Abstract
Transcriptional changes in primary sensory neurons are involved in initiation and maintenance of neuropathic pain. However, the transcription factors in primary sensory neurons responsible for neuropathic pain are not fully understood. Dorsal Root Ganglia Homeobox (DRGX) is a paired-like homeodomain transcription factor necessary for the development of nociceptive primary sensory neurons during the early postnatal period. However, roles for DRGX after development are largely unknown. Here, we report that DRGX downregulation in primary sensory neurons as a result of post-developmental nerve injury contributes to neuropathic pain in rats. DRGX expression was decreased in nuclei of small and medium primary sensory neurons after spinal nerve ligation. DRGX downregulation by transduction of a short hairpin RNA with an adeno-associated viral vector induced mechanical allodynia and thermal hyperalgesia. In contrast, DRGX overexpression in primary sensory neurons suppressed neuropathic pain. DRGX regulated matrix metalloproteinase-9 (MMP-9) and prostaglandin E receptor 2 mRNA expression in the DRG. MMP-9 inhibitor attenuated DRGX downregulation-induced pain. These results suggest that DRGX downregulation after development contributes to neuropathic pain through transcriptional modulation of pain-related genes in primary sensory neurons.
Keywords
Introduction
Neuropathic pain often has a chronic course and therefore can seriously impair quality of life along with associated problems such as a loss of function, anxiety, depression, disturbed sleep, and impaired cognition. 1 However, the pharmacotherapy of neuropathic pain remains unsatisfactory because of limited efficacy and severe adverse effects. 2 Neuropathic pain arises from a lesion or disease of the somatosensory system. 3 Functional changes in primary sensory neurons, a major origin of neuropathic pain, play a substantial role in initiation and maintenance of neuropathic pain. Genetic studies have shown that profound transcriptional changes occur in primary sensory neurons under neuropathic pain.4–7 However, the transcription factors responsible for neuropathic pain are not yet fully understood in primary sensory neurons.
Dorsal Root Ganglia Homeobox (DRGX) is a paired-like homeodomain transcription factor that represses or enhances specific gene transcription depending on cell context.8–10 DRGX is expressed in several areas of the peripheral and central nervous systems during embryonic development and is required for development of the nociceptive system during embryogenesis.11,12 DRGX-knockout mice exhibit extensive death of nociceptive primary sensory neurons.11,13 In the superficial dorsal spinal cord, a reduction in the number of glutamatergic neurons and noxious stimulation-induced Fos-immunoreactive neurons was observed in DRGX-knockout mice. 14 Consistent with these results, DRGX-knockout mice have reduced responses to a broad range of noxious stimuli. 11 After birth, DRGX expression becomes progressively restricted to primary sensory neurons and superficial dorsal spinal cord neurons,15,16 as is the case with a number of homeobox genes.17–20 In general, homeobox genes show dynamic expression changes over the course of development and then exhibit different functions in the adult. 21 However, roles for DRGX after development remain largely unknown in nociceptive primary sensory neurons.
In DRGX-knockout mice, enhanced memory performance and front-hippocampal connectivity were observed in association with expression changes in several genes including matrix metalloproteinase-9 (MMP-9) and prostaglandin E receptor 2 (EP2) mRNA expression in the medial prefrontal cortex, amygdala, and/or hippocampus. 22 MMP-9 and EP2 in the primary sensory neurons were involved in pathological pain. MMP-9 becomes upregulated in primary sensory neurons in the first several days after nerve injury, and MMP-9-null mice exhibit reduced nerve injury-induced mechanical allodynia. 23 MMP-9 in the DRG is also involved in paclitaxel-induced neuropathic pain. 24 EP2 is also increased in primary sensory neurons after nerve injury, and blockade of this receptor relieves pain after nerve injury.25,26
In this study, we explored the function of DRGX in primary sensory neurons after development by regulating gene expression with an adeno-associated viral (AAV) vector and found that DRGX downregulation induced neuropathic pain, possibly through transcriptional regulation.
Materials and methods
Animal models
Sprague-Dawley male rats (six weeks old) were used for all experiments. All experiments were carried out in agreement with the Animal Experiments Ethical Review Committee and approved by the President of Nippon Medical School (Approval number 27–037). All rats had free access to food and water and were allowed to live individually under optimal conditions. For surgery, all rats were subjected to deep anesthesia with isoflurane inhalation (2%–3%). The neuropathic pain model was induced by spinal nerve ligation (SNL), as previously described. 27 Briefly, the left (ipsilateral) lumbar fifth (L5) spinal nerve was exposed and tightly ligated with 4–0 silk thread at two sites separated by about 1 mm. For induction of tissue inflammation, 100 µl of complete Freund’s adjuvant (CFA; Merck GKaA, Darmstadt, Germany) was injected into the left plantar skin of the hind paw. The right (contralateral) side was left intact for a control.
Behavioral tests
Paw withdrawal threshold and latency in hindlimbs in response to mechanical and thermal stimuli, respectively, were measured before and 1, 4, 7, and 14 days after SNL. To evaluate mechanical allodynia, we used the von Frey test (Muromachi Kikai, Tokyo, Japan). 28 For acclimation, rats were placed individually in plastic enclosures with a mesh floor at least 30 min before testing. Next, a von Frey filament was applied vertically to the planter surface of the hindpaw until the filament slightly bent. The weakest force (g) inducing hindpaw withdrawal at least three times in five trials was determined to be the paw withdrawal threshold. To evaluate thermal hyperalgesia, the Plantar test (Ugo Basile, Comerio, Italy) was used. Briefly, rats were placed individually in enclosures with a floor for acclimation. Next, a radiant heat generator placed under the glass floor was positioned directly beneath the hind paw and stimulated twice with at least a 5-min interval. The mean value of the two trials was referred to as the paw withdrawal latency. Both tests were performed in a blind fashion.
Quantitative reverse transcription-polymerase chain reaction
The L5 DRG was removed, frozen in liquid nitrogen, and stored at −80°C until RNA purification. For RNA purification, total RNA was extracted from L5 DRGs on the injured left and intact right sides after SNL using RNAiso Plus (Takara Bio, Shiga, Japan). Total RNA (500 ng) was reverse-transcribed with a random primer using an iScript Select cDNA Synthesis kit (Bio-Rad, Hercules, CA). Purity and concentration of extracted RNAs were measured with a NanoDrop One Spectrophotometer (Thermo Fisher Scientific, Waltham, MA). Quantitative polymerase chain reaction (PCR) analysis was performed using Power SYBR Green PCR Master Mix (Thermo Fisher Scientific) on a StepOnePlus Real-time PCR System (Thermo Fisher Scientific). The PCR program was initiated by 95°C for 10 min, followed by 40 cycles consisting of 95°C for 15 s and 60°C for 1 min. Primer pairs for DRGX, MMP-9, EP2, B-cell lymphoma-2 (Bcl-2), bcl-2 associated X protein (Bax), and caspase-3 were designed using Primer blast (https://www.nih.gov/) as the following sequences: DRGX (forward, 5′-
In situ hybridization
To produce an in situ hybridization probe for DRGX, a fragment of the DRGX nucleotide sequence was amplified from rat DRG-derived cDNA using forward (5′-
Rats were transcardially perfused with phosphate-buffered saline (PBS) followed by 4% paraformaldehyde in PBS. L5 DRGs were excised, post-fixed in the same fixative overnight at 4°C, and cryoprotected in 20% sucrose in PBS overnight at 4°C. Tissues were rapidly frozen in dry ice/acetone and sectioned at a 10-µm thickness using a cryostat (Leica Microsystems, Wetzlar, Germany). Sections were treated with 1 µg/ml proteinase K for 5 min. After incubation in 4% paraformaldehyde/PBS for 20 min, sections were hybridized with the digoxigenin-labeled RNA probe in hybridization buffer (50% formamide, 5 × saline-sodium citrate (SSC) pH 4.5, 1% sodium dodecyl sulfate (SDS), 50 µg/ml heparin sodium, and 50 µg/ml yeast RNA) at 65°C overnight. Sections were washed with a first wash buffer (50% formamide, 5 × SSC pH 4.5, and 1% SDS) at 65°C for 30 min and then three times with a second wash buffer (50% formamide and 2 × SSC pH 4.5) at 65°C for 30 min. Subsequently, sections were incubated with an alkaline phosphatase-conjugated anti-digoxigenin antibody (1:1000; Roche Diagnostics) at 4°C overnight, followed by staining with BM-purple (Roche Diagnostics) at room temperature for five days. The sense probe did not produce any signal in intact L5 DRGs, verifying sequence-specific staining (data not shown). Images were captured using a high-resolution microscope equipped with a computer (Olympus, Tokyo, Japan). To measure cell sizes of primary sensory neurons, six DRG sections (60-µm interval) obtained from individual rats were analyzed. The cell area was calculated using ImageJ software (version 1.52; National Institutes of Health, Bethesda, MD) from the manually drawn outline of primary sensory neurons.
Immunofluorescence
For generation of a polyclonal anti-DRGX antibody (custom-made by Merck GKaA), two rabbits were used for antibody production by immunization with a purified recombinant DRGX protein conjugated to keyhole limpet hemocyanin. Pre-immune sera were collected from rabbits before protein injections and pooled. Antibody production was initiated by subcutaneous injection of recombinant protein and boosted three times at two-week intervals using the same protein dosage. After the fourth immunization, antibody production and specificity were tested using an enzyme-linked immunosorbent assay (data not shown). The antibody was generated against rat DRGX, corresponding to amino acids 92 to 110 (CERGASDQEPGAKEPMAEVT, excluding the homeobox domain).
L5 DRG sections were pre-incubated in PBS containing 5% normal donkey serum and 0.3% Triton X-100 for 30 min, followed by incubation with a rabbit anti-DRGX antibody (1:1000) at 4°C overnight. Sections were washed in PBS and then incubated with a secondary antibody labeled with Alexa Fluor 488 (1:1000; Thermo Fisher Scientific) or Alexa Fluor 594 (1:1000; Thermo Fisher Scientific) at room temperature for 1 h. Fluorescent images were captured using a high-resolution digital camera equipped with a computer (Olympus). Specificity of the polyclonal anti-DRGX antibody was assessed using an antibody-absorption test. DRGX antiserum (1 ml in 1:1000 dilution) was incubated overnight at 4°C with 10 µg of DRGX antigen peptide (CERGASDQEPGAKEPMAEVT), and then processed for immunofluorescence, as described above. Immunoreactivity of DRGX antiserum in L5 DRG was abolished by pre-absorption of the antiserum with the antigen peptide (Supplemental Figure 1(a)) and was decreased by DRGX shRNA (Supplemental Figure 1(b)), indicating specific staining with the DRGX antiserum.
Adeno-associated virus vector production
pAAV plasmid encoding an enhanced green fluorescent protein (EGFP) expression cassette was produced by replacing the bGH pA sequence in the pAAV-MCS plasmid (Agilent Technologies, Santa Clara, CA) with the B19 promoter and EGFP gene and linearized by PCR. To express DRGX, the coding sequence of DRGX was amplified from rat DRG-derived cDNA using forward (5′-
Recombinant serotype 6 AAV vector was generated by transfection using an adenovirus-free triple transfection method. pAAV, AAV packaging (pRepCap 6as), 30 and helper (pHelper; Takara Bio) plasmids were co-transfected into 293EB cells at a ratio of 1:1:1 using polyethylenimine. Culture medium was collected five days after transfection and cell debris was removed by centrifugation at 3000 r/min for 20 min at 4°C. AAV vector was purified using a cesium chloride density-gradient centrifugation at 30,000 r/min for 2.5 h at 16°C. After dialysis with a Slide-A-Lyzer G2 dialysis cassette (Thermo Fisher Scientific), the AAV vector was concentrated using an Amicon Ultra-4 30 K filter (Millipore). Genomic titers of each AAV vector were determined by quantitative PCR. For use, each AAV vector was diluted with PBS to ∼5 × 1013 vector genomes (vg)/ml. AAV vectors (4 µl) were slowly injected into L5 DRG using a microsyringe with a 27-gauge needle after behavioral tests.
Intrathecal drug administration
Intrathecal catheter for drug administration was implanted in rats three days before AAV injection, as previously described. 31 Briefly, polyethylene catheter (PE-10) filled with saline was slowly inserted into the spinal subarachnoid space from cisterna magna to the level of the lumbar enlargement of the spinal cord. Rats without obvious movement disturbances, such as paralysis, were used for further experiments. Seven days after AAV injection, 10 µl of MMP-9 inhibitor or vehicle followed by 10 µl of saline (flush) was intrathecally administered. MMP-9 inhibitor (Abcam plc, Cambridge, UK) was dissolved in in a sterile normal saline containing 20% dimethyl sulfoxide at a concentration of 1 µg/µl.
Statistical analysis
Values are expressed as mean ± standard error of the mean (SEM). SPSS software (IBM, Armonk, NY) was used for statistical analyses. Normality of data was assessed by Shapiro–Wilk test. The paired t-test was used for normally distributed data sets. If normal distribution was not assumed, the Mann–Whitney U test was performed. Equality of variance was assessed by Levene’s test. Welch’s or unpaired t-test was used if homoscedasticity was rejected or not, respectively. For multiple comparisons, normally distributed data sets were analyzed by two-way repeated-measures ANOVAs followed by Bonferroni tests. For non-normally distributed data sets, Mann–Whitney U test with Bonferroni correction was performed. P < 0.05 was considered statistically significant.
Results
DRGX was decreased in injured DRG in neuropathic pain
Paw withdrawal threshold and latency in response to mechanical and thermal stimuli, respectively, were significantly decreased for at least 14 days after SNL (Figure 1(a) and (b)), indicating the rats had developed neuropathic pain. Expression of DRGX mRNA was significantly reduced in the injured L5 DRG from day 4 after SNL (Figure 1(c)). In contrast, DRGX mRNA expression was not changed in the L5 DRG seven days after CFA injection (Supplemental Figure 2), suggesting that DRGX reduction was specifically caused by nerve injury.

Reduced DRGX expression in primary sensory neurons following SNL. (a) and (b) Mechanical allodynia (a) and thermal hyperalgesia (b) were examined on contralateral and ipsilateral sides of rats after SNL (n = 6; *P < 0.05, and ***P < 0.001, vs. value on contralateral side, by Mann–Whitney U test with Bonferroni correction for mechanical allodynia and two-way repeated-measures ANOVA followed by paired t-test with Bonferroni correction for thermal hyperalgesia). (c) Time course of changes in DRGX mRNA expression in L5 DRG after SNL surgery (n = 5–6; *P < 0.05, vs. value on contralateral side by Mann–Whitney U test with Bonferroni correction). All data represent mean ± SEM. DRG: dorsal root ganglion; DRGX: Dorsal Root Ganglia Homeobox; L5: lumbar fifth; SNL: spinal nerve ligation.
DRGX was decreased in small DRG neurons in neuropathic pain
To investigate cell types expressing DRGX, in situ hybridization for DRGX was first performed in contralateral L5 DRG. DRGX mRNA expression was mainly observed in small to medium primary sensory neurons, most of which were nociceptive neurons (Figure 2(a) and (b)). The percentage of DRGX mRNA-positive neurons was significantly decreased 14 days after SNL (79.4% ± 4.27% for contralateral L5 DRG and 28.2% ± 3.13% for ipsilateral L5 DRG; t-test, P < 0.0001; n = 6 rats per group). DRGX mRNA expression was decreased in both small and medium primary sensory neurons, but not in large neurons (Figure 2(a) and (b)). Similarly, DRGX protein was mainly expressed in small to medium primary sensory neurons in contralateral DRG, whereby it was primarily localized in the nucleus, consistent with its known nuclear function (Figure 2(c) and (d)). After SNL, total DRGX protein-positive neurons were also decreased (75.5% ± 4.65% for contralateral L5 DRG and 28.6% ± 2.79% for ipsilateral L5 DRG; t-test, P < 0.0001; n = 5 rats per group). DRGX protein was mainly decreased in small to medium primary sensory neurons, consistent with in situ hybridization (Figure 2(b) and (d)).

Nuclear expression of DRGX protein in small to medium primary sensory neurons was decreased after SNL. (a) and (c) Representative images of in situ hybridization (a) or immunofluorescence (c) for DRGX mRNA and protein expression, respectively, in L5 DRG from contralateral and ipsilateral sides 14 days after SNL surgery. Scale bar = 100 µm. (b) and (d) Size distribution of DRGX-positive neurons in L5 DRG on contralateral and ipsilateral sides was assessed by in situ hybridization (b) or immunofluorescence (d) (n = 5–6). All data represent mean ± SEM. DRG: dorsal root ganglion; DRGX: Dorsal Root Ganglia Homeobox; L5: lumbar fifth; SNL: spinal nerve ligation.
DRGX overexpression suppressed neuropathic pain
To assess a potential role for DRGX in pain, we knocked down DRGX expression specifically in L5 DRG neurons using an AAV vector expressing both shRNA against DRGX and EGFP, an expression marker. Seven days after direct microinjection of AAV vector into intact L5 DRG, EGFP expression was observed in L5 primary sensory neurons of all cell sizes (Figure 3(a)), consistent with previous reports. 32 DRGX expression was significantly decreased compared with control scramble shRNA (Figure 3(b)). DRGX downregulation decreased both paw withdrawal threshold and latency in response to mechanical and thermal stimuli, respectively (Figure 3(c)). Next, to investigate the involvement of DRGX downregulation in neuropathic pain, DRGX was specifically overexpressed in L5 DRG neurons in neuropathic pain. Seven days after AAV-DRGX injection, DRGX expression was markedly increased compared with the control AAV (Figure 3(d)). Nociceptive responses were unaffected by DRGX induction itself seven days after AAV-DRGX vector infection (Figure 3(e)). DRGX overexpression continuously alleviated mechanical allodynia and thermal hyperalgesia from day 4 after SNL, but not at day 1 after SNL (Figure 3(e)), indicating that DRGX downregulation played an important role in the maintenance of neuropathic pain.
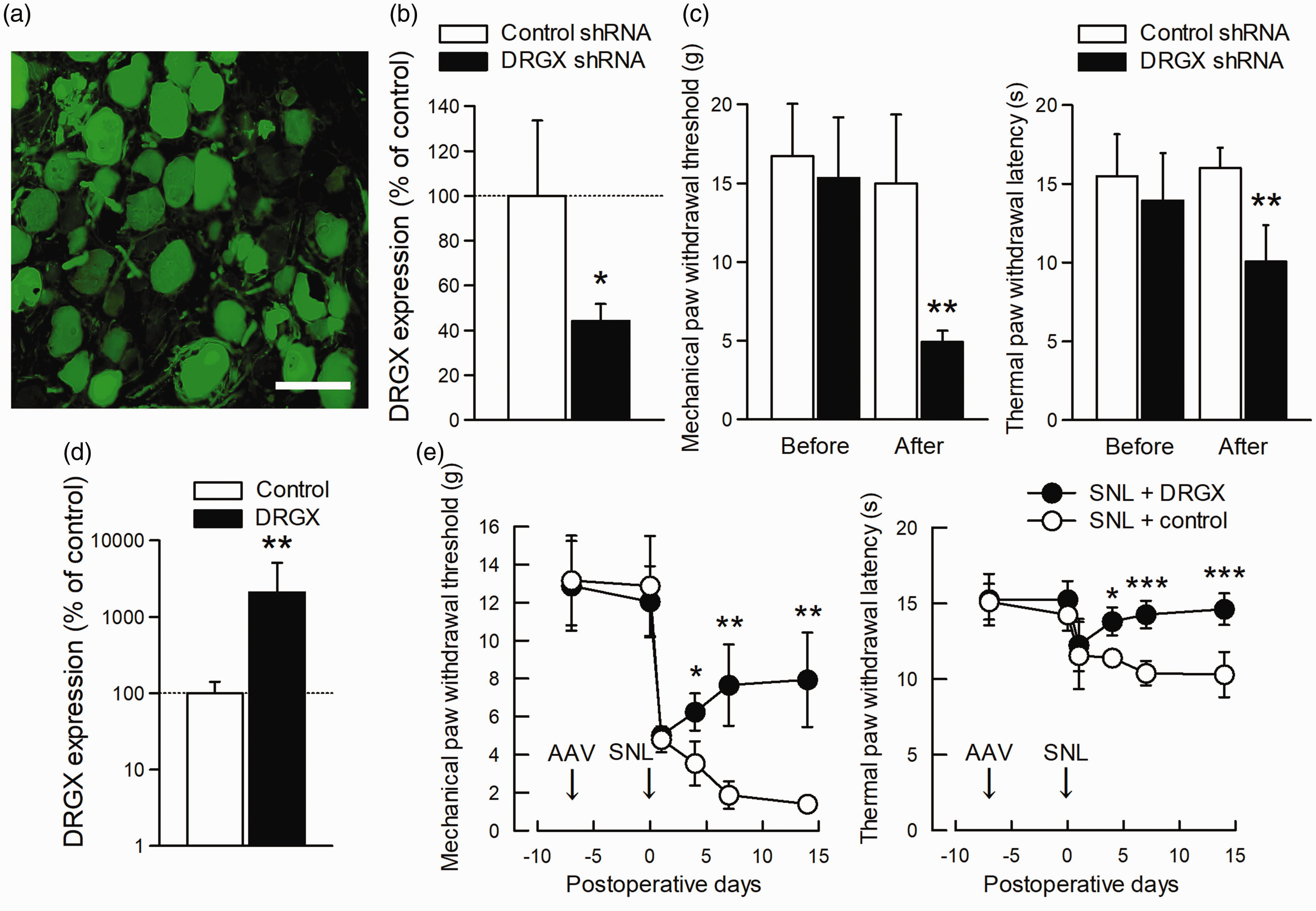
DRGX alleviated neuropathic pain. (a) Representative image of EGFP immunofluorescence in the L5 DRG seven days after control AAV vector injection. Scale bar = 100 µm. (b) DRGX mRNA expression levels in the L5 DRG seven days after injection of a control or DRGX shRNA (n = 6; *P < 0.05, vs. value of AAV injection encoding control scramble shRNA by unpaired t-test). (c) Paw withdrawal response to mechanical or thermal stimuli was evaluated on AAV-injected sides. Behavioral tests were performed before and seven days after injection of an AAV vector encoding control or DRGX shRNA into the L5 DRG of intact rats (n = 6; **P < 0.01, vs. value of AAV injection encoding control shRNA, by Mann–Whitney U test for mechanical allodynia and unpaired t-test for thermal hyperalgesia). (d) DRGX expression levels in the L5 DRG seven days after injection of AAV-control or AAV-DRGX vectors (n = 6; **P < 0.01, vs. value of AAV-control injection by Welch’s t-test). The vertical axis is shown on a logarithmic scale. (e) Paw withdrawal response to mechanical or thermal stimuli was evaluated on the ipsilateral side. AAV-control or AAV-DRGX vector was injected seven days before SNL (n = 6; *P < 0.05, **P < 0.01, and ***P < 0.001, vs. value of AAV-control injection, by Mann–Whitney U test with Bonferroni correction for mechanical allodynia and two-way repeated-measures ANOVA followed by paired t-test with Bonferroni correction for thermal hyperalgesia). All data represent mean ± SEM. AAV: adeno-associated viral vector; DRG: dorsal root ganglion; DRGX: Dorsal Root Ganglia Homeobox; EGFP: enhanced green fluorescent protein; L5: lumbar fifth; shRNA: short hairpin RNA; SNL: spinal nerve ligation.
DRGX modulated MMP-9 expression in DRG under a neuropathic pain condition
To explore pain-relevant target genes of the homeobox transcription factor DRGX, we assessed genes whose expression was reportedly changed in the forebrain of DRGX-knockout mice. 22 Among them, we focused on MMP-9 and EP2, both of which were increased in DRGX-null forebrain and is involved in neuropathic pain.23,25,26 Expression levels of MMP-9 and EP2 were increased in L5 DRG after SNL (Figure 4(a) and (b)). DRGX knockdown by shRNA also increased MMP-9 and EP2 expression in L5 DRG (Figure 4(c) and (d)). DRGX overexpression suppressed SNL-induced MMP-9 upregulation (Figure 4(e)), but not EP2 upregulation (Figure 4(f)), in L5 DRG of rats. On the other hand, an anti-apoptotic Bcl-2 was increased by DRGX knockdown, while a pro-apoptotic Bax and an effector caspase of apoptosis, caspase-3, were unchanged and increased, respectively (Supplemental Figure 3).

DRGX suppressed MMP-9 upregulation by SNL in the L5 DRG. (a) and (b) Expression level of MMP-9 (a) or EP2 (b) mRNA in the L5 DRG of contralateral and ipsilateral sides 14 days after SNL (n = 5; *P < 0.05 and **P < 0.01, vs. value on intact side by paired t-test). (c) and (d) Expression level of MMP-9 (c) or EP2 (d) mRNA in the L5 DRG seven days after injection of a control or DRGX shRNA AAV vector (n = 6; **P < 0.01, by unpaired t-test). (e) and (f) Expression level of MMP-9 (e) or EP2 (f) mRNA in the L5 DRG fourteen days after SNL. AAV-control or AAV-DRGX vector was injected seven days before SNL (n = 6; *P < 0.05, by Mann–Whitney U test). Values are expressed as percentages of values for the contralateral DRG as baseline. All data are mean ± SEM. AAV: adeno-associated viral vector; DRG: dorsal root ganglion; DRGX: Dorsal Root Ganglia Homeobox; EP2: prostaglandin E receptor 2; L5: lumbar fifth; MMP-9: matrix metalloproteinase-9; SNL: spinal nerve ligation.
To further explore the MMP-9 involvement in DRGX-mediated nociceptive modulation, MMP-9 inhibitor was intrathecally administered to rats with DRGX knockdown by shRNA. After MMP-9 inhibitor administration, mechanical allodynia and thermal hyperalgesia were attenuated in rats injected with AAV vector expressing DRGX shRNA (Figure 5).

MMP-9 inhibitor alleviated mechanical allodynia and thermal hyperalgesia induced by DRGX knockdown. Paw withdrawal response to mechanical or thermal stimuli was evaluated on the AAV-injected side. Behavioral tests were performed before and seven days after injection of an AAV vector encoding DRGX shRNA into the L5 DRG of intact rats and 1, 3, and 5 h after intrathecal administration of MMP-9 inhibitor or vehicle (n = 5; *P < 0.05, and **P < 0.01, vs. value on vehicle administration, by Mann–Whitney U test with Bonferroni correction for mechanical allodynia and two-way repeated-measures ANOVA followed by unpaired t-test with Bonferroni correction for thermal hyperalgesia). All data represent mean ± SEM. AAV: adeno-associated viral vector; DRG: dorsal root ganglion; DRGX: Dorsal Root Ganglia Homeobox; i.t.: intrathecal administration; MMP-9: matrix metalloproteinase-9.
Discussion
This study first demonstrates that DRGX, a critical transcription factor for the development of nociceptive primary sensory neurons, has a significant role in normal nociception of adult rats. Moreover, DRGX downregulation in primary sensory neurons led to mechanical allodynia and thermal hyperalgesia and was involved in the maintenance of neuropathic pain. Restoration of DRGX expression alleviated neuropathic pain in association with blockade of MMP-9 upregulation.
DRGX modulated nociceptive responses in primary sensory neurons most likely through transcriptional regulation of pain-related genes. Both DRGX mRNA and protein were mainly expressed and significantly downregulated after nerve injury in small-to-medium primary sensory neurons, most of which are thought to be nociceptive neurons. DRGX protein was distributed in the nuclei of primary sensory neurons, consistent with its role in transcriptional regulation. However, pain-related genes modulated by DRGX, a transcription factor known to repress9,10 or enhance 8 transcriptional activity in a cell-context dependent manner, remain poorly understood, while mechanisms modulating DRGX expression (Phox2b, PRDM12, and Tlx3) or activity (phosphorylation though Tlx3 and conformational change by prolyl isomerase PIN1) have been reported. However, in the medial prefrontal cortex, amygdala, or hippocampus of DRGX-null mice, mRNA levels of dozens of genes were reportedly changed. 22 Therefore, DRGX downregulation-induced pain was most likely mediated by transcriptional regulation of pain-related genes. Consistent with this idea, DRGX downregulation induced upregulation of MMP-9 and EP2 in the DRG, as well as hyperalgesia. Furthermore, DRGX overexpression counteracted the neuropathic pain and MMP-9 upregulation induced by SNL, while hyperalgesia induced by DRGX downregulation was attenuated by MMP-9 inhibitor. In contrast, EP2 upregulation after SNL was not suppressed by DRGX overexpression, suggesting that other mechanisms such as a cAMP/PKA signaling pathway 33 dominantly mediated the EP2 upregulation. MMP-9 and EP2 were previously reported to be increased in small and medium DRG neurons after nerve injury.23,24,26 MMPs are widely involved in inflammation and tissue remodeling by cleaving extracellular matrix proteins, cytokines, and chemokines.34,35 Among them, MMP-9 upregulation in primary sensory neurons upon nerve injury leads to spinal microglial activation through pro-interleukin-1β cleavage, contributing to neuropathic pain. 23 Similarly, EP2, a receptor for prostaglandin E2, is well known to be involved in neuropathic 33 and inflammatory36,37 pain through the cAMP/PKA signaling pathway. 26 However, impact of DRGX downregulation on neuronal activity remains unknown, although functional connectivity between medial prefrontal cortex and dorsal hippocampus during behavioral tasks were increased in DRGX knockout mice. 22 Overall, DRGX likely modulated pain behaviors through transcriptional modulation of pain-related genes.
Roles for DRGX in primary sensory neurons seem to change throughout cell development. Functional significance of DRGX in adult primary sensory neurons has remained speculative, although DRGX was well-characterized as a crucial transcription factor for establishment and maintenance of DRG to dorsal spinal cord circuitry during embryonic development. DRGX-null mutant mice reportedly show a marked loss of small-diameter primary sensory neurons and glutamatergic neurons in the superficial spinal dorsal horn.13,14 Accordingly, DRGX deficiency during development exhibits congenital hypoalgesia in adult mice. However, primary sensory neurons rarely die after peripheral nerve injury in rats following development, 38 despite DRGX downregulation. Consistent with this, a pro-apoptotic Bax and an anti-apoptotic Bcl-2 were unchanged and increased rather than decreased in the adult rat DRG after DRGX knockdown in this study. On the other hand, an effector caspase of apoptosis, caspase-3, which is also involved in neuronal plasticity apart from apoptosis, 39 was increased. Therefore, apoptosis or its associated signaling pathway may have a potential role in DRGX function in pain modulation. Differences in functional significance during and after development are commonly observed, especially for neurotrophic factors. 40 Notably, mice with a null mutation for nerve growth factor (NGF), a key neurotrophic factor for nociceptive primary sensory neurons during development, 41 showed cell death of nociceptive primary sensory neurons during development and resultant diminished responsiveness to pain. 42 However, after development, NGF could reportedly attenuate neuropathic pain 43 or produce hyperalgesia in naïve animals, 44 indicating a developmental change in cellular functions.
In conclusion, we demonstrated the importance of DRGX for normal nociception in adult rats. Moreover, DRGX downregulation in primary sensory neurons was involved in the maintenance of neuropathic pain after nerve injury, possibly through modulation of MMP-9 expression. Analysis of roles for DRGX in primary sensory neurons after development will provide further insights into transcriptional regulation of nociception and neuropathic pain.
Supplemental Material
MPX904462 Supplemental material1 - Supplemental material for Dorsal Root Ganglia Homeobox downregulation in primary sensory neurons contributes to neuropathic pain in rats
Supplemental material, MPX904462 Supplemental material1 for Dorsal Root Ganglia Homeobox downregulation in primary sensory neurons contributes to neuropathic pain in rats by Takaya Ito, Atsushi Sakai, Motoyo Maruyama, Yoshitaka Miyagawa, Takashi Okada, Haruhisa Fukayama and Hidenori Suzuki in Molecular Pain
Supplemental Material
MPX904462 Supplemental material2 - Supplemental material for Dorsal Root Ganglia Homeobox downregulation in primary sensory neurons contributes to neuropathic pain in rats
Supplemental material, MPX904462 Supplemental material2 for Dorsal Root Ganglia Homeobox downregulation in primary sensory neurons contributes to neuropathic pain in rats by Takaya Ito, Atsushi Sakai, Motoyo Maruyama, Yoshitaka Miyagawa, Takashi Okada, Haruhisa Fukayama and Hidenori Suzuki in Molecular Pain
Supplemental Material
MPX904462 Supplemental material3 - Supplemental material for Dorsal Root Ganglia Homeobox downregulation in primary sensory neurons contributes to neuropathic pain in rats
Supplemental material, MPX904462 Supplemental material3 for Dorsal Root Ganglia Homeobox downregulation in primary sensory neurons contributes to neuropathic pain in rats by Takaya Ito, Atsushi Sakai, Motoyo Maruyama, Yoshitaka Miyagawa, Takashi Okada, Haruhisa Fukayama and Hidenori Suzuki in Molecular Pain
Footnotes
Acknowledgments
The authors thank Mayuri Nakajima, Nao Yaegashi, and Risa Kakiuchi for their technical assistance.
Author Contributions
TI, AS, and HS designed the study; TI, AS, and MM conducted experiments; YM and TO generated AAV vectors; TI, AS, HF, and HS analyzed data, prepared figures, and wrote the manuscript. All authors have read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Japan Society for the Promotion of Science KAKENHI (grant numbers: JP16K10986 and JP16H05461).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
