Abstract
Chronic pain is a debilitating symptom with a significant negative impact on the quality of life and socioeconomic status, particularly among adults and the elderly. Major Depressive Disorder (MDD) stands out as one of the most important comorbid disorders accompanying chronic pain. The kynurenine pathway serves as the primary route for tryptophan degradation and holds critical significance in various biological processes, including the regulation of neurotransmitters, immune responses, cancer development, metabolism, and inflammation. This review encompasses key research studies related to the kynurenine pathway in the context of headache, neuropathic pain, gastrointestinal disorders, fibromyalgia, chronic fatigue syndrome, and MDD. Various metabolites produced in the kynurenine pathway, such as kynurenic acid and quinolinic acid, exhibit neuroprotective and neurotoxic effects, respectively. Recent studies have highlighted the significant involvement of kynurenine and its metabolites in the pathophysiology of pain. Moreover, pharmacological interventions targeting the regulation of the kynurenine pathway have shown therapeutic promise in pain management. Understanding the underlying mechanisms of this pathway presents an opportunity for developing personalized, innovative, and non-opioid approaches to pain treatment. Therefore, this narrative review explores the role of the kynurenine pathway in various chronic pain disorders and its association with depression and chronic pain.
Introduction
Pain represents a critical symptom across various disorders and serves as a component of the body’s defense mechanism. Chronic pain, defined as lasting 3 months or longer, constitutes a debilitating symptom with substantial adverse effects on quality of life and socioeconomic status, particularly in adults and the elderly. 1 According to the CDC’s analysis of data from the 2019-2021 National Health Interview Survey (NHIS), an estimated 20.9% of adults in the United States, totaling 51.6 million individuals, experienced chronic pain in 2021. 2 Major Depressive Disorder (MDD) is one of the most significant comorbid disorders associated with chronic pain, with the prevalence of chronic pain among individuals with depression being higher compared to the overall estimate of chronic pain prevalence (39% vs 20.9%). 2 Furthermore, comorbid MDD and chronic pain typically lead to poorer outcomes compared to individuals experiencing chronic pain alone. 3 Moreover, there is a concerning estimate regarding the prevalence of problematic opioid use among adults with chronic pain, reaching 36.3%. 4
The kynurenine pathway serves as the principal route for tryptophan degradation and plays a crucial role in various biological processes, including the regulation of neurotransmitters, immune responses, cancer, metabolism, and inflammation.
5
Various metabolites produced in the kynurenine pathway, such as kynurenine, kynurenic acid, 3-hydroxyanthranilic acid, 3-hydroxykynurenine, and quinolinic acid, exert effects on these processes. For instance, quinolinic acid acts as a specific but weak agonist of the N-methyl-
The mechanisms underlying the development of chronic pain and its risk factors remain incompletely understood. As mentioned, the kynurenine pathway may play a role in pain development, and further understanding of this pathway and its metabolites in pain development and management could be beneficial. Therefore, this review aims to provide an updated examination of the role of the kynurenine pathway in various chronic pain conditions.
Methods
Literature search strategy
A comprehensive literature search was conducted to identify relevant studies exploring the impact of the Kynurenine Pathway across various chronic pain conditions. The search strategy aimed to capture the breadth and depth of research on this topic, utilizing a combination of controlled vocabulary and free-text keywords. Given the interdisciplinary nature of the subject matter, multiple electronic databases were queried, including PubMed, Scopus, and Web of Science. No restrictions were applied regarding publication date or language to ensure inclusivity of all relevant literature.
Selection of keywords
Keywords were selected based on the main themes addressed in the narrative review. These included terms related to the Kynurenine Pathway, chronic pain conditions, and associated mechanisms and outcomes. The initial keyword list encompassed concepts such as “Kynurenine Pathway,” “neuropathic pain,” “depression,” “headache,” “gastrointestinal disorders,” “fibromyalgia,” and “chronic fatigue syndrome.” Additional terms and synonyms were incorporated to capture related concepts comprehensively.
Expansion of keyword list
To refine the search strategy and enhance its sensitivity, the initial keyword list was expanded to include variations and synonyms of key terms. For instance, terms such as “neuroinflammation,” “glial activation,” “serotonin imbalance,” and “oxidative stress” were included to encompass broader aspects of the underlying mechanisms involved in chronic pain conditions.
Boolean operators and search syntax
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Boolean operators (AND, OR, NOT) were utilized to combine keywords effectively and tailor the search strategy to specific research questions. For example, search strings such as “ ('Kynurenine Pathway’ OR 'tryptophan metabolism’) AND ('neuropathic pain’ OR 'neuroinflammation')” were employed to retrieve articles focusing on the intersection between the Kynurenine Pathway and neuropathic pain.
Kynurenine pathway
Tryptophan, an indispensable amino acid, serves as a fundamental component for protein synthesis and as a precursor for the synthesis of various biologically active molecules, including nicotinic acid, melatonin, serotonin, and nicotinamide adenine dinucleotide (NAD+). It undergoes catabolism via two intricate metabolic pathways: the kynurenine pathway and the serotonin pathway. 5 While a minor fraction of tryptophan follows the serotonin pathway, the majority is metabolized through the kynurenine pathway. 7 Tryptophan exerts its influence within the central nervous system (CNS) primarily through serotonin production, a well-known bioactive molecule. The products of the kynurenine pathway modulate numerous biological processes, including host-microbiome signaling, immune cell responses, and neuronal excitability. 6 Tryptophan serves as the substrate for the synthesis of melatonin, serotonin, and indole metabolites.8–10
The kynurenine pathway is regulated by the enzymes indoleamine 2,3-dioxygenase (IDO) and, to a lesser extent, tryptophan 2,3-dioxygenase (TDO), which are the initial and rate-limiting enzymes of this pathway.8–10 Although both TDO and IDO catalyze the same reaction in kynurenine pathway, they exhibit distinct tissue expression patterns. TDO is predominantly expressed in the liver, whereas IDO1 is distributed across several organs in the body, with IDO2 mainly found in the kidney, liver, and antigen-presenting cells.6,11 Formylkynurenine, produced by both IDO and TDO, is converted to kynurenine by kynurenine formidase. Kynurenine undergoes further enzymatic transformations: kynurenine can be converted to kynurenic acid by kynurenine aminotransferase (KAT), while kynureninase and kynurenine 3-monooxygenase (KMO) yield anthranilic acid and 3-hydroxykynurenine, respectively. Kynureninase and KAT further convert 3-hydroxykynurenine to 3-hydroxyanthranilic acid and xanthurenic acid, respectively. Anthranilic acid can be transformed into 3-hydroxyanthranilic acid by 3-hydroxyanthranilic acid hydroxylase.12–14 In mammals, kynureninase predominantly catalyzes the conversion of 3-hydroxykynurenine to 3-hydroxyanthranilic acid. However, within the brain, anthranilic acid is the preferred precursor of 3-hydroxyanthranilic acid compared to its presence in peripheral organs.15,16 The final stages of the kynurenine pathway lead to the production of picolinic acid, quinolinic acid, and nicotinic acid from 3-hydroxyanthranilic acid. Picolinic acid is generated from 3-hydroxyanthranilic acid by 7,2-amino-3-carboxymuconic acid semialdehyde decarboxylase (ACMSD), while 3-hydroxyanthranilate-3,4-dioxygenase produces quinolinic acid. Nicotinic acid is synthesized from 3-hydroxyanthranilic acid by phosphoribosyltransferase (Figure 1).
11
The kynurenine pathway is a metabolic route through which tryptophan undergoes catabolism, along with the serotonin pathway. Formylkynurenine is synthesized by both indoleamine 2,3-dioxygenase (IDO) and tryptophan 2,3-dioxygenase (TDO), with subsequent conversion to kynurenine catalyzed by kynurenine formamidase. Kynurenine is subject to various enzymatic activities. Specifically, kynurenine can be metabolized to kynurenic acid by kynurenine aminotransferase (KAT). Furthermore, kynureninase and kynurenine 3-monooxygenase (KMO) convert kynurenine into anthranilic acid and 3-hydroxykynurenine, respectively. Subsequently, kynureninase and KAT further metabolize 3-hydroxykynurenine to 3-hydroxyanthranilic acid and xanthurenic acid, respectively. Anthranilic acid is subject to conversion to 3-hydroxyanthranilic acid via the enzyme 3-hydroxyanthranilic acid hydroxylase. In the terminal steps of the kynurenine pathway, 3-hydroxyanthranilic acid serves as a precursor for the synthesis of picolinic acid, quinolinic acid, and nicotinic acid. Picolinic acid is produced from 3-hydroxyanthranilic acid by 7,2-amino-3-carboxymuconic acid semialdehyde decarboxylase (ACMSD), while quinolinic acid is generated through the action of 3-hydroxyanthranilate-3,4-dioxygenase. Nicotinic acid synthesis from 3-hydroxyanthranilic acid is facilitated by phosphoribosyltransferase. Additionally, inflammatory cytokines such as tumor necrosis factor-alpha (TNF-α), interferon-gamma (IFN-γ), IFN-α, and IFN-β have been shown to enhance the activity of IDO.
Studies have indicated that among the branches of kynurenine metabolism, the pathways leading to the production of quinolinic acid and kynurenic acid are particularly implicated in chronic pain states.
5
Traditionally, the kynurenine pathway was dichotomized into neurotoxic (quinolinic acid) and neuroprotective (kynurenic acid) branches.
5
However, recent findings suggest that kynurenine metabolites may exhibit multifaceted properties. For instance, kynurenic acid has been associated with cognitive decline in patients with Alzheimer’s disease,
17
yet low doses of kynurenic acid have been reported to contribute to cognitive enhancement.
18
Furthermore, it has been demonstrated that the segregation of kynurenine pathway branches occurs within the CNS, with astrocytes responsible for producing neuroprotective metabolites and macrophages/microglia, activated in various neuroimmunological diseases, synthesizing neurotoxic metabolites.19,20 Dysfunction within the kynurenine pathway leads to elevated levels of neurotoxic metabolites, correlating with inflammatory and neurodegenerative conditions such as 3-hydroxykynurenin, 3-hydroxyanthranilic acid, and quinolinic acid.
21
Although kynurenine-derived metabolites exhibit poor crossing of the blood–brain barrier (BBB),
Inflammatory cytokines have the capability to trigger the activation of IDO. In instances of inflammation, proinflammatory cytokines, particularly tumor necrosis factor-alpha (TNF-α) and interferon-gamma (IFN-γ), are known to potentially elevate tryptophan catabolism by promoting the expression of IDO, consequently altering the metabolism of tryptophan towards the production of kynurenine.23,24 IFN-α has a weak direct effect on IDO induction. 25 Furthermore, IFN-β has been shown to enhance IDO activity and alter the ratio of kynurenine/tryptophan in individuals with multiple sclerosis (MS). 26 It has been shown that neuroinflammation in the hippocampus followed by IDO activation, with a consequent decrease in the levels of serotonin (5-HT), can induce depression in diabetic rats. 27 Cytokines and kynurenines play a significant role in facilitating communication between the brain and the immune system, thereby regulating both neuronal and glial activity within the CNS, as well as modulating immune system function within an integrated network. 28
Kynurenine pathway and neuropathic pain
Neuropathic pain is a significant form of chronic pain, often arising from direct damage to the somatosensory nervous system. Despite understanding several underlying mechanisms, effective treatments remain elusive. 29 Notably, interactions between neurons and glial cells, such as microglia, oligodendrocytes, and astrocytes in the spinal cord, play a crucial role in neuropathic pain by amplifying central sensitization. 30 Mediators released by glial cells that boost glutamatergic transmission in spinal cord neurons can worsen neuropathic pain.30,31 Studies suggest that NMDA antagonists hold promise in alleviating tactile allodynia in neuropathic animals.32–34 Additionally, immune and glial cell activation in both the peripheral and central nervous systems contribute to the upregulation of the kynurenine pathway.14,35
Kynurenine pathway metabolites
Heyliger et al. demonstrated the analgesic effects of various metabolites of this pathway in rodent models. Intraperitoneal administration of tryptophan, kynurenine, anthranilic acid, quinolinic acid, xanthurenic acid, and picolinic acid exhibited significant analgesia, particularly in tail-flick and hotplate tests. 36 Furthermore, intrathecal administration of kynurenine to rats demonstrated dose-dependent antinociceptive effects. 37 Quinolinic acid and xanthurenic acid emerged as frequently elevated biomarkers in samples collected from a substantial cohort of patients suffering from chronic pain. 38 Xanthurenic acid is known neuroactive compounds that exert its effects through glutamatergic receptors. 39
Recent studies have shed light on the intricate role of kynurenic acid, a key metabolite of the pathway, in modulating neuropathic pain. Concurrent administration of
Intraperitoneal injection of probenecid and
It has been shown that CXCL17 can induce strong pain-related behaviors in mice through GPR35, measured by von Frey and cold plate tests. In addition, administration of kynurenic acid and zaprinast diminished CXCL17-evoked pain-related behaviors in both tests. Additionally, kynurenic acid and zaprinast reduce tactile and thermal hypersensitivity in CCI mice and enhance the effectiveness of morphine in neuropathy. 53
Cinnabarinic acid (CA) is a byproduct of the kynurenine pathway, originating from the conversion of 3-hydroxyanthranilic acid. At low doses, CA reduces pain behavior in mice, observed in the formalin test and CCI model. CA-induced analgesia depends on metabotropic glutamate receptor 4 (mGlu4) but not aryl hydrocarbon receptor (AhR) activation. Combined administration of CA and the AhR antagonist, CH223191, shows enhanced analgesic effects in CCI mice, indicating synergistic action. These findings highlight CA’s involvement in pain modulation through mGlu4 and AhR. 54
Kynurenine pathway enzymes
Dysregulation of enzymes in the kynurenine pathway has been linked to the development of neuropathic pain. Increased levels of kynurenine metabolites, especially quinolinic acid and 3-hydroxykynurenine, can cause damage and dysfunction in neurons. This occurs through heightened activation of NMDA receptors and oxidative stress.55,56 Various studies have shown that 3-hydroxykynurenine generates harmful free radicals such as superoxide and hydrogen peroxide, leading to mitochondrial impairment and subsequent neuronal dysfunction.56,57 These mechanisms are crucial in the onset of neuropathic pain.58,59
Maganin et al. demonstrated the accumulation of dendritic cells expressing IDO1 in the dorsal root leptomeninges of mice with spared nerve injury (SNI), contributing to increased kynurenine levels in the spinal cord. They found that while IDO1, implicated in neuropathic pain mechanisms, is not upregulated in resident spinal cord cells, it is present within the vasculature and circulating immune cells. Moreover, IDO1-expressing dendritic cells can infiltrate and accumulate in the dorsal root lesion. Within the spinal cord, astrocytes expressing kynurenine-3-monooxygenase metabolize kynurenine to the pronociceptive metabolite 3-hydroxykynurenine. Additionally, quinolinic acid contributes to neuropathic pain development by activating glutamatergic NMDA receptors. 60
Ciapala et al. revealed increased levels of KMO, kynureninase, and 3-hydroxyanthranilate dioxygenase after CCI. Furthermore, a single intrathecal administration of
The alteration of the kynurenine pathway in neuropathic pain, migraine, and cluster headache.

Abbreviations. QUIN: quinolinic acid, XANA: xanthurenic acid, SNI: spared nerve injury, IDO: indoleamine-2,3-dioxygenase, TDO: tryptophan-2,3-dioxygenase, CCI: chronic constriction injury, KMO: kynurenine monooxygenase, KYNU: kynureninase, 3-HA: 3-hydroxyanthranilate, 3-HK: 3-hydroxykynurenine, DRG: dorsal root ganglia, HAAO: 3-hydroxyanthranilate-3,4-dioxygenase, KYN: kynurenine, TRP: tryptophan, KAT: kynurenine aminotransferase, CRPS: complex regional pain syndrome, SNL: spinal nerve ligation, KYNA: kynurenic acid, PICA: picolinic acid, ANA: anthranilic acid, NTG: nitroglycerin.
Kynurenine pathway and inflammation
CCI was associated with upregulation of kynurenine 3-monooxygenase mRNA in the spinal cord and DRG. Pharmacological inhibition of microglia using minocycline, or direct inhibition of kynurenine 3-monooxygenase using Ro61-6048, decreased neuropathic pain intensity on days 3 and 7 post-CCI. Biochemical analysis showed that Ro61-6048 reduced CD40/IBA1, IL-1β, IL-6, and NOS2 mRNA levels in both DRG and spinal cord tissues. 64
SNI-induced depressive-like behavior, although not mechanical allodynia, was mitigated with intracerebroventricular administration of either KMO inhibitor, Ro 61-8048, or IL-1 receptor antagonist (IL-1RA). 48 However, if Ro 61-8048 had been administered via intrathecal injection, mechanical allodynia would have been alleviated. 64 This underscores the significance, particularly, of the IL-1 signaling pathway for these processes, likely through the KMO-mediated pathway. The IL-1 signaling pathway within the spinal cord is suggested to be pivotal in driving peripheral (liver) IDO-1-mediated mechanical allodynia and depression-like behavior induced by SNI. 65
Activation of the immune system and elevated inflammatory cytokines have been implicated in the development of diabetic neuropathy by activating the kynurenine pathway. Pires et al. observed elevated levels of interleukin (IL)-8 and GM-CSF in patients with type 1 diabetes mellitus and neuropathic pain, suggesting immune cell involvement. 66 According to research findings, GM-CSF has been implicated in neuropathic pain, as it has been shown to enhance the release of pro-inflammatory mediators from spinal glial cells. 67 The chemokine IL-8, primarily released from macrophages, serves as a recruiter for both neutrophils and T lymphocytes. Recent studies have linked IL-8 to neuropathic pain observed in peripheral neuropathies. 68 Furthermore, Pires et al. demonstrated increases in two inflammatory biomarkers: the kynurenine/tryptophan ratio, a marker of IDO activity, and neopterin. The kynurenine/tryptophan ratio was positively correlated with pain intensity. Moreover, activity of KAT was higher in diabetic neuropathic pain patients, indicating increased production of xanthurenic acid. 66 The KYN/TRP ratio stands as a well-established marker for IDO1 activity and inflammatory activation, showcasing an elevated ratio in conditions such as chronic lower back pain and complex regional pain syndrome.69,70
Furthermore, spinal nerve ligation (SNL) in rats was associated with increased levels of IDO1 in the anterior cingulate cortex and amygdala. Administration of PCC0208009, an indirect inhibitor of IDO1, attenuated pain-related behaviors in the formalin test and increased anxiogenic behaviors and cognition in SNL rats, without affecting sleep or locomotor activity. PCC0208009 decreased IDO1 levels in the anterior cingulate cortex and amygdala through inhibition of the IL-6-JAK2/STAT3-IDO1-GCN2-IL-6 pathway, acting as a selective pain suppressor. Additionally, PCC0208009 reversed synaptic plasticity at the structural and functional levels by suppressing NMDA2B receptor and CDK5/Tau and CDK5/MAP2 pathways in the anterior cingulate cortex and amygdala. 71
Therapeutic options
The kynurenine pathway presents a promising avenue for therapeutic intervention in neuropathic pain management, as elucidated by emerging research findings. Various metabolites within this pathway, including kynurenic acid, have demonstrated significant analgesic effects in preclinical models. Intraperitoneal administration of kynurenine pathway metabolites such as kynurenine has shown notable analgesia in rodent models, particularly evident in tailflick and hotplate tests. Furthermore, intrathecal administration of kynurenine has exhibited dose-dependent antinociceptive effects, suggesting the potential for targeting spinal mechanisms involved in neuropathic pain. Notably, the analgesic properties of kynurenic acid have been underscored by its ability to act as an endogenous antagonist for various receptors implicated in pain processing, including NMDA, AMPA, and α7nACh receptors, as well as its modulation of GPR35 receptors found in both glial cells and neurons.
Moreover, dysregulation of kynurenine pathway enzymes has been implicated in the pathogenesis of neuropathic pain, presenting potential targets for therapeutic intervention. Elevated levels of kynurenine metabolites, such as quinolinic acid and 3-hydroxykynurenine, have been associated with neuronal damage and dysfunction through mechanisms involving increased NMDA receptor activation and oxidative stress. Inhibition of kynurenine pathway enzymes, such as kynurenine 3-monooxygenase (KMO), has shown promise in attenuating neuropathic pain intensity and reducing inflammatory cytokine levels in preclinical models. Additionally, targeting IDO1, a key enzyme in the kynurenine pathway, has demonstrated analgesic effects and modulation of synaptic plasticity, suggesting its potential as a selective pain suppressor.
Furthermore, recent investigations into novel compounds targeting the kynurenine pathway have unveiled promising candidates for neuropathic pain management. Systemic administration of the prodrug L-4-chlorokynurenine (4-Cl-KYN) in rodent models has shown remarkable efficacy in mitigating hyperalgesia and allodynia associated with tissue inflammation and nerve injury. Through conversion to 7-chlorokynurenic acid (7-Cl-KYNA), a potent antagonist of the NMDA receptor glycine B site, 4-Cl-KYN demonstrates dose-dependent antihyperalgesic effects, surpassing the efficacy of established analgesics such as gabapentin and NMDA receptor antagonist MK-801. These findings underscore the potential of targeting the kynurenine pathway for neuropathic pain relief, offering novel avenues for therapeutic intervention and improving patient outcomes. 72
Kynurenine links depression to chronic pain
Approximately 10%–20% of the general population may experience major depressive disorder (MDD) at some point in their lifetime. Research conducted in clinical settings indicates bidirectional relationships between depression and chronic pain, with elevated prevalence observed among individuals with chronic pain, and vice versa. Chronic pain is identified as a significant risk factor for MDD development, with prevalence ranging from 20% to 80% among patients with chronic pain. Moreover, a correlation exists between depression severity and pain intensity, suggesting an independent impact of depression on pain perception.73,74
Neuroscientific studies highlight overlapping brain regions involved in both bodily pain perception and mood regulation, including the insular cortex, prefrontal cortex, anterior cingulate cortex, hippocampus, amygdala, and thalamus. 75 This suggests potential neuroplastic alterations contributing to the co-occurrence of pain and depression. 73 Additionally, interactions between inflammatory pathways and neural circuits within the brain play a role in shaping behavioral responses and precipitating depression at the molecular level. 76 Neuroinflammation, an innate immune response of the nervous system to injury, infection, and neurodegeneration, plays a pivotal role in this process. 77 Chronic inflammation is associated with structural changes in neurons, facilitating the transition from acute pain to chronic pain and from sickness to depression. Inflammatory cytokines exert influence on neuroplasticity and neurotransmitter metabolism, thereby affecting regions of the brain implicated in depression. 78
The kynurenine pathway has garnered attention for its involvement in both MDD and chronic pain. For instance, Bai et al. investigated metabolic pathways associated with psychoneurological symptoms (PNS), including pain, fatigue, anxiety, and depressive symptoms, in children undergoing chemotherapy. Among 9276 metabolic features, the tryptophan amino acid pathway correlated with the PNS cluster. 79 Additionally, research has demonstrated that reduced levels of tryptophan and the consequent elevation in the kynurenine/tryptophan ratio are associated with the severity of pain and depression among individuals diagnosed with complex regional pain syndrome. 80
Kynurenine pathway metabolites
The study conducted by Barjandi et al. indicates that low levels of tryptophan and high levels of kynurenine are associated with increased pain intensity, suggesting significant tryptophan metabolism via the kynurenine pathway during inflammation-induced pain. 81 Additionally, kynurenine and its metabolites can induce neurotoxic effects in specific brain regions, particularly the prefrontal cortex and hippocampus. 5
Among breast cancer survivors, co-occurring symptoms such as pain, fatigue, depression, anxiety, and sleep disturbances are commonly observed, potentially influenced by inflammation-induced activation of the tryptophan-kynurenine pathway. Multivariable analysis highlights lower tryptophan levels and a higher kynurenine/tryptophan ratio as significant predictors of increased symptom severity, suggesting a potential role of impaired tryptophan availability in the development of psychoneurological symptoms among these survivors. 82
The hippocampus, a pivotal region implicated in depression, exhibits heightened levels of quinolinic acid in individuals with depression. 83 The hippocampus, a limbic structure, is integral to mood and behavior regulation. While the hippocampus itself does not generate quinolinic acid, it does express NMDA receptor 2A and 2B subtypes, which exhibit a notably high affinity for quinolinic acid. Quinolinic acid serves as a neurotoxic agent on the hippocampus, potentially contributing to the development of depression. 84 Post-mortem examinations of individuals who experienced major depression also revealed higher levels of quinolinic acid in the prefrontal cortex compared to non-depressed individuals. 85
Quinolinic acid has been associated with various detrimental effects, including mitochondrial damage, excitotoxicity, oxidative stress, and disruption of autophagy. 66 Recent research has shown elevated levels of quinolinic acid and reduced levels of kynurenic acid in the pathogenesis of depression and chronic pain.5,86 Similarly, two meta-analyses demonstrated a shift in tryptophan metabolism from serotonin to the kynurenine pathway and heightened levels of quinolinic acid alongside reduced levels of kynurenic acid and kynurenine in patients with depression.21,84
Yun et al. investigated the association between the tryptophan-kynurenine pathway and painful physical symptoms (PPS) in MDD. Their analysis revealed that MDD patients with PPS exhibited higher kynurenine and quinolinic acid levels, along with an elevated kynurenine/tryptophan ratio compared to those without PPS. Significant correlations were found between PPS severity and quinolinic acid levels, as well as inverse correlations with tryptophan levels or kynurenic acid/quinolinic acid ratios. Multiple regression analysis identified kynurenine/tryptophan ratios, kynurenic acid/quinolinic acid ratios, and Hamilton Depression Scale (HAMD) scores as significant predictors of PPS severity. These findings underscore the potential role of the tryptophan-kynurenine pathway in the pathophysiology of pain in MDD, suggesting its importance as a biomarker or therapeutic target warranting further investigation. 87
Kynurenine pathway enzymes and inflammation
As previously discussed, tryptophan plays a crucial role in serotonin production through the serotonin pathway. There exists an inverse correlation between levels of serotonin and IDO. During inflammation, the continuous utilization of tryptophan by IDO through the kynurenine pathway leads to abnormally low serotonin levels. Studies have shown a reduced serotonin/tryptophan ratio and an elevated kynurenine/tryptophan ratio in a rat model of chronic arthritis inflammatory pain. 69
Proinflammatory cytokines like IL-1β, IL-6, and TNF-α have been found to reduce hippocampal neurogenesis, with increased levels of these cytokines observed in individuals with depression. Chronic inflammation, or neuroinflammation, is suggested to escalate levels of inflammatory cytokines and activate the kynurenine pathway. Research indicates that endotoxin-induced depressive-like symptoms can be alleviated through IDO deletion, IDO inhibition, or deletion of IFN-γ receptors. Furthermore, administration of IFN-γ to patients with chronic medical conditions heightens the risk of depression by activating the kynurenine pathway. Treatment with IFN-γ has been shown to elevate levels of kynurenine and quinolinic acid, contributing to the severity of depressive symptoms. Subsequent to interferon therapy, increased concentrations of quinolinic acid in the cerebrospinal fluid have been observed, correlating with the severity of depressive symptoms. Moreover, this finding underscores the role of elevated quinolinic acid levels in exacerbating depressive symptoms, rather than solely depleting serotonin24,88–94
Kim et al. found that a significant proportion of hematologic cancer patients experienced a cluster of psycho-neurological symptoms, including depression, cognitive impairment, fatigue, sleep disturbance, and pain. Older age and elevated levels of IL-1α and IL-6 were associated with this symptom cluster. Additionally, higher activity of IDO was observed in patients with the symptom cluster, positively correlated with IL-6 levels. Symptom severity, IL-1α, IL-6, and IDO activity were significantly higher in cancer patients compared to healthy controls, and these findings were consistent across time points. 95
In the SNI model of neuropathic pain in mice, depressive behaviors were accompanied by an increase in plasma kynurenine/tryptophan ratios and upregulation of IDO1 and IL-1β mRNA in the liver. However, no detectable changes in brain or spinal cord IDO1 mRNA levels were observed. SNI induced inflammatory activity in the spinal cord, as evidenced by increased IL-1β mRNA expression. Interestingly, intrathecal administration of IL-1RA mitigated the increase in liver IDO1 and IL-1β. Intrathecal IL-1RA prevented both depression and allodynia in mice. IDO1 has been demonstrated to be essential for the development of neuropathic pain and depression, as IDO1−/− mice did not develop pain or depression. 65
Similarly, chronic pain induction in rats led to depressive behavior alongside upregulation of IDO1 in the bilateral hippocampus, resulting in altered kynurenine/tryptophan and serotonin/tryptophan ratios. Additionally, elevated plasma IDO activity was observed in patients experiencing pain and depression and in rats displaying anhedonia induced by chronic social stress. In vitro and in vivo experiments demonstrated that IL-6 induces IDO1 expression through the JAK/STAT pathway. Moreover, either genetic knockout or pharmacological inhibition of hippocampal IDO1 activity attenuated both nociceptive and depressive behavior. 69
Additionally, a meta-analysis revealed decreased levels of both kynurenine and kynurenic acid in patients with depression compared to healthy controls. Furthermore, it indicated that both the kynurenic acid/3-hydroxykynurenine and kynurenic acid/quinolinic acid ratios were lower in patients with depression than in healthy controls. However, the authors did not report any differences in the levels of 3-hydroxykynurenine and quinolinic acid between individuals with depression and healthy controls. It has been noted in studies that there is an increase in inflammation in patients with depression, and the aforementioned decrease in the kynurenic acid/3-hydroxykynurenine ratio in patients with depression corroborates this observation, suggesting that depression is associated with the activation of KMO. 86
Laumet et al. demonstrated an upregulation of IL-1β and KMO in the contralateral side of the brain in SNI mice. The upregulation of KMO was associated with increased quinolinic acid levels and decreased kynurenic acid levels in the contralateral hippocampus. Notably, the increase in KMO occurred within NeuN-positive neurons in the hippocampus, rather than in microglia. Their findings revealed that intracerebroventricular administration of IL-1RA after SNI inhibited IL-1β signaling, subsequently inhibiting KMO and preventing depression in mice. However, this treatment did not affect allodynia. Additionally, intracerebroventricular administration of the KMO inhibitor, Ro 61-8048, yielded similar results. Thus, they suggested that the development of depression in SNI mice necessitates brain IL-1β signaling and subsequent activation of neuronal KMO, while pain is independent of this pathway. 48
Therapeutic options
Therapeutic options for conditions involving dysregulation of the kynurenine pathway, such as chronic pain and depression, cover a wide range of interventions targeting crucial mechanisms implicated in their development. For example, Deng et al. explored the potential of Gentiopicroside (Gent) in easing depressive-like behavior induced by lipopolysaccharide (LPS) in mice due to its anti-inflammatory properties and antidepressant effects in animal models. They found that Gent treatment prevented the depressive-like behavior induced by LPS, as shown by reduced immobility time in forced swimming and tail suspension tests. This antidepressant effect of Gent was linked to the normalization of LPS-induced changes in brain inflammatory mediators, including IL-1β and TNF-α. Additionally, Gent prevented excessive activation of IDO and restored GluN2B subunit expression in the prefrontal cortex challenged by LPS. 96
Khan et al.'s study emphasizes the therapeutic potential of targeting microglial activation to alleviate pain sensitivity and depressive-like behaviors associated with adolescent intermittent access to ethanol (AIE) exposure in mice. Chronic minocycline administration was found to relieve hyperalgesia and social deficits post-AIE, suggesting that inhibiting microglia could mitigate these behavioral effects. Moreover, the study highlights decreased tryptophan hydroxylase 2 (Tph2) expression and serotonin (5-HT) levels in the dorsal raphe nucleus (DRN) following microglial activation, suggesting that neuroinflammation-induced serotonin depletion contributes to AIE-induced pain and depression. These findings propose that targeting microglial activation or enhancing serotonin signaling could be promising for individuals with comorbid chronic pain and depression. 97
Dai et al. revealed that maintenance fluoxetine exposure in animal models of comorbid pain and depression upregulated the 5-hydroxytryptamine 1A (5-HT1A) auto-receptor and IDO1 in the brainstem DRN, leading to a shift in tryptophan metabolism away from serotonin biosynthesis. This upregulation of IDO1 was found to be downstream of fluoxetine-induced 5-HT1A receptor expression. Importantly, inhibiting either the 5-HT1A receptor or IDO1 activity sustained fluoxetine’s antidepressant and antihyperalgesic effects, suggesting that 5-HT1A-mediated IDO1 upregulation in the DRN contributes to the reduced efficacy of fluoxetine during maintenance treatment. 97
In addition to natural compounds, pharmacological agents such as selective serotonin reuptake inhibitors (SSRIs) have demonstrated efficacy in alleviating depressive symptoms by influencing the kynurenine pathway. SSRIs like escitalopram have been shown to reduce levels of neurotoxic metabolites such as 3-hydroxykynurenine and quinolinic acid, thus exerting neuroprotective effects. Furthermore, antidepressants like citalopram, fluoxetine, and imipramine have been found to modulate kynurenine pathway enzymes, upregulating KAT and downregulating KMO, thereby elevating the kynurenic acid/3-hydroxykynurenine ratio.98–100
Moreover, emerging research highlights the potential of compounds like trans-astaxanthin and AV-101 (l-4 chlorokynurenine) in managing both chronic pain and depression. It has been revealed that trans-astaxanthin significantly ameliorated both mechanical allodynia and thermal hyperalgesia in mice with CCI, indicative of its analgesic properties, while also demonstrating antidepressant-like effects by reducing immobility time in forced swim and tail suspension tests. Study revealed an increase in IDO expression following CCI surgery in both the hippocampus and spinal cord. This was accompanied by an elevation in the kynurenine/tryptophan ratio, a decrease in the 5-HT/tryptophan ratio, and a reduction in the 5-HT/5-HIAA ratio. Treatment with trans-astaxanthin reversed the aforementioned effects induced by CCI surgery. Furthermore, trans-astaxanthin administration effectively attenuated the expression of IL-1β, IL-6, and TNF-α in the hippocampus and spinal cord of CCI mice. 101
AV-101, a potent NMDA receptor antagonist, has shown efficacy in alleviating allodynia, mechanical hyperalgesia, heat hyperalgesia, and depression in animal models of neuropathic pain, underscoring the therapeutic potential of targeting NMDA receptors in the management of pain and comorbid depression. 102
Overall, therapeutic options for chronic pain and depression associated with dysregulation of the kynurenine pathway encompass a diverse array of interventions, including natural compounds, pharmacological agents, and novel therapeutics targeting key enzymes and receptors involved in tryptophan metabolism and neuroinflammation. Further research into these mechanisms and their modulation may offer new avenues for the development of more effective treatments for individuals with these debilitating conditions.
Kynurenine pathway and headache
Cluster headaches are characterized by unilateral headache attacks occurring multiple times a day, accompanied by ipsilateral autonomic dysregulation such as tearing, a runny nose, and redness of the conjunctiva. These headaches tend to follow the circadian rhythm, especially at night, and can also be influenced by seasonal changes. Current research identifies three key anatomical structures contributing to cluster headaches: the hypothalamus, the trigeminovascular system, and parasympathetic nerve fibers. 103
Migraine, a recurrent headache disorder with complex origins, is often marked by debilitating attacks. It is broadly categorized into two types: migraine without aura and migraine with aura. Migraine without aura involves headache attacks along with heightened sensitivity to light and sound, as well as symptoms like nausea and vomiting. Conversely, migraine with aura presents transient neurological symptoms preceding or accompanying the headache. However, these classifications may not be sharply distinct, potentially representing a spectrum of a single condition with diverse clinical presentations.104,105
The trigeminovascular system, comprising the trigeminal cranial nerve, associated nuclei, and cerebral blood vessels, is crucial in the neurophysiology of migraine attacks. Within this system, nociceptive axonal projections, including Aδ fibers and C-fibers, release various important neuropeptides. Notably, calcitonin gene-related peptide (CGRP) and pituitary adenylate cyclase-activating peptide (PACAP) are implicated in migraine pathophysiology.106,107 CGRP plays a significant role in headache pain perception by enhancing nociceptive transmission. 108 Additionally, PACAP and its receptor may contribute to both cluster headaches and migraines, with elevated PACAP-38 levels observed during the ictal phase of these headaches. Intravenous PACAP-38 administration has been shown to induce migraine attacks in both healthy individuals and migraine patients without aura.109,110
Research indicates that inhibiting NMDA receptor or non-NMDA excitatory amino acid receptor leads to significant reduction in trigeminovascular-evoked responses. This suggests the involvement of both NMDA- and non-NMDA-mediated mechanisms in transmission within the trigeminocervical complex, emphasizing the importance of glutamatergic mechanisms in primary headache syndromes such as migraine and cluster headache. 111
Glutamate plays a pivotal role in nociceptive sensitization induction. 112 Studies have linked migraine with polymorphisms in genes modulating synaptic glutamate uptake and postsynaptic glutamate signaling. 113 Furthermore, compounds from the kynurenine pathway have the potential to interact with glutamate receptors, indicating their possible involvement in migraine and cluster headache pathophysiology (Table 1).
Kynurenine pathway metabolites
Tuka et al. delved into the intricate interplay of glutamatergic neurotransmission and kynurenine metabolites within primary headache disorders, with a focus on episodic cluster headache. They discovered that in comparison to healthy controls, patients experiencing cluster headaches showed a decrease in
Tuka et al. also found significantly lower plasma concentrations of tryptophan metabolites, including tryptophan, kynurenine, kynurenic acid, quinolinic acid, anthranilic acid, xanthurenic acid, and picolinic acid, during the interictal phase of migraines compared to healthy controls, particularly evident in the migraine without aura (MWoA) subgroup. Some metabolites showed a tendency for elevation during the ictal phase, with notable increases observed exclusively for anthranilic acid, 5-hydroxy-indoleacetic acid, and melatonin in MWoA patients. 115
Curto et al. demonstrated significant reductions in various metabolites, such as kynurenine, kynurenic acid, 3-hydroxykynurenine, 3-hydroxy-anthranilic acid, and quinolinic acid, along with increased levels of tryptophan and anthranilic acid in cluster headache patients. Notably, reduced levels of kynurenic acid suggest NMDA receptor overactivity in cluster headache. Moreover, decreased levels of xanthurenic acid, a potential analgesic compound and activator of Glu2 and Glu3 receptors, may contribute to the severity of pain attacks.116,117
In another study, Curto et al. showed significant reductions in the levels of kynurenine, kynurenic acid, and other metabolites like 3-hydroxykynurenine and quinolinic acid in chronic migraine patients compared to controls. Conversely, levels of tryptophan, anthranilic acid, and xanthurenic acid were increased in chronic migraine patients. These findings suggest a one-way metabolism of kynurenine into anthranilic acid at the expense of kynurenic acid and 3-hydroxykynurenine in migraine. The decrease in kynurenic acid levels supports the hypothesis of NMDA receptor overactivity in migraine, while the increase in xanthurenic acid may represent a compensatory mechanism to reinforce endogenous analgesic pathways. 118
Nitroglycerine (NTG) has been established as a method to induce migraine-like symptoms in both human subjects and animal models. NTG can activate and sensitize the trigeminal system in both humans and animals. Studies have demonstrated that intraperitoneal administration of NTG at a dosage of 10 mg/kg leads to downregulation of KAT, resulting in decreased levels of kynurenic acid in the brain. 119 Additionally, intraperitoneal injection of NTG (10 mg/kg) into rats was linked to a reduction in the expression of IDO1, TDO2, kynurenine hydrolase, and KMO in the caudal trigeminal nucleus (TNC). This observation suggests that NTG has the capacity to downregulate the kynurenine pathway, potentially influencing the glutamatergic system. 120
Liu et al. revealed a significant decrease in gut microbiota richness and evenness in pediatric migraine patients compared to healthy controls. In their investigation of plasma concentrations of tryptophan, serotonin, kynurenic acid, kynurenine, and quinolinic acid, along with related ratios, in pediatric migraine patients and healthy controls, notable differences were observed. Migraine children exhibited elevated 5-HT levels, decreased kynurenic acid concentrations, and increased quinolinic acid levels compared to healthy counterparts. Moreover, the kynurenic acid/kynurenine ratio was lower, and the quinolinic acid/kynurenine ratio was higher in migraine patients, indicating altered tryptophan metabolism. Additionally, the kynurenic acid/quinolinic acid ratio was significantly decreased in migraine children and demonstrated excellent diagnostic efficacy for pediatric migraine. 121
Therapeutic options
Various studies have demonstrated that the use of kynurenic acid analogs, such as 2-(2-N,N-dimethylaminoethylamine-1-carbonyl)-1Hquinolin-4-one hydrochloride (KA1) and N-(2-N-pyrrolidinylethyl)-4-oxo-1H-quinoline-2-carboxamide hydrochloride (KA2), along with the co-administration of kynurenine and probenecid, effectively prevents NTG-induced behavioral and morphological changes. These analogs likely target NMDA receptors.122,123 Furthermore, probenecid and KA2 have been found to attenuate the decrease in CGRP and the increase in c-fos, neuronal nitric oxide synthase, and calmodulin-dependent protein kinase II alpha (CamKIIα) expression in C1-C2.123,124
NTG induces alterations in CamKIIα and CGRP expression in the trigeminal nucleus. Rats pretreated with
In a preclinical rat model, NTG administration followed by the orofacial formalin test led to the upregulation of nitric oxide synthase (eNOS), CGRP, and pro-inflammatory cytokines in both the trigeminal ganglia and the TNC. However, administration of the kynurenic acid analogue, N-(2-N,N-Dimethylaminoethyl)-4-oxo-1H-quinoline-2-carboxamide hydrochloride, prevented these changes, suggesting the involvement of glutamate in migraine development at both peripheral and central levels. 126
NTG-induced migraines increase the number of c-fos immunoreactive secondary sensory neurons within the caudal trigeminal nucleus. Although kynurenic acid cannot penetrate the BBB, its precursor, kynurenine, effectively crosses the blood–brain barrier (BBB) when combined with probenecid. Systemic treatment with kynurenine plus probenecid significantly mitigates the nitroglycerin-induced surge in c-fos immunoreactivity within the brainstem. 127
Probenecid has been shown to have an anti-nociceptive effect in the trigeminal inflammatory pain model (orofacial formalin test) in rats. Increasing kynurenic acid levels in the CNS may be a potential mechanism for this effect. 128 Similarly, KA1 and KA2 alleviate both behavioral and morphological changes in the formalin model of trigeminal activation, further supporting the role of kynurenic acid analogs in pain modulation. 129
Intravenous administration of NMDA receptor inhibitors, such as kynurenic acid, its synthetic analog, and MK-801, in a rat model of migraine induced by electrical stimulation of the trigeminal ganglion effectively prevented the overexpression of PACAP, a recognized biomarker of migraine. These inhibitors modulated glutamatergic transmission. 130
Cortical spreading depression (CSD), a phenomenon characterized by an initial excitation wave followed by inhibition in cortical neurons, is believed to underlie aura in migraine. 131 Depletion of 5-HT enhances CSD-induced trigeminal nociception by increasing cortical excitability and trigeminal sensitivity. 132 Kynurenic acid reduces the number of CSD waves and decreases BBB permeability during CSD, offering a potential therapeutic approach for migraines and other conditions associated with CSD. 133
CSD is favored by NMDA receptor activation and increased cortical excitability.
In vivo electrophysiological experiments revealed that NTG administration influences CSD, specifically reducing propagation failure compared to controls. Sumatriptan, a common migraine medication, decreased the number of CSDs but has minimal effect on propagation failure, while novel kynurenic acid analogues (SZR-105 and SZR-109) restored propagation to control levels. 136 Additionally, KA1 and KA2 target glutamate receptors and inhibit the propagation of CSD waves. 134
Cseh et al. demonstrated significant elevations in glutamate, kynurenic acid, and kynurenine in the TNC at 24 h post-complete Freund’s adjuvant (CFA) treatment in rats with CFA model of orofacial pain, subsequently diminishing by 48 h. Additionally, the somatosensory cortex exhibited notable increases in kynurenic acid and 5-HT levels. 137 Moreover, the administration of CFA into the rat dura mater has been linked to the production of phosphorylated extracellular signal-regulated kinases one and two (pERK1/2) and IL-1β in the trigeminal ganglion.138–140 It has been demonstrated that pretreatment with intraperitoneal administration of a novel kynurenic acid analogue (SZR72) can reduce the immunoreactivity of pERK1/2 and IL-1β. 141 Additionally, immunohistochemical studies revealed increased c-fos and glutamate immunoreactivity in TNC and cervical part of the trigemino-cervical complex (C1-C2) neurons following CFA application, which was prevented by SZR72 treatment. 142
As previously mentioned, intraperitoneal administration of NTG downregulates KAT, resulting in decreased levels of kynurenic acid in the brain. Cannabinoid receptor agonist anandamide treatment modulates NTG-induced changes, suggesting its potential role in influencing the activation and central sensitization processes within the trigeminal system, likely through cannabinoid receptors. 119 The interplay between the kynurenine pathway and other systems, such as the endocannabinoid system, underscores the multifaceted nature of migraine pathophysiology and highlights potential avenues for therapeutic intervention. These findings collectively underscore the pivotal role of the kynurenine pathway in headache disorders and offer promising prospects for the development of novel therapeutic strategies targeting this pathway.
Kynurenine pathway and gastrointestinal disorders
The dysregulation of the kynurenine pathway is linked to several gastrointestinal conditions, notably irritable bowel syndrome (IBS) and Inflammatory Bowel Disease (IBD). This pathway is closely tied to the function of gut microbiota and plays a crucial role in the microbiota-gut-brain axis.
IBS is a functional disorder of the gastrointestinal tract, characterized by symptoms like abdominal pain, bloating, flatulence, changes in bowel habits, and fluctuations in mental well-being. Diagnosis mainly relies on symptom assessment after ruling out other organic pathologies.143,144 IBD, which includes Crohn’s disease and ulcerative colitis, is a chronic inflammatory condition affecting the gastrointestinal tract. Common symptoms include pain, fatigue, persistent diarrhea, weight loss, fever, and, in severe cases, rectal bleeding.145,146
The alteration of the kynurenine pathway in gastrointestinal disorders, fibromyalgia, and chronic fatigue syndrome.
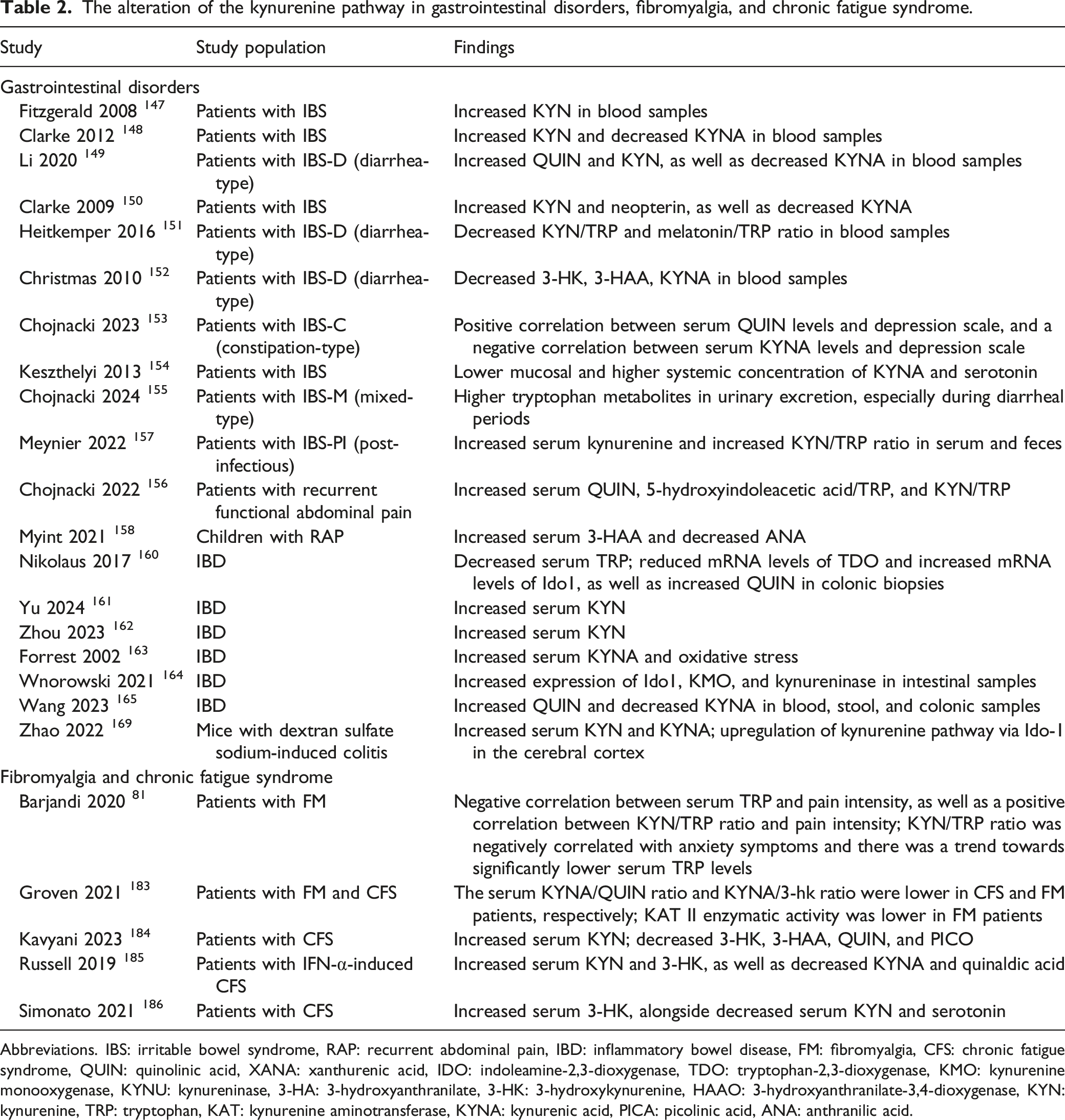
Abbreviations. IBS: irritable bowel syndrome, RAP: recurrent abdominal pain, IBD: inflammatory bowel disease, FM: fibromyalgia, CFS: chronic fatigue syndrome, QUIN: quinolinic acid, XANA: xanthurenic acid, IDO: indoleamine-2,3-dioxygenase, TDO: tryptophan-2,3-dioxygenase, KMO: kynurenine monooxygenase, KYNU: kynureninase, 3-HA: 3-hydroxyanthranilate, 3-HK: 3-hydroxykynurenine, HAAO: 3-hydroxyanthranilate-3,4-dioxygenase, KYN: kynurenine, TRP: tryptophan, KAT: kynurenine aminotransferase, KYNA: kynurenic acid, PICA: picolinic acid, ANA: anthranilic acid.
Kynurenine pathway and irritable bowel syndrome
Research into the kynurenine pathway in IBS unveils a complex interaction among metabolites and their implications for disease progression. Numerous studies have scrutinized these metabolites in patients with either constipation- or diarrhea-type IBS (IBS-C and IBS-D).
Evidence shows that IBS is linked with heightened levels of kynurenine, the kynurenine/tryptophan ratio, and quinolinic acid, alongside reduced levels of kynurenic acid and the kynurenic acid/kynurenine ratio.147–150 Moreover, neopterin levels are elevated in IBS patients compared to controls. 150 These findings suggest heightened activity of IDO in IBS patients, indicating immune-mediated alterations in tryptophan metabolism.148,150
However, Heitkemper et al. reported a potential decrease in the kynurenine/tryptophan ratio (though not statistically significant) and the melatonin/tryptophan ratio in IBS-D patients. A lower melatonin/tryptophan ratio in IBS-D compared to IBS-C and healthy control groups hints at a potential impact on sleep quality in IBS patients, particularly those with diarrhea. 151
On the contrary, another study found no significant variances in the levels of kynurenine, xanthurenic acid, or anthranilic acid between IBS-D patients and controls. Additionally, reduced levels of 3-hydroxykynurenine, 3-hydroxyanthranilic acid, and kynurenic acid were observed in IBS-D patients. 152
Moreover, studies revealed a positive correlation between the severity of IBS symptoms and the kynurenine/tryptophan ratio, particularly in individuals with severe symptoms, who also exhibited an increased likelihood of depression or anxiety. Although IFN-γ levels did not differ among groups, a positive correlation with the kynurenine/tryptophan ratio was noted in IBS subjects. 147 Furthermore, there was a positive correlation between quinolinic acid levels and intestinal symptoms and depression scale scores, and a negative correlation between kynurenic acid levels and intestinal symptoms and depression scale scores in IBS-C and IBS-D patients.149,153
Keszthelyi et al. demonstrated that individuals with IBS displayed lower mucosal concentrations and higher systemic levels of kynurenic acid and serotonin compared to healthy controls. They also found a significant association between mucosal concentrations of serotonin and kynurenic acid and the physiological state in individuals with IBS. 154
Chojnacki et al. evaluated urinary excretion of selected tryptophan metabolites during constipation and diarrhea periods in patients with mixed-type irritable bowel syndrome (IBS-M). They found that all metabolite levels were higher in the patient group and significantly increased during diarrheal periods, particularly 5-hydroxyindoleacetic acid and indican, coinciding with alterations in the gut microbiome. 155
Similarly, Chojnacki et al. investigated tryptophan metabolism in patients experiencing recurrent functional abdominal pain. They discovered significantly higher ratios of 5-hydroxyindoleacetic acid/tryptophan, kynurenine/tryptophan, and levels of quinolinic acid in patients compared to controls. Interestingly, reducing tryptophan consumption led to a significant decrease in quinolinic acid levels. 156
Furthermore, research showed significant changes in the kynurenine pathway post-Citrobacter rodentium infection, with infected mice exhibiting elevated levels of serum kynurenine and an increased kynurenine/tryptophan ratio in fecal and serum samples, suggesting potential alterations in the kynurenine pathway’s regulation in post-infectious IBS (IBS-PI). 157
Myint et al. examined the levels of kynurenine pathway metabolites in children with recurrent abdominal pain (RAP). They found significantly higher levels of serum TNF-α in children with RAP, along with lower levels of IL-10, IL-6, and brain-derived neurotrophic factor (BDNF). Additionally, there was a notable increase in the metabolite 3-hydroxyanthranilic acid and a decrease in anthranilic acid, though the levels of tryptophan, kynurenine, and the kynurenine/tryptophan ratio did not differ significantly between children with RAP and controls. 158
Clarke et al. explored the relationship between toll-like receptor (TLR) activation and kynurenine pathway activity in IBS. Elevated kynurenine/tryptophan ratios were observed in IBS patients compared to controls. Furthermore, differential profiles of kynurenine production were found following TLR activation in IBS patients compared to controls, notably at TLR1/2, TLR2, TLR3, TLR5, TLR7, and TLR8. These findings suggest potential utility of measuring tryptophan metabolites downstream of TLR activation as biomarkers for IBS diagnosis. 148 Additionally, research has demonstrated that TLR4 plays a role in migraine-like behavior and nucleus caudalis neuronal activation through myeloid differentiation primary response gene 88 (MyD88) in mice. 159 Thus, TLR activation may contribute to migraine development through neuroinflammatory pathways. However, further studies are needed to delineate the precise role of TLR activation and its relationship with the kynurenine pathway in IBS, migraine, and associated inflammation.
Given the contradictory findings regarding kynurenine metabolite levels in IBS, it is imperative to conduct further studies with larger participant cohorts to precisely delineate the role of the kynurenine pathway in IBS.
Kynurenine pathway and inflammatory bowel disease
Research has shown that serum tryptophan levels are significantly lower in patients with IBD compared to controls, especially in those with Crohn’s disease. Lower tryptophan levels have been found to correlate with disease activity and C-reactive protein (CRP) levels. In colonic biopsies from IBD patients, there is a reduction in mRNA levels of TDO, while IDO1 mRNA levels are elevated. Furthermore, analysis of tryptophan metabolites has revealed increased activity in the kynurenine pathway during active IBD, particularly with elevated levels of quinolinic acid. 160
In IBD patients, elevated levels of kynurenine have been observed, suggesting its potential as a distinguishing factor between Crohn’s disease and ulcerative colitis patients.161,162 Additionally, researchers have noted increased activity in the kynurenine pathway in IBD patients, alongside heightened oxidative stress even during inactive disease phases. Both Crohn’s disease and ulcerative colitis patients show elevated levels of kynurenic acid, despite unchanged neopterin levels indicating disease quiescence. 163
The dysregulation of the kynurenine pathway in IBD is marked by increased expression of rate-limiting enzymes such as IDO1, KMO, and kynureninase, 164 leading to elevated levels of kynurenine and its metabolites. Metabolomic analyses consistently show elevated kynurenine levels in blood, stool, and colonic biopsies of IBD patients, particularly during active inflammation. Additionally, alterations in the expression of KAT are observed in IBD patients, with implications for the production of neuroactive metabolites such as kynurenic acid and quinolinic acid. Notably, kynurenic acid, with its neuroprotective properties, shows decreased levels in Crohn’s disease patients, while quinolinic acid, a potential neurotoxin, is increased in IBD patients. The dysregulation of kynurenic acid and quinolinic acid ratios may contribute to altered gut-brain signaling and potentially exacerbate symptoms such as intestinal hypermotility and depression observed in IBD patients. 165
A negative correlation has been shown between intestinal inflammation severity in IBD and levels of xanthurenic and kynurenic acids in both mice (with sodium sulphate-induced colitis) and humans. Supplementation with xanthurenic acid or kynurenic acid has been found to alleviate colitis severity through Aryl hydrocarbon Receptor (AhR) activation and modulation of cellular energy metabolism in intestinal epithelial cells and T cells. 166
Wang et al. identified a positive feedback loop involving IL-6, IDO1, and AhR, which accelerated kynurenine pathway metabolism in the gut. Additionally, it revealed a negative feedback mechanism involving AhR and GPR35 that regulates intestinal damage and inflammation, maintaining intestinal integrity and homeostasis. Through virtual screening and biological verification, the study identified vardenafil and linagliptin as GPR35 and AhR agonists, respectively, among approved drugs. Importantly, in vivo experiments demonstrated that vardenafil (GPR35 agonist) and linagliptin (AhR agonist) significantly mitigated chemotherapy-induced intestinal toxicity. 167
Overexpression of IDO1 in the intestinal epithelium led to increased secretory cell differentiation and mucus production, with thicker mucus layers and altered microbial composition. IDO1 interacts with the AhR to inhibit NOTCH1 activation. IDO1 transgenic mice demonstrated reduced bacterial load and protection against inflammatory insults. In Crohn’s disease patients, increased IDO1 expression is correlated with elevated secretory cell markers. 168
Dextran sulfate sodium (DSS)-treated mice exhibited elevated levels of kynurenine and kynurenic acid in serum, regulated by IDO-1 and KAT. In the cerebral cortex, DSS-induced colitis upregulated the kynurenine pathway via IDO-1, leading to a neurotoxic astrocytic phenotype. Gut microbiota alterations in DSS-induced colitis correlated with changes in tryptophan metabolism, implicating a role for intestinal flora in kynurenine pathway dysregulation. Additionally, analysis of ulcerative colitis patient data (using Gene Expression Omnibus datasets) confirmed higher IDO-1 levels in peripheral blood monocytes and colon. 169
Yisiyerili et al. investigated the effects of the angiotensin receptor blocker (ARB) irbesartan on stress-induced intestinal inflammation, considering the association between stress, IBS, and hypertension. In stressed mice, they observed an upregulation of IDO-1 and TDO mRNA, along with a downregulation of tryptophan hydroxylase-1 (TPH-1) mRNA in both the intestine and liver. Additionally, stress was associated with decreased plasma levels of tryptophan and serotonin, and an increase in plasma kynurenine levels. However, in stressed mice, irbesartan demonstrated a dose-dependent restoration of intestinal expression of IDO-1, TDO, and TPH-1. Moreover, irbesartan exhibited a dose-dependent restoration of plasma levels of tryptophan, serotonin, and kynurenine. 170
Puerarin has been found to effectively treat ulcerative colitis by restoring gut microbiota composition and metabolic activities. It reverses 3-hydroxyanthranilic acid levels and increases the abundance of Rhodobacteraceae and Halomonadaceae taxa, which are crucial players in kynurenine metabolism. 171 Bao et al. found that moxibustion, particularly when combined with IDO1 inhibitor 1-methyltryptophan (1-MT), effectively reduced intestinal inflammation and depressive behavior in rats with Crohn’s disease. This effect was associated with downregulation of IDO1 levels in the colon and hippocampus, inhibition of pro-inflammatory cytokines, modulation of kynurenine pathway metabolites, and reduction of hippocampal microglia activation and neuronal activity. 172
Therapeutic options
Therapeutic approaches for IBS and IBD often target the dysregulated kynurenine pathway, a key player in gastrointestinal disorders. For IBS patients, interventions may include reducing tryptophan consumption to decrease quinolinic acid levels, which could contribute to symptom severity and mood disorders.173,174 Additionally, targeting the kynurenine/tryptophan ratio, linked to symptom severity and psychological comorbidities, may prove beneficial. Studies suggest that adjusting kynurenine metabolites like quinolinic acid and kynurenic acid could offer relief, especially for IBS-C patients. Moreover, interventions to restore mucosal concentrations of serotonin and kynurenic acid might rebalance intestinal homeostasis.
In IBD, therapies might focus on inhibiting enzymes involved in the kynurenine pathway, such as IDO1 and KMO, to reduce the production of neurotoxic metabolites like quinolinic acid while promoting the synthesis of neuroprotective compounds like kynurenic acid. Additionally, targeting inflammatory pathways associated with the kynurenine pathway, like IL-6 and AhR signaling, could alleviate intestinal inflammation and oxidative stress in IBD patients. Emerging treatments such as ARBs and remedies like puerarin and moxibustion show promise in modulating the kynurenine pathway and alleviating symptoms of IBD.
Overall, therapeutic strategies for IBS and IBD aim to restore the balance of kynurenine pathway metabolites, mitigate inflammation, and alleviate gastrointestinal symptoms and associated psychological distress.
Kynurenine pathway and fibromyalgia and chronic fatigue syndrome
Evidence is growing to suggest that the kynurenine pathway is implicated in the development of CNS-related disorders. Chronic Fatigue Syndrome (CFS) and Fibromyalgia (FM) are two interconnected disorders whose pathology remains elusive. However, shared symptoms, such as depression, hint at the involvement of the CNS in their development. 175
FM is characterized by persistent symptoms including widespread pain, fatigue, heightened muscle tenderness, and increased sensitivity to pain. 176 Meanwhile, CFS is a debilitating long-term condition with a diverse array of symptoms. 177 Both disorders are prevalent, with CFS affecting 0.5 to 2.5% of the population and FM impacting up to 5%.178,179
Chronic inflammation, commonly observed in patients with Myalgic Encephalomyelitis (ME)/CFS, triggers the activation of microglia and astrocytes. Recent evidence suggests that microglial activation extends across extensive brain regions in individuals diagnosed with ME/CFS. 180 Neuroinflammation is evident in various brain regions of ME/CFS patients and is correlated with the severity of neuropsychological symptoms. 181 Studies indicate correlations between CRP, the kynurenine/tryptophan ratio, insulin resistance, and CFS symptoms, including psychosomatic symptoms in COVID-19 recovery patients. 182
Barjandi et al. investigated tryptophan and kynurenine levels in FM and temporomandibular disorders myalgia (TMDM) patients compared to healthy controls. They discovered a negative correlation between tryptophan levels and pain intensity, alongside a positive correlation between the kynurenine/tryptophan ratio and pain intensity. Additionally, in FM patients, the kynurenine/tryptophan ratio showed a negative correlation with anxiety symptoms, and tryptophan levels tended to be significantly lower compared to healthy individuals. 81
Groven et al. found that the kynurenic acid/quinolinic acid ratio and the kynurenic acid/3-hydroxykynurenine ratio were lower in CFS and FM patients, respectively, compared to healthy controls. This decrease in neuroprotective ratio in FM was associated with increased pain symptoms. Moreover, KAT II enzymatic activity (xanthurenic acid/3-hydroxykynurenine ratio) was lower in FM patients compared to healthy controls. 183
Kavyani et al. reported increased kynurenine production in CFS patients compared to healthy controls, which correlated with symptom severity. They also observed decreased levels of 3-hydroxykynurenine, 3-hydroxyanthranilic acid, quinolinic acid, and picolinic acid, potentially impacting cellular energy levels negatively. 184
Russell et al. demonstrated that an IFN-α-induced model of CFS is linked to increases in kynurenine, 3-Hydroxykynurenine, and the ratio of kynurenine to tryptophan, as well as decreases in kynurenic acid and quinaldic acid. 185 In another study, Simonato et al. revealed elevated levels of IL-17A, fatty acid binding protein-2 (FABP-2), and 3-hydroxykynurenine, alongside reduced levels of kynurenine and serotonin in CFS patients compared to healthy controls. They noted an increased kynurenic acid/kynurenine ratio (kynurenine aminotransferase activity) and 3-hydroxykynurenine/kynurenine ratio (3-monooxygenase activity). Furthermore, patients whose CFS onset followed an infection exhibited lower kynurenine levels compared to those with no infection-triggered onset. 186
Considering these findings (Table 2), it’s evident that the kynurenine pathway may contribute to FM and CFS. While some studies have reported increased kynurenine production in CFS patients compared to healthy controls, others have found decreased levels of kynurenine. Further research incorporating larger, more diverse cohorts and standardized methodologies is necessary to reconcile these conflicting findings and elucidate the precise role of the kynurenine pathway in CFS.
Conclusion
In this review, we emphasized the significance of the kynurenine pathway and its metabolites in the etiology of neuropathic pain, headaches, gastrointestinal disorders, FM, and CFS, as well as its involvement in the comorbidity of depression and chronic pain. A burgeoning body of literature indicates that kynurenine and its derivatives possess the capacity to modulate various classical neurotransmitter systems via direct or indirect pathways. Extensive investigations in animal models of chronic pain have delineated the participation of kynurenine pathway enzymes and metabolites in both peripheral and central nervous system mechanisms, alongside the repercussions of enzyme inhibition. Nonetheless, human evidence remains constrained. Deciphering the underlying mechanisms of this pathway holds promise for the development of individualized, innovative, and non-opioid therapeutic modalities for pain management. Thus, there exists a pressing need to broaden research endeavors in this domain, which could potentially enhance therapeutic efficacy. Furthermore, it is imperative to explore gender-specific disparities in underlying mechanisms. Augmenting understanding of this pathway has the potential to shift focus from symptom management, such as pain alleviation, towards disease prevention.
Footnotes
Authors’ contributions
S.P.E conceptualized the study. All of the authors performed the literature search and wrote the draft. E.H and S.P.E edited the article. E.H and S.P.E have the co-first authorship and contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
