Abstract
Background
Neuropathic characteristics are highly involved in the development of chronic pain both physically and psychologically. However, little is known about the relationship between neuropathic characteristics and brain morphological alteration.
Objectives
The aim of this study is to investigate the mechanisms of chronic pain development by examining the above-mentioned relationships by voxel-based morphometry in patients with chronic pain.
Methods
First, we assessed neuropathic characteristics using the painDETECT Questionnaire in 12 chronic pain patients. Second, to assess the gray matter volume changes by voxel-based morphometry, we conducted magnetic resonance imaging of the brain. We applied multiregression analysis of these two assessment methods.
Results
There were significant positive correlations between painDETECT Questionnaire scores and the gray matter volume in the bilateral anterior cingulate cortex and right posterior cingulate cortex.
Conclusions
Our findings suggest that neuropathic characteristics strongly affect the brain regions related to modulation of pain in patients with chronic pain and, therefore, contribute to the severity of chronic pain.
Keywords
Introduction
Chronic pain is complicated because of its effects on various physical, emotional, and cognitive functions. 1 Therefore, the treatment of chronic pain requires multiple approaches, such as pharmacological therapy, interventional therapy, exercise, rehabilitation, and psychological therapy.
In clinical practice, neuropathic pain is regarded as group of disorders characterized by nerve damage including diabetic polyneuropathy, postherpetic neuralgia, and poststroke syndromes 2 and defined by the International Association for the Study of Pain as “Pain caused by a lesion or disease of the somatosensory nervous system.” Under this definition, chronic pain has been classified into neuropathic pain or non-neuropathic pain. However, Attal 3 provided a novel concept that there are various degrees of neuropathic characteristics in chronic pain, that is, overlapping symptoms such as neuropathic pain and nociceptive pain in various chronic pain conditions. A general population survey4,5 demonstrated that chronic pain patients with neuropathic characteristics showed higher pain intensity, lower quality of life or sleep, and more severe anxiety or depression than those without neuropathic characteristics. Thus, neuropathic characteristics could be present in most chronic pain patients and are highly involved in the development of chronic pain.
Voxel-based morphometry (VBM) is a neuroimaging analytical method of evaluating regional differences in the density or volume of the gray matter throughout the brain. As VBM can assess the anatomical differences throughout the brain, a whole brain analysis can be conducted. 6 VBM is widely used in the studies of pain and psychiatric disorders. 6 The brain signature hypothesis 7 states that chronic pain is closely associated with the structure of the brain. A recent review 8 showed that the gray matter volume in a specific region changes under chronic pain condition, as shown by magnetic resonance imaging (MRI). Although the regions showing gray matter morphological alterations differ among various chronic pain patients, there are overlapping areas such as the cingulate cortex, orbitofrontal cortex, insula, and dorsal pons, 7 which are considered to interact with each other during the pain experience.
Wu et al. 9 reported that the scores of ankylosing spondylitis patients in the painDETECT Questionnaire (PD-Q), a screening questionnaire to identify neuropathic characteristics, positively correlated with the gray matter volume in the anterior cingulate cortex, prefrontal cortex, thalamus, and striatum. Because chronic pain commonly has various degrees of neuropathic characteristics, which play a key role in the development of chronic pain, neuropathic characteristics should not be restricted to ankylosing spondylitis patients, as reported by Wu et al. 9 This consideration led us to hypothesize that neuropathic characteristics may be significantly related to the structure of the pain-modulation areas including the anterior cingulate cortex among chronic pain patients with various pathological processes.
To investigate the relationship between neuropathic characteristics and the structures of pain-modulation areas, we conducted a VBM experiment of 12 chronic pain patients. We recruited patients with various types of chronic pain patients. We expected that the volumes of pain-modulation areas, such as the anterior cingulate cortex, correlate with the scores of PD-Q, which is used to identify neuropathic characteristics.
Methods
General design
All patients were recruited among the outpatients in our pain clinic. Each patient completed the PD-Q. Each patient’s MRI data were subsequently acquired on another scheduled day after answering the questionnaire (average, 23 days; SD, 20 days).
Subjects
Patient characteristics.

CRPS: Complex regional pain syndrome; LSS: lumbar spinal stenosis; FM: fibromyalgia.
This study was approved by the institutional review board of Gunma University Graduate School of Medicine, Maebashi, Japan (approval No. 1010). This study was conducted in accordance with institutional ethical provisions and the Declaration of Helsinki. The subjects were financially compensated for taking part in this study.
Patients are listed in registration order. Complex regional pain syndrome (CRPS); lumbar spinal stenosis (LSS); fibromyalgia (FM); left (L); right (R), both sides (B), and standard deviation (SD).
Questionnaire
All patients completed PD-Q. 10 PD-Q is a screening questionnaire to identify neuropathic characteristics. PD-Q consists of seven neuropathic symptoms such as the pain scale, the pain course, and a section on the existence of radiating pain with a total score between 0 and 38. Each of seven neuropathic symptoms is rated on a 6-point scale: 0 (never) to 5 (very strongly). The pain course is rated from −1 to 1 point and the existence of radiating pain is rated from 0 to 2 points. A score ≥ 19 indicates a high possibility of neuropathic characteristics and ≤ 12 indicates their unlikelihood. 10
Neuroimaging assessments
T1-weighted brain MRI was performed using a 3-Tesla scanner (MAGNETOM Trio, A Tim system, Siemens, Germany) at Gunma University Hospital (Gunma, Japan). Magnetization-prepared rapid gradient echo (MPRAGE) images were acquired (176 slices, 1 mm slice thickness with the following parameters: repetition time (TR) = 2300 ms; echo time (TE) = 2.98 milliseconds; flip angle = 9°; field of view = 256 mm2; voxel size = 1 ×1 × 1 mm3, matrix size = 256 × 256). Each image was examined for artifacts.
VBM protocol and analysis
We used VBM8 toolbox revision 435 (http://dbm.neuro.uni-jena.de/vbm/) with Statistical Parametric Mapping (SPM) 8 revision 5236 (The Wellcome Trust Centre for Neuroimaging; http://www.fil.ion.ucl.ac.uk/spm) in MATLAB 2013a (MathWorks, Inc.) to analyze the obtained structural images.
The structural images were corrected for bias-field inhomogeneity and spatially normalized with diffeomorphic anatomical registration through exponentiated Lie algebra (DARTEL) to the Montreal Neurological Institute (MNI) template, and tissues were classified as gray matter, white matter, or cerebrospinal fluid. In the modulation process, nonlinear deformation was used for normalization so that voxel intensities reflected regional gray matter volumes adjusted for individual brain sizes. Images were then smoothed to a Gaussian kernel of 8-mm full width at half maximum. After the preprocessing of structural images, gray matter segments were subjected to second-level analysis. As a primary outcome, multiple regression analysis, with PD-Q score as the effect of interest and gender, age, and pain duration as the effects of no interest, was applied. Considering a possibility that pain duration affect gray matter volume, 11 pain duration was included as a covariate. We conducted the regression analysis with pain duration as an effect of interest, but as no brain region was found, pain duration was added as the effect of no interest. The statistical threshold for the MRI data analysis were set at family wise error corrected p < 0.05 at the cluster level with the height threshold as uncorrected p < 0.001 at the peak level. This threshold was applied for the whole-brain analysis. In the analysis, we applied nonstationary correction. 12
Results
Scores in painDETECT Questionnaire
The average score in PD-Q was 22.25 (SD = 6.94; range, 10–35). Because we regard chronic pain as having various degrees of neuropathic characteristics, we enrolled all patients regardless of the score.
Correlation analysis between painDETECT score and gray matter volume
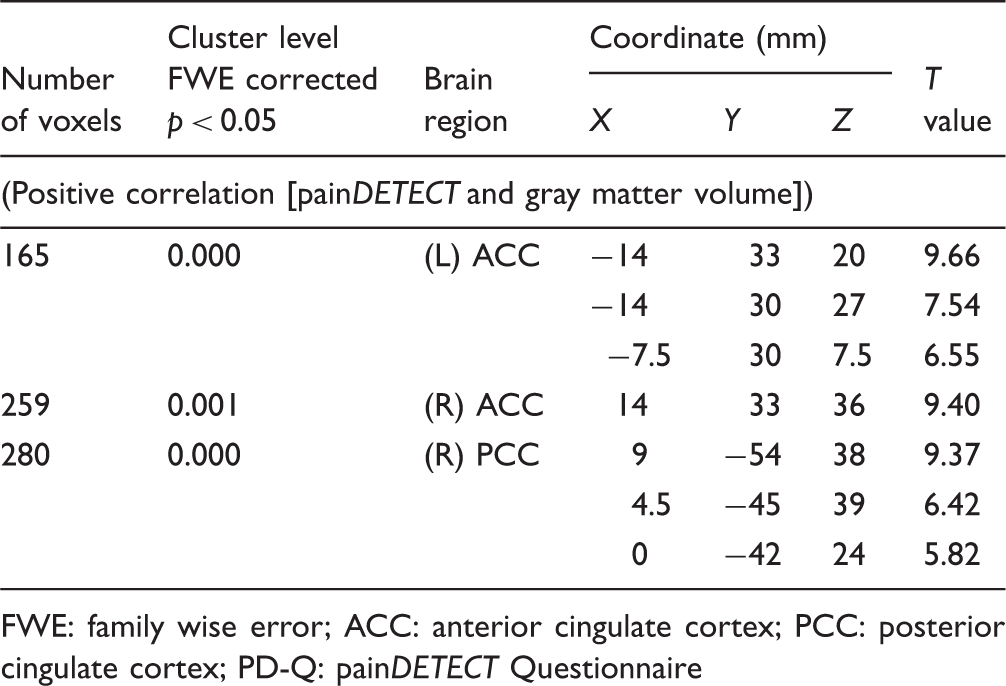
The effect of neuropathic characteristics to gray matter volume was examined by multiple regression analysis. There were significant positive correlations between gray matter volume and painDETECT scores, i.e., increase in gray matter volume with increasing scores) in the right anterior cingulate cortex (ACC), left ACC, and right posterior cingulate cortex (PCC) (Table 2, Figure 1). No significant negative correlation was found between these factors in these regions. For data illustration purpose, we further calculated correlation coefficient between average beta value within each significant cluster and PD-Q scores (Figure 2).
Gray matter volume in three regions was significantly correlated with painDETECT score. ACC: anterior cingulate cortex; PCC: posterior cingulate cortex. Correlation coefficient between average beta value within each significant cluster and PD-Q scores. ACC: anterior cingulate cortex; PCC: posterior cingulate cortex; PD-Q: painDETECT Questionnaire. Brain regions showing significant relationship between PD-Q score and gray matter volume. FWE: family wise error; ACC: anterior cingulate cortex; PCC: posterior cingulate cortex; PD-Q: painDETECT Questionnaire

Discussion
Neuropathic characteristics of participants
In this study, the PD-Q score was 21.5 (median score; interquartile range, 19.0 to 26.3). PD-Q is a screening questionnaire that determines the prevalence of neuropathic characteristics. A high score represents strong neuropathic characteristics. 10 Considering that a score ≥ 19 indicates a high possibility of neuropathic characteristics in PD-Q, most of the patients in this study are considered to have strong neuropathic characteristics.
Effect of neuropathic characteristics on chronic pain
Neuropathic characteristics and the severity of chronic pain are closely related. For example, patients with neuropathic characteristics show higher pain intensity and longer pain duration than those without neuropathic characteristics in chronic low back pain, 13 fibromyalgia, 14 and general chronic pain.15–17 Furthermore, the levels of psychological aspects such as anxiety and depression, as well as pain intensity, were much higher in patients with neuropathic characteristics in chronic pain associated with diabetes mellitus 18 and in general chronic pain patients. 19 Rasmussen et al. 20 showed that general chronic pain and putative neuropathic pain have overlapping neuropathic characteristics. Although the neuropathic characteristics in chronic pain do not always involve neuroanatomical regions and may include psychogenic factors or somatization, these characteristics may be related to the severity of chronic pain. Considering that chronic pain is related to brain morphological changes and neuropathic characteristics are related to the severity of chronic pain, neuropathic characteristics may also be related to brain morphological changes with worsening chronic pain.
Brain regions related to neuropathic characteristics
In this study, we found positive correlations between PD-Q score and gray matter volume in the bilateral ACC and right posterior cingulate cortex by the analysis throughout the brain. Here, we discuss the impact of neuropathic characteristics on the development of chronic pain and the role of these brain regions that show significant positive correlations with neuropathic characteristics.
As we hypothesized, we found significant positive correlations between the volume of ACC and the scores of PD-Q. Findings of functional neuroimaging using recent techniques indicate that ACC is involved in cognition and emotions 21 ; thus, ACC is a key node for the affective pain matrix. 22 It was reported that the analgesic effect of hypnosis and placebo treatment is associated with ACC activity. 23 Salomons et al. 24 showed that the ACC activity was reduced by the manipulation of subjects’ belief that they could control subsequent painful stimulus, although they actually received the same painful stimulus despite their control. These studies suggest that ACC is involved in pain modulation via the cognition of pain. Furthermore, in chronic pain patients as well as healthy subjects, ACC is activated by painful stimulation although the activated area is smaller in chronic pain patients. 25 In our study, the gray matter volume and the neuropathic characteristics positively correlated in ACC. We speculate that cognition and emotions are induced by sudden pain attacks, and typical neuropathic characteristics such as burning pain sensation and allodynia might have activated the ACC and consequently increased the gray matter volume in chronic pain patients.
PCC is considered to regulate attention and cognition. 26 In a study of healthy volunteers, only PCC showed a higher activity during the anticipation period for noxious pain than during the noxious pain period, although several other brain regions were activated during both periods. 27 The patients with a focal region in PCC showed impairment in the performance of multiple tasks that require cognitive ability. 28 These studies support the idea that the role of PCC is related to attention and cognition. Meerwijk et al. 29 in their review stated that PCC is activated in 31.6% of the studies of psychological pain such as current psychological pain and recalled grief or sadness, 29 whereas Apkarian et al. 30 found that it is involved in only about 9% of physical pain studies. Thus, attention and cognition, rather than noxious pain itself, activated PCC.
Morphological alterations in these brain regions in chronic pain patients are also shown in other VBM studies of ACC31–35 and PCC. 36 These studies also support the idea that these brain regions are related to the processing of chronic pain. The above-mentioned studies indicate that these brain regions interact with each other during pain experience, that is, cognition of and attention to pain. 37 Considering that chronic pain involves complex interactions among physical, emotional, cognitive, and social aspects 38 and that neuropathic characteristics are related to the severity of chronic pain, it is reasonable to consider that the brain structure is altered in these brain regions. Our findings suggest that chronic pain with neuropathic characteristics activated these brain regions and might have increased gray matter volume.
Alteration of gray matter volume and its mechanisms
We also discuss the gray matter volume alteration and its mechanisms. Although gray matter volume alteration is observed in many chronic pain diseases,34,39–42 its mechanism still remains unclear. Gray matter volume alteration is presumed to occur thorough the change in cell size, neuronal and glial cell genesis, and angiogenesis. 43 A rat neuropathic pain model showed longer dendrites with more branches in neurons than the sham-operated rats. 44 Stressed mice showed decrease gray matter volume with loss of synaptic spine density of dendrites. 45 These studies indicate that gray matter volume alteration relies on activity-dependent dendritic changes. 46 In this study, gray matter volume positively correlated with the scores of neuropathic characteristics. Considering the candidate mechanism of gray matter volume alteration above, more severe neuropathic characteristics might have induced higher activities in those brain regions and subsequently increased gray matter volume.
Borsook et al. 46 proposed adaptive and maladaptive changes as the brain responses to pain. Adaptive brain responses increase activity-dependent dendritic complexity and maladaptive brain responses decrease dendritic complexity. Several studies indicate that a reduced gray matter volume is reversed to levels seen in healthy states in a relatively short period, for instance, after arthroplasty in painful osteoarthritis patients and improvement of headache in whiplash injury.47–49 These outcomes might have arisen from maladaptive (for reduced volume) and adaptive (for reversed volume) responses. In our study, gray matter volume in brain regions related to pain modulation positively correlated with more severe neuropathic characteristics. It is assumed that our findings might have been derived from adaptive brain responses to activity-dependent dendritic changes. 46
Gray matter volume and pain duration
In the current study, patients with a wide range of pain durations (from 5 months to 27 years) were included. Pain duration and pain intensity could be the factors that affect brain morphological changes. 50 In this study, we found no positive or negative correlation between gray matter volume and pain duration, whereas we found a strong positive correlation between gray matter volume and neuropathic characteristics in ACC and PCC. Schmidt-Wilcke et al. 51 found a correlation between gray matter volume and pain intensity but not between gray matter volume and pain duration, which is similar to our finding. Taking into account, a previous report that daily repeated painful stimulations altered gray matter volume in one week, 52 these pain durations would be long enough for the gray matter volume to alter. These reports support our finding of a strong positive correlation between neuropathic characteristics and gray matter volume, but not between pain duration and gray matter volume.
Limitations
Our study has limitations. First, the small number of subjects, 12 patients, is a limitation of this study because statistical power depends on total sample size. Second, the comparison of gray matter volume between the healthy subjects and patients could not be conducted because we can not distinguish PD-Q effects from subjects’ common effects as the PD-Q scores were all zero. Healthy subjects are expected to score zero in PD-Q, as PD-Q was developed to discriminate between neuropathic and nociceptive pain components in chronic low back pain patients and PD-Q consists of questions that address the quality of neuropathic pain symptoms, the pain patterns, and the existence of radiating pain. 10
Conclusion
In this study, we found positive correlations between PD-Q scores and gray matter volume in brain regions related to pain experience. Our findings imply that more severe neuropathic characteristics induce greater activity and subsequently induce morphological alterations in those brain regions. Our study, therefore, indicates the importance of neuropathic characteristics in chronic pain.
Footnotes
Authors' Note
Acknowledgments
The authors are very grateful to Mr. Koichi Ujita and Mr. Kazuya Asano for devoted support to this project as radiological technologists.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Grants-in-Aid for Scientific Research (KAKENHI) from the Ministry of Education, Culture, Sports, Science and Technology of Japan (Grant Number 25462424).
