV8-hSyn-DIO-hM3Dq (Gq)-mCherry virus followed by clozapine injection. Arrowheads indicated c-Fos/mCherry double-positive neurons. Scale bar, 50 μm. (i) Visualization and quantification of overlap of c-Fos and mCherry expression in laminae I-IV (sections from n = 3 mice were examined for quantification). MI: mechanical itch, CQ: chloroquine.
The effect of activation of NPY neurons on dry skin itch model
To ascertain whether NPY neurons have a role in alloknesis associated with chronic itch, we employed a mouse dry skin model treated with acetone-ether-water (AEW),
29
in which the loss of Piezo2-Merkel cell signaling contributed to alloknesis.
30
We performed chemogenetic activation of NpyCre neurons in the AEW mice, and found that alloknesis and spontaneous scratching behavior were significantly attenuated compared to the control group (Figure 2(a) and (b)). Activation of NPY neurons has the potential to inhibit dry skin-related alloknesis. Activation of NPY neurons in cervical spinal cord could inhibit both mechanical and spontaneous scratching itch behavior in AEW model. (a) The scratching number induced by mechanical stimulation (von Frey 0.04 g, 0.07 g, 0.16 g and 0.4 g) decreased after chemo-activation of NPY neurons in cervical spinal cord of AEW model mice. N = 7 mice per group. ***p < .001 versus the WT group by two-way ANOVA followed by Tukey post hoc test. (b) The spontaneous scratching number decreased after chemo-activation of NPY neurons in cervical spinal cord of AEW model mice. N = 7 mice per group. *p < .05 versus the WT group by two-tailed unpaired Student’s t test.
NPY1 receptor’s role in mechanical and chemical itch
Npy1r is a principal receptor that is abundantly expressed in glutamatergic excitatory neurons in the superficial dorsal horn of the spinal cord.17,18 We postulated that NPY neurons may inhibit mechanical itch through NPY1R. To test this possibility, we first performed i.t. injection of a selective NPY1R agonist, LP-NPY,
31
and found that LP-NPY dramatically reduced mechanical itch (Figure 3(a)). Moreover, it also markedly reduced CQ-, compound 48/80- and histamine-induced chemical itch (Figure 3(b)–(d)). Next, we did the i.t. injection of a selective NPY1R antagonist, BIBO 3304, and discovered that BIBO 3304 could significantly enhance mechanical itch (Figure 3(e)), contrarily. For chemical itch, it also clearly enhanced CQ-, compound 48/80- and histamine-induced chemical itch (Figure 3(f)–(h)). The opposite LP-NPY and BIBO 3304 results indicated that NPY1R participated in the transmission of mechanical and chemical itch. NPY1 receptor’s role in mechanical and chemical itch. (a) The scratching number induced by mechanical stimulation (von Frey 0.07 g, 0.16 g and 0.4 g) decreased after i.t. injection of LP-NPY (1 nmol/10 μL). N = 10 mice per group. *p < .05, ***p < .001 versus the saline group by two-way ANOVA with Tukey post hoc test. (b)–(d) The scratching number induced by chemical itch stimulation CQ 200 μg, compound 48/80 100 μg and histamine 200 μg decreased after i.t. injection of LP-NPY (1 nmol/10 μL). N = 10 mice per group. *p < .05, ***p < .001 versus the saline group by two-tailed unpaired Student’s t test. (e) The scratching number induced by mechanical stimulation (von Frey 0.02 g, 0.04 g, 0.07 g, 0.16 g, 0.4 g, 0.6 g and 1.0 g) increased after i.t. injection of BIBO 3304 (1 μg/10 μL). N = 8 mice per group. *p < .05, ***p < .001 versus the saline group by two-way ANOVA with Tukey post hoc test. (f)–(h) The scratching number induced by chemical itch stimulation CQ 50 μg, compound 48/80 25 μg and histamine 50 μg increased after i.t. injection of BIBO 3304 (1 μg/10 μL). N = 10 mice per group. *p < .05 versus the saline group by two-tailed unpaired Student’s t test. LP-NPY: NPY1R agonist; BIBO 3304: NPY1R antagonist; i.t.: intrathecal.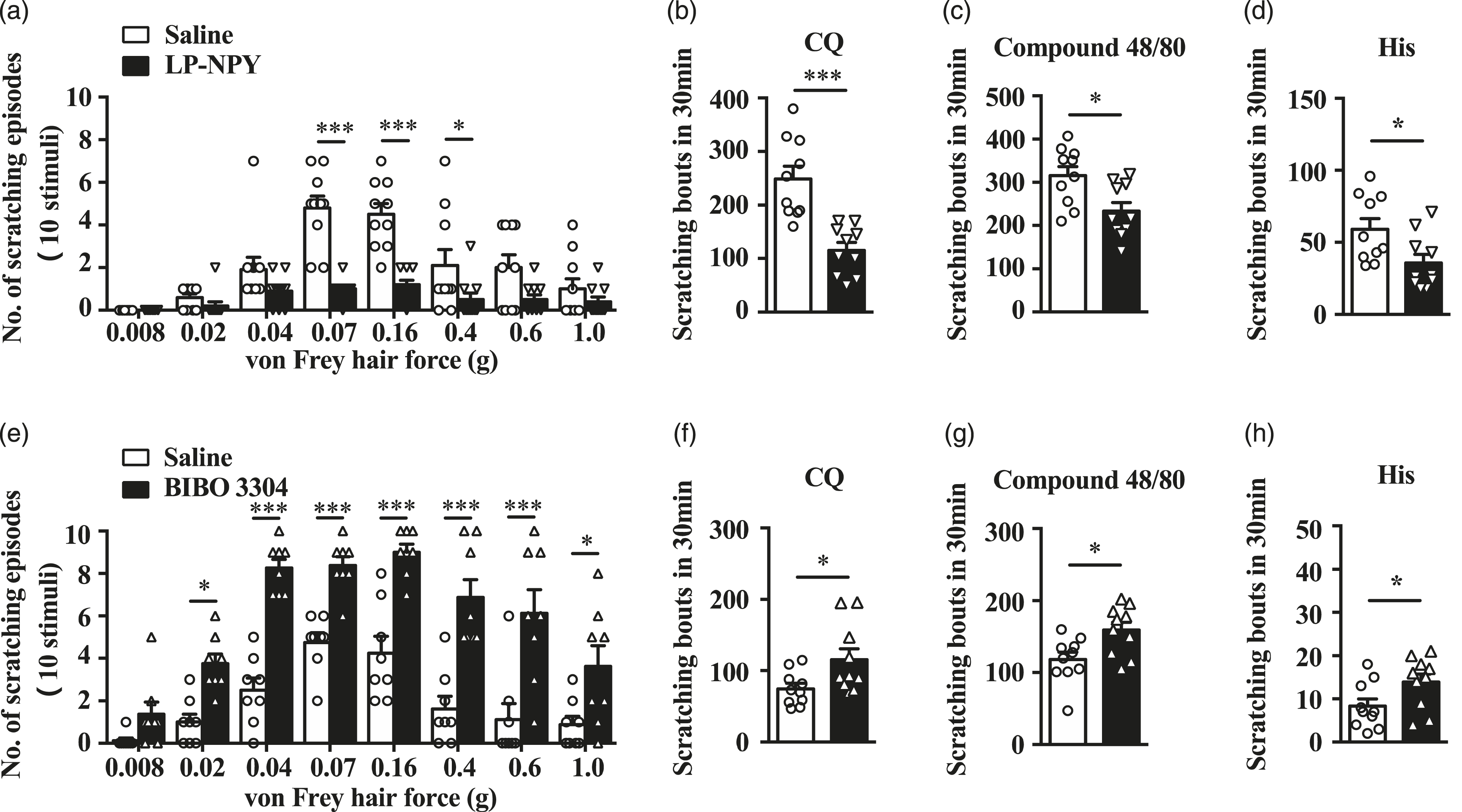
Effect of knock down Npy1r on mechanical and chemical itch
To confirm the function of NPY1R in mechanical and chemical itch, we next knocked down Npy1r by i.t. injection of siRNA against Npy1r and found that mice with Npy1r siRNA treatment exhibited significant increase in mechanical itch and CQ-induced itch as compared to the control siRNA injection (Figure 4(a) and (b)). The specificity of knockdown of Npy1r, but not Npy2r, in the spinal cord was confirmed by qRT-PCR (Figure 4(c)). Neither Npy1r nor Npy2r expression was changed significantly in dorsal root ganglion neurons (Figure 4(d)). Similar enhancing effects on mechanical and chemical itch between Npy1r siRNA and BIBO 3304 treatment confirmed that NPY1R’s role in inhibition of mechanical and chemical itch. Knock down NPY1R’s effect in mechanical and chemical itch. (a) The scratching number induced by mechanical stimulation (von Frey 0.02 g, 0.04 g, 0.07 g, 0.16 g and 0.4 g) increased after consecutively i.t. injection of Npy1r siRNA 6 days. N = 10 mice per group. *p < .05, **p < .01 versus the control group by two-way ANOVA with Tukey post hoc test. (b) The scratching number induced by chemical itch stimulation CQ 50 μg increased after consecutively i.t. injection of Npy1r siRNA 6 days. N = 9 mice per group. *p < .05 versus the control group by two-tailed unpaired Student’s t test. (c) The levels of Npy1r transcripts decreased in the spinal cord following Npy1r siRNA treatment, while not affect the levels of Npy2r transcripts, as detected by qRT-PCR. N = 4 mice per group. ***p < .001, ns: p > .05 versus the control group by two-way ANOVA with Tukey post hoc test. (d) Neither levels of Npy1r nor Npy2r transcripts in the DRG affected by the Npy1r siRNA treatment, as detected by qRT-PCR. N = 4 mice per group. ns: p > .05 versus the control group by two-way ANOVA with Tukey post hoc test. SC: spinal cord; DRG: dorsal root ganglion.
NPY1R’s role in chronic itch of dry skin model
Above-mentioned results indicated that chemo-activation of NPY neurons in cervical spinal cord could inhibit both alloknesis and spontaneous scratching behavior in AEW dry skin itch model. Whether the inhibition effect is through NPY1R like the normal state is unclear. First, we did i.t. injection of NPY1R agonist (LP-NPY) and it could greatly decrease AEW-associated alloknesis (Figure 5(a)). Moreover, it also dramatically attenuated spontaneous dry skin itch (Figure 5(b)). In addition, NPY1R antagonist (BIBO 3304) i.t. injection could reverse the alloknesis phenomenon which was inhibited by NPY1R agonist (LP-NPY) in AEW model. These results indicated that pharmacological activation of NPY1R neurons could also have the potential to inhibit the dry skin itch. NPY1 receptor’s role in mechanical and spontaneous itch behavior of AEW model. (a) The scratching number induced by mechanical stimulation (von Frey 0.02 g, 0.04 g, 0.07 g, 0.16 g, 0.4 g, 0.6 g and 1.0 g) decreased after i.t. injection of LP-NPY (1 nmol/10 μL) in AEW model mice. N = 9 mice per group. ***p < .001 versus the saline group by two-way ANOVA with Tukey post hoc test. (b) The spontaneous scratching number in AEW model mice decreased after i.t. injection of LP-NPY (1 nmol/10 μL). N = 9 mice per group. ***p < .001versus the saline group by two-tailed unpaired Student’s t test. (c) The LP-NPY- inhibited scratching number induced by mechanical stimulation (von Frey 0.04 g, 0.07 g, 0.16 g, 0.4 g, 0.6 g and 1.0 g) in AEW model reversed after i.t. injection of BIBO 3304 (1 μg/10 μL). N = 8 mice per group. ***p < .001, ns: p > .05 versus the saline group by two-way ANOVA with Tukey post hoc test. LP-NPY: NPY1R agonist; BIBO 3304: NPY1R antagonist; i.t.: intrathecal.
Discussion
Our study explored the inhibitory function of NPY neurons in itch and demonstrated that they exerted inhibitory roles in both mechanical and chemical itch via NPY1R. Chemogenetic activation of NPY neurons could inhibit both von Frey filament-evoked mechanical itch and chloroquine-evoked chemical itch, consistently with previous study. 5 Also, pharmacological data suggested that NPY neurons acted an inhibition role in mechanical and chemical itch via NPY1R signaling. One seemingly unexpected finding is that NPY neurons, a gate for mechanical itch, 3 also inhibit chemical itch, similar with previous study. 32 This may be explained as that NPY neurons are distributed throughout the dorsal horn laminae I-IV, 3 while mammal spinal cord dorsal horn lamina I or II, V and ventral parts could also receive unmyelinated C fiber and myelinated Aδ fiber inputs to transmit pain, itch and temperature sensation,33,34 which observation argues that NPY neurons are unlikely to be modality- or function-specific to inhibit mechanical itch sensation. In addition, pervious study has indicated that activation of NPY interneurons generated a powerful direct GABAergic inhibition of GRPR interneurons selectively. 5
Indeed, abundant studies have implicated both NPY and NPY1R in inhibition of nociceptive processes in the spinal cord,6,35–39 particularly participating in widely inhibition of neuropathic and inflammatory pain behavior.40,41 The discrepancies among Bourance et al’s, 3 Acton et al’s 13 and ours may be due to the approaches used. For instance, the spinal ablation or silence of NPY, NPY1R neurons might have masked the phenotype resulting from possible compensatory effect or partial inhibition,3,13 as pointed by this paper. 5 In this regard, pharmacological or chemogenetic activation approach should be considered as a technique of choice for examining the role of inhibitory neurons. Furthermore, the finding that less than 20% of NPY neurons receive mono- or poly-Aβ inputs (most of which are innervated by C/Aδ fibers, 3 mainly transmitting noxious sensory information) also supports a broader role for NPY neurons in inhibition of sensory transduction. Given that NPY1R is enriched in laminae I-II glutamatergic excitatory neurons, 17 which also express genes involved in pain (e.g., somatostatin),18,42 it is conceivable that a significant percentage of NPY1R neurons exerts an anti-nociceptive function, in parallel with the NPY-NPY1R inhibitory pathway for itch. Considering our results that i.t. injection of NPY1R agonist LP-NPY could inhibit the scratching behavior induced by CQ, compound 48/80 and histamine (Figure 3(b)–(d)), and Npy1r siRNA treated mice showed more scratching behavior induced by CQ (Figure 4(b)), we draw the conclusion that NPY-NPY1R pathway could also inhibit the chemical itch sensation. However, the itch-specific neurons involved in the pathway are not clear. It has been proved that tdTomato+ neurons of Y1Cre mice have no overlap with GRPR neurons, while have co-expression with NK1R+ neurons in lamina I and laminae III-IV. 13 Some studies indicated that NK1R+ projection neurons conveyed chemical itch sensation to key supraspinal regions.43–45 Consistently with previous studies, ablation of NK1R + neurons could significantly decrease chloroquine-evoked itch behavior. 13 Moreover, Boyle et al have showed that GRPR neurons have no response current to LP-NPY. 5 For these reasons, more ample evidences should be exhibited to replenish the NPY-NPY1R signaling in inhibiting the chemical itch transmission.
Meanwhile, the majority of inhibitory NPY interneurons in spinal cord displayed a tonic firing pattern, 3 which could play a strong tonic inhibition role in hairy skin receiving low-threshold inputs. Therefore, related excitatory neurons could be over activated to show spontaneous scratching behavior after removing the inhibitory effect caused by ablation of NPY neurons. Moreover, it has been proved that neuropeptide NPY involved in the inhibition of neuropathic and inflammatory pain behavior.40,41 These findings demonstrated that the inhibitory NPY interneurons in dorsal horn may act two separated functions, for itch and pain. It still need much deeper study to determine whether it exists two different groups of NPY neurons or the same neuropeptide and interneurons could display different functions according to the noxious or innoxious stimuli.
In Feng et al’s study, they indicated that the loss of Piezo2-Merkel cell signaling in peripheral nervous system contributed to alloknesis. 30 However, what central nervous system’s role in alloknesis of AEW model is unclear. According to gate control theory in pain sensation transmission,33,46–48 the disinhibition of inhibitory interneurons in dorsal horn could activate excitatory transmission neurons to induce allodynia phenomenon. Similarly, the alloknesis caused by dry skin model may due to the disinhibition of NPY neurons gating transmission of mechanical itch in dorsal horn.3,5,13 Furthermore, our results, both chemogenetic activation of NPY neurons (Figure 2(a) and (b)) and NPY1R agonist LP-NPY i.t. Treatment (Figure 5(a) and (b)) could attenuated alloknesis and dry skin spontaneous itch in AEW model, indicated that NPY neurons still had reserve function to inhibit alloknesis even though its weakened inhibitory effect. Therefore, NPY neurons could be the new therapeutic targets to chronic itch patients’ alloknesis symptom clinically.
In conclusion, we demonstrate that NPY neurons not only inhibit mechanical itch but also chemical itch by NPY-NPY1R signaling pathway, which pathway also participates in alloknesis and dry skin itch of AEW model. However, which itch-specific excitatory neurons inhibited by NPY-NPY1R signaling in dorsal horn to transmit chemical itch is unknown. It’ll still worth to study much deeply in the mechanism of itch transmission.
Footnotes
Author contributions
S Chen and J Chen did virus injection, behavior tests, and wrote the manuscript. D Tang did the immunofluorescence staining and RT-PCR test. W Yin, S Xu and P Gao provided advices on the project. Y Jiao and W Yu conceived and supervised the project. All authors read and approved the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project has been supported by Shanghai Sailing Program (Grant No. 21YF1425100 to S Chen), National Natural Science Foundation of China (Grant No. 82101287 to S Chen, Grant No. 82371478 to Y Jiao, Grant No. 32030043 to W Yu, Grant No. 82101290 to W Yin), Innovation Program of Shanghai Municipal Education Commission (2019-01-07-00-01-E00074 to W Yu), Innovative Research Team of High-level Local Universities in Shanghai (SHSMU-ZDCX20211102 to Y Jiao), Shanghai Engineering Research Center of Peri-operative Organ Support and Function Preservation (20DZ2254200 to W Yu) and Shanghai Science and Technology Innovation Action Plan Popularization of Science Special Project (21DZ2315300 to W Yu).
