Abstract
The anterior cingulate cortex (ACC) is critical for pain perception, emotion and cognition. Previous studies showed that the ACC has a complex network architecture, which can receive some projection fibers from many brain regions, including the thalamus, the cerebral cortex and other brain regions. However, there was still a lack of whole-brain mapping of the ACC in adult mice. In the present study, we utilized a rabies virus-based retrograde trans-monosynaptic tracing system to map whole-brain afferents to the unilateral ACC in adult mice. We also combined with a new high-throughput, high-speed and high-resolution VISoR imaging technique to generate a three-dimensional whole-brain reconstruction. Our results showed that several principal groups of brain structures send direct monosynaptic inputs to the ACC, including the cerebral cortex, amygdala, striatum, the thalamus, and the brainstem. We also found that cortical neurons in the ACC mainly receive ipsilateral monosynaptic projections. Some cortical areas and forebrain regions also bilaterally projected to the ACC. These findings provide a complete analysis of the afferents to the ACC in adult mice, and whole-brain mapping of ACC afferents would provide important anatomic evidence for the study of pain, memory, and cognition.
Keywords
Introduction
The anterior cingulate cortex (ACC) is known to be involved in pain perception, emotional disorders, and cognition.1–3 The ACC is located in the frontal part of the cingulate cortex and has a complex network architecture. Previous studies have shown that cortical neurons in the ACC receive direct inputs from various cortical and subcortical structures, such as the thalamus, amygdala and somatosensory cortices, which convey nociceptive information as well as information involved in the mediation of emotional states from somatic and visceral organs to the ACC.1,4 The thalamocortical pathways are essential for many key physiological functions, including sensory perception, motor coordination and emotion. 5 Our previous anatomic studies have shown that the ACC receives major inputs from different thalamic nuclei. 6 Experimental activation of mediodorsal thalamic-ACC projections involves in pain processing and pain-related aversion.6,7 In addition, activation of the ascending locus coeruleus-ACC norepinephrinergic projections facilitates behavioral responses to pain and itch by enhancing glutamatergic synaptic transmission and neural excitability in the ACC. 8 In recent years, studies have also reported that cortico-cingulate connections are enhanced during chronic pain. Activation of the primary somatosensory cortex (S1) axon terminals increase the response of ACC neurons to noxious stimuli and this S1-ACC neuronal circuit can mediate aversive responses to pain. 9 Despite these reports showing functional characteristics of different ACC-related neuronal circuits, more systematic anatomic evidence is still needed for the study of pain, memory, and cognition.
Most of the efforts about afferents to the ACC have been reported in monkeys, rats and rabbits.10–12 Early in 1987, Vogt and Pandya 13 utilized horseradish peroxidase (HRP) to retrogradely trace thalamic and cortical afferents to the ACC in Rhesus Monkey. Recently, Fillinger et al. 4 utilized the FluroGold (FG)/β-subunit of choleric toxin (CTb) dyes to map whole-brain afferents to the ACC of mice, and found the area 24a and 24b receive different projection fibers from many cortical and subcortical areas. However, a systematic analysis was still lacking in adult mice. Due to the limitations of traditional fluorescence imaging techniques, previous studies on brain connectome required proportional sampling on serial thin brain sections, which results in a lack of continuity and axonal tracing. Recently, A new microscopy method of Volumetric Imaging with Synchronized on-the-fly-scan and Readout (VISoR) made breakthroughs in tissue clearing, scanning speed and imaging resolution, and achieved a high-resolution, high-throughput and high-speed brain mapping.14,15 Previously, we utilized VISoR imaging to trace the efferents of the ACC in the adult mice,16,17 however, further investigation is still needed for the whole-brain afferents to the ACC.
Here, we utilized a rabies virus-based trans-monosynaptic retrograde tracing system and combined the VISoR imaging method to systematically map three-dimensional (3D) whole-brain presynaptic partners of the ACC in adult mice by injecting the virus into the unilateral ACC. Our results showed that several principal groups of brain structures can send direct monosynaptic inputs to the ACC, including cortical areas such as different layers of the ACC, contralateral ACC, primary and secondary sensory cortex (S1 and S2), insular cortex (IC), retrosplenial cortex (RSC) and hippocampus CA1, amygdala, striatum, different thalamic nucleus and the brainstem such as the ventral tegmental area (VTA). The ACC mainly receives ipsilateral monosynaptic inputs. Some cortical areas and forebrain regions can also bilaterally project to the ACC. Besides, we also summarized functional characteristics of ACC-related neuronal circuits, which are mainly involved in pain perception, pain-related aversion and anxiety, itching, memory consolidation and cognition, and other behaviors such as sociability and consolation. Our results provide more extensive anatomic evidences for ACC-related circuitry mechanisms and functions, and provide possibilities for the development of novel drugs and clinical therapy for relevant neuropsychiatric diseases.
Methods
Animals
Adult (aged 6-8 weeks) male C57BL/6 mice were purchased from the Experimental Animal Center of Xi’an Jiaotong University. All experimental mice were randomly housed in plastic cages under an artificial 12-h day/night cycle, with enough food and water freely available at the temperature of 22–25°C. Mice were raised in an experimental environment for at least 1 week before carrying out experiments. All experimental procedures involving animals were in accordance with the guidelines of the Ethics Committee of Xi’an Jiaotong University.
Viral tracing and surgery
To map whole-brain afferents to the ACC in mice, we micro-injected AAV helper viruses (AAV2/9: rAAV-hSyn-EGFP-2a-TVA-2a-RVG-WPREs-pA; 2.0 × 1012 genomics copies per mL) and rabies viruses (RV: RV-EnvA-ΔG-DsRed; 2.0 × 108 genomic copies per mL) bought from Brainvta company (Wuhan, China) into the right ACC. Viral injection was performed as previously described.6,18 Briefly, the experimental mice were anesthetized with 2% isoflurane and placed on a stereotaxic apparatus. The right ACC of the mouse brain was located according to the Mouse Brain in Stereotaxic Coordinates, 4th edition (0.90 mm anterior to the bregma, 0.30 mm lateral to the midline, 1.40 mm ventral to the surface of the skull). Using a glass pipette and micro syringe pump, AAV helper viruses with a volume of 200 nL were first stereotaxically injected into the right ACC with equal speed (23 nL/min, once every 10 s). An additional 10 min was kept to allow diffusion of viral particles before the glass electrode was slowly withdrawn. Next, the surgical wound was carefully sutured and disinfected. After a 3-week expression of AAV helper viruses, 200 nL of RV-EnvA-ΔG-DsRed was injected into the same location and was expressed for 1 week.
Brain slice separation
The fixation solutions containing 4.0% paraformaldehyde (PFA, pH 7.4) with or without 4.0% hydrogel monomer solution (HMS, 4.0% w/v acrylamide, 0.05% w/v bisacrylamide, 4.0% w/v PFA, 0.25% w/v VA-044 thermal initiator) in phosphate buffered saline (PBS) were prepared and stored at 4°C before using. One week after viral expression, the experimental mice were deeply anesthetized with 2% isoflurane and then perfused intracardially with 0.01 M PBS followed by 4.0% PFA. The whole brain was separated and stored in 4.0% PFA solution. Next, the whole brain was transferred into 4.0% HMS at 4°C overnight to 2 days and then embedded with an equal volume of mixed solution containing 4.0% HMS and 20% bovine serum albumin (BSA). After three times washing in PBS, the embedded block was trimmed and vertically fixed on the vibroslicer. 300 μm-thickness coronal brain slices were cut and transferred into 50 mL tubes in sequence (Compresstome VF-300, Precisionary Instruments; 40-50 slices for one mouse).
Tissue clearing and fluorescence imaging
These brain slices were then treated with a clearing solution (5% PBS-Triton) at 37°C and gently shaken for 24 h. After clearing, these brain slices were washed with PBS three times and mounted onto the quartz slides in sequence. To maintain stability, these brain slices were fixed with 4% HMS at 37°C for 4 h and then immersed into refractive-index-matching solution (50% iohexol, 23% urea, 11% 2,2′,2″-nitrilotriethanol, and 16% distilled water) with a refractive index of 1.52 for 4 h to facilitate optical transparency. Next, these cleared brain slices were imaged at 1 × 1 × 2.5 μm 3 voxel resolution using the VISoR2 technique, and synchronized beam-scan illumination and camera-frame readout generated sixteen-bit images with the sample stage moving linearly in the X direction. Custom programs and Imaris software were used for whole-brain image reconstruction and visualization.14,15
Results
Overview of whole-brain monosynaptic inputs to the ACC neurons using a rabies virus-based system
In the past, due to the limitations of fluorescence imaging techniques, a systematic whole-brain connectome of the ACC was still lacking in mice. The VISoR system made breakthroughs in tissue clearing, scanning speed, and imaging resolution, and achieved high-resolution, high-throughput, and high-speed brain mapping. Here, we utilized VISoR imaging combined with a rabies virus-based trans-monosynaptic retrograde tracing system to systematically map three-dimensional (3D) whole-brain afferents to the ACC. As shown in Figure 1(a), the AAV helper virus was first stereotaxically micro-injected into the right ACC of five adult male mice. After viral expression for 21 days, we injected the rabies virus into the same location. 6 With the help of TVA and RVG proteins, the rabies virus-infected ACC neurons and retrogradely spread to the upstream neurons. This strategy can achieve whole-brain retrograde and trans-monosynaptic mapping of afferents to the ACC. With viral expression for 7 days, five mice were deeply anesthetized and intracardially perfused with 0.01 M PBS followed by 4% PFA in PBS (pH 7.4). A series of 300 μm-thickness coronal brain slices (about 40-50 slices for one mouse) were prepared and cleared for VISoR imaging as previously described. In the viral injection site of the ACC, we found that these viruses were expressed from the rostral to the caudal ACC. These neurons only infected by rAAV were EGFP+ (green), which can anterogradely spread to the axonal terminals of infected ACC neurons. Those starter neurons (yellow) were infected by both rAAV (EGFP+) and RV (DsRed+). Besides, these DsRed+ neurons (red) indicated presynaptic inputs, which can send direct projection fibers to starter neurons. We also found that ACC neurons formed monosynaptic connections between different layers (Figure 1(b)). 3D reconstruction of the whole brain was performed according to the volumetric imaging data. The horizontal, sagittal, and coronal views of the 3D-reconstructed whole brain were displayed in Figure 1(c-e).

Whole-brain VISoR imaging with the rabies virus injected into the unilateral ACC of adult mice. (a) Experimental timeline for retrograde tracing projections to the ACC. The AAV helper virus with TVA receptor, RVG and EGFP; and the glycoprotein (G)-deleted rabies virus with EnVA and DsRed. (b) A typical sample of coronal brain sections showing the combination of two viruses micro-injected into the ACC. The green represents the neurons labeled by helper virus (EGFP+); the red indicates presynaptic inputs that can directly project to the ACC (DsRed+); the yellow represents the starter neurons, which are infected by both the helper virus and rabies virus. The blue and white boxed views indicated enlarged views. (c–e) The horizontal (c), sagittal (d) and coronal (e) views for one 3D-reconstructed whole brain. a: anterior; d: dorsal; r: right. Scale bars: 1 mm (white); 50 μm (black); 20 μm (red).
To map rabies virus-labeled whole-brain presynaptic partners to the ACC, we imaged serial whole-brain coronal sections after viral tracing. The input area was identified based on the Mouse Brain in Stereotaxic Coordinates, 4th edition, and we found that the ACC neurons integrate monosynaptic inputs from widespread brain regions, ranging from the olfactory bulb to the brainstem (Bregma AP +3.5 mm to −7.0 mm). The results showed that most of the whole-brain afferents to the ACC originated from the cortex and thalamus. Additionally, hippocampal formation and amygdala accounted for minor direct projections. There are also a few other areas in the subcortical forebrain and brainstem that were weakly labeled (Figure 1(c-e)).
ACC neurons receive widespread cortical inputs
The cerebral cortex is made up of billions of neurons, which can form a complex neuronal network with the cortex and subcortical areas. 19 The ACC serves as a key cortical area for sensation and cognition, previous studies showed that the ACC forms direct connections with many other cerebral cortices.1,4 In our present study (Figure 2; Table 1), we found that the ACC received significant monosynaptic inputs across the whole bilateral neocortical areas (the white arrows represent the cortical areas containing DsRed+ neurons), with an overall ipsilateral preference.

Widespread cortical and diencephalic distributions of efferent neurons to the unilateral ACC. (a-l) Representative coronal sections showing cortical and thalamic labeling of monosynaptic inputs to the ACC neurons. The approximate AP level from bregma is indicated for each section. The white arrows indicate cortical inputs to the ACC; the yellow arrows indicate diencephalic inputs to the ACC. Scale bar: 1 mm (white).
Network connections from widespread cortical areas and the hippocampus to the ACC.
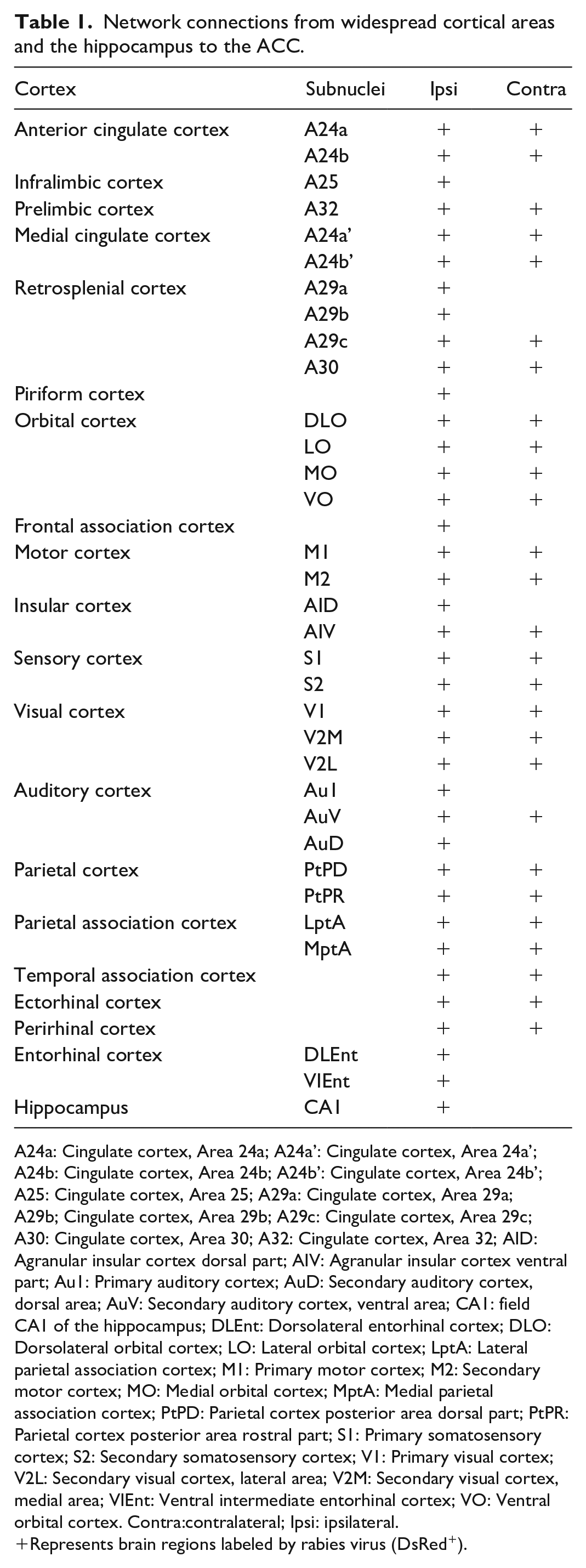
A24a: Cingulate cortex, Area 24a; A24a’: Cingulate cortex, Area 24a’; A24b: Cingulate cortex, Area 24b; A24b’: Cingulate cortex, Area 24b’; A25: Cingulate cortex, Area 25; A29a: Cingulate cortex, Area 29a; A29b; Cingulate cortex, Area 29b; A29c: Cingulate cortex, Area 29c; A30: Cingulate cortex, Area 30; A32: Cingulate cortex, Area 32; AID: Agranular insular cortex dorsal part; AIV: Agranular insular cortex ventral part; Au1: Primary auditory cortex; AuD: Secondary auditory cortex, dorsal area; AuV: Secondary auditory cortex, ventral area; CA1: field CA1 of the hippocampus; DLEnt: Dorsolateral entorhinal cortex; DLO: Dorsolateral orbital cortex; LO: Lateral orbital cortex; LptA: Lateral parietal association cortex; M1: Primary motor cortex; M2: Secondary motor cortex; MO: Medial orbital cortex; MptA: Medial parietal association cortex; PtPD: Parietal cortex posterior area dorsal part; PtPR: Parietal cortex posterior area rostral part; S1: Primary somatosensory cortex; S2: Secondary somatosensory cortex; V1: Primary visual cortex; V2L: Secondary visual cortex, lateral area; V2M: Secondary visual cortex, medial area; VIEnt: Ventral intermediate entorhinal cortex; VO: Ventral orbital cortex. Contra:contralateral; Ipsi: ipsilateral.
Represents brain regions labeled by rabies virus (DsRed+).
We firstly explored intracingulate network connections which directly projected to the ACC and observed bilateral labelings of DsRed+ neurons from both A24a and A24b, indicating that there exists different types of monosynaptic connections within the ACC, including contralateral-ipsilateral neuronal connections and direct projections from different layers. Among them, the contralateral ACC neurons send direct projections to the ipsilateral region through the corpus callosum (Figure 2(c) and (d)). In the rostral cingulate, the prelimbic cortex (A32) and the infralimbic cortex (A25) were also densely labeled, with bilateral labeled neurons in the A32 (Figure 2(a) and (b)). In the medial cingulate cortex (MCC), the A24a’ and the A24b’ send dense projections to the ACC, which were thought to be related to cognition and movement. Caudally, the RSC corresponding to areas 29 and 30, also presented a specific pattern of connections, with dense labeling in the A30 and A29c (Figure 2(d-j)).
Next, the non-cingulate cortical areas that projected to the ACC were analyzed in our study. As displayed in Figure 2, the piriform cortex (Pir), as the largest component of the primary olfactory cortex, showed light DsRed+ labeling. Rostrally and ipsilaterally, the dorsolateral (DLO), lateral (LO), medial (MO) and ventral (VO) orbital cortices were densely labeled. And the frontal association cortex (FrA), the primary (M1) and secondary (M2) motor cortex were also observed to contain DsRed+ neurons. The insular cortex was thought to be important for emotional and cognitive functions. 20 We found that DsRed+ neurons were mainly distributed in the dorsal (AID) and ventral (AIV) part of the agranular insular cortex, and mostly located in the rostral region. Additionally, the ACC also received direct projections from more posterior regions, including the somatosensory (S1, S2), visual (V1, V2M, and V2L), auditory (Au1, AuV, and AuD), parietal (PtPD and PtPR) and parietal associative (LPtA and MPtA) cortices. In the temporal areas, the temporal association (TeA), ectorhinal (Ect) and perirhinal (PRh) cortices were lightly labeled, which constitutes the entorhinal-hippocampal system with the hippocampus, the dentate gyrus, the subicular areas and the entorhinal cortex (Ent), is one of the most important brain network for memory and emotion. 21 In the present study, we found that the Ent and CA1 also directly innervate ACC neurons.
Except for ipsilateral labeling, light projections were also observed from the contralateral brain regions, such as the orbital cortex (LO, MO, and VO), AIV, motor cortex, somatosensory cortex, visual (V1, V2M, and V2L), auditory (AuV) cortices, etc. These results showed that ACC neurons receive widespread cortical inputs from bilateral hemispheres, which may provide the anatomic foundations for further investigation of cortico-cortical circuit mechanisms.
ACC neurons receive widespread inputs from the diencephalon
The diencephalon acts as a relay station between the sensory, central, and endocrine systems, and is one of the most elaborate structures in the vertebrate brain, which is made up of the thalamus, the hypothalamus, epithalamus, subthalamus, and the pituitary. The thalamocortical pathways are essential for many key physiological functions, including sensory perception, motor coordination and emotion. 5 Our previous studies have conducted a detailed analysis of thalamic-anterior cingulate monosynaptic inputs. 6 Here, we gave a brief overview of different thalamic nuclei that directly projected to the ACC. As shown in Figure 2(d-i) and Table 2, we utilized the yellow arrows to map the diencephalon nuclei with DsRed+ neurons. First, in the anterior thalamic nucleus, the anteromedial part (AM) displayed dense labeling in the ipsilateral side of the injection site, while the anteroventral (AV), anterodorsal (AD) and interanterodorsal (IAD) were lightly labeled. We also observed direct projections from the medial thalamic nucleus, which contained the central (MDc) or lateral (MDl) part of the mediodorsal thalamic nucleus and the submedius (Sub) thalamic nucleus, with a high density in the MDl. In the ventral group, the ventromedial (VM), ventral anterolateral (VAL) and posterolateral (VPL) thalamic nucleus can send presynaptic inputs to the ACC. Among the lateral thalamic nuclei, DsRed+ neurons were observed in the dorsomedial part (LDDM) or the ventrolateral part (LDVL) of the laterodorsal thalamic nucleus, and the mediorostral part (LPMR) or laterorostral part (LPLR) of the lateral posterior thalamic nucleus. Also, the midline and intralaminar thalamic nucleus, such as the paraventricular (PV), reuniens (Re), rhomboid (Rh), centrolateral (CL), centromedial (CM) and parafascicular (PaF) thalamic nucleus, displayed DsRed+ neuronal labelings. However, these labeled nuclei mainly focused on the ipsilateral thalamus, and only the Re contained minor contralateral labelings, which indicated that nociceptive information from somatic and visceral organs is conveyed indirectly to the ACC through the ipsilateral thalamus.
Network connections from diencephalon to the ACC.
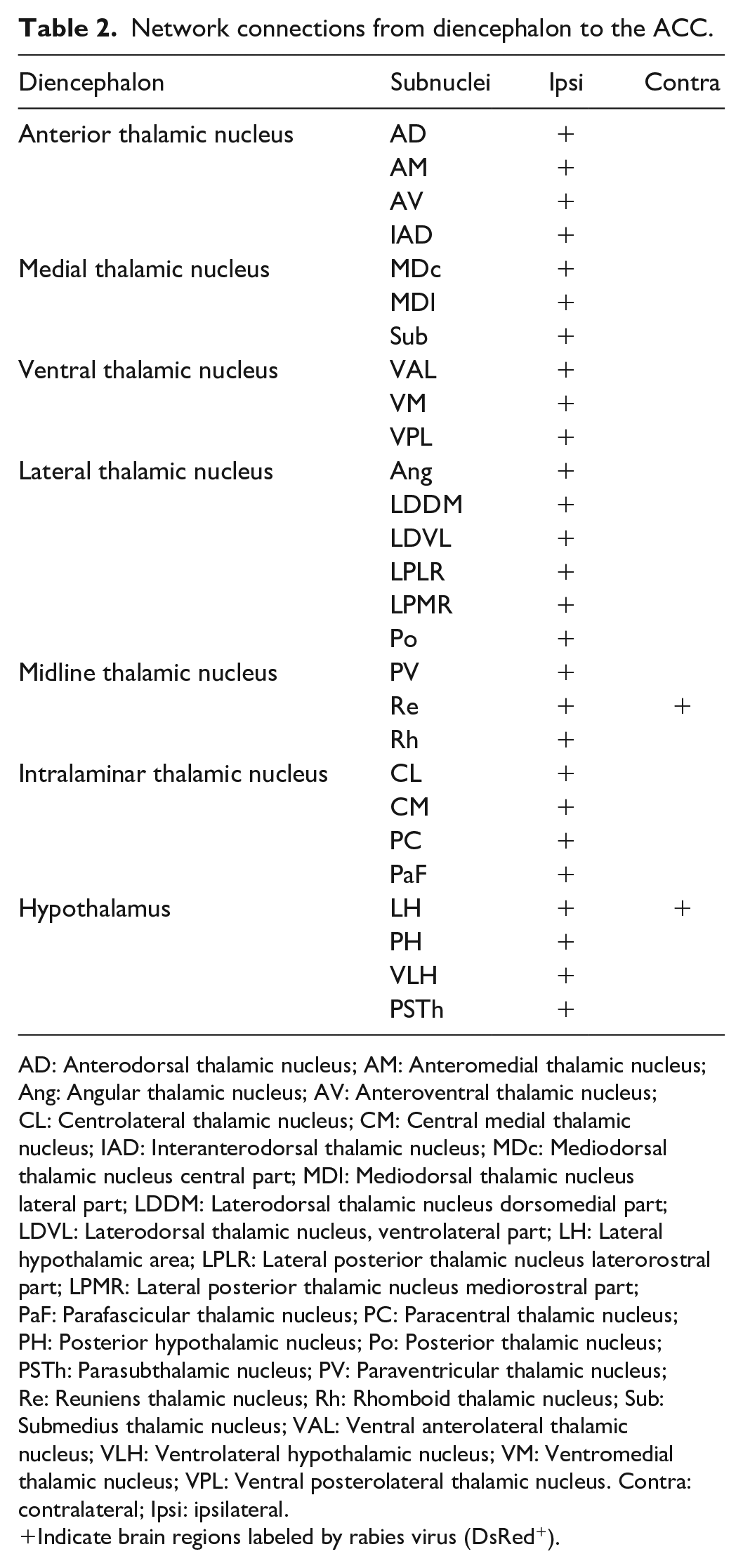
AD: Anterodorsal thalamic nucleus; AM: Anteromedial thalamic nucleus; Ang: Angular thalamic nucleus; AV: Anteroventral thalamic nucleus; CL: Centrolateral thalamic nucleus; CM: Central medial thalamic nucleus; IAD: Interanterodorsal thalamic nucleus; MDc: Mediodorsal thalamic nucleus central part; MDl: Mediodorsal thalamic nucleus lateral part; LDDM: Laterodorsal thalamic nucleus dorsomedial part; LDVL: Laterodorsal thalamic nucleus, ventrolateral part; LH: Lateral hypothalamic area; LPLR: Lateral posterior thalamic nucleus laterorostral part; LPMR: Lateral posterior thalamic nucleus mediorostral part; PaF: Parafascicular thalamic nucleus; PC: Paracentral thalamic nucleus; PH: Posterior hypothalamic nucleus; Po: Posterior thalamic nucleus; PSTh: Parasubthalamic nucleus; PV: Paraventricular thalamic nucleus; Re: Reuniens thalamic nucleus; Rh: Rhomboid thalamic nucleus; Sub: Submedius thalamic nucleus; VAL: Ventral anterolateral thalamic nucleus; VLH: Ventrolateral hypothalamic nucleus; VM: Ventromedial thalamic nucleus; VPL: Ventral posterolateral thalamic nucleus. Contra: contralateral; Ipsi: ipsilateral.
Indicate brain regions labeled by rabies virus (DsRed+).
Additionally, the hypothalamus serves as the primary control center for the endocrine and autonomic nervous system, which is responsible for regulating many key physiological functions, such as body temperature, hunger, thirst and stress, etc. 22 Here, we found that the ACC received presynaptic afferents from the lateral (LH), posterior (PH) and ventrolateral (VLH) hypothalamic nucleus. In particular, the LH bilaterally projects to the ACC. Also, the parasubthalamic nucleus (PSTh), a small nucleus located on the lateral edge of the posterior hypothalamus, displayed light labeling that directly projected to the ACC. These neuronal circuits may play important roles in regulating pain-induced feeding behaviors and sleeping disorders, even insomnia.
ACC neurons receive inputs from subcortical forebrain
Except for the cerebral cortex, we also found that other subcortical forebrain regions send direct projections to the ACC (Figure 3; Table 3). Among them, the claustrum (Cl) displayed dense labelings mostly at its rostral level, and lighter labelings were observed in the contralateral Cl. Bilateral labelings were also observed in the nucleus of the horizontal limb of the diagonal band (HDB) with ipsilateral preference. In the midline of the brain, the lambdoid (Ld) and medial septal zone (MS), the nucleus of the vertical limb of the diagonal band (VDB) also displayed bilateral DsRed+ neuronal labelings. Different from previous studies, 4 only unilateral labelings were observed in the amygdala, which contains the anterior and posterior parts of the basolateral amygdaloid nucleus (BLA and BLP), the medial part of central amygdaloid nucleus medial part (CeM) and the Extension of the amygdala (EA). Scattered DsRed+ neurons were always observed in the ipsilateral globus pallidus (GP) and the internal capsule (ic). In these subcortical connections, only partial neuronal circuits, such as BLA-ACC and Cl-ACC, were reported which can regulate pain, social and cognitive functions,23-26 further studies were still needed to explore functional characteristics of these neuronal circuits.

ACC neurons receive inputs from the subcortical forebrain. (a-f) Representative coronal sections indicate widespread labeling from the subcortical forebrain. Bilateral labeling was observed in the Cl, HDB/VDB, and the globus pallidus. Ipsilateral presynaptic inputs were distributed in the amygdala, the striatum and the internal capsule. The white arrows indicate presynaptic inputs from the subcortical forebrain to the ACC. Scale bar: 1 mm (white).
Network connections from subcortical forebrain to the ACC

BLA: Basolateral amygdaloid nucleus anterior part; BLP: Basolateral amygdaloid nucleus posterior part; CeM: Central amygdaloid nucleus medial part; Cpu: Caudate putamen; EA: Extension of the amygdala; GP: Globus pallidus; Ld: Lambdoid septal zone; MS: Medial septal nucleus. Contra: contralateral; Ipsi: ipsilateral.
Indicate brain regions labeled by rabies virus (DsRed+).
ACC neurons receive inputs from the brainstem
The brainstem was categorized into three major parts: midbrain, pons, and medulla oblongata. There were a few areas in the brainstem that provided weak but important innervations, which were located almost exclusively in monoaminergic centers (Figure 4; Table 4). 27 Similar with previous reports, the ipsilateral ventral tegmental area (VTA) showed light staining that directly projected to the ACC (Figures 4(a)). More caudally, the dorsal (DR), median (MnR) and paramedian (PMnR) raphe nucleus, as well as the rostral linear nucleus (RLi) were moderately labeled. Especially the PMnR, can bilaterally project to the ACC (Figure 4(b)). Finally, in the pons, the laterodorsal tegmental nucleus (LDTg) also showed light staining (Figure 4(c)). Simultaneously, the locus coeruleus (LC) is the main nucleus in the brain responsible for norepinephrine (NE) release, minor labeled neurons were observed in our present study (Figure 4(d)). These results indicated that the tegmental, raphe nuclei and the locus coeruleus are main regions of the brainstem which send direct inputs to the ACC.

Distributions of efferent neurons from the brainstem that can send direct projections to the ACC. (a–d) Representative coronal sections indicate widespread labeling from the brainstem. Light labeling were distributed in the brainstem that contained the LC, raphe and tegmental nuclei. The white arrows indicate presynaptic inputs from the brainstem to the ACC. Scale bar: 1 mm (white).
Network connections from brainstem to the ACC.

DR: Dorsal raphe nucleus; LDTg: Laterodorsal tegmental nucleus; MnR: Median raphe nucleus; PMnR: Paramedian raphe nucleus; RLi: Rostral linear nucleus; VTA: Ventral tegmental area. Contra: contralateral; Ipsi: ipsilateral.
Indicate brain regions labeled by rabies virus (DsRed+).
Discussion
The ACC is essential to some of the most vital functions of the brain, including pain processing, mood disorders and cognition.1,2,28 In order to understand the circuitry mechanisms of how the ACC modulates these behaviors, it is necessary to investigate the whole-brain inputs to ACC neurons. Traditional fluorescence imaging techniques on brain connectome required proportional sampling on serial thin brain sections, which results in a lack of continuity and axonal tracing. In this study, we provide a detailed analysis of whole-brain afferents to the ACC combined rabies virus-based trans-monosynaptic retrograde tracing system with the VISoR imaging method. These afferents arise mainly from widespread cortices and the thalamus, with light contributions from the forebrain and brainstem. These presynaptic inputs showed preferences for ipsilateral projections. Compared with traditional methods, our present study is more precise and efficient which provides a comprehensive map of the presynaptic partners that send direct projections to the ACC.
Implications for the role of ACC in pain and itching processing
Pain is an unpleasant sensory and emotional experience that serves a vital physiological function in all vertebrates by alerting the organism to the potential for tissue damage.1,29 Central sensitization is thought to be an important pathogenesis of chronic pain.1,30,31 Previous studies have revealed that many brain regions can be activated by various painful stimuli. In particular, the ACC formed complex neuronal networks with many cerebral cortices and subcortical regions, which were involved in pain and emotion processing. 1
We first summarized ACC-related neuronal circuits involved in pain and itching processing (Figure 5(a) and (b)). In the cerebral cortex, the cross-callosal projection of the ACC between two hemispheres contributes to mirror-image pain, 32 and this bilateral projection can also regulate pain-related anxiety. 33 Activation of the S1 axon terminals increases the response of ACC neurons to noxious stimuli, and modulation of this S1-ACC projection regulates chronic pain-related aversive responses. 9 Recent studies showed that activation of the V2MGlu-ACCGABA-Glu circuit mimics green light-induced antinociception in both neuropathic and inflammatory pain in mice. 34 Consistent with these studies, we also observed contra-ipsilateral projection within the ACC, and dense staining was also labeled in the S1 and V2M. In addition, other cerebral cortices, including the A25, insular cortex especially the anterior insula, M1, MCC, and the RSC,35-39 were also related to pain processing. Our present study provides sufficient anatomic evidence and found that these nuclei directly projected to ACC neurons. However, the functional characteristics and molecular mechanisms of these cortico-cortical neuronal circuits are still poorly understood, more studies are still needed for further explorations.
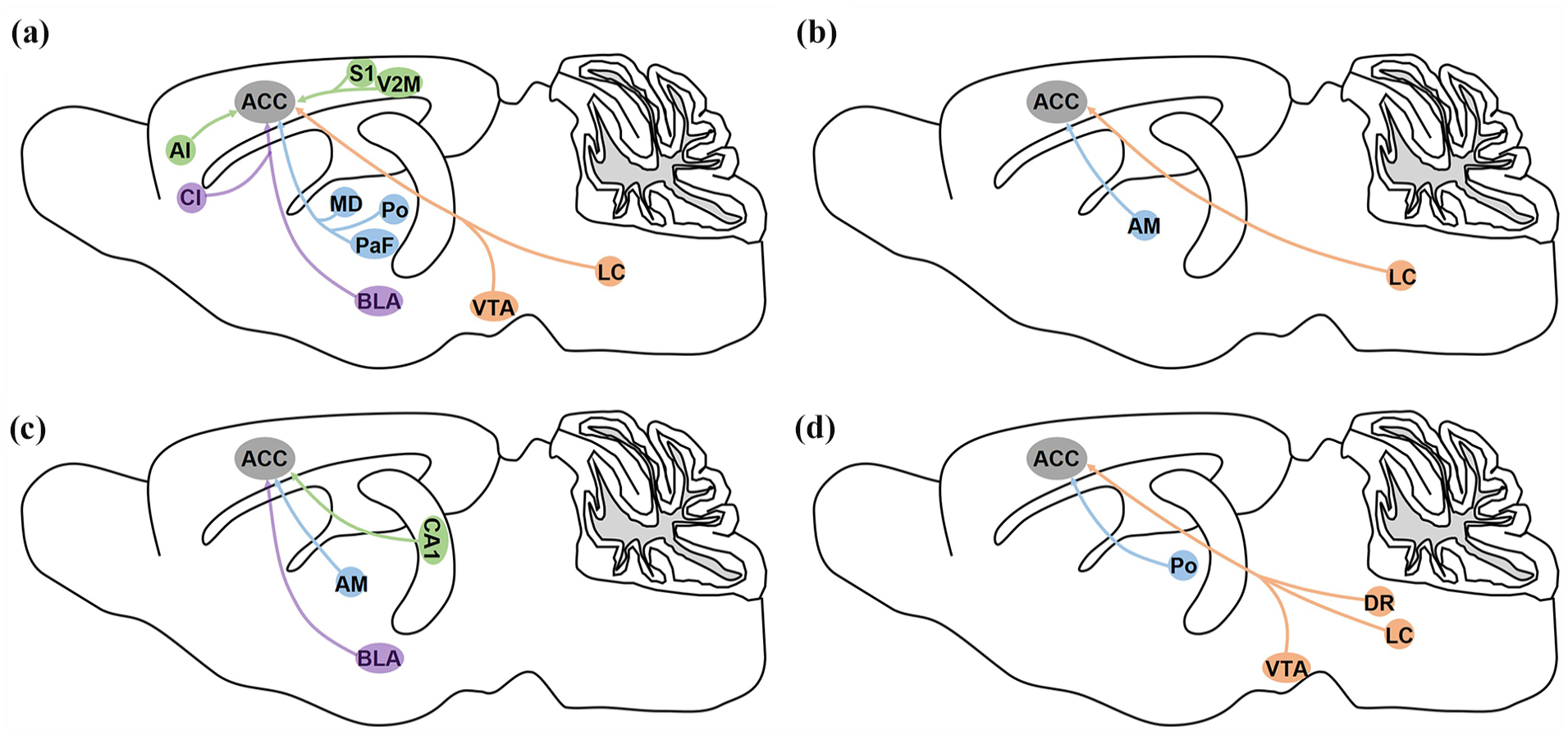
Summarized functional characteristics of whole-brain monosynaptic inputs to ACC neurons. (a) Afferents to the ACC are involved in pain sensation and pain-related behaviors. (b) Neuronal circuits from the AM or LC to the ACC are involved in itching. (c) Presynaptic inputs to the ACC can modulate memory and cognition. (d) The reward and sociability behaviors can be regulated by ACC-related neuronal circuits. The green circles represent neuron ensembles located in the cerebral cortex and the hippocampus; the blue circles indicate neuron ensembles located in the thalamus; the orange circles represent neuron ensembles located in the brainstem ; the purple circles indicate the forebrain regions.
Under pain processing, peripheral nociceptive information from somatic and visceral organs is conveyed indirectly to the ACC through the thalamus, mainly the medial thalamus. Pain experience can remodel the MD-ACC projection to modify nociceptive sensitivity and promote pain-related aversion. 40 Previous studies also showed that PaFGlu-ACCGABA-Glu mediates depression-induced allodynia. 41 Furthermore, the Po-ACC circuit is thought to be associated with migraine-related affective/motivational behaviors. 42 Then, the ACC also innervated to the spinal cord dorsal horn directly or indirectly through the periaqueductal gray (PAG) and rostral ventral medulla (RVM), which played an important role in the descending modulation of pain.43-45 Together, these neuronal connections form spinal dorsal horn-thalamus-cortex-spinal dorsal horn long-distance feedback loop for pain processing. Our results also showed that the ACC received a great number of presynaptic inputs from the AM, and the AM-ACC neuronal circuits can modulate histaminergic itch sensation. 46 Our previous works showed pruritogen-induced scratching corresponded with the enhanced glutamatergic excitatory transmission in layers II/III in the ACC which regulated downstream inhibitory circuits by specific kainate receptors (KARs). 47 Therefore, it is likely that AM-ACC circuits modulate itching behaviors through the activation of postsynaptic KARs. The circuit-related neuronal types will be specifically labeled for itching-related studies.
In addition to the cerebral cortex and thalamus, the BLA and the claustrum located in the forebrain were also labeled. The BLA, largely involved in pain and emotional processing, establishes connections with cortical pyramidal neurons but may also influence feed-forward inhibition by projecting to local parvalbumin inhibitory interneurons. Previous studies showed that the neuronal circuit from the BLA to the ACC was involved in depression-like behaviors and comorbidity with chronic pain. 24 The Cl-ACC played important roles in visceral and inflammatory pain processing.25,26 In the brainstem, an ACCGlu-VTAGABA-VTADA-ACCGlu positive-feedback loop-mediated the progression and maintenance of neuropathic pain and comorbid anxiodepressive-like behavior. 48 Noradrenergic projections from the LC innervated the ACC, which facilitated brain responses to pain and itch.8,49 However, in our present study, only weak staining was observed in the brainstem. As mentioned above, the brainstem contains rich monoaminergic neurons, however, our present study did not map cell-specific neuronal circuits, further studies are needed to explore these circuitry characteristics and underlying mechanisms in pain and itching processing.
Implications for the role of ACC in memory and cognition
The ACC plays an important role in shaping the plasticity of neuronal circuits linked to memory consolidation and cognitive functions, which are intricately linked with neurogenesis of the hippocampus and other memory-encoding brain regions. 50 We gave brief overview of ACC-related neuronal circuits involved in memory and cognition in Figure 5(c). Previous studies showed that activating Gi of CA1 astrocytes impaired memory acquisition and expression by inhibiting the activation of CA1 neurons which directly projected to the ACC. 51 Among the anterior nuclei, the AM forms the “extended hippocampal circuit” with the CA1, ventral subiculum, Ent and Ect, and with the RSC, which is largely involved in social defeat-associated contextual fear memory, spatial navigation, and spatial learning. Consistent with previous studies, our present study also found that the AM represents the main source of projection to the ACC. The anteromedial thalamus selects strong memories and connects with the ACC to selectively stabilize remote memories, which provides insights into how memories are selected and stabilized into long-term cortical storage. 52 The BLA-ACC circuit was also proved to be associated with memory and cognition.53,54 Nevertheless, there was still limited research on the role of ACC neuronal circuits in cognition and memory.
Besides, as described in Figure 5(d), ACC-related neuronal circuits, especially dopaminergic were also involved in reward and social recognition. Different from pain processing, dopaminergic projection of VTA-ACC neuronal circuits was also involved in the acquisition and maintenance of μ-opioid-induced place preference. 55 The LC-ACC neuronal circuits modulate parental pup retrieval behavior, inactivation of this noradrenergic pathway may disrupt parental care. 56 Furthermore, the serotonergic projection from the DR to the ACC is implicated in modulating consolation and sociability. 57
In summary, our results provide exact anatomic evidences for the whole-brain connectome of the ACC, which is a necessary step to address their roles in normal and pathological conditions. Our future study aims to map cell-specific neuronal circuits of the ACC by introducing cre-dependent transgenic mice and to clarify the underlying functional characteristics and circuitry mechanisms.
Footnotes
Acknowledgements
We would like to thank the EJLB-CIHR Michael Smith Chair in Neurosciences and Mental Health in Canada, Canada Research Chair, Ontario-China Research and Innovation Fund (OCRIF), Canadian Institute for Health Research operating and project Grants (MOP-124807; PJT-148648 and 419286) for funding support to M.Z.
Authors’ contributions
M.X., F.X., G.Q.B., X.H.L., X.X.Y., J.S.L., and M.Z. designed the experiments. M.X., Q.Y.C., W.T.S., and Z.X.Z., performed experiments and analyzed data. M.X., Q.Y.C., X.H.L., J.S.L., and M.Z. drafted the manuscript and finished the final version of the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
