Abstract
Background
While the PKCγ neurons in spinal dorsal horn play an indispensable part in neuropathic allodynia, the exact effect of PKCγ neurons of brain regions in neuropathic pain remains elusive. Mounting research studies have depicted that the anterior cingulate cortex (ACC) is closely linked with pain perception and behavior, the present study was designed to investigate the contribution of PKCγ neurons in ACC to neuropathic allodynia and pain-related emotion in newly developed Prkcg-P2A-Tdtomato mice.
Methods
The c-fos expression in response to innocuous stimulation was used to monitor the activity of PKCγ in CCI (chronic constriction injury of the sciatic nerve) induced neuropathic pain condition. Activating or silencing ACC PKCγ neurons by chemogenetics was applied to observe the changes of pain behavior. The excitability of ACC PKCγ neurons in normal and CCI mice was compared by patch-clamp whole-cell recordings.
Results
The PKCγ-Tdtomato neurons were mainly distributed in layer III-Vof ACC. The Tdtomato was mainly expressed in ACC pyramidal neurons demonstrated by intracellular staining. The c-fos expression in ACC PKCγ neurons in response to innocuous stimulation was obviously elevated in CCI mice. The patch clamp recordings showed that ACC PKCγ-Tdtomato neurons were largely activated in CCI mice. Chemogenetic activation of ACC PKCγ neurons in Prkcg-icre mice induced mechanical allodynia and pain-related aversive behavior, conversely, silencing them in CCI condition significantly reversed the mechanical allodynia and pain-related place aversive behavior.
Conclusion
We conclude that the PKCγ neurons in ACC are closely linked with neuropathic allodynia and pain-related emotional behaviors.
Introduction
Neuropathic pain is a pathological process characterized with allodynia, hyperalgesia, and spontaneous pain, which is always related with negative emotional reactions, tending to cause great disturbances in the life of patients. The PKCγ neurons in spinal dorsal horn (SDH) and medullary dorsal horn (MDH) have been proposed to be an imperative part of the neural circuits involved in neuropathic mechanical allodynia.1–5 By using a newly developed Prkcg-P2A-Tdtomato mice line, 2 we have found that spinal PKCγ neurons mainly received inputs from Aβ myelinated primary afferents carrying low-threshold mechanical information. The feed forward inhibitory circuit composed of PKCγ neurons and glycinergic neurons in SDH is accountable for the occurrence of allodynia.2,3 The electrophysiological and morphological characters of the feed forward inhibitory circuit have remarkable adaptive changes after peripheral nerve injury.2,4 In addition, the activation and silence of PKCγ neurons in spinal cord are closely correlated with allodynia.5–8
The PKCγ neurons are exclusively located in entire central nervous system (CNS) including spinal cord and diverse brain regions.9,10 Despite considerable advances have been made in researches that PKCγ neurons are highly related with chronic pain at the spinal level, their exquisite role for supraspinal pain modulation is poorly understood. It is universally acknowledged that the anterior cingulate cortex (ACC) is a prominent brain region, responsible for mood disorders, motivation, cognition, and action.11–16 It also has been consistently reported that the ACC acts an essential part in pain modulation, and 17,18 neurons in ACC are continuously excited during nociception and become overactive in chronic pain condition.19–23 Recent evidence also indicates that the variation of Glutamate and GABAergic neurotransmitter levels in ACC of animals is related with acute and chronic pain.24–26 A number of studies have demonstrated the prominent contribution of ACC to pain perception, however, the imperial role of PKCγ neurons in ACC with neuropathic pain remains to be illuminated, only a micro report supports that the PKCγ as synaptic protein is closely correlated with pain behavior by influencing the synaptic plasticity. 27
Therefore, we want to clarify the function of PKCγ neurons in ACC in neuropathic allodynia and pain-related emotion. We first confirmed the distribution of PKCγ neurons in brain regions including ACC in Prkcg-P2A-Tdtomato mice. Then combined with immunofluorescence, patch clamp recording and Chemogenetic methods investigated the effect of PKCγ neurons activities on neuropathic pain behavior. We provided the initial evidence that ACC PKCγ neurons contributed to neuropathic pain associated allodynia and pain-related emotional behaviors.
Materials and method
Animals
The Prkcg-icre mice and the Prkcg-P2A-Tdtomato mice were developed in our lab. 2 Prkcg-icre mice (6–8 weeks old) mice were utilized for behavioral experiments; Prkcg-P2A-Tdtomato mice (4–6 weeks old) were required in electrophysiological experiments. All mice were bred in SPF level laboratory room with 12h light–dark circle, and the temperature and the environment humidity were maintained at 22∼24°C and 20%, respectively. The usage and disposal of mice were in accordance with the requirement for caring laboratory animals. The experimental processes were approved by Fourth Military Medical University Ethics Committee. The mice were separated into different groups randomly, and all behavioral experiments were performed between 8:00 AM and 6:00 PM.
Patch clamp recording
The brain of Prkcg-P2A-Tdtomato mice (4–6 weeks old) was rapidly transferred into cold NMDG-HEPES artificial cerebrospinal fluid (aCSF: 92 NMDG, 92 HCL, 25 glucose, 1.2 Na2HPO4, 30 NaHCO3, 20 HEPES, 2.5 KCl, 5 Sodium ascorbate, 2 Thiourea, 3 Sodium pyruvate, 10 MgSO4, and 0.5 CaCl2) with precharged mixture (95% O2, 5% CO2). Coronal slices (300 μm) of ACC (1.7–0.8 mm rostral to the Bregma) were obtained by oscillating slicer (VT1000, Leica). Then the slices were removed into the incubation chamber full of HEPES holding aCSF, consisting of (in mM): 92 NaCl, 2.5 KCl, 2 CaCl2, 2 MgSO4, 20 HEPES, 1.2 NaH2PO4, 30 NaHCO3, 25 glucose, 5 Sodium ascorbate, 2 Thiourea, and 3 Sodium pyruvate, which was pre-filled with mixture (95% O2, 5% CO2) at 35°C. Finally, slice was removed into recording flume after incubating for at least 60 min, perfusing it with constantly aerated recording aCSF (124 NaCl, 2.5 KCl, 2 CaCl2, 2 MgSO4, 5 HEPES, 1.2 NaH2PO4, 24 NaHCO3, and 12.5 glucose), at a speed of 1–2 ml/min. Neurons in ACC with autofluorescence were selected for patch clamp whole cell recordings. The solution in recording electrodes was made up by (in mM): 145 potassium gluconate, 5 HEPES, 0.5 EGTA, 2 MgCl2, 5 K2ATP, and 5% biocytin (pH 7.2–7.4). The whole cell recording was completed by breaking the membrane after high resistance sealing.
Data was collected after stabilizing at least 5 min. Neurons would be abandoned if resistance was more than 20 MΩ or the resting membrane potential (RMP) was higher than −50 mV. The membrane test was conducted in V-Clamp program, RMP was obtained in “I=0” mode. The intensity of step current (25 ms) to induce first action potential (AP) was named rheobase. The threshold of AP was defined as the amplitude in 1/3 of the derivative of AP. The amplitude of AP referred to the difference between maxima and baseline of the AP. Signals were acquired by Axopatch 200B amplifier (Molecular Devices, USA), digitized at 10 kHz with a digitizer (Digidata 1440A, Molecular Devices) and analyzed with pClamp10.0 software (Molecular Devices).
Immunofluorescence
The adult male Prkcg-P2A-Tdtomato mice (6–8 weeks old) in sham and CCI group received stimulations given by 0.4 g von-Frey fiber with six circles at 5-minute interval. Stimulus was given to each mouse 10 times at 5s intervals in one circle, and the duration of stimulus would be no more than 3 s. Two hours after mechanical stimulation, the mice were deeply anesthetized with pentobarbital sodium (0.5 mg/10g), and then perfused by 20 mL saline and 40 mL 4% paraformaldehyde (PFA). After that, the brain was continually fixed and dewatered with 20% and 30% sucrose successively at 4°C. Then the tissues were cut into 20 μm slices by Cryostat Microtome (Leica). After washing three times in the 0.1×PBS, the sections first reacted with rabbit anti-c-fos antibody (diluted 1:1000; SYSY) for or rabbit anti-PKCγ antibody (diluted 1:200; GENETEX) 12–18 h at 4°C. Then they were incubated in secondary donkey anti-rabbit IgG conjugated with Alexa Fluor 488 (diluted 1: 500, Molecular Probes-A21206, USA) for 2–3 h at RT. Finally, sections were washed for three times and covered with anti-fluorescence reagent. In refer to our published study, 2 once the electrophysiological experiment finished, the slices were fixed and then dehydrated, washed with Tris-Triton (TT) buffer for three times. Time of blocking in 4% normal goat serum TT buffer and incubation in SA5001 (1:500; Vector labs) at 4°C were 1 and 24 h, respectively. On alternate days, washing slices by Tris buffer for three times, prepared for confocal analysis.
Viral injection
To chemogenetically activate or inhibit the PKCγ neurons in ACC, the adeno-associated viruses (AAVs) (0.2 μL) rAAV-Ef1a-DIO-hM3D (Gq)-mCherry-WPREs or rAAV-Ef1a-DIO-hM4D (Gi)-mCherry-WPREs (0.2 μL) or rAAV-Ef1a-DIO-mCherry-WPREs (0.2 μL) were microinjected into ACC (AP: +1.2; ML+/-0.3 DV: −1.2) of three groups of Prkcg-icre mice (0.1 μL/min). At least 10 min was kept before withdrawing needle to guarantee the sufficient diffusion. The expression of AAVs requires 3 weeks.
Surgery
CCI models (chronic constriction injury of the sciatic nerve) were conducted, referring to methods published before. 28 Briefly, the mouse (3 weeks) was anesthetized by the 3% isoflurane in oxygen, which should be reduced to 1.5% to ensure the anesthesia state. The unilateral sciatic nerves were quickly exposed at mid-thigh level, 5 mm of that was freed of surrounding connective tissue carefully. Then three knots were laced up loosely round the sciatic nerve with 5–0 suture from distal to proximal, 1 mm apart, with the first knot next to the trifurcation. The proper tightness depended on the slight tremors of the hind limb. After that, the skin was sutured. The mice would behave as mild valgus or have slight limp if operated correctly. Mice with severe motor dysfunction of operational limb were abandoned.
Behavioral measurement
The mice were placed on the experimental environment to habituate for 30 min in following 3 days before the formal testing. Results were compared before and after CNO (ip 0.5 mg/mL 0.2 mL) or saline (ip 0.2 mL) injection.
Conditioned position preference
The following experimental procedures referred to the protocols described in the latest articles with little modification.18,30 Two large conditioning partitions (20 cm × 20 cm × 20 cm), which is recognized by distinct visual, tactile stimuli, constitute the test apparatus. A door in the middle allows the mice to move freely between the two spaces. In preconditioning days (Days 1), the mice could explore freely for 30 min at beginning, only the last 15 min were recorded. Analysis software (Yuyan technology) was used to record and analyze data blindly. Once mice spent more than 70% time in one compartment, they would be excluded from the following research studies. Days 3–6 were conditioning days, on days 2 and 4, mice received CNO (ip 0.5 mg/mL 0.2 mL) or saline (ip 0.2 mL) were restricted to one chamber for 45 min. On days 3 and 5, no treatment was applied and mice were restrained in another partition for 45 min. On the sixth day, mice were allowed to move freely in two chambers for 15 min. We recorded the time mice spent in each compartment and compared with that in the same chamber in the day 1.
Imaging
Nine slices from three Prkcg-P2A-Tdtomato mice were used to analyze the co-expression of Tdtomato and PKCγ, 18 slices from three normal, and 3 CCI model mice were used to analyze the co-expression of PKCγ-Tdtomato and c-fos. Images were obtained by the Olympus FV1200 confocal microscope. Quantification of overlay was performed on FIJI using the Cell Counter Plugin. The brain slices were scanned in the z stack model to obtain the complete neuronal images with 2 μm thick per step. The sholl analysis was widely used to quantify the complexity of neuronal dendrites, and was an essential tool in neurobiology. The morphologic characteristics of PKCγ-Tdtomato neurons in ACC of normal and CCI mice were evaluated by sholl analysis with FIJI software.
Data analysis
All data were demonstrated as mean ± SEM. Unpaired Student’s t tests was used to analyze single-variable differences. One-way or Two-way analysis of variance (ANOVA) followed by Bonferroni posttest was used to evaluate differences in three or more groups. Chi-square test or Fisher exact test was applied to assess differences in proportion between groups. Prism GraphPad8.0 software was used to prepare the diagram. Data analysis was conducted by SPSS22.0 software.
Results
The PKCγ neurons are abundant in anterior cingulate cortex
We first observed the whole brain mapping of the PKCγ neurons in Prkcg-P2A-Tdtomato gene knock-in mice. The fluorescent protein was found in the following regions (Figure 1(a)): the olfactory area (anterior olfactory area), the hippocampal formation (CA1 region), the amygdala (basolateral amygdaloid nucleus), the cerebellum (pyramidal layer), medulla oblongata (superior vestibular nucleus; vestibulocerebellar nucleus; lateral vestibular nucleus; superior vestibular nucleus), and so on. The results also revealed that the PKCγ-Tdtomato neurons were widely distributed in anterior cingulate cortex (layers II–VI), while the cell bodies of these neurons were mainly located in layers III–V (Figures 1(b) and (c)). Double staining showed that 95.6 ± 0.7% (
Based on the abundant distribution of PKCγ neurons in ACC and the close association between ACC and pain perception, we first compared the rate of activated PKCγ neurons in ACC between the control and neuropathic pain model groups by applying c-fos which is a common marker for detecting neuronal activities. Six male adult mice were separated into control and CCI groups randomly. The mice in both groups were anesthetized with pentobarbital sodium (0.5 mg/10g) and perfused 2 h after innocuous mechanical stimulation. The results of immunofluorescence staining were shown in Figure 2(a) and (b). There is little difference in total number of PKCγ neurons in normal and CCI mice (117.333 ± 7.641 vs 138 ± 13.736, 
The results above suggested that the PKCγ neurons in ACC were obviously activated in response to innocuous stimulation in CCI mice, so we next wanted to further clear the changes in the excitability of PKCγ neurons in mice with neuropathic pain by whole-cell recordings (Figure 3(a)). The electrophysiological and morphological features of PKCγ neurons in ACC of normal mice and neuropathic pain model mice were compared. We observed that the negative and positive membrane properties in normal and CCI mice exhibited no statistical difference (Figures 3(d)–(k)), except discharge frequency (paired t test, **** Electrophysiological features of PKCγ neurons. Data are shown as means ± SEM, Comparison of holding potential (Hold), membrane resistance (Rm), input resistance (Ra), membrane capacitance (Cm), resting membrane potential (RMP), rheobase, action potential threshold, amplitude, duration and half-width of PKCγ ACC neurons between normal and CCI mice. Students t-test, normal versus CCI, **



Chemogenetic activation or inhibition of the PKCγ neurons in ACC induces or alleviates mechanical allodynia
To explore the influence of ACC PKCγ neurons activities on pain-related behavior, we adopted chemogenetics to either activate PKCγ neurons by hM3Dq or inhibit them by hM4Di in Prkcg-icre mice. The injection site was chosen according to the location of PKCγ neurons in ACC as above mapping results depicted (Figure 6(a)). The experimental process was described in Figure 6(b). After behavioral experiment, all mice were perfused to confirm the virus expression. For normal mice, the instant activation of PKCγ neurons by CNO injection (ip) led to mechanical allodynia in 1–6 h after the CNO injection (figure 6(c) and (d)), rather than the thermal pain hypersensitivity (Figure 6(e)), while the acute silence of the PKCγ neurons had not detectable changes in both mechanical pain and thermal pain threshold, as same as the mCherry group (Figures 6(c)–(e)). For neuropathic pain model mice, we got the basal values of animals the day before CCI surgery. At the seventh day after surgery, all mice were tested again to ensure the success of pain model, following by CNO injection (ip). The CNO injections in hM4Di group largely alleviate the mechanical allodynia (figure 6(f) and (g)), while the activation of PKCγ neurons has no effect on the PWMT (paw withdrawal mechanical threshold), dynamic score, and PWTL (paw withdrawal thermal latency) of mice with established neuropathic pain. There was also no significant change in the mCherry group after CNO injection (Figure 6(h)). Taken together, these results demonstrated that the activation or inhibition of PKCγ neurons in ACC significantly exacerbates or alleviates neuropathic allodynia.
The activities of PKCγ neurons in ACC were also associated with pain-related emotion
As pain sensations are usually accompanied with emotional reactions, we wanted to test whether PKCγ neurons in ACC are associated with pain-related emotion. In the current research, chemogenetics-based methods, that activation of PKCγ neurons in normal mice and inhibition of PKCγ neurons in CCI models, were used to investigate the pain-related aversive and preferable behavior in Prkcg-icre mice. The operational process was shown in the Figure 7(a). The normal mice expressed with hM3Dq spent apparently less time in the chamber paired with CNO injection after conditioning treatment (figure 7(b) and (c), 116.2 ± 22.41, ****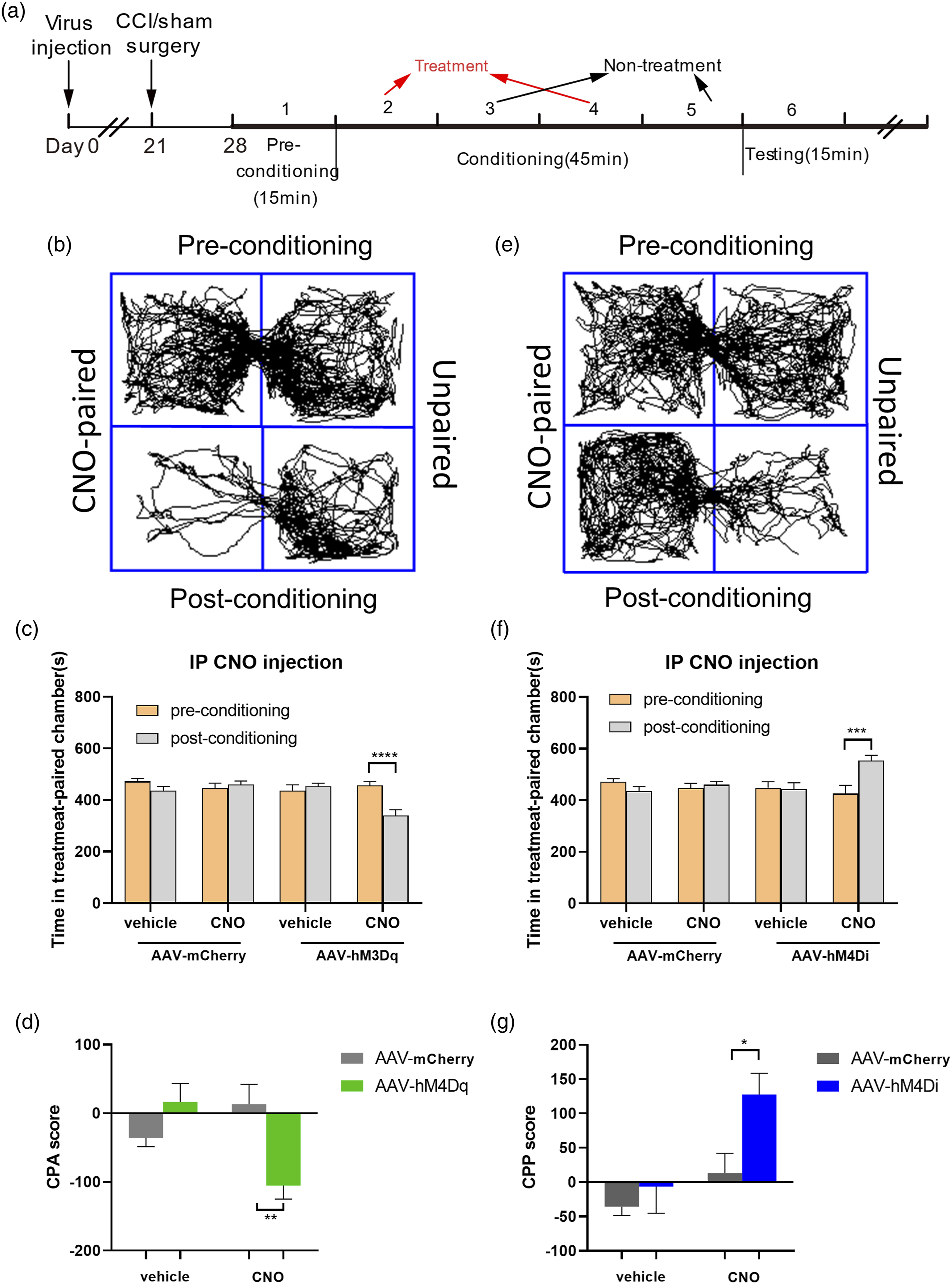
Discussion
The present study explored the contribution of PKCγ neurons in ACC to neuropathic allodynia and pain-related emotion in newly developed Prkcg-P2A-Tdtomato mice. The PKCγ-Tdtomato was mainly expressed in pyramidal neurons located in layers III–Vof ACC demonstrated by intracellular staining. The innocuous stimulation evoked more c-fos expression in ACC PKCγ neurons of CCI mice than that of normal mice. The ACC PKCγ neurons exhibited hyperexcitability in CCI mice as reviewed by patch clamp recordings. Chemogenetic activation of ACC PKCγ neurons in Prkcg-icre mice induced mechanical allodynia and pain-related aversive behavior, conversely, silencing them in CCI condition significantly reversed the mechanical allodynia and pain-related aversion.
Previous studies demonstrated that ACC plays a pivotal part in modulation of pain; however, few studies have been able to define the part of contribution by different subtypes of neurons in ACC. In addition, the role of PKCγ neurons in ACC in mechanical allodynia is rarely mentioned equally. In this article, consistent with the critical function of the ACC in pain modulation, we further revealed the imperial relationship between PKCγ neurons in ACC and neuropathic pain. We proposed that the PKCγ neurons in ACC function as a manager of enhancing or alleviating neuropathic pain behavior under normal and pathological states.
Widely acknowledged theory indicated that the PKCγ was distributed in the CNS, and greatly existed in many important functional structural areas like the hippocampus, amygdala complex, and SDH, implying its vital part in the corresponding function. 32 However, the specific location of it in CNS is not well elucidated. In previous research, autoradiography (labeled ligands or GTPγS), in situ hybridization, and antibody-based techniques are usually used,1,9,10 which offers limited information due to technical defects and human factors. Here, we applied fluorescent-labeled animals and achieve co-expression of PKCγ with td-tomato by using CRISPA-CAS9 technique, inserting td-tomato gene into PKCγ gene order. 2 In this research, we mainly elucidate the location of PKCγ in the CNS including ACC, laying a good foundation for further understanding of the function of PKCγ neurons in brain.
As we all know, c-fos is a classic marker of neuronal activity 33 ; in this article, we find that no matter in normal or pathological condition, the activation rate of neurons in ACC was very high, but the co-expression rate of PKCγ and c-fos was higher in neuropathic pain group, demonstrating that the PKCγ neurons were motivated in neuropathic pain condition. The ACC is a vital brain region in charge of advanced cognitive and affection function,18,34 various daily activities like crawling, feeding, and drinking cannot be separated with neuronal activities in ACC, let alone the mechanical stimulation. What’s more, in vitro experiment, our electrophysiological study demonstrated that the excitability of PKCγ neurons in ACC of CCI mouse was truly elevated.
In research of exploring the effect of descending serotonergic (5-HT) pathways to mechanical allodynia, the morphological restructure of PKCγ neurons by 5-HT2AR activation was found to contribute to open the gate for allodynia. 35 For this reason, we therefore investigated whether PKCγ neurons in ACC had morphological changes during neuropathic pain by comparing the structural morphology of PKCγ neurons naïve and CCI model mice. But no significant differences were found, indicating that the morphology of PKCγ neurons in ACC is not related with neuropathic pain.
We applied hM3Dq and hM4Di (hM4Di can silence neurons, while the hM3Dq can activate them) in this experiment, so that we can testify the therapeutical effect by acutely suppressed PKCγ neurons in ACC to the developed neuropathic pain. Moreover, our result that the silence of PKCγ neurons in normal condition has no effect on pain threshold suggested the PKCγ neurons in ACC are at rest state and do not take part in maintaining normal mechanical threshold. Meanwhile, the effect of activating PKCγ neurons in CCI mice demonstrated that the activation of PKCγ neurons in ACC can fully induce allodynia.
Although many research studies have suggested that the activity of neurons in ACC can simultaneously raise or decrease the mechanical and thermal pain threshold of animals, most of them are based on the non-selective activating or inhibiting neurons in ACC.36,37 Meanwhile, it has been proved that the regulation of mechanical pain and thermal pain in ACC is independent of each other or definitely be opposite from each other. For example, T2DM (Type 2 diabetes mellitus) mice developed increased thermal pain threshold and reduced mechanical pain threshold which can be regulated by NRSF/REST levels in ACC. 38 Additionally, it was reported that sleep deprivation or pharmacologic enhancement of EEG δ power can dramatically decrease mechanical pain thresholds, but not thermal thresholds, in a partial sciatic-nerve ligation model of neuropathic pain mice. 39 Stimulus in Hargreaves test could be regarded as a noxious stimulation; however, the result based on the complete activation or inhibition of PKCγ gene shows that it has no effect on the occurrence of acute pain.40,41 Therefore, it’s possible that the PKCγ neurons in the ACC are only involved in mechanical pain processing instead of thermal pain, but a definite conclusion needs further investigation.
In conclusion, this study identified that the PKCγ neurons in ACC are also closely related with the development of neuropathic allodynia and pain-related emotion.
Supplemental Material
sj-pdf-1-mpx-10.1177_17448069211061973 – Supplemental Material for The PKCγ neurons in anterior cingulate cortex contribute to the development of neuropathic allodynia and pain-related emotion
Supplemental Material, sj-pdf-1-mpx-10.1177_17448069211061973 for The PKCγ neurons in anterior cingulate cortex contribute to the development of neuropathic allodynia and pain-related emotion by Xiao Zhang, Peng Liu, Xiaolan He, Zhenhua Jiang, Qun Wang, Nan Gu, and Yan Lu in Molecular Pain
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by Yan Lu grants from the National Natural Science Foundation of China (31530090, 81971058).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
