Abstract
Cerulein is an orthologue of cholecystokinin, which is often used to induce acute pancreatitis in pre-clinical studies. In these models, animals show signs of pain, and this is the most common complaint of patients with acute pancreatitis. However, little is known about how this pain is mediated, the role of cerulein murine pain responses, or its relevance to human pancreatitis pain. We injected 25 or 50 µg/kg cerulein intraperitoneally into male and female mice and assessed pain behaviors using the von Frey test of mechanical hypersensitivity. The excitability of mouse and human visceral dorsal root ganglia (DRG) neurons was assessed using whole-cell patch-clamp electrophysiology. Pharmacology was performed using commercial antagonists of cholecystokinin (CCK) A or B receptors. We show that pain behaviors developed similarly in male and female cerulein-injected mice and that visceral DRG from these mice exhibited increased excitability compared to controls. Direct application of cerulein to T8-L2 mouse and human DRG showed increased excitability compared to controls consistent with DRG from cerulein-injected mice. The actions of cerulein on visceral DRG neurons were attributed to CCK-A, but not CCK-B receptor. A similar response to cerulein was observed in human thoracic DRG neurons. These findings highlight the importance of the cholecystokinin system, particularly the CCK-A receptor, to visceral pain including pancreatitis through direct sensitization of visceral DRG neurons from mice or humans.
Keywords
Introduction
The cerulein-induced acute pancreatitis model is widely used as an effective tool by researchers for studying both acute and chronic pancreatitis.1–6 Cerulein, an orthologue of cholecystokinin (CCK), is a secretory peptide released by specialized intestinal cells, visceral nerves and other cell types. CCK release after a meal modulates the secretion of digestive enzymes by the exocrine pancreatitis. Acute pancreatitis is an acute inflammatory disease most often caused by alcohol misuse or gallstones obstructing the pancreatic duct. Chronic pancreatitis is characterized by chronic pancreatic inflammation and is most often caused by alcohol misuse combined with cigarette smoking. 7 A common feature of acute and chronic pancreatitis is severe abdominal pain. Treatment of rodents and other animal models with supra-physiologic concentrations of CCK or cerulein for a day or two causes acute pancreatitis. 6 If supra-physiologic concentrations of CCK are repeated over weeks, it can lead to chronic pancreatitis. In both cases, the animals show signs of pain.
Whether endogenous CCK participates in acute or chronic pancreatitis injury in humans has been discussed. 8 However, one preliminary study found that a CCK-A receptor antagonist reduced pain in patients with acute pancreatitis. 9 A second found that the same antagonist significantly reduced pain in chronic patients experiencing an acute attack. 10 Although the role of CCK in the pathogenesis of injury and inflammation in acute and chronic pancreatitis remains unclear, CCK likely has a role in mediating the associated pain.
The CCK receptor system has been a historic target for pain management and is found throughout the viscera and peripheral and central nervous systems. 11 In addition, there is substantial evidence to suggest that cerulein induces a condition in rodents similar clinically to that experienced by patients. 8 For example, treatment of rodents with supramaximal doses of cerulein induces pro-inflammatory cytokines, such as interleukin-1β, interleukin-6, tumor necrosis factor-α, and chemokines as seen in clinical pancreatitis.12–18 Cerulein also produces reactive oxygen species in pancreatic acinar cells. 19 These actions stimulate a robust inflammatory response in the pancreas and surrounding tissues. The result is the pathological sensitization of pancreatic sensory neurons and a vicious cycle of neurogenic inflammation that causes transmission of the signal to the spinal cord via small myelinated and unmyelinated fibers20–22 and is, therefore, expected to produce significant functional changes in the nociceptive system serving this organ.
In neuronal physiology, CCKAR/CCKBR leads to the activation of various downstream targets, many of which are ion channels. Voltage-gated ion channels play a fundamental role in controlling neuronal excitability. The upregulation of Na+ currents and suppression of K+ currents appear to contribute to peripheral sensitization.20–22 In addition, ionic conductances, which underlie the generation of action potentials and regulate excitability of T8-L2 dorsal root ganglia (DRG) neurons, are fundamentally important in the transduction of pain signals from the GI tract to the CNS.23,24 Furthermore, it is more likely that changes in ion channel function in nociceptive DRG neurons are organ/disease-specific, which further modulates the intrinsic properties of DRG neurons and is important in the genesis of visceral pain.25,26
In this study, we investigated changes in the electrophysiological properties and excitability of visceral DRG neurons in response to the cerulein-induced acute pancreatitis model in mice. To better characterize this phenomenon, we also investigated the direct effect of cerulein on mouse DRG neurons. We also examined human visceral DRG neurons using the same paradigm. Finally, we examined the differential contribution of CCK-A and CCK-B receptors in mediating changes in mouse and human visceral DRG neurons. We find that cerulein can induce visceral pain and changes in DRG responses. We also demonstrate a direct effect of cerulein on DRG neurons. Our studies highlight the need to understand changes in DRG electrophysiology and the cholecystokinin system in mediating visceral pain states in mice and humans.
Methods
Animals
All animal research was conducted with the approval of the University of New Mexico Institutional Animal Care and Use Committee. Our facilities are accredited by AAALAC and possess the necessary federal certifications: Animal Welfare Assurance #D16-00,228 (A3350-01) and USDA Registration # 85-R-0014. Male and female C57BL/6NJ mice (20–25 g; 5–6 weeks) were obtained from Jackson Laboratories. Rodents were housed in a well-ventilated room maintained at 20–22°C, with a reversed 10/14 h dark/light cycle to facilitate testing during their active phase. They were acclimated for 1 week prior to the commencement of the studies. They had unrestricted access to food and water throughout the experimental period, receiving a standard chow diet with low soybean content.
Cerulein-induced acute pancreatitis model
The acute pancreatitis model was induced by repeated injections of the cholecystokinin analog cerulein (Sigma-Aldrich, Cat# C9026, St. Louis, MO) dissolved in 0.9% sterile saline. Animals were randomly divided into three groups: cerulein 25 µg/kg, cerulein 50 µg/kg, and naïve control. In brief, animals received hourly intraperitoneal (i.p.) injections of 100 µL of 0.9% sterile saline containing cerulein, 6 times a day for 2 days. Naïve control animals were not injected. Mice were sacrificed on day 2 after the last cerulein injection, and the T8-L2 DRG were harvested for primary culture intended for electrophysiological study.
In vivo experimental pain-related behavioral testing
Sensitivity on the left hind paw was assessed before cerulein administration to establish a baseline threshold and at the pre-determined time points after the first cerulein dose using the “up and down” approach. 27 Hypersensitivity is measured by the reflexive withdrawal response time to mechanical stimulation of the left hind paw 28 using graded, thin nylon von Frey filaments with defined bending forces (tensile strength). Five applications of several mid-range von Frey filaments, applied once every 3–4 s, comprised the trial. The next stronger filament was used if no affirmative reaction was received. Reduced sensitivity threshold, or “hypersensitivity,” was demonstrated by responses to a lower gram force than in the control group.
Mouse dorsal root ganglion (mDRG) culture
Persistent or ongoing pain in acute pancreatitis is thought to be initiated and maintained by activating and sensitizing pancreatic afferents. 29 Since the behavioral data did not show a significant difference in withdrawal threshold between the cerulein 25 µg/kg and cerulein 50 µg/kg groups, mice from the cerulein 25 µg/kg group were sacrificed on day 2 after the last cerulein injection. The T8-L2 dorsal root ganglia (DRG) were harvested for primary culture intended for electrophysiological study. Mouse DRG cultures were performed as previously described by our group and others with some modifications.30–32 Briefly, mice were deeply anesthetized with 3% isoflurane and then euthanized by decapitation prior to dissecting the dorsal root ganglia (DRG) for primary cultures intended for electrophysiological study. Bilateral T8-L2 DRG were carefully removed and put into an enzymatic solution containing sterile, magnesium/calcium-free HBSS (Stem Cell Technologies, Cat# 37250), papain (Worthington, Cat# LS003126), dispase (Sigma, Cat# D4693), and collagenase (Worthington, Cat# LS004176). Enzymatic digestion was carried out at 37°C, 5% CO2 for 40 min, with trituration every 20 min. Complete DMEM-based media (2 mL DMEM supplemented with 10% fetal bovine serum, 1% antibacterial/antimycotic with 100 units/mL of penicillin, 100 μg/mL of streptomycin, and 25 μg/mL of amphotericin B) was added to the enzymatic digestion. The solution was then strained through a 100 μm cell strainer and rinsed several times with an additional 6 mL complete media. The digested DRG cell suspension was rinsed by gentle centrifugation at 300 × g for 5 min and resuspended in 1 mL complete media. 125 μL of the mouse DRG cell suspension was added to each 12 mm coverslip pre-coated with poly-d-lysine (Gibco, Cat# A38904-01) and coated with 50 μg/mL laminin (Sigma, Cat# L2020), allowed to attach for 30–60 min before gently flooding the wells with enough media to fill each well, 1–2 mL for a 12 well plate. Electrophysiological recordings were done 16–20 h after mDRG culture completion.
Human dorsal root ganglion (hDRG) culture
hDRG culture was performed as described by our lab and others and cultured for up to 11 days in vitro (DIV).32–34 Thoracic hDRG were obtained from an ethically consented, recently deceased organ donor at the University of New Mexico Hospital in coordination with New Mexico Donor Services. Study activities were approved by the Human Research Review Committee at the University of New Mexico Health Sciences Center (Institutional Review Board (IRB) approved study #23-205. Electrophysiological recordings took place between DIV 2 and 5. The donor was a 47-year-old white male, and the cause of death was intracranial hemorrhage.
Whole-cell patch clamp electrophysiology
Whole-cell patch-clamp electrophysiology of cultured mDRG (in vivo and in vitro, respectively) and hDRG was performed as previously described by our group.30,34 Electrophysiological recordings of cultured mDRG neurons of cerulein-treated mice were done without further treatment in the dish. To evaluate the direct effect of cerulein and associated mechanism of action on mDRG and hDRG neurons, in vitro treatment of cultured naïve mouse DRG and hDRG was done with cerulein (0.025 and 0.05 µM), cerulein (25 µM)+CCKA antagonist loxiglumide (330 nM; Cayman Chemical, Cat# 25534), as well as cerulein (25 µM)+CCKB antagonist LY225910 (100 nM) (Tocris Biosciences, Cat# 1018) in culture media for 1 h prior to recording. Recordings were done at room temperature, with the recording chamber perfused with artificial cerebrospinal fluid (aCSF) containing 113 mM NaCl, 3 mM KCl, 25 mM NaHCO3, 1 mM NaH2PO4, 2 mM CaCl2, 2 mM MgCl2, and 11 mM Dextrose bubbled with 95% O2/5% CO2.32,34 Neurons were identified with differential interference contrast optics connected to an IR-2000 digital camera (Dage MTI, Indiana City, MI) or an Olympus digital camera. Cell diameter was measured using Dage MTI camera software or ImageJ (NIH, Bethesda, MD). Current-clamp recordings were performed using a Multiclamp 700B (Molecular Devices, San Jose, CA). Signals were acquired as previously described using a Digidata 1550B converter (Molecular Devices, San Jose, CA) and recorded using Clampex 11 software (Molecular Devices, San Jose, CA). Patch pipettes with 3–7 MΩ electrode resistance were made fresh with a Zeitz puller (Werner Zeitz, Martinsried, Germany) from borosilicate thick glass (GC150F, Sutter Instruments, Novato, CA). Intracellular patch-pipette solution contained 120 mM K-gluconate, 11 mM KCl, 1 mM CaCl2, 2 mM MgCl2, 10 mM HEPES, 11 mM EGTA, 4 mM Mg-ATP. Cells that did not fire APs or had an RMP of >−35 mV were excluded from further analysis. Bridge balance was applied for all recordings.
Electrophysiology analysis methods
Current-clamp recordings started with 25 ms of the cell at rest, followed by 500 ms current pulse increasing in 10 pA increments from −100 pA in increments until they reached inactivation up to 4 nA, and finally 500 ms recovery between each current-clamp step. Electrophysiological analysis was done using Easy Electrophysiology as previously described.32,34 Rheobase is measured at the current injection, eliciting firing while not considering rebound firing or spontaneous activity. Thus, the minimum possible value is 10 pA. Cells were continually stimulated stepwise until they were observed to reach rheobase and then further activated until they reached inactivation (the point where the cell was no longer firing or started having fewer APs after the maximum number of APs), up to 4 nA. Resting membrane potential (RMP) was calculated from the average of 10 sweeps minimum and was not corrected for junction potential. The hyperpolarizing −100 pA step was used to determine input resistance (Rin). The first spike latency (FSL) was determined by measuring the time of the rheobase spike fired after the start of current injection. Cells that fired more than 1 AP during any current injection step at rheobase or higher were considered multi-firing. APs from spontaneous activity were excluded when determining multi-firing cells. Spontaneous activity was assessed with either no current injection or enough current injection to hold the membrane voltage at −45 mV during a 30 s recording. If the neuron had a minimum of one AP during the 30 s, it was recorded as having spontaneous activity. Rebound firing was defined as the neuron firing an AP during the 500 ms recovery in any hyperpolarizing current injection steps. The firing frequency plots were generated by considering only multi-firing cells. The number of APs generated at the current above rheobase was plotted and analyzed in GraphPad Prism v10.0.2 at each current level.
Statistical and data analysis
Easy Electrophysiology v.2.5.1 (London, UK) and Clampfit 11.2 (Molecular Devices, San Jose, CA) were used for the analysis. Statistical analysis was conducted with GraphPad Prism v10.0.2 (Boston, MA). Error bars denote the mean ± standard error of the mean (SEM) unless otherwise specified. A p-value of less than 0.05 was considered statistically significant. Statistical tests are shown in the figure legends.
Results
Intraperitoneal cerulein elicits pain-like behaviors in male and female mice
To establish the cerulein-induced visceral pain model in mice, we performed 6 hourly intraperitoneal injections for 2 days of the cholecystokinin analog cerulein (25 and 50 µg/kg). At baseline, mechanical withdrawal threshold was not different between groups (Figure 1). Cerulein induced secondary mechanical hypersensitivity on the hindpaw. The hourly intraperitoneal injections of cerulein caused mice to develop mechanical hypersensitivity first observed 3 h following the first cerulein injection, which persisted until the last observation on day 2. The force needed to elicit a hindpaw withdrawal response was significantly reduced in 25 µg/kg cerulein (0.13 ± 0.04) and 50 µg/kg cerulein (0.07 ± 0.02) groups, in comparison to naïve control (4.66 ± 0.63; n = 4 mice per group, p < 0.0001, two-way ANOVA followed by Tukey’s multiple comparison test, Figure 1). No significant differences were found between 25 and 50 µg/kg cerulein-treated groups. No significant differences were found between male and female cerulein or naïve groups.

Intraperitoneal cerulein elicits pain-like behavior in male and female mice. Intraperitoneal administration of cerulein elicits hypersensitivity to von Frey stimulation. Analyzed with two-way ANOVA followed by Tukey’s multiple comparison test (n = 4 per group, ****p < 0.0001) compared to control.
In vivo cerulein treatment elicits increased excitability of male and female mouse DRG neurons
Changes in neuronal excitability are typical of sensitization to pain. To determine whether changes in the excitability of DRG neurons innervating the viscera (T8-L2) are changed in the acute cerulein-induced visceral pain model, we performed whole-cell patch-clamp electrophysiology of dissociated T8-L2 DRG neurons from male and female cerulein (25 µg/kg) intraperitoneal-injected mice. Current clamp recordings were obtained from T8-L2 small diameter (<30 µm) DRG neurons. We observed that intrinsic properties, including resting membrane potential, rheobase, input resistance, and first spike latency on rheobase, did not change significantly following cerulein treatment (Figure 2(a)–(d)).

DRG neuronal excitability is increased in cerulein intraperitoneal-injected male and female mice with pain-like behavior. (a) Resting membrane potential (RMP), (b) rheobase, (c) input resistance, and (d) latency of first spike on rheobase were not significantly changed following cerulein (25 µg/kg) treatments (naïve male n = 15 cells, naïve female n = 10 cells, cerulein treated male n = 20 cells, cerulein treated female n = 11 cells, One way ANOVA followed by Tukey’s multiple comparison test). (e) Cell image of mice DRG neuron. Significant difference was found in the percentage of cells that displayed (f) multi-firing property and (g) rebound firing following cerulein treatment (***p < 0.001, **p < 0.01; Fisher’s exact test) compared to control. (h) No significant effect was observed on the spontaneous activity (holding at −45 mV). A significant difference was found in the (i) firing frequency (restricted to multi-firing cells) during the current step protocol (10 pA stepwise increase of 500 ms current injection window) as current injection increased from rheobase (naïve male n = 15 cells, naïve female n = 9 cells, cerulein treated male n = 17 cells and cerulein treated female n = 10 cells, ****p < 0.0001; two-way repeated measures ANOVA followed by Tukey’s multiple comparison test) compared to naïve controls. Inset: Example traces of multi-firing mice DRG neurons current step protocol, naïve male (blue), naïve female (green), cerulein-treated male (red), and cerulein treated female (purple).
We observed a significant reduction in the prevalence of multi- compared to single firing neurons in cerulein versus naïve male mice (85% vs 100% of naïve, ****p < 0.0001, Fisher’s exact test, Figure 2(f)) and a reduction in the prevalence of rebound firing in cerulein versus naïve male mice (p < 0.01, Fisher’s exact test Figure 2(g)). We did not observe any differences in spontaneous activity (Vh = −45 mV; Figure 2(h)). We observed a statistically significant increase in action potential firing frequency in cerulein-injected mice DRG neurons (****p < 0.0001, two-way repeated measures ANOVA followed by Tukey’s multiple comparison test) compared to naïve control mice (Figure 2(i)). Therefore, cerulein injection (compared to vehicle) was sufficient to increase excitability of visceral DRG neurons in male and female mice with underlying pain behaviors.
Cerulein directly acts on mouse DRG neurons to increase excitability
To demonstrate a direct effect of cerulein on nociceptors, we cultured visceral T8-L2 DRG neurons from naïve male mice and treated them either with 0.025 µM and 0.05 µM cerulein, or remain untreated, separately, in culture media for 1 h prior to recording. Doses of cerulein for in vitro use were calculated based on the assumption that 1 mg/kg in vivo is roughly equivalent to 1 µM in vitro as performed in a previous study. 35 Current clamp recordings were obtained from small diameter (<30 µM) neurons. The results showed that the cerulein significantly enhanced the excitability of cultured mouse DRG neurons. We observed a significant change in the resting membrane potential following cerulein 0.05 µM treatment (*p < 0.05; one-way ANOVA followed by Tukey’s multiple comparison test) compared to the control (Figure 3(a)). Rheobase, input resistance, and latency of the first spike on rheobase did not change significantly following cerulein treatments (Figure 3(b)–(d)). In addition, we found a significant difference in the percentage of cells that displayed multi-firing following cerulein treatment (*p < 0.05; Fisher’s exact test) compared to control (Figure 3(f)). No significant effect was observed on the rebound firing (Figure 3(g)). The percentage of cells that displayed spontaneous activity (holding at −45 mV) was significantly decreased following cerulein treatment (*p < 0.05; Fisher’s exact test) compared to control (Figure 3(h)). We found a statistically significant increase in action potential firing frequency in cerulein-treated neurons (****p < 0.0001; two-way repeated measures ANOVA followed by Tukey’s multiple comparison test) compared to control (Figure 3(i)). In summary, direct cerulein application was sufficient to increase the excitability of naïve visceral DRG neurons in a manner consistent with in vivo administration.

Cerulein acts directly on mouse sensory neurons to increase excitability. Cerulein increased excitability of cultured mice DRG neurons treated in vitro prior to recording. There was a significant change in the (a) RMP following cerulein 0.05 treatment (Untreated n = 16 cells, Cerulein (0.025 µM) treated n = 15 cells and Cerulein (0.05 µM) treated n = 12 cells, *p < 0.05; one-way ANOVA followed by Tukey’s multiple comparison test) compared to control. (b) Rheobase, (c) input resistance, and (d) latency of first spike on rheobase not significantly changed following cerulein treatments. (e) Cell image of mice DRG neuron. A significant difference was found in the percentage of cells that displayed (f) multi-firing property following cerulein 0.025 µM treatment (*p < 0.05; Fisher’s exact test) compared to control. No significant effect was observed on the (g) rebound firing. The percentage of cell that displayed (h) spontaneous activity (holding at −45 mV) was significantly lower following cerulein treatment at 0.025 µM, but unchanged at 0.05 µM (**p < 0.01, *p < 0.05; Fisher’s exact test). (i) A significant difference was found in the firing frequency (restricted to multi-firing cells) during the current step protocol (10 pA stepwise increase of 500 ms current injection window) as current injection increased from rheobase (Untreated n = 14 cells, Cerulein (0.025 µM) treated n = 14 cells and Cerulein (0.05 µM) treated n = 11 cells, ****p < 0.0001; two-way repeated measures ANOVA followed by Tukey’s multiple comparison test) compared to control. Inset: Example traces of multi-firing mice DRG neurons current step protocol, untreated blue, cerulein (0.025 µM) red, cerulein (0.05 µM) green.
Loxiglumide reduced cerulein-induced excitability of mouse nociceptors
Our data demonstrates that cultured DRG neurons became hyperexcitable following cerulein administration either in vivo or in vitro. Two CCK receptor subtypes, CCKAR and CCKBR, mediate the actions of cerulein. To determine which of these mediated increased DRG excitability, we treated cultured DRG neurons from naïve male mice with cerulein (0.025 µM) as described, along with either CCKAR antagonist loxiglumide (330 nM) or CCKBR antagonist LY225910 (100 nM), separately, in culture media for 1 h before recording. Current clamp recordings were obtained from small diameter (<30 µm) neurons. The results showed that CCKAR antagonist loxiglumide (330 nM) reduced the cerulein-induced excitability of cultured mouse DRG neurons. Our data demonstrated a significant increase in the rheobase and latency of the first spike on rheobase following loxiglumide treatment (**p < 0.01, *p < 0.05; one-way ANOVA followed by Tukey’s multiple comparison test) compared to the control (Figure 4(a) and (b)). The resting membrane potential did not change significantly following loxiglumide or LY225910 treatment (Figure 4(c)). In addition, we observed a statistically significant decrease in action potential firing frequency in loxiglumide-treated neurons (****p < 0.0001, **p < 0.01; two-way repeated measures ANOVA followed by Tukey’s multiple comparison test) but not in CCKBR antagonist LY225910-treated neurons, compared to control (Figure 4(d)). We found a significant effect on rebound firing following loxiglumide and LY225910 treatment, respectively (****p < 0.0001; Fisher’s exact test) compared to control (Figure 4(e)). The results showed a significant reduction in the percentage of cells that displayed multi-firing following loxiglumide treatment (57% vs 93% of control, ****p < 0.0001; Fisher’s exact test) compared to control (Figure 4(f)). We observed a significantly lower proportion of loxiglumide and LY225910 treated cells (21% and 61%, respectively vs 76% of control, ****p < 0.0001, *p < 0.05; Fisher’s exact test) that exhibited spontaneous activity at a holding potential of −45 mV (Figure 4(g)).
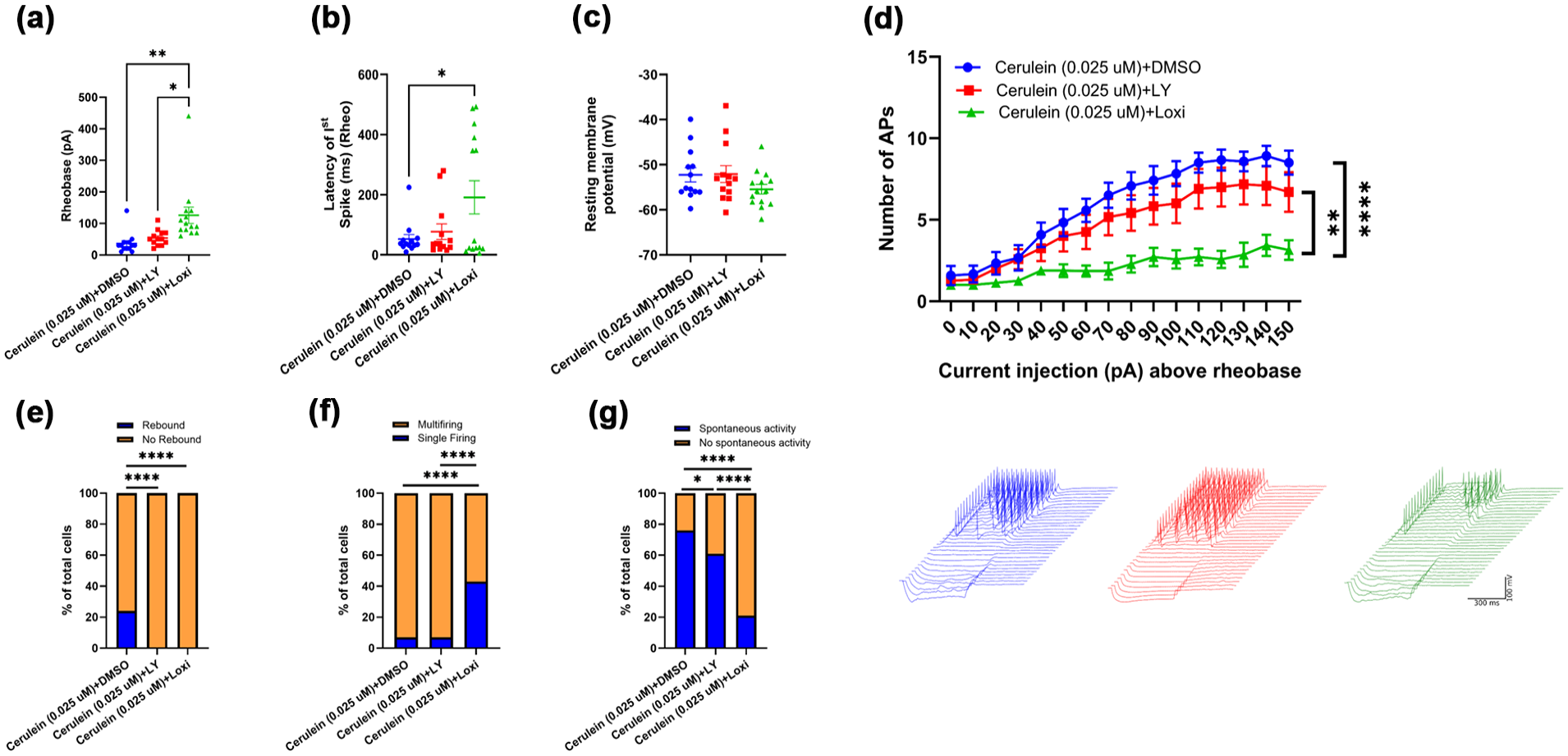
CCK-AR antagonist, loxiglumide, acts directly on mouse sensory neurons to reduce cerulein-induced excitability. Loxiglumide (330 nM) reduced cerulein induced excitability of cultured mice DRG neurons treated in vitro for 1 h prior to recording. There was a significant change in the (a) rheobase, and (b) latency of first spike on rheobase following loxiglumide (330 nM) treatment (Cerulein (0.025 µM)+DMSO treated n = 13 cells, Cerulein (0.025 µM)+LY225910 treated n = 13 cells and Cerulein (0.025 µM)+Loxiglumide treated n = 14 cells, **p < 0.01, *p < 0.05; one-way ANOVA followed by Tukey’s multiple comparison test) compared to control. (c) RMP was not significantly changed following loxiglumide treatment. (d) A significant difference was found in the firing frequency (restricted to multi-firing cells) during the current step protocol (10 pA stepwise increase of 500 ms current injection window) as current injection increased from rheobase (Cerulein (0.025 µM)+DMSO treated n = 12 cells, Cerulein (0.025 µM)+LY225910 treated n = 12 cells and Cerulein (0.025 µM)+Loxiglumide treated n = 8 cells, ****p < 0.0001, **p < 0.01; two-way repeated measures ANOVA followed by Tukey’s multiple comparison test) compared to control. Inset: Example traces of multi-firing DRG neurons current step protocol, cerulein+DMSO (blue), cerulein+LY225910 (red), cerulein+Loxiglumide (green). In addition, following loxiglumide and LY225910 treatment, significant differences were found in the percentage of cells that displayed (e) rebound firing, (f) multi-firing compared to single firing, and (g) spontaneous activity (SA) when held at −45 mV (****p < 0.0001, *p < 0.05; Fisher’s exact test) compared to control.
Cerulein directly acts on human nociceptors to increase excitability, and this effect was reduced by loxiglumide
Having demonstrated that the cultured mouse DRG neurons become hyperexcitable following cerulein treatment and that the CCKAR antagonist loxiglumide reduced the effect, we next tested these findings in cultured human DRG neurons to determine whether these effects could be translatable to human visceral pain. To do so, we treated cultured human thoracic DRG neurons with cerulein (0.025 µM) and loxiglumide (330 nM) in culture media for 1 h prior to recording. Current clamp recordings demonstrated that the loxiglumide (330 nM) reduced the cerulein-induced excitability of cultured human DRG neurons. We observed a significant increase in the rheobase (p = 0.011) following loxiglumide (330 nM) treatment (*p < 0.05; one-way ANOVA followed by Tukey’s multiple comparison test) compared to control (Figure 5(a)). The resting membrane potential, input resistance, number of action potentials, and latency of the first spike at rheobase did not change significantly following loxiglumide treatments (Figure 5(b)–(e)). We found a significant effect on the percentage of cells that displayed multi-firing following loxiglumide treatment (****p < 0.0001; Fisher’s exact test) compared to control (Figure 5(f)). No significant effect was observed on the spontaneous activity (held at −45 mV) and rebound firing following loxiglumide treatment compared to the control (Figure 5(g) and (h)).

Loxiglumide acts directly on human thoracic sensory neurons to reduce cerulein-induced excitability. Loxiglumide (330 nM) reduced cerulein induced excitability of cultured human DRG neurons treated in vitro for 1 h prior to recording. There was a significant increase in the (a) rheobase (minimum current required to elicite firing) following loxiglumide (330 nM) treatment (DMSO treated n = 6 cells, Cerulein (0.025 µM)+DMSO treated n = 6 cells and Cerulein (0.025 µM)+Loxiglumide treated n = 6 cells, *p < 0.05; one-way ANOVA followed by Tukey’s multiple comparison test) compared to control. (b) Resting membrane potential, (c) input resistance, (d) maximal number of action potential elicited above rheobase, and (e) latency of first spike on rheobase were not significantly changed following loxiglumide treatments. Example traces of multi-firing human DRG neurons current step protocol, DMSO (blue), cerulein+DMSO (red), cerulein+Loxiglumide (green). Significant differences were found in the percentage of cells that displayed (f) multi-firing property and (g) spontaneous activity (SA) when held at −45 mV following loxiglumide treatment (****p < 0.0001; Fisher’s exact test) compared to control. (h) No significant effect was observed on the rebound firing. (i) Cell image of human DRG neuron.
Discussion
Our findings investigate the behavioral and electrophysiological modifications in mice induced by intraperitoneal cerulein, a CCK analog, and sensitizer that elicits visceral pain and is used as a commonly used pancreatitis model for pre-clinical studies. We demonstrate that male and female mice injected intraperitoneally with cerulein showed similarly increased excitability of visceral DRG neurons. We also show we can recapitulate this increased excitability by applying cerulein directly to visceral neurons from naïve male mice. We also show that cerulein affects neuronal excitability more prominently through CCKA than CCKB receptors in both mouse and human visceral DRG neurons. Furthermore, we demonstrate that this effect is conserved across species by showing increased excitability of human thoracic DRG neurons following in vitro cerulein application. This study suggests that cerulein mediates visceral pain in mice, and likely in humans, through CCKA receptors on visceral DRG neurons.
The cerulein-induced acute pancreatitis model is reported to produce secondary mechanical hypersensitivity measured on the hindpaws. 6 The behavioral changes and development of mechanical allodynia we observed in cerulein-injected mice are consistent with previous findings. This may be associated with constant peripheral activation of visceral nociceptors which can lead to central sensitization, an increase in ascending pain transmission, and activation of numerous higher pain modulation centers, as well as decreased descending pain inhibition and neurogenic inflammation.36–38 Further, no significant differences were detected between two doses, that is, 25 and 50 µg/kg of cerulein, on mechanical allodynia response, indicating that repeated cerulein administration at a dose of 25 µg/kg, which causes milder pancreatitis than 50 µg/kg, is sufficient for pain model development.
Our findings from current-clamp recordings demonstrated that repeated intraperitoneal cerulein administration results in an excitability increase in an unbiased sample of visceral DRG neurons. The CCK receptor system, a historic target for pain management, is a gastrointestinal and neuropeptide signaling network found throughout the viscera and peripheral and central nervous systems. 10 Increased pancreatic stimulation due to loss of feedback inhibition of CCK has been proposed to contribute to the pain of some patients with chronic pancreatitis. 39
To confirm that CCK could have direct effects on mouse neurons and human neurons, we investigated effects on excitability of visceral DRG neurons from naïve mice and human donor neurons treated in vitro with cerulein. The electrophysiological recordings of visceral DRG neurons from naïve mice treated in vitro with cerulein (0.025/0.05 µM) indicate increased excitability. These findings are supported by previous studies.30,40–42 Similar results were observed in dissociated human thoracic DRG neurons treated in vitro with cerulein (0.025 µM). Previous studies have reported that intravenous administration of CCK octapeptide increases rectal hypersensitivity in humans, possibly underlying irritable bowel syndrome.43,44 Nociceptive signaling relies on various ion channels in sensory neurons that regulate nociceptive pain by modulating neuronal excitability. In neuronal physiology, CCK receptor signaling leads to the activation of many downstream targets, many of which are ion channels. Voltage-gated ion channels play a fundamental role in controlling neuronal excitability. In nociceptive small-diameter DRG neurons, cerulein decreased A-type K+ currents and increased neuronal excitability, but did not affect delayed-rectifier K+ currents. 42 CCK receptor activation by cerulein binding has been shown to increase cytosolic Ca2+ via IP3 receptors and other Ca2+ channels, which are involved in increasing neuronal excitability in DRG neurons.45–47 Our results indicate that different ion channels regulating excitability may be involved in the action of cerulein in mouse and human visceral DRG neurons. The actions of cerulein on mouse visceral DRG neurons were mainly on frequency-current relationships, whereas those on human visceral DRG neurons were mainly on rheobase. These suggest slightly different mechanisms in human and mouse DRG neurons. However, the clinical history of the donor could have influenced these results. Nevertheless, a more detailed study of the role of CCK in human DRG neurons in naïve and pain states is warranted for future work.
We further investigated the mechanisms of cerulein by using CCKA/CCKB receptor antagonists. Our patch-clamp electrophysiology data showed that in vitro treatment with CCKA, but not CCKB, receptor antagonists significantly attenuated the excitability of mouse and human visceral DRG neurons sensitized in vitro with cerulein. There is substantial evidence of CCKA receptor involvement in various visceral pain models.48–51 This effect may be correlated with CCKA receptor expression in the primary afferent neurons, which mediate activation of NMDA receptors, possibly via NMDA receptor phosphorylation, and a greater Ca2+ response to cerulein.51–53 The use of pharmacologic blockers of CCKA/B receptors may be problematic as non-specific actions could be an issue such as those observed on rebound firing that cerulein itself did not affect, which implicates actions on T-type voltage-gated calcium channels 54 . Future studies may require CCK receptor knockout techniques in specific sensory neurons subtypes. In addition, labeling of pancreas-innervating afferents using a tracer or genetic approaches would aid in the understanding of these results as it relates to pancreatitis per se.
Though many features of acute and chronic cerulein-induced pancreatitis appear to be shared with human disease, whether CCK has a role in clinical pancreatitis has been a topic of discussion. In rodent models, including those not induced by cerulein such as the CDE diet and ductal injection of taurocholate in mice, have shown that CCK receptor antagonists reduce the severity of experimental pancreatitis.55,56 A similar reduction in acute pancreatitis injury was observed when the CCK-A receptor was genetically eliminated in rats. The role of CCK in humans remains unclear. Although CCK blood levels can be elevated in gallstone pancreatitis, 57 this response does not occur with other causes of acute pancreatitis, such as alcohol. The role of CCK in chronic pancreatitis has also been discussed. 55 Though some have reported that CCK levels are increased, others have not. These differences may be dependent on the stage of the disease. In addition to possible roles in pancreatic injury, CCK’s role in the pancreatitis pain response are seen in experimental models such as ours and supported by at least one clinical study. Our finding suggests that in addition to inflammatory pathways, CCK-stimulated pathways, including those on DRG neurons, contribute to pancreatitis pain. One implication of our studies is the short-term use of CCK-A receptor antagonists for reducing acute pancreatitis pain should be reconsidered.
This study highlights the need to investigate electrophysiological changes associated with nociceptors innervating the viscera, thereby elucidating the mechanisms of visceral pain. Future studies should build upon these characterizations and define the cholecystokinin system as a significant component of visceral pain disorders including pancreatitis.
Footnotes
Acknowledgements
We extend our deepest gratitude to the organ donors and their families, whose generous contributions made this study possible.
Abbreviations
m/hDRG: mouse or human dorsal root ganglia; CCK: cholecystokinin; CCKA/BR: cholecystokinin A or B receptor; RMP: resting membrane potential; Rin: input resistance; FSL: first spike latency.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: G.V.D. is a named inventor of patents issued related to renalase’s discovery and therapeutic use. Renalase is licensed to Bessor Pharma, and Gary V. Desir holds an equity position in Bessor and its subsidiary, Personal Therapeutics. None of the other authors has any conflicts of interest, financial or otherwise, to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We acknowledge funding from the Department of Defense Peer Reviewed Medical Research Programs, Investigator-Initiated Research Award (Partnering PI Option), Award #HT9425-23-1-0329/0330 (FG, KNW, SRAA, GVD), Award #HT9425-23-1-0329/0330 and VA Merit Award (BX003250) to FG, and the Research Endowment Fund of the Department of Anesthesiology and Critical Care Medicine, University of New Mexico Health Sciences Center.
