Abstract
Different brain areas have distinct roles in the processing and regulation of pain and thus may form specific pharmacological targets. Prior research has shown that AMPAkines, a class of drugs that increase glutamate signaling, can enhance descending inhibition from the prefrontal cortex (PFC) and nucleus accumbens. On the other hand, activation of neurons in the anterior cingulate cortex (ACC) is known to produce the aversive component of pain. The impact of AMPAkines on ACC, however, is not known. We found that direct delivery of CX516, a well-known AMPAkine, into the ACC had no effect on the aversive response to pain in rats. Furthermore, AMPAkines did not modulate the nociceptive response of ACC neurons. In contrast, AMPAkine delivery into the prelimbic region of the prefrontal cortex (PL) reduced pain aversion. These results indicate that the analgesic effects of AMPAkines in the cortex are likely mediated by the PFC but not the ACC.
Introduction
Chronic pain is highly prevalent, and it is associated with a range of morbidities. 1 Meanwhile, many of the currently used analgesics, such as opioids, are sedating and can even cause life-threatening acute respiratory depression.2–6 Thus, there is an urgent need for new, effective, and safe analgesics.3,7
Previous studies have shown that pain triggers changes in synapses and circuits in the cerebral cortex.8–10 In particular, the prefrontal cortex (PFC) has been shown to play an important role in the top-down regulaition of pain.11–14 The anterior cingulate cortex (ACC), in contrast, is involved in the processing of the aversive response to pain,15–19 and activity in this region increases in response to noxious stimuli.20–25 The activation of ACC, in turn, enhances pain aversion.23,26
Excitatory glutamate signaling in neurons from different regions of the brain strongly impacts the regulation of pain behaviors, 7 and this is especially relevant for signaling through α-Amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptors, which are the primary glutamate receptors in the central nervous system. 27 AMPAkines are a class of synthetic compounds developed to enhance glutamate transmission in a use-dependent manner by binding to an allosteric site on the AMPA receptor.28,29 There is substantial animal data to suggest that systemic delivery of AMPAkines can reduce pain,7,30,31 and that at least some of its analgesic effects are mediated by increasing glutamatergic signaling in the PFC and nucleus accumbens, likely through descending inhibition.30–32 It is not known, however, if AMPAkines can alter nociceptive processing in the ACC.
Here, we studied the effect of AMPAkines on the pain-aversive processing of neurons in the ACC of rats. We found that direct delivery of CX516, a commonly used AMPAkine, into the ACC did not alter pain-aversive responses in rats. This is in contrast to the anti-aversive effect of this drug when delivered to the prelimbic PFC (PL-PFC). Furthermore, we imaged time-lapse calcium (Ca2+) activity in the ACC and found that systemic delivery of AMPAkines did not result in significant changes in nociceptive activity in ACC neurons. These results suggest that AMPAkines likely have site-specific effects in the brain.
Results
AMPAkine administration in the ACC does not modulate acute pain aversion
AMPAkines are known to produce analgesic effects when delivered systemically.7,30,31 To investigate the impact of this class of drugs in the ACC, we tested the analgesic effects of delivery of CX516, a well-known AMPAkine, directly into the ACC, using a conditioned place aversion (CPA) assay.23,33 In the CPA assay, rats were first placed in the two-chambered apparatus and allowed to move freely (Figure 1(a)). During the conditioning phase, one chamber was paired with a noxious pin prick (PP) stimulus to the hind paw, and the other chamber was paired with no external stimuli (NS) (Figure 1(a)). The rats were then allowed free access to both chambers during the testing phase with no further stimuli applied, and avoidance of either chamber during this phase indicates a pain-aversive response. In order to investigate the effects of CX516 on pain-aversive behaviors, we injected CX516 versus saline (control) bilaterally into the ACC before starting the CPA assay (Figure 1(b)). Naïve rats that received an intracranial CX516 injection did not demonstrate any differences in their mechanical withdrawal threshold compared to naïve rats injected with saline (Figure 1(c)). Rats that received a control injection showed an avoidance of the PP-paired chamber during the testing phase, demonstrating expected pain-aversive response.23,33–36 However, rats that received CX516 also showed this avoidance, indicating that this drug in the ACC does not provide any analgesic or anti-aversive effects (Figure 1(d), (e)). To quantify the difference between the saline and CX516 groups, we compared the CPA scores of rats that received saline injections versus rats that received CX516 injections (Figure 1(f)). CPA scores are calculated by taking the time a rat spent in the PP chamber during testing phase and subtracting that from the time the rat spent in the same chamber during the preconditioning phase. A higher CPA score indicates greater avoidance of and thus aversion to the PP treatment. We did not find a significant difference in the CPA scores between saline and CX516 groups, indicating a lack of anti-aversive effect of CX516 in the ACC. Intracranial injection of CX516 into the ACC does not significantly affect aversive response to acute pain (a) Timeline of conditioned place aversion behavioral assay (CPA) and diagram illustrating the CPA apparatus and treatments during conditioning. A noxious pin prick (PP) stimulus was paired with one chamber of the CPA apparatus, while the other chamber had no noxious stimuli (NS). The pin prick was applied every 30 s to the rat’s hind paw. (b) Schematic detailing the intracranial injections into the anterior cingulate cortex (ACC); rats either received saline or the AMPAkine (CX516). (c) An intracranial CX516 injection to the ACC did not decrease the withdrawal threshold compared to saline in naïve rats. (n = 4 per group, two-way ANOVA test) (d) Rats intracranially injected with saline in the ACC displayed avoidance of the chamber associated with PP. (***p = .0003, n = 8, paired t test) (e) Rats intracranially injected with CX516 in the ACC displayed avoidance of the chamber associated with PP. (*p = .0234, n = 8, paired t test) (f) Intracranially injecting CX516 or saline did not produce a significant change in CPA scores. (p = .1751, n = 8, unpaired t test). Data are represented as mean ± SEM.
AMPAkine administration in the PL-PFC provides a reduction in pain aversion
CX516 has been shown previously to provide analgesic effects when delivered directly into the PL-PFC of rats.
31
To confirm these findings, we injected CX516 versus saline directly into the PL-PFC, followed by CPA tests (Figure 2(a)). Rats that received saline injections still avoided the PP chamber, demonstrating aversion to the nociceptive treatment (Figure 2(b)). However, after local infusions of CX516 into the PL-PFC, rats did not show a preference for either chamber during the testing phase, indicating a decrease in pain aversion (Figure 2(c)). The CPA scores for saline and CX516 further qualified the anti-aversive or analgesic effects of this drug in the PL-PFC (Figure 2(d)). These CPA results strongly indicate that AMPAkines target AMPA receptors in the PL-PFC to decrease pain aversion to acute noxious stimuli, in contrast to its lack of therapeutic activity in the ACC. Intracranial injections of CX516 into PL-PFC decreases aversive response to acute pain (a) Schematic detailing the intracranial injections into the prelimbic-prefrontal (PL-PFC); rats either received saline or CX516. (b) Rats intracranially injected with saline in the PL-PFC displayed avoidance of the chamber associated with PP. (***p = .0003, n = 8, paired t test) (c) Rats intracranially injected with CX516 in the PL-PFC displayed decreased aversion to the chamber associated with PP. (p = .8170, n = 8, paired t test) (d) Intracranial injections of CX516 in the PL-PFC induced a decreased aversive response, as shown by the decreased CPA score with the rats with CX516 injections. (**p = .0041, n = 8, unpaired t test). Data are represented as mean ± SEM.
Systemic AMPAkine administration does not alter calcium activity in the ACC
To further study the effect of AMPAkines on ACC, we used in vivo time-lapse calcium (Ca2+) imaging to measure how CX516 can modulate the activity of individual neurons in the ACC in response to the noxious (PP) stimulus in awake, freely-moving rats (Figure 3(a)). We injected GCaMP6f into one side of the ACC and implanted a gradient-index (GRIN) lens in each rat (Figure 3(b)). We then attached a single-photon miniscope to the GRIN lens to track Ca2+ activity within CAMKII-expressing neurons, which are mostly excitatory neurons.37,38 For each recording session, we measured Ca2+ activity for 5 min with no external stimuli for a baseline measurement (Figure 3(c)). We then measured Ca2+ activity after the delivery of a non-noxious von Frey (vF) mechanical stimulus versus the delivery of the PP stimulus. We repeated these measurements following a systemic injection of CX516 to compare neural activity before and after AMPAkine injections. We identified sub-populations of neurons that responded to non-noxious (vF) or noxious (PP) inputs, and analyzed the Ca2+ activity of these neurons (Figure 3(d)–(g)). First, we examined at the average peak of ΔF of Ca2+ response to noxious versus non-noxious stimuli and found that, as expected from previous studies,23,33,39 the neural response in the ACC was significantly higher to PP than vF (Figure 3(h)–(i)). However, we did not find any substantial differences in Ca2+ activity in response to noxious stimuli after CX516 injection (Figure 3(g), (j)). We then selected a subset of pain-responsive neurons, and found that there were no substantial differences in Ca2+ activity in response to noxious stimuli after CX516 injection in these pain-responsive neurons either (Figure 3(k)). These findings are in sharp contrast to the modulation of the nociceptive responses of neurons in the PL-PFC by AMPAkines shown in previous studies,
31
and they further support our behavioral findings that AMPAkines do not significantly modulate nociceptive response in ACC neurons to impact pain-aversive behaviors. CX516 does not significantly affect ACC peak calcium fluorescence after noxious and non-noxious stimuli. (a) Schematic illustrating calcium imaging experiments. (b) Gradient-index (GRIN) lens placement in the ACC. (c) Timeline of the calcium imaging experiments. (d and e) Map of sample identified contours in the maximum projection image after motion correction and (e) their activity traces. (f) ACC neurons regions of interests (ROIs) with their corresponding contours over the imaging field of view. (g) Representative traces of neuronal ROIs in response to vF and PP, before and after CX516 administration. (h) Enhanced peak activity of ACC neurons in response to PP stimuli compared to vF stimulation without CX516 injection. (n = 331 neurons from five rats; p = .0165, paired t test). (i) Neurons in the ACC demonstrated increased peak activity after PP stimuli versus vF stimuli, post-systemic injection of CX516. (n = 343 neurons from five rats; p = .0457, paired t test). (j) After CX516 injection, neurons in the ACC did not show a difference in peak Ca2+ fluorescence in response to noxious PP stimulations. (Baseline: n = 331 neurons from five rats; CX516: n = 343 neurons from five rats; p = .4989, unpaired t test). (k) After CX516 injection, PP-responsive neurons in the ACC did not show a difference in peak Ca2+ fluorescence. (Baseline: n = 47 neurons from five rats; CX516: n = 60 neurons from five rats; p = .8366, unpaired t test.)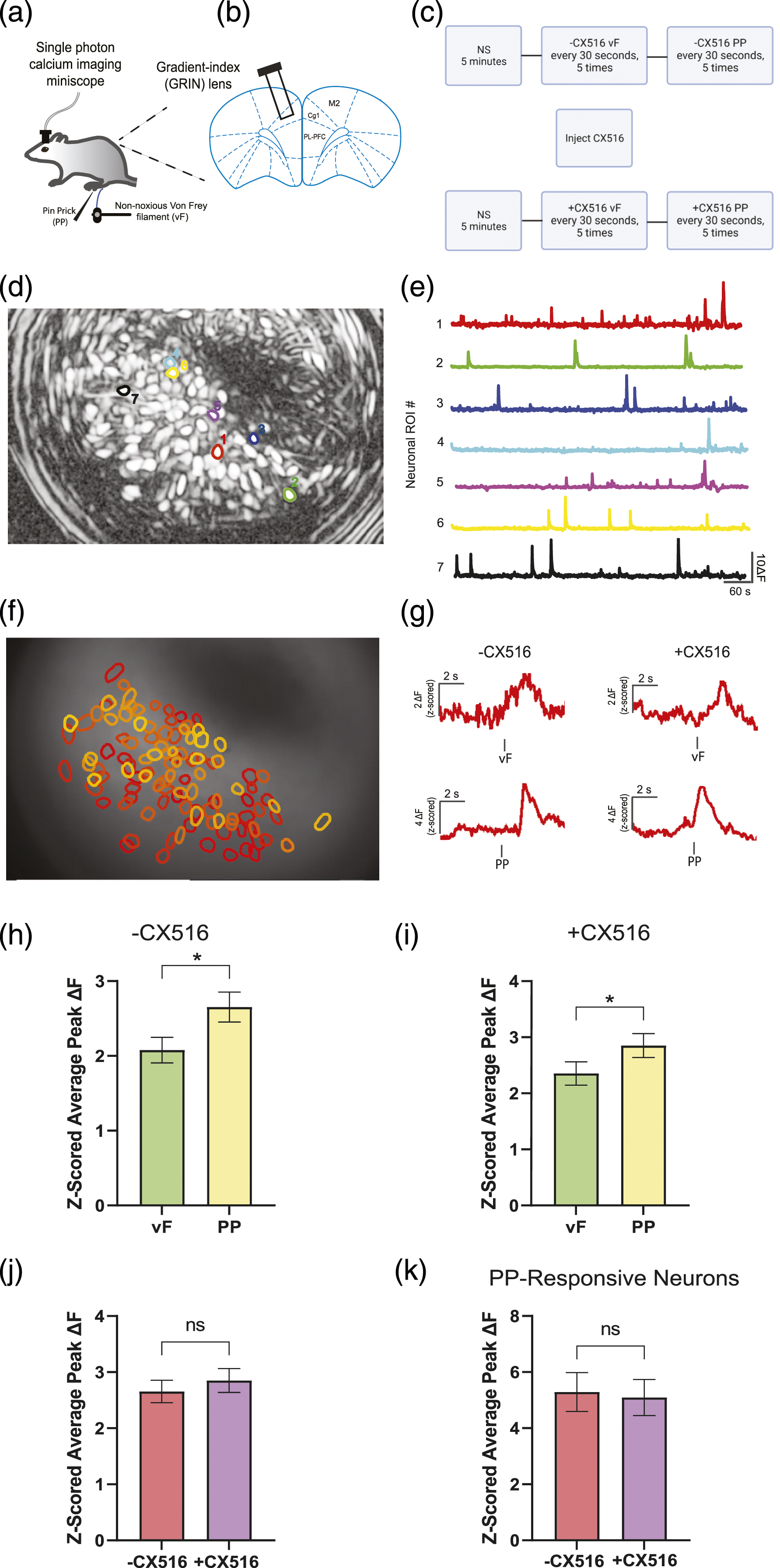
Discussion
In this study, we found that CX516, a well-known AMPAkine, has differential effects in the PL-PFC and ACC. Although it reduced pain aversion in the PL-PFC, it demonstrated no analgesic effects in the ACC, nor did it modulate nociceptive response in ACC neurons. These results indicate that this class of analgesics likely have site-specific effects in the brain.
Sedation and respiratory depression caused by opioids and other sedatives are serious side effects,40–45 raising the need for new and safe analgesics. By increasing excitatory neurotransmission in the pre-Botzinger complex in the medulla, AMPAkines can even stimulate the respiratory drive46–50 to treat or prevent hypoventilation caused by sedative medications often used to treat pain.50–53 Recently, AMPAkines have also been shown to treat both sensory and affective components of pain in animal models, and thus, studies of their analgesic mechanisms and sites of action can further support their therapeutic potential.
A key mechanism of AMPAkines is enhancement of glutamate signaling through AMPA receptors. Indeed, glutamate signaling in the central nervous system (CNS), especially signaling through AMPA receptors plays an important role in regulating pain sensitivity as well as mood. 54 However, AMPA receptor signaling can have both pro- and anti-nociceptive effects, depending on the target region in the CNS. Activation of the AMPA receptors in the spinal dorsal horn and amygdala has been shown to increase pain sensitivity.55–63 In contrast, AMPA receptor signaling in the PL-PFC, PAG, and NAc provides descending inhibition to the spinal cord to relieve pain.7,30,64–67 Our results here support the analgesic effects of AMPAkines in the PL-PFC but not the ACC, and these results are compatible with previous results showing that AMPAkine delivery into the PL-PFC could alter sensory allodynia associated with chronic pain. 31 Whereas activation of PL-PFC is known to produce anti-nociceptive effects, activity of ACC neurons, on the other hand, plays a key role in processing the aversive response to pain.15–20,26 Previous studies have shown that AMPAkines can specifically increase the nociceptive response in the PL-PFC, 31 and our results on the CPA assays indicate that their activity in the PL-PFC can also reduce pain-aversion. In contrast, we found that CX516 did not change the nociceptive response in individual excitatory neurons of the ACC. An interesting pharmacological property of the AMPAkines is that these drugs have differential affinity for neurons in the CNS, with higher affinity in the PFC, the NAc, and brain stem. 68 Thus, it is possible that AMPAkines do not demonstrate sufficient binding to the ACC neurons to modulate the nociceptive processing of these neurons. Another possibility is that AMPAkines act on both excitatory and inhibitory interneurons in the ACC, producing a negligible net effect on the outputs from the ACC.
There are several limitations to this study. Due to the close proximity of the ACC and PFC in the rat brain, there may be technical limitations on how specifically the AMPAkines bind only to the target area. Future studies of in vivo or in vitro whole-cell recordings and/or two-photon calcium imaging could provide further clarification on differential neuronal responses to AMPAkines in the cortex. In addition, future studies including both male and female sexes are needed to investigate any potential sex-related differences in the effects of AMPAkines on pain regulation.
In conclusion, we have shown that AMPAkines have differential effects in nociceptive processing in the CNS. Whereas these drugs have analgesic effects on neurons in the PL-PFC, they do not appear to modulate the nociceptive response in ACC neurons. Future studies may be needed to further elaborate the molecular mechanisms for these pharmacological differences.
Experimental model and subject details
Animals
All procedures were performed in accordance with the New York University School of Medicine (NYUSOM) Institutional Animal Care and Use Committee (IACUC) guidelines to ensure minimal animal use and discomfort, as consistent with the National Institute of Health (NIH) Guide for the Care and Use of Laboratory Animals. Male Sprague-Dawley rats were purchased from Taconic Farms (Albany, NY) and held at the vivarium facility in the NYU Langone Science Building with controlled humidity, temperature, and 12 h (6:30 a.m. to 6:30 p.m.) light-dark cycle. Food and water were provided ad libitum. All animals were about 7 to 8 weeks old upon arrival at the animal facility and were given 10–14 days to adjust to the vivarium facility before beginning any surgeries or behavioral assays.
Virus construction and packaging
The recombinant adeno-associated virus (AAV) vectors were serotyped with AAV1 coat proteins and packaged at Addgene viral vector manufacturing facilities. Viral titers were around 1 × 101³ particles per milliliter for pENN.AAV1. CamkII.GCaMP6f.WPRE.SV40.
Drugs
CX516 (MedChemExpress) was suspended in dimethyl sulfoxide (DMSO) to make a 20 mg/kg solution for systemic injection. CX516 was also resuspended in 0.9% saline to a concentration of 800 uM for intra-PL-PFC and intra-ACC infusions in rats. For intracranial injections, 0.5 ul of the drug or saline solution was injected to each side of the brain through a 33G Hamilton’s syringe. Rats were allowed at least 1 week to recover from cannula implantation surgery before intracranial injections began. After the injections, rats were given up to 30 min to rest before starting behavioral assays.
Intracranial viral injections and cannula implantation
For rats subjected to the gradient-index (GRIN) lens implantation, rats were anesthetized with isoflurane (1.5%–2%) and were unilaterally injected with 0.65 μL viral vectors in to the ACC at a rate of 0.1 μL/30 s. Injections were made using a 32G 1 μL Hamilton syringe at anteroposterior (AP) +2.7 mm, mediolateral (ML) ±1.6 mm, and dorsoventral (DV) −2.0 mm with the syringe tips angled 22° toward the midline. After the injection, the needle was left in place for 10 min before it was raised 0.5 mm to allow for the viral particles to diffuse and to minimize particle dispersion along the injection tract. The needle was held in place for an additional 5 min before being slowly raised from the brain entirely.
For cannula implantations, rats were anesthetized with isoflurane (1.5%–2%) and were stereotactically implanted with two 26 gauge guide cannulas (Plastics One Technologies, USA) bilaterally in the ACC (AP + 2.7 mm, ML ± 1.6 mm, DV -1.0 mm, with tips 22° toward the midline) or PL-PFC (AP + 2.9 mm, ML ± 1.6 mm, DV -2.7 mm, with tips 17° toward the midline). Cannulas were held in place by dental acrylic and capped until the intracranial injection.
Intracranial pharmacology
For intracranial injections, solutions were loaded into a 30 cm length of PE-50 tubing, with one end attached to a 10 µL Hamilton’s syringe filled with distilled water and the other end to a 33 gauge injector cannula, which extended 1 mm past the implanted cannula guides. Delivery of 0.5 µL of the solution to each side took place over the course of 100 s and the injector cannulas were left in place for another 60 s to allow for diffusion the solution into the brain region. Behavioral tests were completed up to 30 min after intracranial injections. After behavioral experiments were completed, the cannulas were recapped to prevent any blockage.
Gradient-Index lens implantation and mounting
Rats were stereotactically implanted with the gradient-index (GRIN) lens (1.0 mm diameter, ∼9.0 mm length, Inscopix) at AP +2.7 mm, ML ± 1.6 mm, and DV -1.8 mm, with the tip of the lens angled 22° towards the midline, 4-6 weeks after the intracranial injection of the GCaMP6f virus. The region between the lens and the site of the open craniotomy was held by silicone elastomer (Kwik-Sil, World Precision Instruments). Dental acrylic was used to secure the lens in place.
3-4 weeks after the GRIN lens implantation, rats were anesthetized with 0.5–1% isoflurane and were checked for GCaMP6f fluorescence and Ca2+ transient activity. The miniature microscope (nVoke, Inscopix) was attached to a baseplate and was stereotactically adjusted to the lens implant to find the optimal field of view (FOV) for neural activity imaging. Both auditory (clapping) and sensory (tail pinching) stimuli were used to produce neural activity, and for rats that had an optimal Ca2+ response, the baseplate was mounted and secured above the lens. After confirming the location of the baseplate, the anesthesia was raised to 1.5–2% isoflurane, and the baseplate was secured with adhesive cement (Metabond Quick! Adhesive Cement System, C&B). After the baseplate was secured, a baseplate cover (Inscopix) was magnetically attached to the baseplate in order to protect the lens.
GRIN lens imaging procedure
The rat was placed in a chamber over a metal mesh table at the beginning of the imaging procedure. The miniature microscope was mounted and secured to the baseplate. The rat was allowed to habituate to the chamber for about 10 min before beginning the imaging procedure. During the imaging procedure, spontaneous neural activity was recorded for 5 min while the rat moved freely within the chamber without any external stimulus. Non-noxious peripheral stimulation was delivered to the plantar surface of the hind paw by using a 4 g von Frey (vF) filament. Noxious peripheral stimulation was delivered to the plantar surface of the hind paw contralateral to the grin lens implantation by using a 27G needle pin prick (PP). The noxious stimuli were terminated upon withdrawal of the paw. Both the noxious and non-noxious stimuli were delivered every 30 s, totaling five stimuli for each type. Experiments were recorded by a video camera (HC-V550, Panasonic). No physical damage to the paws was observed.
GRIN lens data acquisition and preprocessing
The miniature fluorescent microscope videos were recorded at a frame rate of 20 Hz, with a laser power of 0.6-0.8mW/mm2. Raw videos were spatially down sampled by a binning factor of 4 (16x spatial downsample) and temporally down sampled by a binning factor of 2 (down to 10 frames per second) using the Inscopix Data Processing Software (Inscopix). Using the Inscopix Data Processing Software, the videos were then motion-corrected relative to a reference frame to match the XY positions of each video frame in the whole video. The motion-corrected 10 Hz video of raw Ca2+ activity was saved as a. TIFF file and used to investigate the rat’s neural activity to the stimuli. Using modified constrained non-negative matrix factorization scripts (CNMF_E) in MATLAB, the Ca2+ signals were obtained to estimate temporally constrained instances of calcium activity for each neuronal region of interest (ROI)69,70.
Imaging data analysis
The peak Ca2+ fluorescence ΔF of each neuron was calculated using the post stimulus range from 0 to 5 s, with 0 indicating the time of the external stimulation. Each trial was z-scored by subtracting the mean and dividing by the standard deviation of a baseline range of −5 to −3 s before the external stimulation. The z-scored trial was then binned into 100 ms bins. A 2 s moving window with step size 100 ms was used to find the maximum average Ca2+ fluorescence for each trial. The maximum average Ca2+ fluorescence was then averaged across all the trials to calculate the peak post-stimulus activity for any given ROI during a session.
Behavioral assay
Conditioned place aversion (CPA) experiments were conducted in a standard two-chambered device, comprising of two compartments of equal size connected by a large opening for rats to move and explore freely. The walls of each chamber contained a different scented balm to provide the rat with contextual cues. The CPA protocol consisted of preconditioning (baseline), conditioning, and testing phases. The preconditioning phase was 10 min, and rats were allowed to freely travel between the two chambers. Rats spending more than 500 s or less than 100 s in each chamber during the preconditioning phase were removed from the behavioral test. After the preconditioning phase, the rats underwent the conditioning phase, which was 20 min in total. One of the two chambers was paired with a noxious stimulus (PP) and the other chamber was paired with no external stimuli (NS). We used a 27-gauge syringe to deliver PP as noxious stimuli to rats' plantar region of the right hind paw, and each pin prick stimulation was terminated when the rat withdrew its paw. The peripheral stimulus was delivered at 30 s intervals. The rat was allowed 10 min in each of the two chambers. The order of the PP stimulus and the NS were counterbalanced, so that about half of the rats received the PP stimulation first and the other half received NS first in the conditioning stage. During the testing phase, the rats were allowed free access to move between the two chambers for 10 min without any external stimuli. Preconditioning, conditioning, and testing phases were conducted on the same day. Animal movements during the CPA procedure were recorded by a camera from above the apparatus and analyzed with the AnyMaze Version 6.32 software (Stoelting Co., Wood Dale, IL, USA). More time spent in a particular chamber in the testing phase compared to baseline indicated a preference for that particular chamber, and decreased time spent in a chamber during testing phase, as compared to the preconditioning phase, indicated aversion of that chamber. The CPA score is a quantification of the rat’s aversion to a chamber and is found by subtracting the time the rat spent in the chamber associated with the PP stimulation during the testing phase from the time it spent in the corresponding chamber during the preconditioning stage. A higher CPA score indicated greater pain aversion to the PP stimulus.
Footnotes
Acknowledgments
Cartoon in Figure 3(c) was created with ![]() .
.
Author contributions
J.W. conceived, designed and supervised the study. E.Z., D.M., H.J.J., M.S., and W.L. collected the data. E.Z., Q.Z. and J.W. analyzed data. E.Z. and J.W. wrote the manuscript with inputs from other authors.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Jing Wang has a patent on the use of α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor modulators in pain regulation (U.S. 10702513).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health Grants R01-GM115384 (JW).
