Abstract
Bone cancer pain (BCP), one of the most intractable symptoms in patients with cancer, remains poorly understood and lacks effective therapeutic interventions. In this study, we employed an established rat model of BCP induced by intratibial injection of MRMT-1 mammary carcinoma cells. Transcriptomic profiling of the L4-L6 dorsal root ganglia (DRGs) revealed an upregulation of the amino acid transporter SLC38A3. This finding was further confirmed by time-dependent increases in both its mRNA and protein levels. Immunofluorescence co-localization indicated that SLC38A3 was expressed in NF200-, CGRP-, and IB4-positive neurons within the L4-L6 DRGs, and its expression was upregulated in the BCP model. Concomitantly, the transient receptor potential vanilloid 1 (TRPV1) expression in BCP rat DRGs was dynamically upregulated at both the mRNA and protein levels, aligning temporally with pain hypersensitivity. Lentivirus-mediated overexpression or knockdown of SLC38A3 in the DRGs led to a corresponding upregulation or downregulation of TRPV1-expression. Activation of the PI3K/AKT signaling pathway corresponds with BCP-related pain behaviors and expression patterns of SLC38A3 and TRPV1. Bexarotene alleviates BCP in rats by suppressing the aberrant overexpression of SLC38A3, thereby blocking the PI3K/AKT signaling pathway-mediated upregulation of TRPV1. These findings indicate that SLC38A3, through its downstream PI3K/AKT-TRPV1 axis, may serve as a potential molecular mechanism for analgesia in BCP.
Introduction
Bone cancer pain (BCP), one of the most common and intractable symptoms in patients with cancer, is characterized by persistent severe pain and limited therapeutic efficacy, considerably impairing patients’ quality of life.1,2 Current clinical analgesic strategies (e.g., acetaminophen, nonsteroidal anti-inflammatory drugs, and opioids) exhibit suboptimal efficacy and notable adverse effects, severely restricting their clinical application.3–5 Hence, elucidating the molecular mechanisms of BCP and identifying novel therapeutic targets remain critical research priorities.6,7
Glutamine, the most abundant amino acid in plasma, plays a pivotal role in cellular metabolism and signaling regulation. 8 Furthermore, studies have demonstrated that glutamine metabolism can alleviate chronic pain, suggesting its potential as a therapeutic target for pain management.9,10 Particularly noteworthy is the observed involvement of the glutamate-glutamine cycle in the modulation of chronic neuropathic pain in rat models. 11 These findings collectively highlight the pivotal role of glutamine metabolic pathways in pain mechanisms. Glutamine must be transported into or out of cells via active transporters to ensure functional delivery. 12 Members of the solute carrier family 38 (SLC38) are recognized as primary glutamine transporters in mammalian cells, facilitating the transmembrane transport of neutral amino acids. 13 SLC38A3 specifically transports glutamine, asparagine, histidine, and other nitrogen-rich amino acids. 14 Studies have shown that cellular damage or pathological conditions increase the demand for glutamine, which may be met by the upregulation of SLC38A3 (combined with other transporters). In this study, transcriptomic sequencing revealed an upregulation of SLC38A3 in the DRGs of BCP model rats; however, its precise mechanistic role in BCP remains to be elucidated.
Transient receptor potential vanilloid 1 (TRPV1), a member of the TRPV subfamily of the TRP ion channel superfamily, is a cation channel specifically expressed in sensory neurons. These bacteria are sensitive to noxious heat, extracellular acidification, and capsaicin. 15 TRPV1 expression is associated with pain in bone cancer. Following tumor cell inoculation, tumor infiltration induces significant upregulation of TRPV1 expression in the DRGs, thereby exacerbating pain sensitization.16,17 The structural specificity of the peripheral nervous system (e.g., DRGs) renders it a more promising target for analgesia, compared to the central nervous system. 18 However, the side effects of TRPV1 antagonists limit their clinical applications. 15 After tissue inflammation or peripheral nerve injury, the expression and release of interleukin-6 (IL-6) in the DRGs activates the PI3K/AKT signaling pathway, leading to the functional upregulation of TRPV1. Enhanced TRPV1 activity promotes calcium influx, ultimately resulting in neuronal sensitization and hyperalgesia. 19 Based on these findings, we hypothesized that SLC38A3 participates in BCP progression by regulating the PI3K/AKT-mediated TRPV1 activation.
This study employed an established rat model of BCP 20 to demonstrate the critical role of SLC38A3 in BCP pathogenesis and elucidated that it functions by activating the PI3K/AKT-TRPV1 signaling axis. These findings provide novel therapeutic targets and a theoretical foundation for clinical management of BCP.
Material and methods
Reagents and antibodies
Bexarotene (B125743) and MK2206 (M129684) were purchased from Shanghai Aladdin Biochemical Co. Ltd. The rabbit monoclonal anti-SLC38A3 antibody (14315-1-AP) was purchased from Proteintech. Rabbit monoclonal anti-TRPV1 antibody (Ab305299) was purchased from Abcam (Cambridge, MA, USA). Rabbit monoclonal anti-phospho-AKT (Ser473) (#4060) and anti-Akt (#4685) were purchased from Cell Signaling Technology (Danvers, MA, USA).
Cell lines and cell culture
Rat breast cancer cells (MRMT-1) and human embryonic kidney cells (HEK293T) were obtained from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China) (Supplemental Table 1–1). The cells were cultured in Dulbecco’s modified Eagle’s medium (Gibco, Carlsbad, CA, USA) containing 10% fetal bovine serum (Gibco), penicillin (100 units/mL), and streptomycin (100 units/mL) at 37 °C in a humidified incubator containing 5% CO2.
Plasmid construction and lentivirus construction and concentration
The SLC38A3-OEcDNA was inserted into the pCDH-CMV-MCS-EF1-CopGFP-T2A-Puro vector to construct a lentiviral overexpression vector for SLC38A3. Oligonucleotide fragments containing SLC38A3 shRNA were cloned into the hU6-MCS-CBh-gcGFP-IRES-puromycin vector to generate a lentiviral knockdown vector. Lentiviral particles were produced by co-transfecting 293T cells with the envelope plasmid pMD2.G and packaging plasmid psPAX2 (Supplemental Table 1–2). The lentiviral supernatant was mixed with the lentiviral concentration solution in a 5:1 ratio and incubated on a shaker at 0–4 °C for 2 h or overnight. The viral pellet was resuspended in PBS at 1/10 to 1/100 of the original sample volume. The concentrated viral samples were aliquoted and stored at −80 °C for future use.
Real-time PCR
Total RNA was extracted from rat L4-L6 DRGs using TRIzol reagent (Invitrogen, Carlsbad, CA, USA). Complementary DNA was synthesized using the Takara PrimeScript TM RT Master Mix kit (Takara, Tokyo, Japan) according to the manufacturer’s instructions. Real-time PCR was conducted on an ABI7500 sequence detection system (Applied Biosystems) using 2×SYBR Green PCR Master Mix (Promega). The reaction conditions were as follows: 95 °C, pre-denaturation for 5 min, 95 °C for 15 s, 60 °C for 1 min, for a total of 40 cycles. The relative expression levels of genes were calculated using the 2-ΔΔCt method. Rat-specific primers were as follows: SLC38A3,5’-TATCTTCGCCCCCAACATCTT-3’(F), SLC38A3,5’-TGGGCATGATTCGGAAGTAGA-3’(R); TRPV1,5’-ATGACACCATCGCTCTGCTC-3’(F), TRPV1,5’-GTGCTGTCTGGCCCTTGTAG-3’(R); and Actin,5’-TGTCACCAACTGGGACGATA-3’(F), Actin,5’-GGGGTGTTGAAGGTCTCAAA-3’(R).
Protein extraction and western blot analysis
Total protein was extracted from rat DRGs using ice-cold RIPA lysis buffer containing 50-mM Tris-HCl (pH 8.0), 150-mM NaCl, 0.5% sodium deoxycholate, 0.1% SDS, 1% NP-40, 5-mM EDTA, 0.25-mM phenylmethylsulfonyl fluoride, 1% protease inhibitor cocktail and 1% phosphatase inhibitors (to preserve the phosphorylation status of proteins). The protein concentration was measured using a BCA assay kit (Pierce, Rockford, IL, USA). Subsequently, 25–50 μg of protein was separated via sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred onto polyvinylidene fluoride membranes (Millipore, CA, USA). After blocking, the membranes were incubated with primary antibodies (1:1000 dilution) overnight at 4 °C (Supplemental Table 1–3). They were then washed with TBST and incubated with horseradish peroxidase (HRP)-conjugated secondary antibodies (1:1000 dilution) at room temperature for 1 h. Protein bands were visualized using enhanced chemiluminescence reagents and digitally captured using the FluorChem E imaging system (ProteinSimple, CA, USA). Band intensities were quantified from 8-bit linear TIF images using the LabWorks software (Bio-Rad, USA). Densitometric analysis of the Western blot results was performed using Quantity One software (Bio-Rad, v4.6.2). Background subtraction was applied to all target bands. To minimize potential variations in sample loading and transfer efficiency, the integrated optical density of each target protein band was normalized to that of its corresponding β-actin band using the formula: Normalized value = optical density of target protein band / optical density of β-actin band. Following standard normalization procedures, within each individual blot, the density values of target bands were normalized to the corresponding internal control (β-actin), with the normalized value of the control group set to 1.0 to calculate the relative expression levels of experimental groups. The final data represent the average of multiple independent experiments.
Animals and experimental design
In this study, all animals used were Sprague-Dawley rats purchased from Beijing Vital River Laboratory Animal Technology Co., Ltd. (SPF grade, female, 5–6 weeks old, body weight 180–220 g). The rats were housed under standard conditions with a 12-h light/dark cycle and provided free access to food and water. All animals were acclimatized for 1 week prior to the experiments.
Baseline pain thresholds were measured before surgery, and only rats with qualified thresholds were selected for the procedure to employed an established rat model of BCP.21,22 After anesthesia and thorough disinfection, a 1.5-cm transverse incision was made near the knee joint of the left hind limb to expose the tibial bone surface (inverted triangular area). A 5-mL syringe needle was inserted into the tibial bone marrow cavity at a 45° angle, and 4 μL of MRMT-1 rat mammary carcinoma cell suspension (approximately 4 × 10⁵ cells) was injected using a 10-μL microsyringe. Sham-operated rats received an equal volume of PBS at the same location.
For the SLC38A3 overexpression rat model, a 3-cm longitudinal incision was made along the midline of the back at the L4–L5 intervertebral space. The L4 and L5 vertebrae were identified, and a needle was inserted vertically into the subarachnoid space. A PE-10 catheter was advanced for 2–3 cm into the subarachnoid space to the L4–L5 level, and 10 μL of lentivirus overexpressing SLC38A3 (titer: 1 × 10⁹ TU/mL) was intrathecally injected. Control rats received an equal volume of the lentiviral vector. Rats exhibiting paralysis, lameness, abnormal behavior, or local infection were excluded, and the remaining animals underwent behavioral testing. Using the same procedure, the SLC38A3 knockdown rat model was employed.
Mechanical pain test
Before starting animal pain behavior testing, the animals were maintained under normal conditions. All pain behavior experiments were conducted under double-blind conditions daily from 8:00 to 17:00. Behavioral tests were conducted before tumor inoculation and on days 2, 4, 7, 10, 14, and 21 post - inoculation. Rats were placed in transparent plexiglass chambers with a perforated metal grid floor and allowed 10–15 min to acclimate to the environment until they remained calm. The paw withdrawal mechanical threshold (PWMT) was then assessed using Von Frey filaments (NC12775, Certified, USA). Each filament was applied vertically to the mid-plantar surface of the operated hind paw through the grid until the filament bent into an “S” shape, and held for approximately 2 s. A positive response was recorded if the rat withdrew or licked the paw, indicating that the stimulus had exceeded the pain threshold; otherwise, a negative response was noted. A series of calibrated filaments (0.4, 0.6, 1.0, 2.0, 4.0, 6.0, 8.0, and 15.0 g) were used. Testing began with the middle-intensity filament (2.0 g). If no positive response was observed, denoted as “O,” the next stronger filament was applied; if a positive response (“X”) occurred, the next weaker filament was used. A cutoff force of 15.0 g was set to prevent tissue damage. Successive stimuli were separated by 5–10 second intervals. The 50% paw withdrawal threshold (PWT) was calculated using the up-down method, with the following formula applied: 50% threshold (g) = 10^ (X f + k δ).
Heat test
Behavioral testing was performed prior to tumor cell inoculation and on days 2, 4, 7, 10, 14, and 21 post-inoculation. A thermal pain stimulator (ZH-200; Anhui Zhenghua Biological Instrument Co., Ltd., China) was used to stimulate the hind paws of rats. Rats were placed individually in plexiglass chambers on a transparent glass platform that allows light transmission and allowed 10–15 min to acclimate to the test environment. After the animals settled, the plantar surface of the operated hind paw was exposed to a focused radiant heat source using a Hargreaves-type analgesia meter. The paw withdrawal latency (PWL) was defined as the time from the onset of heat stimulation to the occurrence of a withdrawal or licking response. A cutoff time of 20 seconds was automatically applied to prevent tissue damage. Each rat was tested 4–5 times with an inter-trial interval of 3–5 min, and the average value of these measurements was used for subsequent analysis.
Immunofluorescence staining
Briefly, rats were euthanized with an overdose of sodium pentobarbital. Following confirmation of death, DRGs were dissected from the lumbar segments. 23 The harvested L4–L6 DRGs from rats were immediately immersed in 4% paraformaldehyde for fixation, followed by paraffin embedding, sectioning (3–4 μm thick), and staining. To ensure adequate sampling intervals and avoid analyzing adjacent sections, every third section was selected for immunofluorescence staining. The sections were dewaxed and rehydrated using a graded alcohol series. Endogenous peroxidase activity was quenched with 3% hydrogen peroxide (H₂O₂), and antigen retrieval was conducted using freshly prepared sodium citrate buffer. The sections were blocked with 10% goat serum working solution. Primary antibodies (1:200 dilution) were applied to the tissue surface and incubated overnight at 4 °C. Subsequently, species-specific fluorescent secondary antibodies (mouse or rabbit origin, diluted 1:200) were applied to tissues within the immunohistochemical circle and incubated at room temperature for 1–2 h in the dark. Between each antibody incubation step, the sections were washed three times for 5 min each with PBS. All sections were mounted with an anti-fade mounting medium. The prepared sections were imaged using a laser scanning confocal microscope (Model TY2021006721, Nikon Corporation), and the images were captured for further analysis.
Statistical analysis
All experimental data were analyzed using GraphPad Prism software (version 8.0). The results are presented as the mean ± standard error of the mean (mean ± SEM). Statistical significance was set at
Results
Validation of the BCP rat model
As an established model of BCP, cancer was induced by inoculating MRMT-1 mammary carcinoma cells into the left tibial bone marrow cavity of rats (Figure 1a). We first assessed changes in pain-related behaviors before and on days 2, 4, 7, 10, 14, and 21 after modeling to validate the successful construction of the model. The results demonstrated that compared to the sham group, the mechanical and thermal pain thresholds in the model group began to decline significantly on day 7 post-modeling and persisted until the end of the experiment (Figure 1b and c). Subsequently, the tibias were extracted on days 7, 14, and 21 post modeling for macroscopic imaging and hematoxylin and eosin staining. The results revealed that progressive bone destruction was observed in the model group from days 14 to 21, which was accompanied by an increase in the number of cancer cells within the tibia. Furthermore, the trabecular bone showed significant disruption owing to tumor cell infiltration (Figure 1d and e). These findings collectively confirm the validity of the BCP model.

Validation of the BCP rat model.
Significant upregulation of SLC38A3 in DRGs of BCP rats
The DRGs serve as the primary relay station for numerous sensory pathways, including thermal, nociception, mechanosensation, and proprioception. 24 Accumulating evidence suggests that changes in the expression of ion channels and signaling pathway genes in the DRGs play critical roles in the induction and maintenance of BCP. 25 Therefore, we first performed transcriptomic sequencing of L4-L6 DRGs from the sham and model groups (day 21 post-modeling). Differential gene expression analysis demonstrated that SLC38A3 expression was upregulated in the BCP group compared to the sham group (Figure 2a).

Significant upregulation of SLC38A3 in DRGs of BCP rats.
To further validate these findings, we repeated the BCP model construction and extracted L4-L6 DRGs on days 7, 14, and 21 post-modeling to evaluate SLC38A3 expression at both the mRNA and protein levels. Real-time PCR and western blotting results demonstrated that SLC38A3 expression was markedly elevated from day 7 post-modeling, peaking on day 21 (Figure 2b, c). This trend was consistent with the changes in mechanical and thermal pain thresholds in BCP rats, indicating that the upregulation of SLC38A3 at both the mRNA and protein levels may contribute to the development and progression of BCP. To further explore the role of SLC38A3 in the DRGs, we employed double immunofluorescence staining to co-localize SLC38A3 with three neuronal markers in the DRGs from the sham and model groups (day 21 post-modeling). These markers included NF200 (a marker of large neurons), CGRP (a marker of peptidergic small-to-medium neurons), and IB4 (a marker of non-peptidergic small-to-medium neurons). The results showed that SLC38A3 was co-expressed with all three neuronal markers (NF200, CGRP, and IB4), and its co-localization with these markers was significantly higher in the BCP group than in the sham group (Figure2d–f, Supplemental Figure 1). These results indicate that SLC38A3 is significantly upregulated in the DRGs of BCP rats and is associated with pain.
SLC38A3 promotes the development and progression of BCP
We first intrathecally injected normal rats with lentiviral vectors encoding SLC38A3 overexpression plasmids to investigate whether upregulation of SLC38A3 alone is sufficient to elicit pain-related hypersensitivity (Figure 3a). Mechanical and thermal pain thresholds were measured before and on days 2, 4, 7, 10, 14, and 21 after the lentiviral injection. The results revealed that mechanical and thermal pain thresholds began to decline significantly on day 7 post-injection and persisted until day 21 (Figure 3b), indicating that SLC38A3 overexpression enhanced the sensitivity to mechanical and thermal pain. Furthermore, real-time PCR and western blot analyses confirmed that the intrathecal injection of SLC38A3 overexpression lentivirus significantly increased its expression in the DRGs (Figure 3c).

SLC38A3 promotes the development and progression of bone cancer pain.
Next, we intrathecally injected normal rats with the SLC38A3 knockdown lentivirus, allowing it to infect DRG neurons for approximately 1 week. Subsequently, we established the BCP model in these rats (Figure 3d). Real-time PCR and western blot analyses confirmed that intrathecal injection of the SLC38A3 knockdown lentivirus significantly decreased its expression in L4-L6 DRGs (Figure 3e, f). Similarly, mechanical and thermal pain thresholds were measured before and on days 2, 4, 7, 10, 14, and 21 after the lentiviral injection. The results revealed that, compared with rats the BCP group, rats with SLC38A3 knockdown exhibited significantly attenuated reductions in mechanical and thermal pain thresholds (Figure 3g, h), indicating that SLC38A3 knockdown alleviates pain sensitivity in BCP rats. Collectively, these findings provided strong evidence that SLC38A3 upregulation plays a critical role in the development and progression of BCP.
SLC38A3 promotes BCP by upregulating TRPV1 expression
The nociceptive receptor TRPV1 plays a pivotal role in the initiation, maintenance, and progression of BCP, as it detects noxious stimuli, transmitting signals to the spinal cord, and ultimately relaying pain perception to the cerebral cortex. 26 To explore whether the SLC38A3-mediated promotion of BCP is associated with TRPV1 expression, we first evaluated the changes in TRPV1 levels in the L4-L6 DRGs of BCP rats. Real-time PCR (Figure 4a) and western blotting (Figure 4b) analyses demonstrated that TRPV1 expression at both the mRNA and protein levels was significantly upregulated in the BCP group starting from day 7 post-modeling and peaking on day 21, compared to the sham group. Moreover, double immunofluorescence staining demonstrated the colocalization of SLC38A3 with TRPV1, with both expression and colocalization significantly higher in the BCP group than those in the sham group (Figure 4c). These findings indicate that SLC38A3 contributes to BCP pathogenesis by regulating TRPV1 expression.
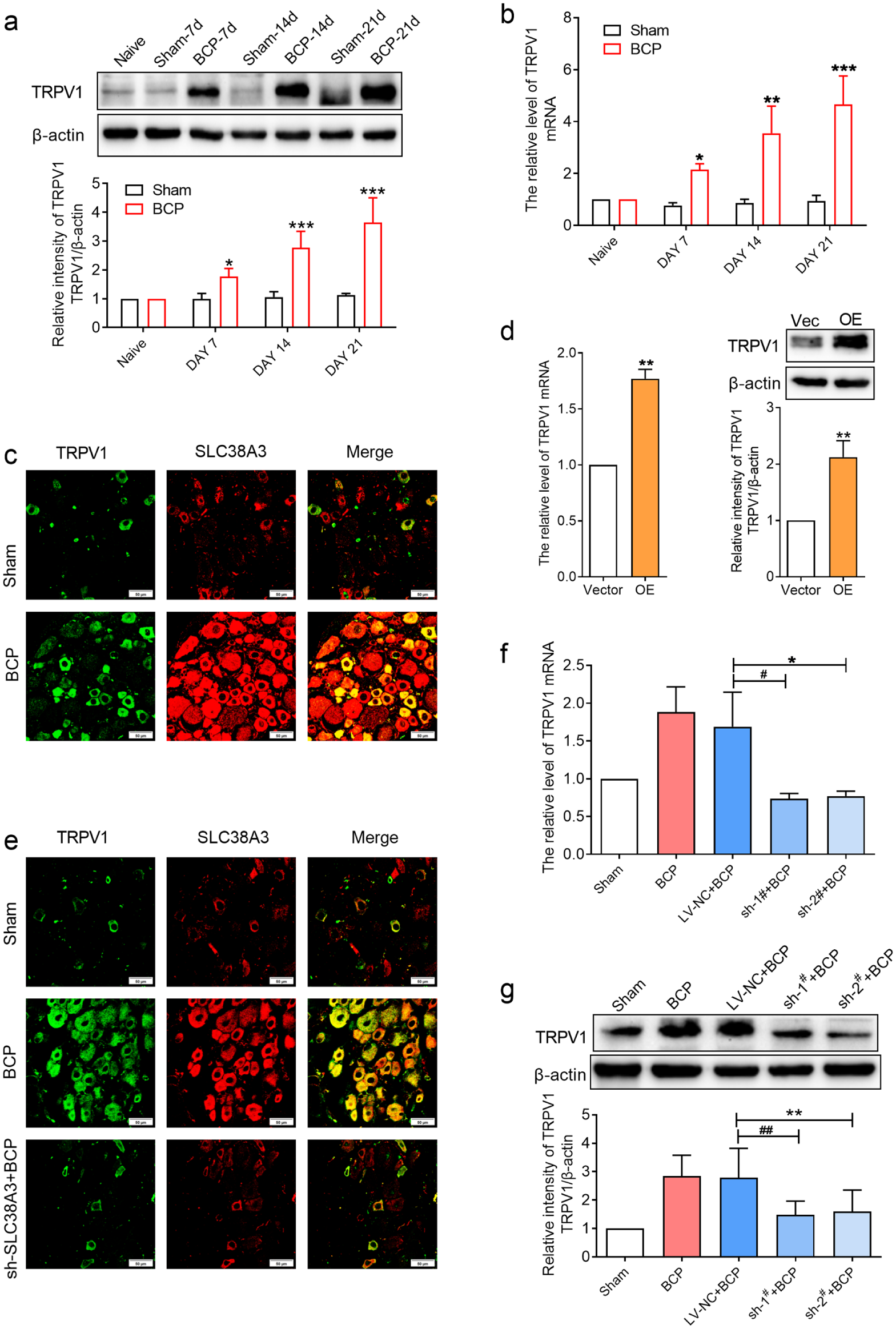
SLC38A3 promotes bone cancer pain by upregulating TRPV1 expression.
To further validate whether SLC38A3 upregulation contributes to the development and maintenance of BCP through TRPV1 modulation, we intrathecally injected lentiviral vectors to overexpress SLC38A3 in the DRGs. Results revealed that TRPV1 expression at both mRNA and protein levels was markedly upregulated following SLC38A3 overexpression (Figure 4d). We intrathecally injected an SLC38A3 knockdown lentivirus into normal rats and established a BCP model 1 week later to further clarify the regulatory effect of SLC38A3 on TRPV1. Immunofluorescence staining revealed that TRPV1 expression and its co-localization with SLC38A3 were significantly reduced in the SLC38A3 knockdown group compared to those in the BCP group (Figure 4e). Subsequent real-time PCR and western blotting analyses confirmed that SLC38A3 knockdown substantially inhibited TRPV1 expression in the DRGs (Figure 4f, g). Collectively, these results indicated that SLC38A3 drives BCP development by promoting TRPV1 expression in the DRGs of BCP rats.
SLC38A3 promotes TRPV1 expression via the PI3K/AKT pathway activation
Previous studies have reported that activation of the PI3K/AKT signaling pathway in DRG neurons of tumor-bearing rats induces functional upregulation of TRPV1, 19 whereas SLC38A3 overexpression promotes epithelial-mesenchymal transition and migration of non-small cell lung cancer cells by activating the PI3K/AKT pathway. 27 According to these findings, we hypothesized that SLC38A3 regulates TRPV1 expression through the PI3K/AKT pathway, thereby driving BCP pathogenesis. To test this hypothesis, we first established a BCP rat model and examined the changes in p-AKT and total AKT protein levels in the L4-L6 DRGs on days 7, 14, and 21 post-modeling. The results showed that p-AKT expression in the DRGs of BCP rats was significantly upregulated starting from day 7, and peaking on day 21, with no notable changes observed in the sham group (Figure 5a). Subsequently, lentiviral vectors were intrathecally injected to overexpress SLC38A3 in the DRGs. Western blot analysis showed that p-AKT expression markedly increased following SLC38A3 overexpression (Figure 5b). To further confirm the effect of SLC38A3 on p-AKT, we intrathecally injected an SLC38A3 knockdown lentivirus into normal rats and established a BCP model 1 week later. Western blotting results demonstrated that SLC38A3 knockdown significantly reduced p-AKT expression in DRGs compared to the BCP group (Figure 5c). These findings indicate that SLC38A3 robustly activates AKT in the DRGs of BCP rats.

SLC38A3 promotes TRPV1 expression via the activation of the PI3K/AKT pathway.
To investigate whether SLC38A3-mediated TRPV1 upregulation was dependent on AKT activation, we administered the AKT inhibitor MK2206 intrathecally to BCP rats on day 14 post-modeling. The treatment was given for four consecutive days, followed by behavioral assessments (Figure 5d). Results showed that both control and MK2206-treated rats exhibited significant mechanical and thermal hyperalgesia compared with rats in the sham group prior to MK2206 treatment. However, after MK2206 intervention, the mechanical and thermal pain thresholds in the treatment group were significantly elevated (Figure 5e, f), indicating that AKT activation is critical for BCP progression. Subsequent western blotting and immunofluorescence analyses showed that p-AKT and TRPV1 expression in the DRGs of the treatment group was significantly lower than that in the BCP group (Figure 5g, h). Collectively, these results demonstrate that SLC38A3 promotes TRPV1 expression by activating the PI3K/AKT pathway, thereby mediating BCP development and progression. This discovery provides novel insights into the molecular mechanisms underlying BCP.
Bexarotene alleviates BCP by suppressing SLC38A3 expression
Previous studies have shown that bexarotene, a retinoid X receptor (RXR) agonist, exhibits enhanced antitumor effects when SLC38A3 is overexpressed in cancer cells. 28 To determine whether bexarotene could mitigate BCP by inhibiting SLC38A3 upregulation, we established a BCP rat model and randomly divided the rats into four groups on day 7 post-modelling: sham group, BCP group, bexarotene-treated group (oral gavage, 50 mg/kg), and vehicle control group (oral gavage with equivalent solvent). Treatments were administered once daily for two weeks, and pain-related behaviors were monitored throughout the intervention period (Figure 6a). Results demonstrated that bexarotene treatment significantly elevated the mechanical and thermal pain thresholds in BCP rats compared to those in the BCP group (Figure 6b, c), indicating its potent analgesic efficacy. Real-time PCR and western blotting analyses revealed that bexarotene treatment markedly suppressed SLC38A3 expression in the DRGs of BCP rats (Figure 6d, e). Subsequent protein-level assessments of p-AKT/AKT and TRPV1 demonstrated that bexarotene significantly reduced their expression compared to that in the vehicle control group (Figure 6f, g), consistent with the observed behavioral improvements.

Bexarotene alleviates BCP by suppressing SLC38A3 expression.
In conclusion, bexarotene alleviated BCP by inhibiting SLC38A3 overexpression, thereby suppressing the PI3K/AKT pathway-mediated upregulation of TRPV1. These findings provide experimental evidence supporting bexarotene as a potential therapeutic agent for BCP and further elucidate the critical role of the SLC38A3-PI3K/AKT-TRPV1 signaling axis in BCP pathogenesis.
Discussion
BCP, characterized by persistent pain, pathological fractures, and motor dysfunction, severely impairs patients’ functional status, quality of life, and survival.29–32 Therefore, elucidating the molecular mechanisms of action of BCP and identifying novel therapeutic targets are of critical clinical significance. In this study, we showed that SLC38A3 promoted BCP by activating the PI3K/AKT signaling pathway, leading to the upregulation of TRPV1 expression. Conversely, suppressing SLC38A3 overexpression significantly relieved BCP. These findings uncover, for the first time, the involvement of SLC38A3 in BCP and provide initial evidence supporting its potential as a promising therapeutic target for BCP treatment.
The solute carrier (SLC) superfamily is comprised of 65 families and 439 transporters, rendering it as one of the most important membrane protein families in the human genome. SLC38A3, a member of the solute carrier family, plays a pivotal role in metastasis of numerous cancers. Studies indicate that SLC38A3 promotes the progression of colorectal cancer (CRC) by positively regulating the function of Hsp70, thereby enhancing tumor cell proliferation and migration. 33 Moreover, SLC38A3 modulates the levels of glutamine, glutamate, asparagine, aspartate, alanine, and glutathione in breast cancer cells, facilitating breast cancer metastasis through the GSK3β/β-catenin/EMT pathway. 34 Previous studies have shown that SLC38A3 mediates glutamine uptake and release of glutamine. 35 Furthermore, treatment with D-aspartate attenuated demyelination and accelerated remyelination in a mouse model of myelin injury and repair. 36 Biallelic variants of SLC38A3, which encodes a glutamine transporter, disrupt the glutamate/GABA-glutamine cycle, leading to epileptic encephalopathy. 37 These findings indicate that SLC38A3 plays a significant role in damaged neural cells. Based on these findings, we established a rat model of BCP and observed significant upregulation of SLC38A3 expression in the L4-L6 DRGs. Furthermore, SLC38A3 expression were positively correlated with pain-related behaviors in rats. These findings suggest that SLC38A3 may be involved in BCP development and progression.
Certain sarcoma cell lines produce soluble lipophilic factors that activate the peripheral nociceptor transduction system by stimulating TRPV1, which contributes to cancer pain. 38 Studies have shown that treatment of DRG neurons with TNF-α increases TRPV1 protein expression through the p38/ mitogen-activated protein kinase (MAPK) and PKC (protein kinase C) signaling pathways, leading to thermal hyperalgesia and nociceptor sensitization. 39 In DRG neurons, the IGF-1 receptor is co-expressed with TRPV1, and treatment with IGF-1 increases both total and membrane-bound TRPV1 protein levels. Conversely, IGF-1 receptor inhibition alleviates BCP behaviors.40,41 These findings indicated that TRPV1, a key regulator of pathological pain. 42 Recent studies have shown that Walker256 cell transplantation exacerbates mechanical allodynia in BCP rats with significantly improved TRPV1 expression in the L4-L6 DRGs. Intrathecal administration of CPZ (a TRPV1 inhibitor) significantly alleviated pain sensitivity in BCP rats.43,44 In this study, we confirmed that both mRNA and protein levels of TRPV1 were significantly upregulated in the DRGs of BCP model rats, with expression trends aligned closely with pain behaviors. Notably, SLC38A3 overexpression further activated TRPV1, whereas SLC38A3 knockdown significantly suppressed its expression, indicating a regulatory relationship between them.
The PI3K/AKT signaling pathway, a central regulator of neuronal plasticity and sensitization, exacerbates BCP-related allodynia when activated.45–47 Moreover, the total protein levels of SLC38A3 and mTOR signaling pathway-related proteins, including AKT, ribosomal protein S6 kinase B1 (RPS6KB1), and ribosomal protein S6 (RPS6), increased with Met supplementation. 48 LSW increased pain thresholds and reduced inflammatory cytokine levels in BCP rats by inhibiting PI3K/Akt and TRPV1 signaling in astrocytes and microglia. 49 TRPV1 and the PI3K/AKT/mTOR pathway are involved in the pathogenesis of chronic postoperative pain (CPOP) at the spinal level. 50 Maresin1 regulates neural growth through the PI3K-AKT-mTOR signaling pathway in vitro and in vivo, and its effects are associated with TRPV1 expression. 51 The dual PI3Kδ/γ inhibitor duvelisib inhibits paclitaxel-induced sensitization of TRPV1 receptors and boosts PI3K/Akt signaling in small-diameter dorsal root ganglion (DRG) neurons, while also increases the population of CD68-positive cells within the DRGs. 52 The endogenous chemokine CCL2 improves the function and mRNA levels of TRPV1 channels and Na(v)1.8 sodium channels in small DRG sensory neurons through activation of the PI3K/Akt signaling pathway. 53 Our results confirmed that SLC38A3 drive TRPV1 functional upregulation by activating the PI3K/AKT pathway. However, it should be noted that this study did not fully elucidate the specific molecular mechanisms by which SLC38A3, as a glutamine transporter, modulates neuronal excitability through amino acid metabolism, warranting further investigation. 27 However, our study has several limitations. First, the sample size for transcriptome sequencing analysis was relatively small, and future studies with larger sample sizes are needed to enhance the reliability of the results. Second, the intrathecal administration method employed precludes precise delineation of the relative contributions of SLC38A3 in the DRG versus the spinal cord. Consequently, the potential role of SLC38A3 expressed in spinal neurons or glial cells in pain processing remains to be elucidated. Furthermore, we cannot exclude the involvement of other ion channels in bone cancer pain. This is particularly relevant for P2X7 54 and NaV1.7, 55 which have been strongly implicated in nociception across various pain models. These limitations highlight critical directions for future research: developing DRG-specific interventions and conditional knockout models will help to precisely dissect the mechanistic role of SLC38A3 in pain regulation, thereby providing new avenues for the targeted treatment of bone cancer pain.
Bexarotene, a selective RXR agonist, regulates cell growth, apoptosis, and differentiation by binding and activating RXR.56,57 Research has demonstrated that bexarotene enhances astrocyte phagocytic activity via the ABCA1-mediated pathway and facilitates neurobehavioral recovery in mice after subarachnoid hemorrhage. 58 Furthermore, bexarotene alleviates neuroinflammation and improves neurological deficits after subarachnoid hemorrhage by activating RXR and modulating the PPARγ/SIRT6/FoxO3a pathway. 59 Additionally, bexarotene promotes the expression of microglia/macrophage-specific brain-derived neurotrophic factors and axonal sprouting, thereby improving neurological function after traumatic brain injury. 60 Bexarotene, a selective RXR agonist, alleviates hyperalgesia in lipopolysaccharide-induced pain models by suppressing the TLR4/MyD88/TAK1/NF-κB/COX-2 signaling pathway. 61 Moreover, bexarotene mitigates spinal neuroinflammation and neuropathic pain induced by chronic constriction injury by targeting MAPK phosphatase-1. 62 In this study, we demonstrated, for the first time, that bexarotene intervention significantly suppressed the expression of SLC38A3, p-AKT/AKT, and TRPV1 in the DRGs of BCP rats, accompanied by the alleviation of mechanical allodynia and thermal hyperalgesia. These findings indicate that bexarotene exerts its analgesic effects on BCP by targeting the SLC38A3-PI3K/AKT-TRPV1 signaling axis. However, the suboptimal results of bexarotene in human trials indicate that therapeutic strategies validated in mouse models must undergo systematic optimization and clinical translation before they can be applied to human disease treatment. 63
In summary, this study reveals a novel mechanism by which SLC38A3 promotes BCP progression by activating the PI3K/AKT pathway to drive TRPV1 expression. Furthermore, we validated that bexarotene alleviates BCP by inhibiting this signaling axis. These findings provide a theoretical foundation and novel therapeutic targets for the precise treatment of BCP.
Supplemental Material
sj-docx-1-mpx-10.1177_17448069251395641 – Supplemental material for Amino acid transporter SLC38A3 mediates bone cancer pain in rats via the PI3K/AKT/TRPV1 signaling pathway
Supplemental material, sj-docx-1-mpx-10.1177_17448069251395641 for Amino acid transporter SLC38A3 mediates bone cancer pain in rats via the PI3K/AKT/TRPV1 signaling pathway by Yu-Na Cheng, Ming-Zhu Zhang, Cui-Qing Xie, Zhi-Qi Li, You-Feng Ge, Zhao-Hui Jin, Zhi-Hua Luo, Chen-Yang Zheng, Liang Chen, Dong Fang, Chen-Guang Niu and Jin-Jin He in Molecular Pain
Footnotes
Acknowledgements
The authors have nothing to report.
Author Contributions
Yu-Na Cheng: Methodology, Data curation, Writing- original draft. Ming-Zhu Zhang: Formal analysis, Methodology. Cui-Qing Xie: Visualization, Data curation. Zhi-Qi Li: Investigation, Formal analysis. You-Feng Ge: Formal analysis, Investigation. Zhao-Hui Jin: Methodology. Zhi-Hua Luo: Data curation. Chen-Yang Zheng: Formal analysis. Liang-Chen: Formal analysis. Dong Fang: Conceptualization, Funding acquisition. Chen-Guang Niu: Methodology, Conceptualization, Funding acquisition. Jin-Jin He: Formal analysis, Investigation, Writing-original draft.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Key R&D Program of China (2021YFA0909600), Key projects of Henan Science and Technology Department (242102311032), National Natural Science Foundation of China (82103514), the scientific and technological project of Science and Technology Department of Henan Province (242102311055) and the Key Project of Science and Technology Research Funded by the Educational Commission of Henan Province (25A310011).
Disclosure
None of the authors of this manuscript is currently an Editor or Editorial Board Member of
Ethics Statement
Approval of the research protocol by an institutional review board: N/A.
Informed consent
N/A.
Registry and the registration no. of the study/trial
N/A.
Animal studies
The animal studies were approved by the Institutional Animal Care and Use Committee of Henan University (HUSOM2022-459)
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
