Abstract
Bone cancer pain is one of the most severe and intractable complications in patients suffering from primary or metastatic bone cancer and profoundly compromises the quality of life. Emerging evidence indicates that the dorsal root ganglion play an integral role in the modulation of pain hypersensitivity. However, the underlying molecular mechanisms during dorsal root ganglion-mediated bone cancer pain remain elusive. In this study, RNA-sequencing was used to detect the differentially expressed genes in dorsal root ganglion neurons of a rat bone cancer pain model established by intratibial inoculation of Walker 256 breast cancer cells. Gene ontology and Kyoto Encyclopedia of Genes and Genomes analysis showed that the differentially expressed genes (fold change > 1.5; false discovery rate < 0.05) were enriched in the bone morphogenetic protein (BMP) signaling pathway, transforming growth factor-β signaling pathway, and positive regulation of cartilage development. Importantly, serum deprivation-response protein (
Introduction
Various tumor types including breast, prostate, and lung carcinomas preferentially metastasize to the skeleton, where they induce significant bone remodeling, destruction, and bone cancer pain (BCP).1–3 BCP is often severe and intractable and is a significant contributing factor to morbidity and loss of quality of life. 4 Most of the current pharmacological treatments provide nonselective relief for pain syndromes. Even for palliative radiotherapy, the rate of complete pain relief remains low. 5 Therefore, it is pivotal to investigate the pathogenesis of BCP and identify novel therapeutic targets for translational medicine.
Inflammatory, neuropathic, and/or ischemic components have been reported to be involved in cancer-induced bone pain. 6 In rodent BCP models, bone metastasis of cancers alters the structure, physiology, and biochemistry of the bone and concurrently induces mechanical and inflammatory hyperalgesia. 7 In particular, hyperexcitability of nociceptive dorsal root ganglion (DRG) neurons is associated with inflammatory pain in rodent models. 8 Emerging evidence indicates that alterations in gene expression9–15 and signaling pathways16–18 contribute to induction and maintenance of BCP. For example, upregulation of P2X3 receptors, 9 sodium channels Nav1.813 and Nav1.9, 10 brain-derived neurotrophic factor,11,19 acid-sensing ion channel 3, 12 protease-activated receptor 2 and 4, 14 and activation of cAMP-PKA, 16 PI3K/Akt, 17 and Hedgehog signaling 18 were involved in DRG-mediated BCP in a rat model. Moreover, several studies have shown attenuation of BCP in rats. These studies included overexpression of suppressor of cytokine signaling 3 in DRG, 15 suppression of asparaginyl endopeptidase in cancer cells, intrathecal injection of genetically engineered human bone marrow stem cells expressing the proenkephalin gene, 20 and intraperitoneal administration of a somatostatin receptor type 4 selective agonist, J-2156. However, comprehensive gene expression profiling of DRGs in a BCP model has not been reported. Such profiling could help identify novel therapeutic targets for the relief of BCP.
In this study, next-generation RNA-sequencing (RNA-seq) was performed to detect the differentially expressed genes (DEGs) in DRGs from a rat BCP model. The results showed that DEGs were enriched in signaling pathways associated with bone pain, inflammation, and degeneration. In particular,
Materials and methods
Animal husbandry
Female Sprague-Dawley rats (200 g–250 g, B&K Universal Group Limited) were used for this study. Animals were housed in separate cages at a temperature of 24 ± 1°C under a 12-h/12-h light-dark cycle and had free access to food and water. All animal procedures were carried out in accordance with the guidelines of the International Association for the Study of Pain 21 and were approved by the Animal Care and Use Committee of Shanghai Chest Hospital, Shanghai Jiao Tong University (Permission No. KS (Y)1616).
BCP model
The BCP model was established as previously described. 22 Briefly, three female Sprague-Dawley rats weighing 70 g to 80 g received an intraperitoneal inoculation of Walker 256 cells. After one week, cells in the ascites were collected and resuspended in saline to a final concentration of 2 × 107 cells/mL. Then, female Sprague-Dawley rats weighing 200 g to 250 g were anesthetized with chloral hydrate (300 mg/kg, i.p.). A 23-gauge needle was inserted into the intramedullary canal of the left tibia, in the lower one third, to create a cavity for the injection of the cells, and 10 μL vehicle (sham group) or Walker 256 cells (2 × 105 cells) (BCP group) were injected into the bone cavity. The cavity was sealed using bone wax prior to closure of the incision. The rats were returned to their home cages upon waking. Rats were sacrificed 21 days after tumor inoculation, and tibias were collected for gross examination.
Pain-related behaviors
Mechanical allodynia was determined by a significant decrease in the threshold of hind paw withdrawal to mechanical stimulation applied to the plantar surface of each hind paw. 23 Briefly, the animals were individually placed beneath an inverted ventilated cage with a metal-mesh floor. Von Frey monofilaments (0.6-, 1.0-, 1.4-, 2-, 4-, 6-, 8-, 10-, and 15-g), starting with 1 g and ending with 15 g in ascending order, were applied perpendicular to the plantar surface of each hind paw from beneath until the paw was withdrawn. The duration of each stimulus was approximately 2 s. Each monofilament was applied five times at 5-min intervals. Quick withdrawal or paw flinching was considered a positive response. The paw withdrawal mechanical frequency (PWMF) to each monofilament was calculated based on five applications. The paw withdrawal mechanical threshold (PWMT) was determined by the force at which PWMF ≥ 60%; 15 g was recorded as the PWMT if PWMF < 60% to all filaments.
Movement-evoked pain was assessed by limb use score. Rats were permitted to move spontaneously on a smooth plastic table (50 cm × 50 cm). The limb use during spontaneous ambulation was scored on a scale of 4 to 0 (4. normal use; 3. slightly limping; 2. clearly limping; 1. no use of the limbs (partial); and 0. no use of the limbs (complete)).
RNA-seq
Rats were promptly sacrificed by cervical dislocation. L3-L5 DRGs were quickly harvested from sham and BCP rats (
Western blotting
The expression of
Real-time quantitative polymerase chain reaction
Rats were anesthetized with sodium pentobarbital (50 mg/kg i.p.), and the L3-L5 DRGs were quickly removed at the defined timepoint for each group. Total RNA was extracted using a total RNA Kit (invitrogen), and complementary DNA (cDNA) was generated using a cDNA Synthesis Kit (Promega, Madison, WI), according to the manufacturer’s instructions. Real-time quantitative polymerase chain reaction (RT-qPCR) was carried out using a SYBR Green assay (Roche Ltd, Basel, Switzerland) and Rotor-Gene 3000 system (Corbett Research, Australia) based on the 2–△△Ct method. β-actin was used as a housekeeping gene. The primer sequences for
Immunohistochemistry
Animals were deeply anesthetized with sodium pentobarbital (50 mg/kg, i.p.) and underwent sternotomy, followed by intracardiac perfusion with 200 mL saline and 200 mL 4% ice-cold paraformaldehyde in 0.1 M phosphate-buffered saline. The spinal cord and L3-L5 DRGs were removed, postfixed in 4% paraformaldehyde for 4 h, and subsequently allowed to equilibrate in 30% sucrose in phosphate-buffered saline overnight at 4°C. Immunohistochemical analyses were performed with the Histostain-SP kit (Zymed, San Francisco, CA). After rehydration, inhibition of endogenous peroxidase with 3% hydrogen peroxide, and blocking (10% rabbit serum) for 30 min, the tissue sections were incubated with a specific antibody against
Targeting Bmp2 by siRNA in BCP model
Statistics
Data are expressed as the mean ±
Results
Pain behaviors of the rat BCP model
Rats inoculated with Walker 256 tumor cells displayed a gradual increase in sensitivity to Von Frey filament stimulation. On day 21 postinoculation of Walker 256 cancer cells, the PWMT from the BCP group decreased from the baseline level of 14.3 ± 1.4 g to 0.9 ± 0.3 g (Figure 1(a)). The limb use score for the BCP group decreased from the baseline level (4.0±0.0) to 0.3 ± 0.23 (Figure 1(b)). The anatomic structure of the tibia appeared thicker, and palpable tumors were noted outside the bone upon gross examination of BCP rats 21 days postinoculation of Walker 256 cancer cells (Figure 1(c)).

The establishment of BCP model by Walker 256 cancer cells inoculation in rats. Rats were inoculated with live or heat-killed Walker 256 cells as described in Materials and method section. (a) Paw withdrawal mechanical threshold (PWMT) and (b) paw withdrawal thermal latency for pain behavior test in sham and BCP group. (c) Anatomic structure of tibia bone from sham and BCP mice on 21 days postinoculation of Walker 256 cancer cells in rats. Data (a and b) are presented as mean ±
Gene expression profiling of DRG neurons from BCP rats by RNA-seq
RNA-seq of rat DRG neurons was performed after confirming the BCP phenotype by radiographic imaging and pain-related behavior measurement at day 21 postinoculation of Walker 256 cancer cells. Eighty DEGs (fold change (FC) > 1.5; false discovery rate < 0.05) were identified in the BCP group (
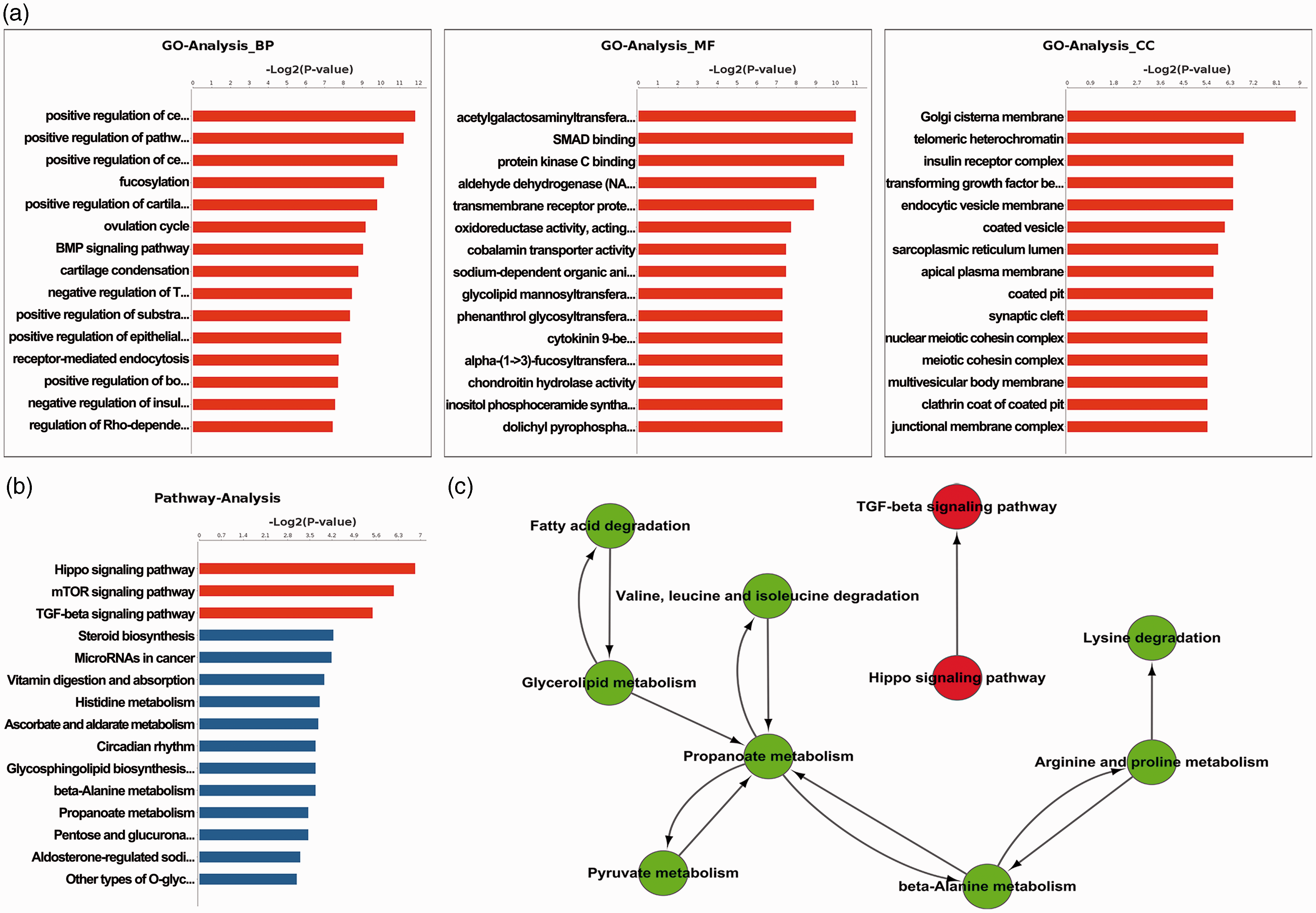
GO and KEGG analysis of DEGs in DRGs (BCP vs. Sham). (a) Gene ontology showed the enrichment of DEGs (BCP vs. Sham) in cellular component, molecular function, and biological process. (b) and (c) KEGG pathway analysis of DEGs (BCP vs. Sham).
Differentially expressed genes (false discovery rate < 0.05) in DRG neurons (BCP vs. Sham) by RNA-seq.

Note: The references cited in this table are shown in Supplementary Table 1.
Upregulation of Bmp2 in DRG is associated with BCP

Upregulation of the expression of Bmp2 in DRGs of BCP rats. The expression of Bmp2 in DRGs from BCP rats was validated by immunohistochemistry (a), RT-qPCR (b), ELISA (c), and western blotting (d). Immunofluorescent microscopy was done using anti-BMP2 antibody as described in Materials and method section. The mRNA level of BMP2 was measured by real-time RT-PCR and normalized by β-actin. The protein level of BMP2 in DRG samples was measured by ELISA and western blotting signals. Representative protein bands are presented on the top of the responding histogram. Data (b and c) are presented as mean ±
Targeting Bmp2 ameliorates BCP in vivo
Information arising from RNA-seq data and subsequent validation prompted us to further investigate the role of

Targeting Bmp2 by intrathecal siRNA administration in BCP rats. (a) The knockdown effect of Bmp2 siRNA was tested by RT-qPCR (
Discussion
Primary and metastatic cancers that affect the bone are frequently associated with severe and intractable BCP including nociceptive, neuropathic, and inflammatory pain.1,24 Although the etiology of BCP remains unclear, animal models of BCP make it possible to unravel neuropathological processes that occur in the region of tumor growth. In recent years, next-generation sequencing including RNA-seq has been used to analyze the cellular transcriptome for mechanistic study and translational medicine. However, gene expression profiling by RNA-seq of DRGs during BCP and peripheral hypersensitivity has not been reported. Here, we made use of RNA-seq and identified eighty DEGs in DRGs from BCP compared to sham rats. Functional annotation showed that nine of the DEGs were associated with bone pain. For example,
BMPs are a group of growth and morphogenetic factors originally discovered by their ability to induce the formation of bone and cartilage. Among the BMP family members, BMP2-7 belongs to the TGF-β superfamily of proteins. BMP2, 4, 6, and 7 are commonly referred to as the osteogenic BMPs, based on their potent bone-inducing activity.
30
BMP3 is a negative regulator of bone density.
31
BMP4 is an important factor in early nervous system development and regeneration after injury and a critical mediator of adult physiological plasticity.
32
In this study, we found that
There are several limitations for the current study. First, we did not include double labeling of the nociceptive neurons and
Despite their limitations, our results confirm the importance of
Supplemental Material
Supplemental material for Upregulation of bone morphogenetic protein 2 (Bmp2 ) in dorsal root ganglion in a rat model of bone cancer pain
Supplemental Material for Upregulation of bone morphogenetic protein 2 (
Footnotes
Acknowledgments
The authors thank Dr. Xingji You and Dr. Hui Sheng (Department of Physiology, Second Military Medical University, Shanghai, China) for their advice and helpful suggestions.
Author Contributions
WJX designed and supervised the research project and wrote the paper. WW executed the rat bone cancer model and performed western blotting and data analysis. JQL performed the paw withdrawal threshold and flinching behavior tests. TW assisted with the rat bone cancer model. XMY aided in data analysis.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by the National Natural Science Foundation of China (Project 81671078).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
