Abstract
Paclitaxel (PTX) treatment induces a pathological pain state that is often associated with neuroinflammation in the central nervous system. The available interventions for PTX-induced pathological pain encounter adverse effects and limited efficacies. Recent studies have shown the significant effectiveness of Electroacupuncture (EA) in pain management as a simple and safe alternative medical treatment. Here, we evaluated the analgesic effect of EA on pain behaviors in PTX-treated rats and investigated its potential analgesic mechanisms. In this study, a pathological pain model was established in SD rats via intraperitoneal (i.p.) injection of PTX. EA or Sham EA treatments were applied every other day for PTX-treated rats. Pain behaviors of mechanical allodynia and thermal hyperalgesia in rats were measured, followed by analysis of the spinal cord tissue via using molecular biology methods. Here, we show that EA treatment is capable to alleviate PTX-induced mechanical allodynia and thermal hyperalgesia in rats. In addition, EA regulated the abnormal protein expression of astrocytes, microglia, neurons, TLR4-MyD88/TRIF signaling pathway and cytokines in the lumbar spinal cord of PTX-treated rats. Furthermore, we investigated the spinal co-expressions of TLR4 in astrocytes, microglia, and neurons respectively in rats and the regulatory effect of EA on TLR4 and cells mentioned above. In summary, EA shows analgesic properties as it ameliorates PTX-induced mechanical allodynia and thermal hyperalgesia probably by reducing central neuroinflammation. Therefore, we consider EA as a potential therapeutic candidate for the treatment of PTX-induced pathologic pain. Notably, this study provides the first morphological evidence that EA may concurrently influence TLR4-mediated neuroimmune interactions across multiple spinal cell types, suggesting a potential central mechanism distinct from previously reported peripheral actions.
Introduction
Paclitaxel (PTX) is a first-line taxane-derived chemotherapeutic agent, originally isolated from Taxus genus, used in treating various malignancies including breast, ovarian, prostate, and lung cancers.1,2 PTX is one of the most commonly used taxanes and has been applied to a large number of cancer patients each year. Unfortunately, PTX treatment can result in a series of side effects, such as dizziness, neutropenia, diarrhea, alopecia, and peripheral neuropathy.2,3 Generally, most of these adverse events are tolerable and can be managed with medical agents, however, paclitaxel-induced peripheral neuropathy (PIPN) is an intractable condition, which occurs in up to 80% of all treated patients, interfering with their quality of life, and leading to the discontinuation of treatment.2,4 The symptoms of PIPN include numbness, tingling, cold-sensitivity, burning pain, balance disorder, stocking-glove distribution, and allodynia.2,4 Among these symptoms, the presence and persistence of neuropathic pain are the main reason for dose reduction and discontinuing PTX treatment in the clinical practice. To date, no optimal therapeutic strategies have been proven effective for the prevention or minimization of PTX-induced neuropathic pain. So that, it is highly required to develop potential analgesic approach to manage PTX-induced pathological pain.
As a traditional Chinese medical practice, acupuncture involves insertion of needles into specific acupoints within human body, and manipulation of needles by manual or electrical stimulation.5–7 Among its diverse therapeutic applications, the most marked and well-known effect of acupuncture relates to its involvement in relieving and managing pain, including PTX-induced neuropathic pain. Clinical studies have demonstrated that electroacupuncture (EA), either as a standalone therapy or combined with pharmacological agents, effectively reduces PTX-induced neuropathic pain and the relevant symptoms.8–11 Preclinical studies further support these findings, showing that EA remarkably alleviated mechanical and/or thermal allodynia/hyperalgesia in animal models of PTX-induced pathological pain.12–14 Recent mechanistic investigations have identified key pathways and targets mediating EA’s analgesic effects in PTX models, including Nrf2-ARE pathway, oxidative signals, 5-HT 1A receptors, p-CaMKII, TRPV1, TLR4, opioid receptor, alpha 2- and beta-adrenoceptors.12–15
In our previous study, 16 we established a rat model of PTX-induced pain. The experimental results showed that EA but not sham EA treatment significantly attenuated PTX-induced mechanical allodynia in rats, with a potential mechanism related to the inhibition of spinal glia and TLR4/NF-κB proteins. In addition, we revealed the significant role of neuroinflammation in the occurrence and development of PIPN, and the effectiveness of EA in regulating the inflammatory response. Nevertheless, more evidence and further studies are required to elucidate the potential mechanism of EA in attenuating PTX-induced pathological pain.
Toll-like receptors (TLRs) are well-known for participating in the regulation of neuroinflammation and nociception.17,18 TLR4 signaling pathway is reported to play an important role in the process of inflammation and hyperalgesia during pathological conditions. 19 In particular, regulation of TLR4 signaling and its downstream signals such as MyD88 and/or TRIF produces therapeutic effects in the treatment of chemotherapy-induced peripheral neuropathy (CIPN).20–22 Thus, TLR4 signaling pathway can be considered as a potential target in the treatment of PTX-induced neuropathic pain and it requires particular attention and profound studies. TLR4 receptors are located on microglia, astrocytes and neurons.23–25 The activation of astrocytes and microglia in the spinal cord is associated with PTX-induced neuropathic pain, which is confirmed by our previous study.16,26 However, the interrelationships between the TLR4 and astrocytes, or TLR4 and microglia during their activation by PTX are require further investigation. Besides, the involvement of spinal neurons may also play an important role in response to PTX-induced pain.
In the present study, we established a rat model of PTX-induced pain, and further evaluated the analgesic effect of EA treatment. Moreover, we investigated the possible mechanisms underlying the antinociceptive action of EA against PTX-induced pain, with a particular emphasis on the TLR4 signaling pathway as well as the involved neuroinflammatory response (Figure 1).

Flow chart illustrating experimental design and body weight of experimental animals. (a) Schematic illustration of the establishment procedure of PTX-induced pain model; application of EA and Sham EA. (b) Body weight and (c) AUC of body weight of rats in Control, PTX, PTX+EA, and PTX + Sham EA groups. (d) The basic structure and important mechanisms of the present study.
Materials and methods
Animals
Male adult Sprague-Dawley rats (150–180 g) were used in this study. All animals were housed 3–4 per cage under conditions of controlled temperature (22–25℃) with a light/dark cycle of 12:12 hours light (7:00 on, 19:00 off). Standard chow and water were provided ad libitum. All manipulations and procedures complied with the ARRIVE guidelines (Animal Research: Reporting of In Vivo Experiments) and the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals. The protocol was approved by the Institutional Animal Care and Use Committee (IACUC) of the China Academy of Chinese Medical Sciences (Approval No. 2015010801). All efforts were made to minimize discomfort and the number of animals used. At the end of experiments, rats were deeply anesthetized and euthanized by injection of sodium pentobarbital (60 mg/kg) intraperitoneally (i.p.) at the end of experiments.
Experimental protocols
Grouping: Based on a random number table, a total of 24 rats were randomly divided into 4 groups: Control group (Control, n = 6), Paclitaxel-induced pain model group (PTX, n = 6), PTX model + EA group (PTX+EA, n = 6), PTX model + Sham EA group (PTX + Sham EA, n = 6).
Modeling: Paclitaxel agent (6 mg/mL, Beijing Union Pharmaceutical Factory, Beijing, People’s Republic of China) was diluted with phosphate buffered saline (PBS) to make PTX solution (2 mg/mL). Rats in PTX, PTX+EA, and PTX + Sham EA groups received injections of PTX solution (2 mg/kg, i.p.) every other day (cumulative dose 8 mg/kg, i.p.) to induce paclitaxel-induced neuropathic pain. Control rats were received i.p. administrations of vehicle (PBS). 16
Others: Body weight was recorded every other day for all rats. Rats in the PTX+EA received EA treatment at bilateral ST36 for 30 min every day. Rats in PTX + sham EA needled superficially at bilateral ST36 points without electrical stimulation. Behavioral tests were performed every other day for 9 days following the initiation of PTX. Following the final test, animals were euthanized, and the spinal tissues were harvested for western blot assays and immunofluorescence detection.
A flow diagram, time chart and experimental designs of the present study are shown in Figure 1(a).
Electroacupuncture treatment
ST36 (Zusanli) acupoints on bilateral hind limbs of rats were used for EA treatment. ST36 in rats is located 10 mm below the knee joint and 5 mm lateral to the anterior tubercle of the tibia (Figure 1(a)). Acupuncture needles at both ST36 acupoints were inserted as follows: one stainless steel needle was inserted into the ST36 acupoint and the other one 5 mm below ST36 acupoint (5 mm in depth). The tips of the electrode cables from an electroacupuncture apparatus (HANS-200A) were connected to a pair of steel acupuncture needles, and constant current pulses (10 Hz, 1 mA) were applied for 30 min. 16 For sham EA, acupuncture needles were inserted into superficially at bilateral ST36 points without electrical stimulation. Both EA and sham EA treatments were performed under isoflurane anesthesia and were applied once every other day for 9 days following the initiation of PTX. Behavioral tests for mechanical allodynia and thermal hyperalgesia were conducted 2 h after the scheduled EA intervention.
Mechanical allodynia
Animals were habituated to the environment for 3 days before behavioral testing. Baseline measurements of mechanical withdrawal latencies for both hind paws (left and right) of rats were performed prior to PTX or vehicle administration. Mechanical withdrawal latencies were measured using a Dynamic Plantar Aesthesiometer (Ugo Basile, Varese, Italy) with a metal filament (0.5 mm in diameter). Rats were placed on an elevated metal wire mesh floor and confined in individual Perspex boxes, where they were allowed to acclimatize for 30 min. Mechanical withdrawal latencies (response time, s) of each hind paws were measured. The aesthesiometer filament was programmed to apply a gradually increasing force (2 g/s, cut-off: 50 g) to the plantar surface of the hind paw until withdrawal occurred. Each hind paw was tested three times at 10-min intervals. Behavioral testing was conducted every other day for 9 days. The area under the curve (AUC) of withdrawal latencies for both hind paws in each group was calculated to investigate the overall effect. 16
Thermal hyperalgesia
Thermal hyperalgesia was evaluated by measuring paw withdrawal latency in response to heat stimulation using a hotplate analgesia meter (Univ. California, San Diego, CA, USA). Each rat was placed in a plexiglas chamber containing a smooth, temperature-controlled glass floor and was acclimated to the testing apparatus prior to the application of thermal stimulus. Paw withdrawal latencies to a radiant source of heat in both right and left hind paws of rats were measured to assess nociceptive thresholds to acute thermal noxious stimulus by using a paw thermal stimulator. The withdrawal latencies of left and right paws to each thermal stimulus were recorded from the start of the radiant heat until the withdrawal reaction occurs. To prevent tissue damage, the heat source was automatically terminated at 20 s if no withdrawal occurred. The area under the curve (AUC) of withdrawal latencies for hind paws in each group was calculated to investigate the overall effect.
Western blot assay
After 9 days following the initiation of PTX or vehicle administration, rats were killed and the bilateral spinal tissues (L3-L6) were harvested, snap-frozen, and stored at −80°C. Whole-cell lysates were prepared from the lumbar region of the spinal cord. Protein concentrations were determined using a BCA protein assay kit (Thermo Scientific, USA). Equal amounts of protein extract were resolved by 10% SDS-PAGE gels and transferred to a polyvinylidene difluoride (PVDF) membrane (Merck Millipore, USA). Membranes were then incubated with anti-TLR4 (Rabbit monoclonal, Abcam, diluted 1:1000), anti-MyD88 (Rabbit monoclonal, CST, diluted 1:1000), anti-TRIF (Rabbit monoclonal, Abcam, diluted 1:1000), anti-GFAP (Rabbit polyclonal, Abcam, diluted 1:5000), anti-TMEM119 (Rabbit polyclonal, Invitrogen, diluted 1:5000), anti-NeuN (Rabbit monoclonal, Proteintech, diluted 1:2000), anti-tumor necrosis factor (TNF-α; Mouse monoclonal, Protein-tech, diluted 1:1000), anti- Interleukin-10 (IL-10; Mouse monoclonal, Protein-tech, diluted 1:1000), anti-β-actin (Rabbit monoclonal, CST, diluted 1:1000), and anti-GAPDH (Rabbit monoclonal, CST, diluted 1:1000). All membranes with antibodies were incubated overnight at 4°C and then incubated with anti-rabbit IgG (1:5000; CST, USA) or anti-mouse IgG (1:5000; CST, USA) for 1 h at room temperature. Protein bands were detected with the use of a chemiluminescent substrate (ECL Prime Western Blotting Detection Reagents; Amersham, UK) and visualized using a luminescent image analyzer (ChemiDoc Imaging System, Bio-Rad, USA).
Immunofluorescence staining
Rats were deeply anesthetized with sodium pentobarbitone (60 mg/kg, i.p.) and were intra-cardially perfused with freshly sterile saline and 4% paraformaldehyde in 0.1 M phosphate buffer (PB). Subsequently, L3-6 spinal cord tissues were quickly removed after perfusion immediately. Spinal cord sections were post-fixed in 4% paraformaldehyde for 4 h, and permeated with 25% sucrose solution overnight at 4℃. The spinal cord sections were then frozen in embedding compounds. The collected tissue was sliced into 30-μm thick sections using a freezing microtome (REM-710, Yamato Koki Industrial, Japan). The L4-L5 spinal slices were used for staining in the present study. For each rat, three transverse sections from the left side and three from the right side of the L4-L5 spinal cord were randomly selected for immunofluorescence staining and subsequent quantitative analysis. The average value of these six sections was calculated for statistical purposes. The frozen spinal slices were blocked with PBS containing 3% goat serum, and then incubated with the primary antibodies to TLR4 (mouse monoclonal, Proteintech, diluted 1:200), GFAP (rabbit monoclonal, Abcam, diluted 1:800), Iba1 (rat monoclonal, GeneTex, diluted 1:200), and NeuN (rabbit polyclonal, Proteintech, diluted 1:200). Slices were then rinsed in PBS and incubated with secondary antibodies (goat anti-rat IgG, Alexa FluorR 568, Abcam, diluted 1:600; goat anti-rabbit IgG, Cross-Adsprbed secondary antibody, Alexa Fluor™ 594, Thermo Fisher, diluted 1:1000; goat anti-mouse IgG, Cross-Adsprbed secondary antibody, Alexa Fluor™ 488, Thermo Fisher, diluted 1:1000). Images were obtained and examined using a confocal fluorescence microscopy (FV1000, Olympus, Tokyo, Japan) and stored on digital media. The fluorescence density value of TLR4, GFAP, Iba1, and NeuN was measured using a computer-assisted system (Image J). The co-stained cells were calculated and assessed with Image J as well.
Statistical analysis
Statistical analysis was performed using GraphPad Prism 8.0 (La Jolla, CA, USA). All data were presented as mean ± SEM and analyzed with repeated-measures one-way ANOVA followed by Dunnett’s post-hoc test, and two-way ANOVA followed by Bonferroni’s multiple comparison test. In all cases, p value of <0.05 was considered a statistical significant.
Results
General status
The general status of experimental rats includes the mental state, limbs, eating/drinking conditions, and body mass. 27 All groups exhibited normal health parameters throughout the study. As shown in Figure 1, body weights in all groups increased steadily over time, with no significant differences observed in body weight trajectories or AUC values between groups (Figure 1(b) and (c)).
Effects of EA on PTX-induced mechanical allodynia
Paw mechanical withdrawal latency (PMWL) was assessed bilaterally in hind paws of rats every other day for 9 days post-PTX administration. There was no significant difference in the basal PMWLs in both left and right hind limbs among all four groups (p > 0.05, Figure 2(a) and (c)). Rats in PTX group showed mechanical allodynia in both left and right hind limbs. In addition, PMWLs of both-side hind paws in PTX, PTX+EA, and PTX + sham EA groups presenting significant downward trend on days 0−9 compared to the Control (Figure 2(a) and (c)). The downward trend can be quantified and measured by AUC. In comparison with Control, AUC values of PMWL in both left and right sides of hind paws in PTX, PTX + EA, and PTX + sham EA were significantly decreased respectively (p < 0.01, Figure 2(b) and (d)).

Electroacupuncture (EA) attenuated mechanical allodynia in rat model of PTX-induced neuropathic pain. (a) Paw mechanical withdrawal latency (PMWL) of the left hind limb in rats was evaluated in a time-course study. (b) Normalized area under the curve (AUC) analysis of Figure 2(a). (c) PMWL of the right hind limb in rats was evaluated in a time-course study. (d) AUC analysis of Figure 2(c). (e) Comparison between left and right hind paws on PTX-induced mechanical allodynia. **p < 0.01 versus Control; #p < 0.05, ##p < 0.01 versus PTX. n = 6 rats/group. One-way or two-way ANOVA followed by Tukey post hoc test was used for statistical analysis.
To evaluate the efficacy of EA in attenuating PTX-induced mechanical allodynia, the values of PMWLs in both-side hind paws that measured from day 1 to day 9 were compared among PTX, PTX+EA, and PTX + sham EA, respectively. As shown in Figure 2(a) and (c), EA treatment significantly attenuated decreasing PMWLs in both-side hind paws induced by PTX at different mechanical allodynia test days. Compared to the PTX group, PMWLs of left paws in PTX+EA were significant increased at day 3 (p = 0.0185), 5 (p = 0.0457), and 9 (p = 0.0448). Compared to the PTX group, PMWLs of right paws in PTX+EA were significant increased at day 1, 3, 5, 7, and 9 (p < 0.01, respectively). As shown in Figure 2(b) and (d), AUC analysis further confirmed the analgesic effect of EA in PTX-treated rats (p < 0.01, both sides of hind paws) in comparison with PTX group. Whereas, sham EA showed no analgesic effect on both-side hind paws, neither in PMWLs nor in AUC analysis (p > 0.05).
In particular, there was no significant differences in PMWLs (evaluated by AUC) between left and right hind paws, suggesting a symmetrical mechanical allodynia induced by PTX (Figure 2(e)).
Effects of EA on PTX-induced thermal hyperalgesia
Paw thermal withdrawal latency (PTWL) was assessed on both left and right hind paws of rats every other day for 9 days following the initiation of PTX. There were no significant differences in the basal PTWLs in both left and right hind limbs among all four groups (p > 0.05, Figure 3(a) and (c)). In addition, the data revealed that PTWLs of both-side hind paws in PTX, PTX+EA, and PTX + sham EA groups presenting significant decreasing trend on days 0−9 compared to the Control group (Figure 3(a) and (c)). This trend was quantified using the AUC. Compared to the Control group, the AUC values of PTWL for both left and right sides of hind paws in PTX, PTX + EA, and PTX + sham EA were significantly decreased respectively (p < 0.01, Figure 3(b) and 3(d)).
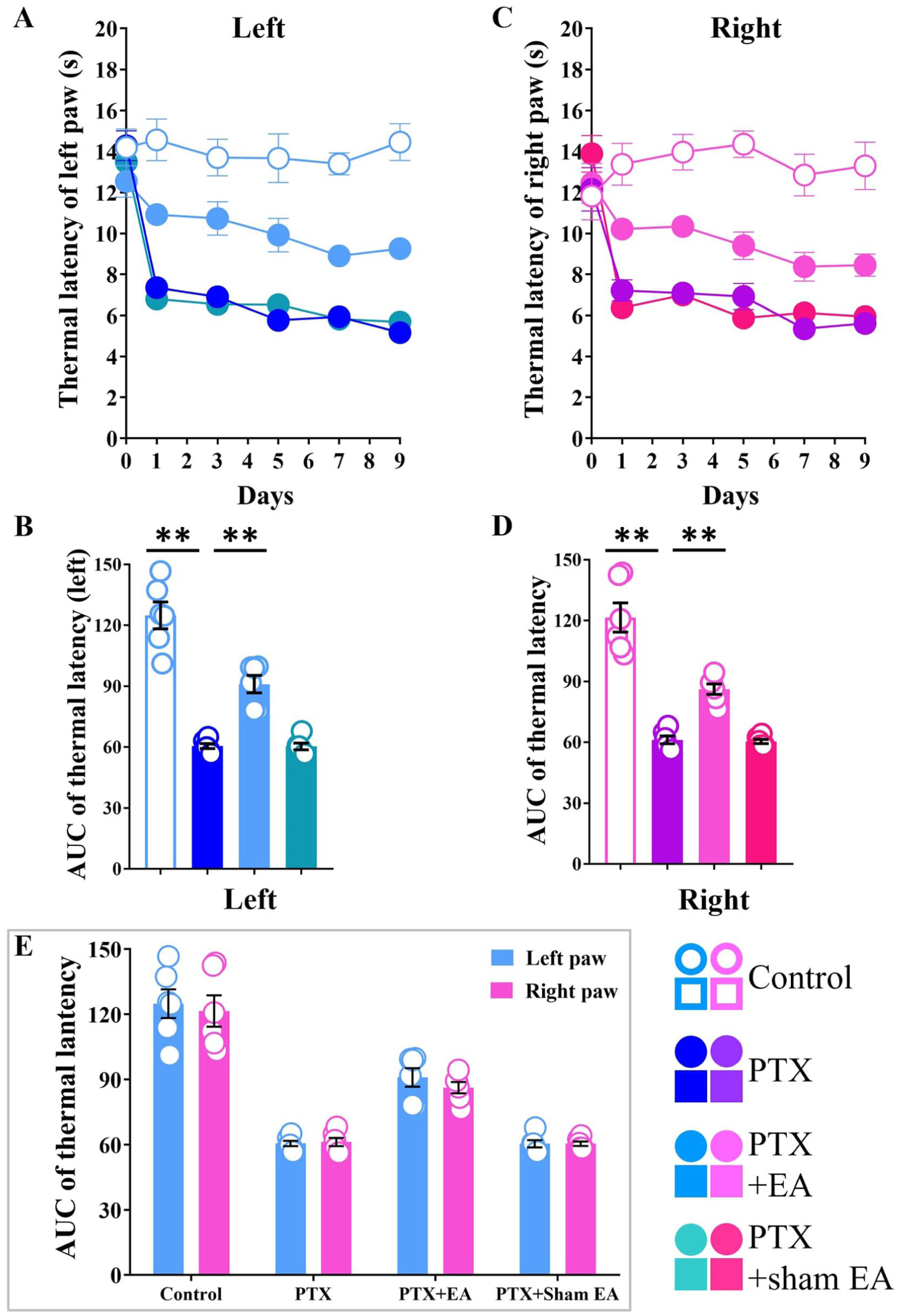
Electroacupuncture (EA) attenuated thermal hyperalgesia in rat model of PTX-induced neuropathic pain. (a) Paw thermal withdrawal latency (PTWL) of the left hind limb in rats was evaluated in a time-course study. (b) AUC analysis of Figure 3(a). (c) PTWL of the right hind limb in rats was evaluated in a time-course study. (d) AUC analysis of Figure 3(c). (e) Comparison between left and right hind paws on PTX-induced thermal hyperalgesia. **p < 0.01 versus Control; #p < 0.05, ##p < 0.01 versus PTX. n = 6 rats/group. One-way or two-way ANOVA followed by Tukey post hoc test was used for statistical analysis.
To evaluate the efficacy of EA in attenuating PTX-induced thermal hyperalgesia, the values of PTWLs in both-side hind paws that measured from day 1 to day 9 were compared among PTX, PTX+EA, and PTX + sham EA, respectively. As shown in Figure 3(a) and (c), EA treatment significantly attenuated decreasing PTWLs in both-side hind paws induced by PTX at different thermal hyperalgesia test days. Compared to the PTX group, PTWLs of left paws in PTX+EA were significant increased at day 1 (p = 0.0008), 3 (p = 0.0003), 5 (p < 0.0001), 7 (p = 0.0074), and 9 (p < 0.0001). Compared to the PTX group, PTWLs of right paws in PTX+EA were significantly increased at day 1 (p = 0.0146), 3 (p = 0.0064), 7 (p = 0.0131), and 9 (p = 0.0222, respectively). As shown in Figure 3(b) and (d), AUC analysis further confirmed the analgesic effect of EA in PTX-treated rats (p = 0.0002, left; p = 0.0013, right) compared to the PTX group. Whereas, sham EA showed no analgesic effect on both-side hind paws, neither in PTWLs nor in AUC analysis (p > 0.05).
In particular, there was no significant differences in PTWLs (evaluated by AUC) between left and right hind paws, suggesting a symmetrical thermal hyperalgesia induced by PTX (Figure 3(e)).
Effects of EA on protein expressions of glia and neurons in the lumbar spinal cord
The spinal cord is made up of neurons and glia (mainly composed of microglia and astrocytes). In the present study, the protein expressions of astrocytes (marker GFAP), microglia (marker TMEM119), and neurons (marker NeuN) in the lumbar spinal cord of rats were detected by western blot assay. PTX administration caused a significant increased in the protein expressions of astrocytes and microglia. At the same time, PTX led to a remarkable decreased of neural protein expressions (Figure 4(a)).
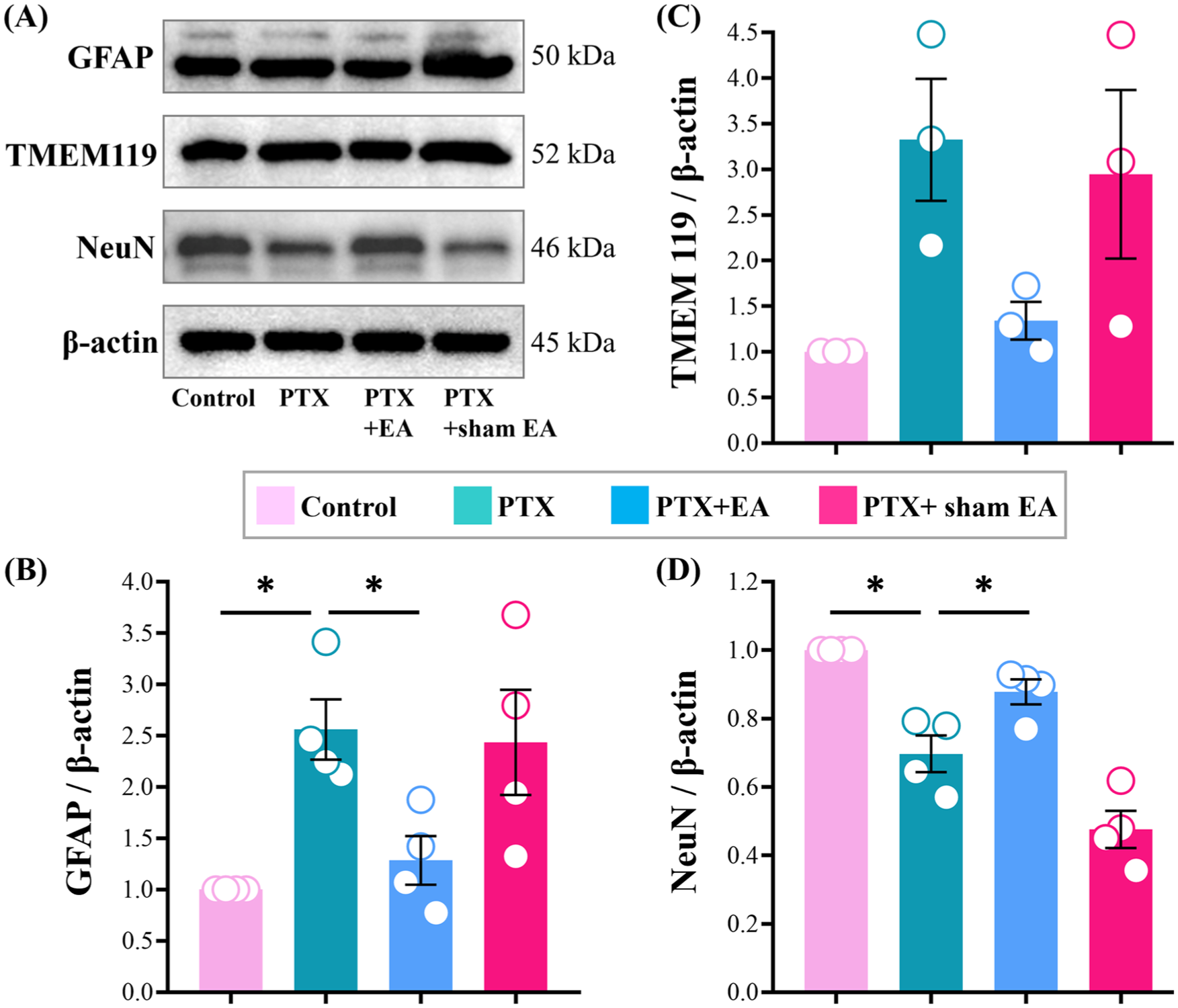
Protein expressions of GFAP, TMEM119, and NeuN in lumbar spinal cord from experimental rats. Representative (a) Western blots of GFAP, TMEM119, and NeuN protein, and relative levels of (b) GFAP, (c) TMEM119, and (d) NeuN in lumbar spinal cord. *p < 0.05, **p < 0.01. n = 3–4 rats/group. One-way or two-way ANOVA followed by Tukey post hoc test was used for statistical analysis.
Compared to the Control group, the protein expressions of astrocytes in lumbar spinal cord were increased significantly in PTX group (GFAP, p = 0.0385, Figure 4(b)). The protein expression of microglia in lumbar spinal cord presented a significant upward trend in PTX group (TMEM119, Figure 4(c)). In contrast, neural protein expression was significantly reduced in the PTX group (NeuN, p = 0.0328, Figure 4(d)).
As shown in Figure 4(a), EA treatment attenuated the overexpression of astrocytes and microglia in the lumbar spinal cord of PTX-treated rats. Compared to the PTX group, the expression levels of spinal GFAP were significantly decreased in PTX+EA group (GFAP, p = 0.0241, Figure 4(b)), and the protein expression of TMEM119 showed an obvious downward trend in PTX+EA group compared to the PTX group (TMEM119, Figure 4(c)). In addition, EA treatment alleviated the inhibition of neural protein expression induced by PTX (Figure 4(a)). Compared to the PTX group, the expression levels of spinal NeuN were significantly increased in PTX+EA group (NeuN, p = 0.0243, Figure 4(d)). Whereas, sham EA treatment showed no regulatory effect on protein expression of GFAP, TMEM119, and NeuN in lumbar spinal cord.
Effects of EA on protein expressions of TLR4 signal pathway in the lumbar spinal cord
Three key indicators (TLR4, MyD88, TRIF) of TLR4 signaling pathway were detected by western blot. Data illustrated PTX administration increased the protein expression levels of TLR4, MyD88, and TRIF in spinal cord of PTX-treated rats (Figure 5(a)).

Protein expressions of TLR4, MyD88, TRIF, TNF-α, and IL-10 in lumbar spinal cord from experimental rats. Representative (a) Western blots of TLR4, MyD88, and TRIF protein, and relative levels of (b) TLR4, (c) MyD88, and (d) TRIF in lumbar spinal cord. Western blots of (e) TNF-α and (g) IL-10 protein, and relative levels of (f) TNF-α and (h) IL-10 in lumbar spinal cord. *p < 0.05, **p < 0.01. n = 3–4 rats/group. One-way or two-way ANOVA followed by Tukey post hoc test was used for statistical analysis.
Compared with Control group, the protein expression of TLR4, MyD88, and TRIF in spinal cord were increased significantly in PTX group (TLR4, p < 0.0001, Figure 5(b); MyD88, p = 0.0004, Figure 5(c); TRIF p < 0.0001, Figure 5(d)).
As shown in Figure 5(a), EA treatment suppressed the PTX-induced increases of TLR4, MyD88 and TRIF in spinal cord in PTX-treated rats. Compared to the PTX group, the expression levels of spinal TLR4, MyD88, and TRIF were significantly decreased from 1.884 ± 0.1158, 1.399 ± 0.1077, and 1.810 ± 0.1005 in PTX group, to 0.7129 ± 0.1463 (TLR, p = 0.0001, Figure 5(b)), 0.9616 ± 0.0752 (MyD88, p = 0.0130, Figure 5(c)), and 0.8402 ± 0.0739 (TRIF, p = 0.0005, Figure 5(d)) in PTX+EA group, respectively. Whereas, sham EA treatment showed no inhibitory effect on protein expression of TLR4, MyD88, and TRIF in spinal cord.
The pro-inflammatory cytokine tumor necrosis factor (TNF-α) and anti-inflammatory cytokine Interleukin-10 (IL-10) were detected by western blot. Data illustrated that PTX administration increased the protein expressions of TNF-α, whereas decreased the protein expressions of IL-10 in the spinal cord of PTX-treated rats (Figure 5(e) and (g)).
Compared to the Control group, the protein expression of TNF-α in spinal cord were increased significantly in PTX group (p = 0.0003, Figure 5(f)), whereas the protein expression of IL-10 in spinal cord were unchanged in PTX group (Figure 5(h)).
EA treatment inhibited the increased expression of TNF-α (Figure 5(e)) and upregulated the protein expression of IL-10 (Figure 5(g)) in lumbar spinal cord PTX-treated rats. Compare to the PTX group, the expression levels of spinal TNF-α was significantly decreased in PTX+EA group (p = 0.0055, Figure 5(f)). Compared to the PTX group, the expression levels of spinal IL-10 was significantly increased (p = 0.0235, Figure 5(h)) in PTX+EA group. Whereas, sham EA treatment showed no effect on spinal protein expression of TNF-α and IL-10.
Effects of EA on fluorescence expression of glia, neurons, and TLR4 in the lumbar spinal cord
Immunofluorescence was used to define the location and expression of astrocytes (marker GFAP), microglia (marker Iba1), and neurons (marker NeuN) in dorsal horn of L3-6 spinal cord, and the cellular localization of TLR4 inside as well.
As shown in Figure 6(a), 7(a), and 8(a), TLR4 was coexisted with GFAP-positive astrocytes, Iba1-positive microcytes, and NeuN-positive neurons in dorsal horn of L3-6 spinal cord mainly in PTX and PTX+EA groups.

Immunolabeled expressions of GFAP, TLR4, DAPI and Merge in the bilateral dorsal horn of lumbar spinal cord from experimental rats. Representative (a) Immunofluorescence staining of GFAP, TLR4, DAPI and Merge, and relative levels of (c) GFAP, (d) TLR4 and (b) Schematic diagram of sampling and cell localization. Scale bar, 50 µm. *p < 0.05, **p < 0.01. n = 4 rats/group. One-way or two-way ANOVA followed by Tukey post hoc test was used for statistical analysis.

Immunolabeled expressions of Iba1, TLR4, DAPI and Merge in the bilateral dorsal horn of lumbar spinal cord from experimental rats. Representative (a) Immunofluorescence staining of Iba1, TLR4, DAPI and Merge, and relative levels of (c) Iba1, (d) TLR4, and (b) Schematic diagram of sampling and cell localization. Scale bar, 50 µm. *p < 0.05, **p < 0.01. n = 4 rats/group. One-way or two-way ANOVA followed by Tukey post hoc test was used for statistical analysis.
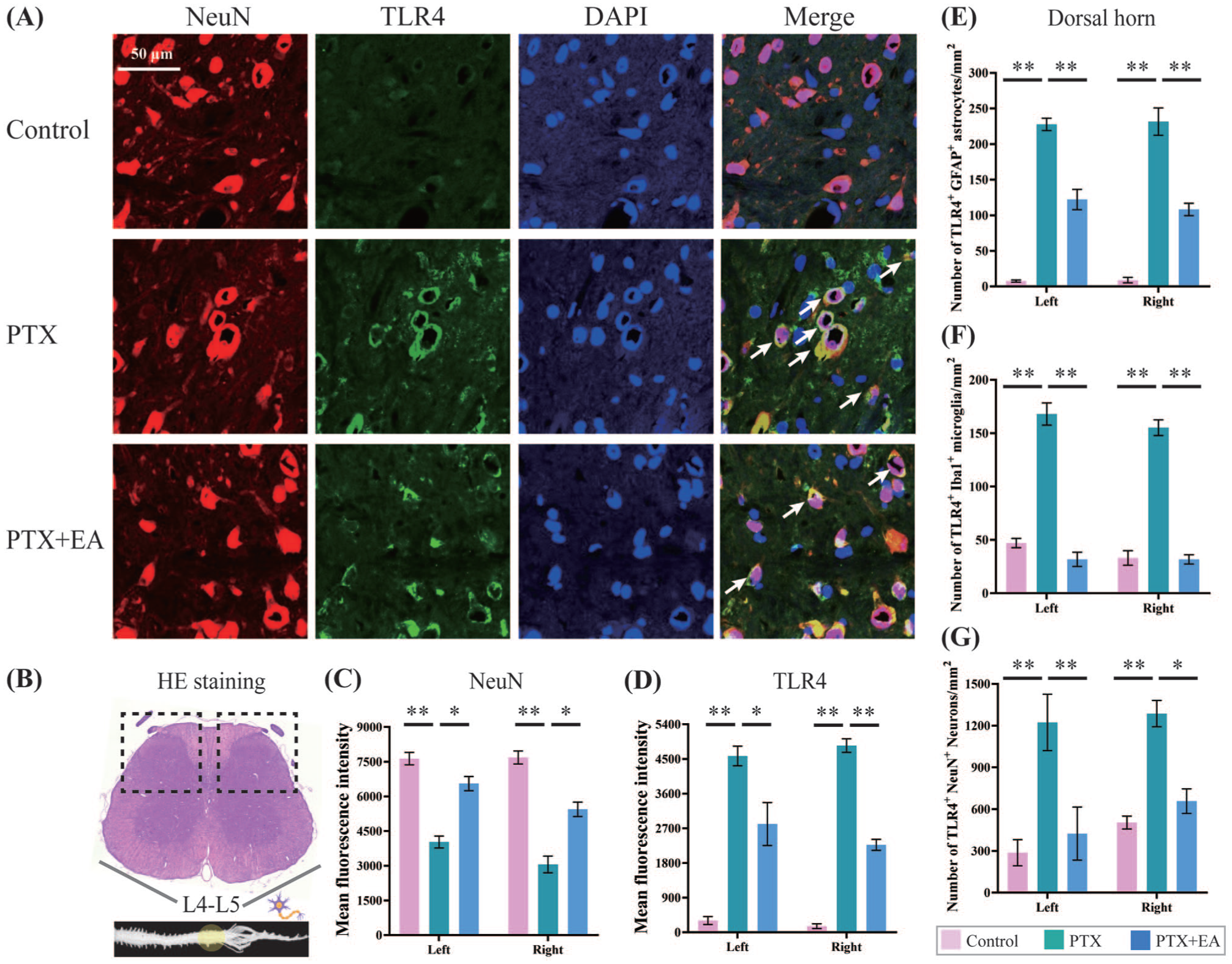
Immunolabeled expressions of NeuN, TLR4, DAPI and Merge in the bilateral dorsal horn of lumbar spinal cord from experimental rats. Representative (a) Immunofluorescence staining of NeuN, TLR4, DAPI and Merge, and relative levels of (c) NeuN, (d) TLR4 and (b) Schematic diagram of sampling and cell localization. Summary data in (e), (f), and (g) show the number per mm2 of the stained TLR4+ in stained astrocytes (GFAP+), microglia (Iba1+) and neurons (NeuN+) respectively. Scale bar, 50 µm. *p < 0.05, **p < 0.01. n = 4 rats/group. One-way or two-way ANOVA followed by Tukey post hoc test was used for statistical analysis.
Compared to the Control group, the immunofluorescence intensity of TLR4, GFAP (GFAP expressed in the spinal dorsal horn of left side (in the left), p = 0.0276; TLR4 expressed in the left, p = 0.0011; GFAP expressed in the spinal dorsal horn of right side (in the right), p = 0.0312; TLR4 expressed in the right, p = 0.0004; Figure 6(c) and (d)), and Iba1 (Iba1 expressed in the left, p = 0.0003; TLR4 expressed in the left, p < 0.0001; Iba1 expressed in the right, p = 0.0012; TLR4 expressed in the right, p = 0.0049; Figure 7(c) and (d)) were increased within the dorsal horn of spinal cord in PTX-treated rats. As shown in Figure 6(a) and 7(a), EA inhibited the increased expression of GFAP, Iba1, and TLR4 in bilateral dorsal horn of lumbar spinal cord in PTX-treated rats. Compared to the PTX group, the immunofluorescence intensity of GFAP and TLR4 in PTX+EA group were significantly decreased (GFAP, p = 0.0135 in the left, p = 0.0418 in the right; TLR4, p = 0.0139 in the left, p = 0.0010 in the right; Figure 6(c) and (d)) within bilateral dorsal horn of lumbar spinal cord in rats. Similarly, compared to the PTX group, the over immunofluorescence intensity of spinal Iba1 and TLR4 in PTX+EA group were significantly inhibited (Iba1, p = 0.0012 in the left, p = 0.0009 in the right; TLR4, p = 0.0158 in the left, p = 0.0270 in the right; Figure 7(c) and (d)) as well.
Consistent with the western blot data of neural protein expression, PTX significantly inhibited neural immunofluorescence intensity in the dorsal horn of lumbar spinal cord in PTX-treated rats (Figure 8(a)). Compared to the Control group, the immunofluorescence intensity of NeuN was decreased (p = 0.0061 in the left, p = 0.0003 in the right, Figure 8(c)) within the bilateral dorsal horn of spinal cord in PTX-treated rats. Whereas, the immunofluorescence intensity of TLR4 was increased (p = 0.0005 in the left, p = 0.0003 in the right, Figure 8(d)) in PTX group campared to the Control group. As shown in Figure 8(a), EA regulated the abnormal expression of NeuN and TLR4 in dorsal horn of lumbar spinal cord in PTX-treated rats. Compared to the PTX group, the immunofluorescence intensity of NeuN in PTX+EA group was significantly increased (p = 0.0237 in the left, p = 0.0276 in the right, Figure 8(c)) within bilateral dorsal horn of lumbar spinal cord in rats. Similarly, compared to the PTX group, the over immunofluorescence intensity of spinal TLR4 in PTX+EA group were inhibited in bilateral dorsal horn (Figure 8(d)) as well.
In addition, we calculated and analyzed the number of TLR4-expressed astrocytes (TLR4+-GFAP+), microglia (TLR4+-Iba1+), and neurons (TLR4+-NeuN+) per mm2 respectively. As shown in Figure 8(e) to (g), PTX increased the number of astrocytes (GFAP+, p = 0.0003, left side; p = 0.0029, right side), microglia (Iba1+, p = 0.0008, left side; p = 0.0017, right side), and neurons (NeuN+, p = 0.0023, left side; p = 0.0046, right side) expressing TLR4 in the dorsal horn of lumbar spinal cord in comparison to Control. EA inhibited the over-activated number of the co-stained cells in all three types (TLR4+-GFAP+, p = 0.0017, left side; p = 0.0057, right side, Figure 8(e), TLR4+-ba1+, p = 0.0025, left side; p = 0.0006, right side, Figure 8(f), TLR4+-NeuN+, p = 0.0043, left side; p = 0.0104, right side, Figure 8(g)).
Discussion
The current study presents evidence that i.p. administration of PTX induced pathological pain in both left and right hind paws of rats, which mainly manifested as mechanical allodynia and thermal hyperalgesia. The results of PMWL and PTWL from the detections of pain behavior (tests of von Frey, hotplate analgesia) showed that PTX-induced neuropathic pain occurred in both left and right hind limbs but without significant difference between them. Both PTX-induced mechanical allodynia and thermal hyperalgesia showed a gradually increasing trend over time. In addition, PTX activated spinal astrocytes, microglia while inhibiting neurons, which was accompanied by the activation of TLR4 signaling pathway, manifested by overexpression of TLR4, MyD88, and TRIF proteins in spinal cord as well as the upregulation of protein expressions of the pro-inflammatory cytokine TNF-α in lumbar spinal cord in PTX-treated rats.
Following PTX administration, EA treatment was applied once every other day for 9 consecutive days in total. Previous studies have demonstrated the effective analgesic actions of EA stimulation at ST36 against various pain including PTX-induced neuropathic pain.28–30 In our previous study, 16 we have proved that EA at ST36 acupoint attenuated the pain-related behaviors of rats with PIPN model. As a continuation of our previous work, the present data confirmed the significant anti-nociceptive effects of EA (at ST36 acupoint) treatment on attenuating PTX-induced neuropathic pain including mechanical allodynia and thermal hyperalgesia. The results demonstrated that EA treatment produced a continuous analgesic effect against both mechanical allodynia and thermal hyperalgesia in both hind limbs and persisted throughout the treatment period. In contrast, sham EA treatment had no analgesic effects on PTX-induced nociceptive behaviors in both left and right hind paws, indicating the specific therapeutic actions of EA intervention.
Paclitaxel-induced neuropathic pain is always associated with neuroinflammation in the central and peripheral nervous system, 31 and it is a consequence of neuroinflammatory process characterized by triggering inflammatory cascades and activating pro-inflammatory cytokines and chemokines in the spinal cord. 32 TLRs function in the immune system and are present in immune, epithelial, neural, and glial cells. 33 Upon activation, TLRs initiate adaptor proteins including myeloid differentiation primary response protein 88 (MyD88), Toll/interleukin-1 receptor domain-containing adapter protein (TIRAP) and TIRAP-inducing IFN-β (TRIF), and subsequently activate downstream signals resulting in cytokine production, chemokines release and immune cascades. 34 TLR4 is mainly expressed in immune cells and also in some non-immune cells, such as nociceptive neurons contributing to pain and inflammation signaling.35,36 Its actions are mediated by two major intracellular signaling pathways namely the MyD88-dependent and/or TRIF-dependent pathways. 37 TLR4 can detect extracellular matrix molecules and damage-associated molecular patterns (DAMPs) which are released by injured sensory neurons, and thus activate immunocompetent cells and cause neural pain. 38 In addition, activation of TLR4 results in increased expression of the transcription factor nuclear factor kappa B (NF-kB) signals and pro-inflammatory cytokines such as TNF-α. These inflammatory factors deteriorate the hyperactivation of the immune response and ultimately lead to nociceptive behaviors and pain. Therefore, the suppression of TLR4 could be considered as a potential therapeutic for attenuating neuropathic pain, and which has been reported previously.37–40 TNF-α is a key pro-inflammatory cytokine that exerts actions on mammalian immunity and cellular homeostasis. TNF-α exerts a positive regulatory feedback loop that enhances NF-κB signals and in turn induces further expression of TNF-α, and followed by a cascade of pro-inflammatory cytokines.41,42 TNF-α is activated in both the peripheral nervous system and central nervous system including the spinal cord in various animal pain models to mediate the initiation and persistence of neuropathic pain.43,44 As a classic and powerful anti-inflammatory cytokine, IL-10 contributes to reduced bioavailability of pro-inflammatory cytokines such as IL-1 and TNF-α. IL-10 signals regulate neuroinflammatory processes in central and peripheral nervous systems relevant to pain reduction and transmission,45–47 and participate in the analgesic effects to against pathological pain.41,47
Our results in the present study showed that EA treatment significantly downregulated the key component expression levels of TLR4 signaling pathway in the spinal cord, with a remarkable inhibition of TLR4, MyD88, and TRIF protein expression, indicating the possible analgesic mechanism of EA treatment might be through its down-regulation of TLR4 itself and both of its two major intracellular signaling pathways in central nervous system. Moreover, the western blot results showed a remarkable increase of TNF-α protein expressions in the lumbar spinal cord in PTX-treated rats, suggesting the important role of spinal TNF-α in PTX-induced neuropathic pain. EA treatment significantly blocked the spinal TNF-α protein expression, indicating that inhibition of the nociceptive cytokine TNF-α might be one of the critical points to potentiate the analgesic effect of EA against PTX-induced neuropathic pain. Besides, the present data showed no significant change of IL-10 protein expressions in lumbar spinal cord between PTX and Control, whereas EA treatment obviously upregulated protein expression of the antinociceptive cytokine IL-10, suggesting that the enhancement of spinal IL-10 possibly mediate the antinociceptive actions of EA in treating PTX-treated rats.
Neurons, astrocytes, and microglia in the central nervous system play an essential role in the occurrence and development of neuropathic pain. 48 Spinal projection neurons form parallel ascending circuit modules that convey noxious cutaneous signals from the spinal cord to the brain. 49 Besides neurons, spinal glia including astrocytes and microglia plays a critical role in processing nociceptive signals. Spinal glial activation is an important component in the initiation and maintenance of hyperalgesia and allodynia in chronic pain.50,51 In our previous study, the spinal astrocytes and microglia were activated in rats following PTX treatment, and their activity were attenuated by EA at ST36. 16 In the present research, we repeated the western blot assays and once again confirmed the inhibitory effect of EA on PTX-induced activation of astrocytes and microglia in the lumbar spinal cord. Importantly, we visualized the location of astrocytes and microglia by introducing immunofluorescence staining in the dorsal horn of lumbar spinal cord, and conduct semi-quantitative and quantitative analysis on it. The immunofluorescence data served as an important supplement to western blot results. Besides, neurons, as the major cells in the spinal cord, was also focused in the present study. We found that PTX increased the protein expression of spinal astrocytes and microglia but inhibited spinal neuron protein levers, and these results were consistent with the trend in fluorescence density expression. EA significantly attenuated both the abnormal protein expressions and immunofluorescence density of spinal astrocytes, microglia, and neurons. As the essential point of the present study, TLR4 was co-stained with spinal astrocytes, microglia, and neurons. The immunofluorescence results indicated that TLR4-expressing astrocytes, microglia, and neurons were activated by PTX but were inhibited following EA treatment, which is consistent with our previous western blot results.
Our present study confirmed the antinociceptive effects of EA in treatment of PTX-induced mechanical allodynia and thermal hyperalgesia in rat PIPN model of neuropathic pain. We gave particular emphasis on the pivotal role of TLR4 in regulating and mediating neuropathic pain so as to investigate and confirm the key target for managing and alleviating neuropathic pain. The current study identified that EA treatment can significantly inhibited the protein expression of TLR4 and its major intracellular signaling pathways (MyD88 and TRIF) in the central nervous system, downregulate pro-inflammatory cytokine TNF-α, and upregulated anti-inflammatory cytokine IL-10 in both central and peripheral systems (the data of peripheral plantar tissue is not presented in the present study). Combined with our previous research, 16 the data manifested the regulatory effects of EA on PTX-induced protein- and immunofluorescence over-expressions of neuroglia, neurons, and the downstream NF-κB and TLR4 signaling pathway in spinal cord, we established a relatively complete chain of evidence to reveal one of the possible mechanism in potentiating the analgesic therapeutics of EA treatment against PTX-induce neuropathic pain, that is EA attenuates PTX-induced mechanical allodynia and thermal hyperalgesia through modulating spinal neuroglia, neurons, and TLR4-MyD88/TRIF-NF-κB signaling pathway, as well as inhibiting pro-inflammatory cytokine whereas activating anti-inflammatory cytokine (Figure 9).

Schematic diagram of potential mechanism in EA-mediated analgesia of PTX-induced neuropathic pain.
Conclusion
Altogether, our findings indicate that EA treatment ameliorated PTX-induced neuropathic pain behaviors in a rat model, possibly through regulating the abnormal expressions of spinal astrocytes, microglia, neurons, and the TLR4-MyD88/TRIF-cytokine signaling pathway expressed within them. However, the exploration of mechanisms on the analgesic actions of EA in treatment of PTX-induced neuropathic pain in our current study remains preliminary, thus the further in-depth research on the possible mechanisms underlying EA analgesic effects against PTX-induce painful neuropathy should be given more emphasis and investigations.
Footnotes
Acknowledgements
We express our appreciation to Mr. Fa-Zhong Zhao for fostering a positive and supportive environment that consistently encourages us to explore the potential of our ideas and learn from our setbacks.
Author contributions
WMN: Data curation, Formal analysis, Methodology, Resources, Software, Writing–review & editing. Zhou-YX: Data curation, Software, Writing–review & editing. Zhao-YX: Conceptualization, Funding acquisition. Data curation, Formal analysis, Methodology, Resources, Software, Writing-original draft, Writing–review & editing. TJW: Data curation, Software, Writing–review & editing. SX: Data curation, Software, Writing–review & editing.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Natural Science Foundation of Beijing (7252245 and 7202141); Fundamental Research Funds for the Central Public Welfare Research Institutes (ZZCZ2025-033, ZZ-JQ2023005 and ZZ13-YQ-067); Scientific and Technological Innovation Project of China Academy of Chinese Medical Sciences (CIZJS2025006); China Scholarship Council (No. 202005350004).
Ethics statement
In the present study, all procedures complied with the guidelines of ARRIVE (Animal Research: Reporting of In Vivo Experiments) and were conducted in accordance with the National Institutes of Health Guide for the care and use of laboratory animals, and approved by the Institutional Animal Care and Use Committee of China Academy of Chinese Medical Sciences (reference No. 2015010801). All efforts were made to minimize discomfort and the number of animals used.
