Abstract
Dynorphin A (1-17) (DynA17) has been identified as a key regulator of both sensory and affective dimensions of chronic pain. Following nerve injury, increases in DynA17 have been reported in the spinal and supraspinal areas involved in chronic pain. Blocking these increases provides therapeutic benefits in preclinical chronic pain models. Although heavily characterized at the behavioral level, how DynA17 mediates its effects at the cellular physiological level has not been investigated. In this report, we begin to decipher how DynA17 mediates its direct effects on mouse dorsal root ganglion (DRG) cells and how intrathecal administration modifies a key node in the pain axis, the periaqueductal gray These findings build on the plethora of literature defining DynA17 as a critical neuropeptide in the pathophysiology of chronic pain syndromes.
Introduction
Endogenous opioid Dynorphin A (1-17) (DynA17), and its degradation products are dramatically upregulated throughout the pain axis in animal models of chronic pain. 1 DynA17 protein upregulation in the spinal dorsal horn and limbic areas results in sensitization of nociceptive pathways and negative affect, respectively, through kappa opioid receptor (KOR) and non-opioid receptor mechanisms.2–4 Animal models of chronic pain show increased DynA17 accumulation for several months in the spinal cord, with high correlation of DynA17 content to mechanical allodynia and thermal hyperalgesia. 5 Exogenous intrathecal administration of DynA17 results in a similar phenomenon and in both cases the mechanism involves NMDA and/or bradykinin receptors.6,7 Furthermore, animals lacking the prodynorphin gene (Pdyn) develop hypersensitivity after induction of preclinical neuropathic pain models, but recover rapidly compared to wild type animals. 8 Interestingly, this phenotype is recapitulated almost identically in animals in which either µ-opioid receptor positive (MOR+) cells in the rostral ventromedial medulla are ablated 9 or when the dorsolateral funiculus (DLF) is lesioned. 10 Decreased levels of DynA17 in the spinal cord are observed following DLF lesions. This suggests a role for DynA17 in the maintenance of a pain state, but not the initial acute pain. KOR is expressed in peripheral afferents as well, thus dynorphin secreted within the CNS does not account for all of the activity of dynorphin as it relates to sensory physiology and pain processing. In the present study we examined the mechanism of DynA17 effects, both the direct effect on DRG, and effects mediated through the spinal cord resulting in altered excitability of PAG neurons, persistent mechanical allodynia, and anxiety-like behaviors.
DynA17 is also upregulated in brain regions after induction of neuropathic models. These increases have been demonstrated to mediate much of the negative affect and aversive components of chronic pain.3,11,12 Within the central nucleus of the amygdala, increased DynA17 signaling through KOR promotes an aversive component of chronic neuropathic pain, as assessed by the conditioned place preference paradigm. 12 Within the nucleus accumbens, decreased inhibitory tone on DynA17-releasing cells promotes increased DynA17 inhibition on target cells resulting in negative affect. 3 Recently, prefrontal cortex DynA17 signaling was demonstrated to cause cognitive disruptions, potentially adding another aspect to effects of DynA17 in chronic pain states. 13 However, this link has yet to be made definitively. Thus, DynA17 plays an instrumental role in both sensory and affective measures that occur in chronic pain states.
Although DynA17 signaling mechanisms within the brain have been moderately characterized, the link between increased spinal DynA17 and changes to the electrophysiological properties of cells due to DynA17 have not been thoroughly characterized. In this study, we began to address this question by first observing the electrophysiological changes induced by DynA17 signaling in vitro in mouse dorsal root ganglion (DRG) cell cultures. We next validated the hypersensitivity induced by intrathecal treatment with DynA17 and performed patch clamp recordings from neurons from ex vivo slices of the periaqueductal gray area (PAG), a key node in both ascending and descending pain modulatory pathways.
Materials and methods
Animals
All animal studies were performed in accordance with approvals from the University of New Mexico Institutional Animal Care and Use Committee. Our facilities are AAALAC accredited and hold the following federal approvals: Animal Welfare Assurance #D16-00,228 (A3350-01) and USDA Registration # 85-R-0014. Male BALB/cAnNHsd mice and female FosCre mice (12 weeks old; Envigo Harlan or JAX Labs) were used for experiments.
A single injection of DynA17 was given at a dose of 3 nmol to mice via intrathecal injection after anesthetizing the mouse with isoflurane and gently shaving the injection area at L4-L5. Using a 30G, 0.5 inch needle attached to a 25 μL Hamilton syringe inserted into the intravertebral space, the DynA17 was injected slowly into the intrathecal space at 5 μL maximum volume. 14
Behaviors
Mechanical hypersensitivity was tested on the hindpaw, with von Frey filament stimulation at baseline and weekly thereafter as has been previously established with the ‘up-down method’ of analysis.15–20 Experimenter was blinded to treatment groups. Mice were allowed to acclimate for 30 minutes in the room prior to testing. A single trial consisted of five applications of several selected mid-range von Frey filaments applied once every 3–4 s. If no positive response was evoked, the next stronger filament was applied. 20 Responses to decreased gram force filaments indicated increased hypersensitivity. Mechanical hypersensitivity testing was performed at 2, 5 and 7 days post-injection and then weekly thereafter.
The light/dark place preference test is an established model of examining anxiety, wherein a mouse is given access to a box with two chambers, with an open door between to allow the mouse free access to either chamber.14,21 One chamber is lit by bright light, while the other remains dark. Collected variables in this test included time spent in each chamber, number of transitions between chambers, number of rearing events, and entry latency into the light chamber from the start of the test. Increased time in the dark chamber, fewer rearing events, and fewer transitions were indicated as measures of increased anxiety. The sucrose splash test is another established model for examining depression, wherein mice are sprayed with a 30% sucrose solution, then monitored for 10 minutes for frequency, duration, and latency of grooming behaviors. Decreased grooming behavior was defined as a measure of depression-like behavior.22,23
Both anxiety- and depression-like behavior tests were performed at 28 days post-DynA17 treatment.
Dorsal root ganglion cultures
Animals were deeply anesthetized with 3% isoflurane and then decapitated prior to dissection of dorsal root ganglia (DRG) for primary cultures. Bilateral L3-L5 DRG were removed and placed in Hank’s Balanced Salt Solution without Ca2+/Mg2+ (HBSS) on ice. A scalpel was used to mechanically disrupt the ganglia and cut them into smaller pieces. The ganglia were digested for 20 min in an enzymatic solution containing HBSS, 1 mg protein/mL papain (Worthington, Cat# LS003126), 5.5 mM L-Cysteine (Cat# C7352-25g, Sigma). This was followed by a second 20-min digestion in a solution of HBSS containing dispase II (4 mg/mL, Cat# D4693-1g, Sigma), and collagenase type 2 (6 mg/mL, Cat# LS004176; Worthington). The digested ganglia were resuspended in Dulbecco’s Modified Eagle Medium (DMEM, Gibco 11,995-065) containing 10% Fetal Bovine Serum (Gibco, Cat# 26,140,079) and 1% Penicillin-Streptomycin (Gibco, Cat# 15,140,148). Suspension was triturated using flame-polished Pasteur pipettes until the suspension passed easily through the bore. Cells were then plated on 12 mm glass coverslips that were coated with HBSS without Ca2+/Mg2 containing 20 ug/mL Poly-D-Lysine (Gibco, Cat# A3890401) and 16ug/mL Laminin (Sigma-Aldrich, Cat# L2020). 24
Preparation of ex vivo brain slices
The animals were anesthetized with 3% isoflurane followed by trans-cardiac perfusion with at least 20 mL of ice-cold N-methyl D-glucamine (NMDG) solution containing (in mM) 92 NMDG, 2.5 KCl, 1.25 NaH2PO4, 30 NaHCO3, 20 HEPES, 25 glucose, 2 thiourea, 5 Na-ascorbate, 3 Na-pyruvate, 0.5 CaCl2, and 10 MgSO4, pH balanced to pH 7.4 using HCl (NMDG cutting solution). The solution was bubbled for 20 min with carbogen (95% O2, 5% CO2) prior to perfusion. After perfusion the brain was removed and fixed to an agar block using cyanoacrylate glue and submerged in ice-cold NMDG cutting solution, continuously bubbled with carbogen. The 300–400 μM transverse brain slices obtained were transferred to warm NMDG solution and held at 32°C for a minimum of 10 min. Slices were then transferred and held in room temperature aCSF containing (in mM) 92 NaCl, 2.5 KCl, 1.25 NaH2PO4, 30 NaHCO3, 20 HEPES, 25 glucose, 2 thiourea, 5 Na-ascorbate, 3 Na-pyruvate, 2 CaCl2, and 2 MgSO4, pH balanced to 7.4, continuously bubbled with carbogen. Slices were held in this solution for a minimum of 1 h prior to recording. 25 vlPAG was identified by examining the slice under ×4 magnification and targeting the ventrolateral region (pictured in Figure 3). Slices were chosen at random and cells are distributed through the rostral/caudal axis of the PAG.
Whole-cell patch clamp electrophysiology
Neurons were identified by infrared differential interference contrast (IR-DIC) connected to an IR2000 camera (Dage MTI, IN). Current-clamp recordings were performed using a Molecular Devices Multiclamp 700B (Scientifica, UK). Signals were filtered at 5 kHz, acquired at 50 kHz using a Molecular Devices 1550B converter (Scientifica, UK), and recorded using Clampex 11 software (Molecular Devices, Scientifica, UK). Electrodes were pulled with a Zeitz puller (Werner Zeitz, Martinsreid, Germany) from borosilicate thick glass (GC150 F, Sutter Instruments). Electrode resistance was 5–8 MΩ. Bridge balance was applied to all recordings. For DRG culture recordings intracellular solution contained (in mM) 125 K-gluconate, 6 KCl, 2 CaCl2, 10 HEPES, 10 EGTA, 2 Mg-ATP, pH 7.3 with KOH. Artificial cerebrospinal fluid (aCSF) contained (in mM) 113 NaCl, 3 KCl, 25 NaHCO3, 1 NaH2PO4, 2 CaCl2, 2 MgCl2, and 11 D-glucose. For brain slice recordings intracellular solution contained (in mM) 120 K-gluconate, 11 KCl, 1 CaCl2, 2 MgCl2 10 HEPES, 11 EGTA, 4 Mg-ATP, 0.5 Na-GTP pH 7.3 with KOH. aCSF contained (in mM) 113 NaCl, 3 KCl, 25 NaHCO3, 1 NaH2PO4, 2 CaCl2, 2 MgSO4, HEPES 5 mM and 11 D-glucose. Recordings were not corrected for junction potential.
Statistical and data analysis
Electrophysiology data analysis was performed using Easy Electrophysiology (v.2.5.1) and Clampfit 11.2. All statistical analysis was performed using GraphPad Prism (v9.2.0). A p-value <0.05 was considered statistically significant. Statistical tests are shown in the figure legends.
Results
DynA17 directly acts on nociceptors to increase excitability and spontaneous activity
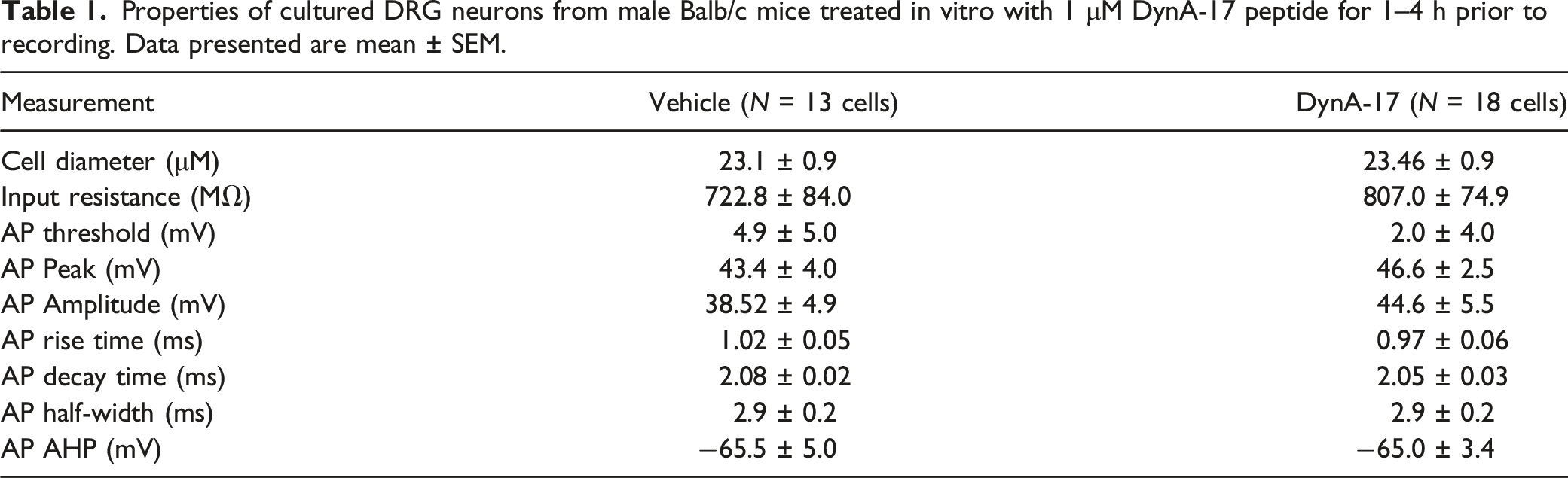
To demonstrate a direct effect of DynA17 on nociceptors, we cultured DRG neurons from naïve male mice (8–12 weeks old) and treated them for with 1 μM DynA17 in culture media for 1–4 h prior to recording. Current clamp recordings were obtained from small (<30 micron) neurons. Rheobase, input resistance, RMP were not significantly changed following DynA17 treatment (Figure 1(a) and (b), Table 1). We observed a significant increase of 61.8% (p = 0.039, Mann-Whitney test) in the number of action potentials elicited in a 500pA ramp (Figure 1(c)). In addition, we observed a significantly greater proportion of DynA17 cells (75% vs. 25% of vehicle, p = 0.020, Fisher’s exact test; Figure 1(d)) exhibited spontaneous activity either without stimulation or with holding current to bring potential to −45 mV. We observed a statistically significant increase of 23.5% (p = 0.0034, Wilcoxon matched pairs signed rank test) in action potential firing frequency in DynA17-treated neurons (Figure 1(e)) compared to vehicle-treated controls. Action potential waveform properties were not significantly changed by DynA17 treatment. AP Waveform and other properties data are summarized in Table 1. DynA17 acts directly on sensory neurons to increase excitability. Dynorphin increased excitability of cultured DRG neurons treated in vitro prior to recording. (a) No significant effect on resting membrane potential or (b) rheobase (minimum current required to elicit firing). (c) There was a significant increase in the number of action potentials seen during 500pA ramp protocol. Vehicle n = 11 cells, DynA17 n = 13 cells, Mann-Whitney test, *p < 0.05. Right: representative traces for the ramp protocol (top: vehicle, bottom: DynA17 treated). (d) There was a significantly greater proportion of DynA17 treated cells that displayed spontaneous activity (SA) at baseline or when held at −45 mV. Value within bar indicates number of cells comprising segment of column. Vehicle n = 12 cells, DynA17 n = 16 cells, Fisher’s exact test, *p < 0.05. Right: example traces illustrating SA in a DynA17 cell with quiescent vehicle cell recording overlaid. (e) A significant difference was found in the firing frequency during the current step protocol (10pA stepwise increase of 500 ms current injection window) as current injection increased from rheobase. Vehicle n = 11 cells, DynA17 n = 12 cells, Wilcoxon matched-pairs signed rank test, **p < 0.01. Line graph illustrating increased mean difference in firing frequency as current increases. Inset: bar graph comparing mean firing frequency. Right: example traces of multi-firing DRG neurons current step protocol, vehicle left, DynA17 right. Properties of cultured DRG neurons from male Balb/c mice treated in vitro with 1 μM DynA-17 peptide for 1–4 h prior to recording. Data presented are mean ± SEM.
Intrathecal DynA17 elicits chronic pain-like behaviors in mice
In order to determine whether DynA17 can elicit chronic pain in mice, we performed intrathecal injection of DynA17 (3 nmol). A single intrathecal injection of DynA17 caused mice to develop mechanical hypersensitivity within 2 days, which persisted for at least 7 days (Figure 2(a)). We also observed significant changes in anxiety- and depression-like behaviors. In the sucrose splash test, DynA17-injected mice spent less time grooming (n = 5 mice per group, p < 0.0001, unpaired t test, Figure 2(b)). In the light-dark maze, DynA17-injected mice refrained from entry to the light for a greater duration compared to vehicle (n = 5 mice per group, p < 0.01, unpaired t test, Figure 2(c)). Intrathecal DynA17 elicits chronic pain-like behaviors in mice. (a). intrathecal injection of Dynorphin elicits hypersensitivity in hindpaw to Von Frey stimulation. Analyzed with two-way ANOVA, ***p < 0.001, ****p < 0.0001. 
Ventrolateral periaqueductal gray (vlPAG) neuronal excitability is increased in DynA17-intrathecal-injected mice with chronic pain-like behaviors
Since the vlPAG is a region heavily implicated in chronic pain and endogenous opioid signaling,
26
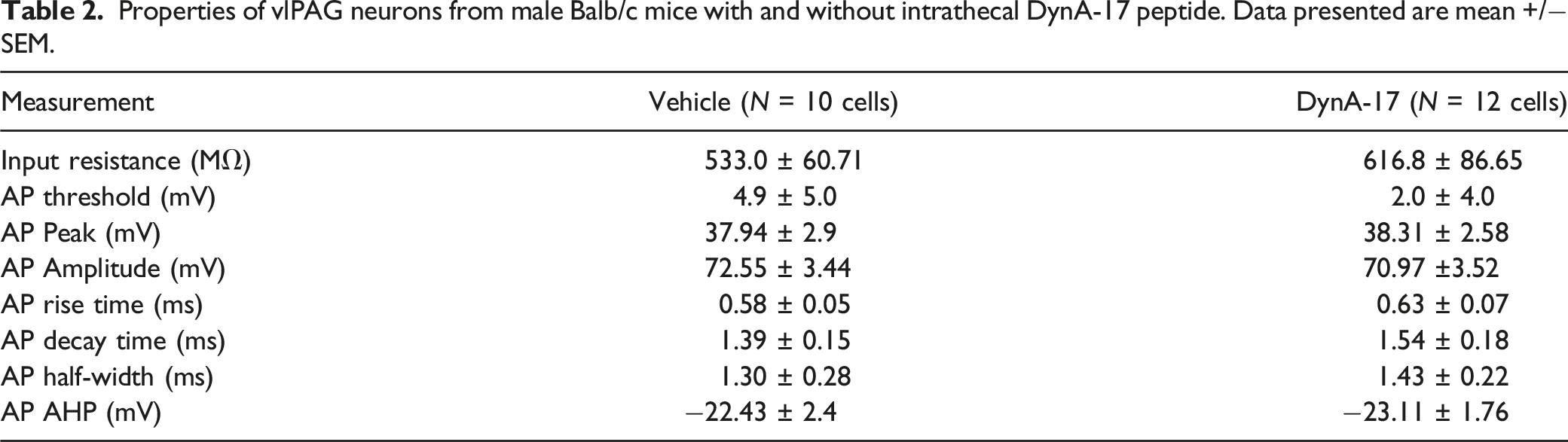
we studied vlPAG neurons in ex vivo slices from male DynA17-intrathecal-injected mice with chronic pain-like behaviors. Using ex vivo slice electrophysiology, we observed that rheobase, input resistance, RMP were not significantly changed following DynA17 treatment (Figure 3(a), Table 2). We observed a statistically significant increase of 22.2% (p < 0.0001, Wilcoxon signed ranked pairs) in action potential firing frequency in DynA17-injected mice (Figure 2(b)) compared to vehicle-injected control mice. Action potential waveform properties were not significantly changed by DynA17 treatment (Table 2). McPherson et al. recently published distinct firing patterns observed in vlPAG neurons in rats, and found electrophysiologically distinct alterations associated with inflammation.
27
We classified our cells according to their scheme. Although we did not observe any so-called onset or random firing patterns, these represent the smallest proportion of their data (10% and 9% of neurons, respectively). They define phasic firing neurons (accounting for 48% of their data) as cells with the duration of firing decreasing at higher current injections. Tonic firing neurons (33% of their cells) fire throughout the duration of injected current even as the current increases. Our data follow a similar distribution, however we did not observe any of the onset or random patterns. In neurons from vehicle-injected mice, we found 6 phasic and 4 tonic firing patterns. In neurons from DynA17-injected mice, we found 8 phasic and 6 tonic firing patterns (Figure 3(c)). Ventrolateral periaqueductal gray (vlPAG) neuronal excitability is increased in DynA17-intrathecal-injected mice with chronic pain-like behaviors (a). Resting membrane potential (right) and rheobase (left) of neurons was not significantly different between groups. Vehicle n = 10 cells, DynA17 n = 12 cells (b). A significant difference was found in the firing frequency during the current step protocol (10pA stepwise increase of 500 ms current injection window) as current injection increased from rheobase. Inset: bar graph comparing mean firing rate. Wilcoxon matched-pairs signed rank test, ****p < 0.0001. Right: example traces of current step recordings for vehicle (top) and DynA17 (bottom). the vehicle recording is representative of a phasic firing pattern, and the dynorphin recording is representative of a tonic firing neuron. (c). Distribution of cells according to phasic or tonic firing pattern. (d). Photo of PAG with the approximate boundaries for the ventrolateral region (thick dashed lines) and the patch pipette highlighted (thin dashed line). Properties of vlPAG neurons from male Balb/c mice with and without intrathecal DynA-17 peptide. Data presented are mean +/− SEM.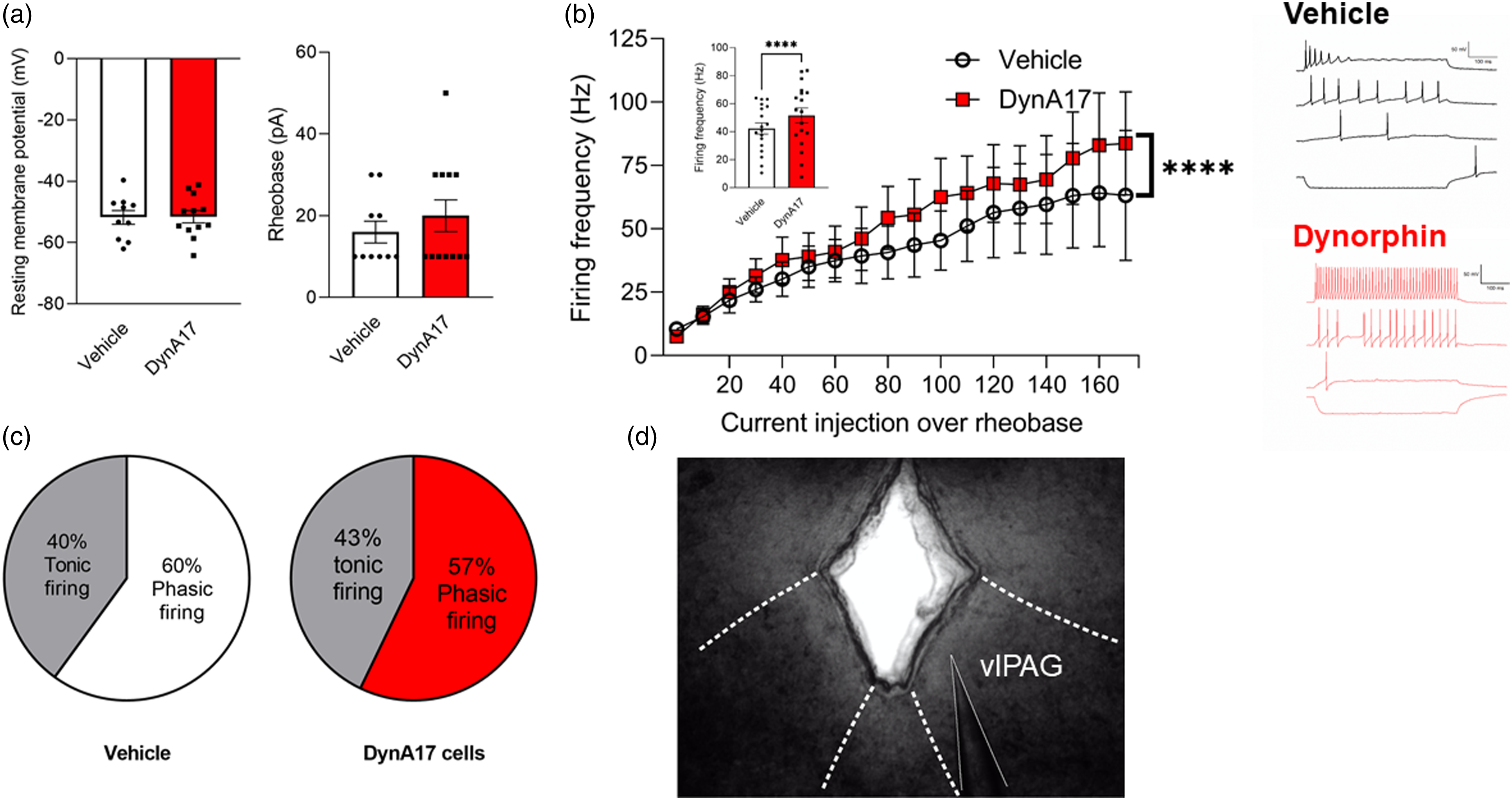
Discussion
These data provide a preliminary exploration of the electrophysiological and behavioral modifications that arise from intrathecal DynA17 in mice. Further, we observed effects in excitability of dissociated DRG neurons from naïve mice treated in vitro with DynA17. The changes observed were consistent with a role for dynorphin as a critical neuropeptide maintaining sensory and affective components of pain. In electrophysiology experiments we characterized actions of DynA17 in both the central and peripheral nervous systems. This study provides characterization of the changes that arise from intrathecal and in vitro dynorphin.
The DRG recordings indicate an increase in the excitability in DynA17-treated DRG neurons. Recent literature indicates KOR is expressed in nociceptive peptidergic afferents as well as low-threshold mechanoreceptors. 26 This study found application of dynorphin decreases voltage-gated calcium channel (VGCC) currents, a canonical effect of KOR activation, exclusively in DRG neurons labelled using a KOR-cre mouse line. A process that dampens calcium influx would suggest attenuated excitability via KOR, while the data presented here suggest increased firing rates in the DRG. It is possible that the effect in excitability is not specific to KOR-expressing neurons. Another key difference in the present study is that the neurons were incubated for 1–4 h in the presence of DynA17 prior to recording, whereas the previous study examined the instantaneous effects of dynorphin on calcium currents, rather than after prolonged exposure in vitro. It is possible that prolonged exposure to DynA17 is driving the altered firing independent of KOR or prolonged rebound changes in cells due to prolonged KOR activation. This remains to be determined. We did not examine neuronal actions of dynorphin in the spinal dorsal horn, which have previously been characterized as inhibitory.28,29 Therefore, dynorphin may have different actions depending on cellular and physiological context as well as the time course of application i.e. minutes versus hours.
The behavioral changes and development of mechanical allodynia we observed in DynA17-injected mice are consistent with previously published data.6,7 The sucrose splash test for depression-like behavior is comparable to effects reported in a study that used kappa agonist Salvinorin A and found reduction in grooming behavior that was restored with KOR antagonists. 30 However, KOR-independent DynA17 effects and inherent differences in the behavioral pharmacology between Salvinorin A and DynA17 make this similarity difficult to interpret. The light-dark maze has not been used in a study of behavioral effects of dynorphin, although the anxiogenic effects of dynorphin have been studied using other measures, such as elevated plus maze, open field test, and forced swim tests. 31 Our results are consistent with this previous work.
The periaqueductal gray is a key node for reception of ascending nociceptive information, as well as an integration site initiating descending pain modulatory signaling. In light of several recent studies it is clear that there are multiple levels of processing that occur in the vlPAG that relate to opioid signaling and descending modulation of pain.32–34 Our findings from current-clamp recordings demonstrate that the intrathecal dynorphin effect results in an excitability increase in an unbiased sample of vlPAG neurons. The pronociceptive effect of chemogenetic activation of vlPAG GABA 32 neurons may relate to our findings, as we observed the increase in pain behavior along with increased vlPAG excitability. However, this same study reports excitatory outputs from this region were reported to drive antinociception and recorded neurons were not defined into excitatory and inhibitory populations in our study. We sorted our cells according to the firing patterns identified in recently published literature. 27 The relative proportion of phasic and tonic firing patterns among our cells parallels their findings, and is nearly identical between the vehicle and DynA17 groups. This suggests that we selected an unbiased sample of neurons from the vlPAG, and the results are not skewed by disproportionate representation of neuronal subtypes. The complexity of the vlPAG output circuitry driving descending inhibition and facilitation has been a long term and ongoing research effort by a number of investigator teams. 35 Future research into the role of dynorphin in chronic pain is necessary to unravel its role in nociception and pain-related affective behaviors. Recently published spatial transcriptomics of the periaqueductal gray 36 will be a helpful tool for unraveling the diverse roles of dynorphin in this brain region. While this study provides some aspects of the electrophysiological profiling of vlPAG neurons following intrathecal dynorphin, future work can expand on this to probe deeper into the excitatory and inhibitory neuronal subtypes that exist in the PAG and other levels of the nervous system to interrogate the role of dynorphin in chronic pain. As a small study in both DRG cultures and a single key node in the nociceptive axis, this study highlights the complexity of DynA17 signaling as more than a KOR ligand. It also highlights the need to investigate electrophysiological changes in other regions such as the spinal cord dorsal horn and parabrachial nucleus in addition to many others. Future studies should build upon these characterizations, and define DynA17 as a major component to not only chronic neuropathic pain pathology, but other affective disorders.
Footnotes
Declaration of conflicting interest
The authors declare there are no conflicting interests, except KNW is consultant with NeuroChronix.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Research Endowment fund of the Department of Anesthesiology & Critical Care Medicine, University of New Mexico School of Medicine.
