Abstract
Objective:
This study aimed to investigate the specific manifestations and differences in brain network activity and functional connectivity between brain networks in patients with trigeminal neuralgia and migraine, aiming to reveal the neural basis of these two diseases.
Background:
Head and facial pain, including trigeminal neuralgia and migraine, is prevalent globally. However, the underlying neural mechanisms of these conditions remain unclear. Resting-state functional connectivity studies based on independent component analysis (ICA) may offer new insights into these diseases.
Methods:
The study involved 23 chronic migraine, 37 episodic migraine, 21 trigeminal neuralgia patients, and 33 age- and gender-matched controls. Resting-state functional magnetic resonance imaging was performed, and five sets of brain network components were extracted through ICA. Neuronal activity indicators were calculated for each participant’s independent components, including amplitudes of low-frequency fluctuation (ALFF) and regional homogeneity (ReHo). Functional connectivity was also assessed and compared among the four groups.
Results:
Trigeminal neuralgia patients showed reduced ALFF in the dorsal attention network versus episodic migraine patients and controls. Both trigeminal neuralgia and chronic migraine patients had decreased ReHo in this network. Migraine patients had weaker connectivity between the default mode and visual networks than controls. Trigeminal neuralgia patients also showed higher connectivity between the somatosensory motor and dorsal attention networks. Compared to episodic migraine, trigeminal neuralgia, and chronic migraine patients had increased connectivity between the visual and dorsal attention networks.
Conclusion:
The study provides evidence that long-term chronic head and facial pain may contribute to abnormalities in the activation and connectivity of the dorsal attention network. Compared to migraine patients, trigeminal neuralgia patients exhibit abnormal brain network connectivity, particularly within the somatomotor network, which may explain the presence of significant “trigger points.” These findings offer new perspectives for understanding the characteristics of different head and facial pain subtypes.
Keywords
Background
Trigeminal neuralgia and migraine are two clinically common chronic pain conditions that have a profound impact on patients’ quality of life.1,2 Although both diseases manifest as head and facial pain and are accompanied by some different clinical symptoms,3,4 the specific differences in their pathophysiology are not yet clear. Revealing the pathological characteristics of these two different types of headaches is significant for deeply understanding the pathogenesis of headaches and formulating targeted therapeutic strategies.
Previous studies have indicated that the pathological mechanism of trigeminal neuralgia is primarily attributed to abnormalities in the trigeminal nerve pathway.5,6 Similarly, in migraine research, some researchers believe that the pathogenesis of migraine is also related to abnormalities in the trigeminal nerve pathway.7,8 An article recently published in the journal Science points out that cerebrospinal fluid (CSF) can establish non-synaptic signal transmission between the brain and trigeminal neurons. After the occurrence of cortical spreading depression (CSD), a transient neurological dysfunction associated with headaches in migraine patients, the proteomic composition of CSF changes, which further activates trigeminal neurons. 9 In addition, the significant efficacy of drugs targeting calcitonin gene-related peptide (CGRP) in treating migraines indicates that the peripheral trigeminal system may be an important target for migraine pathology. 10 Besides the commonality of trigeminal nerve pathway abnormalities, both trigeminal neuralgia and migraine exhibit abnormalities in pain processing pathways, further revealing their complexity and interrelation in pathological mechanisms.11,12 Taken together, multiple studies have detected abnormalities in various brain functions among patients with trigeminal neuralgia, including the prefrontal lobe, temporal lobe, parietal regions, posterior cingulate cortex, insula, and cerebellum.13–15 In migraine research, a review has indicated that regions such as the thalamus, insula, brainstem, periaqueductal gray, primary somatosensory cortex, and hippocampus have been repeatedly implicated in the pathophysiology of migraine across 114 studies. 16 These studies suggest that while the two types of headaches exhibit similar patterns of changes in some brain regions, there are also non-overlapping brain regions with their specific changes.
In recent years, neuroscience and imaging technology have continuously progressed, enabling many researchers to conduct in-depth studies on the brain network function of patients with trigeminal neuralgia and migraine during resting state.17–19 Androulakis et al.20,21 discovered that patients with chronic migraine exhibit reduced functional connectivity in the default mode network, salience network, and executive control network during resting state compared to healthy controls, suggesting a potential link between the connectivity of these intrinsic networks and the chronic development and persistence of migraine. A recent review on brain functional connectivity in migraine patients also pointed out that, in addition to the significant involvement of the default mode network and salience network, the visual and sensorimotor networks also exhibited abnormal activity patterns. 16 Similarly, patients with trigeminal neuralgia demonstrate distinct functional connectivity patterns in the default mode network, sensorimotor network, and salience network compared to healthy individuals, with abnormalities in the salience network correlating negatively with the degree of pain. 22 Research also emphasizes the important role of dynamic pain connectome regions in the pathophysiology of trigeminal neuralgia, including the default mode network, cognitive control network, and salience network. 15 Although some studies have separately explored the brain network characteristics of trigeminal neuralgia and migraine, a systematic and direct comparative analysis remains absent.
This study aims to identify brain networks through independent component analysis using resting-state neuroimaging techniques, thereby deeply analyzing the differences between trigeminal neuralgia and migraine regarding brain network activation and connectivity. We divided migraine patients into two groups, chronic migraine and episodic migraine, to distinguish between patients with long-term chronic pain and those with relatively fewer attacks. 4 We speculate that the impact of headaches on patients’ brain function may involve multiple brain functional networks, including the attentional network, sensorimotor network, default mode network, and frontoparietal network, and there may be differences in the patterns of influence on these networks between trigeminal neuralgia and migraine. By comparing and analyzing brain activity data from patients with different diseases, we hope to reveal their unique manifestations at the neural network level. This will help us better understand the pathogenesis of these two diseases and provide a more accurate and scientific basis for clinical diagnosis and treatment.
Methods
Participants
Initially, our study recruited 146 participants, but due to inadequate image quality, 19 participants were excluded, and another 13 were disqualified owing to excessive head movement. Consequently, a cohort of 114 participants was finalized for this investigation. This cohort encompassed 23 chronic migraine patients (mean age: 44.7 ± 12.4 years), 21 individuals with trigeminal neuralgia (mean age: 50.62 ± 7.98 years), 37 episodic migraine patients (mean age: 43.16 ± 10.40 years), and 33 healthy controls matched for age and gender (mean age: 45.24 ± 9.67 years). All patients were sourced from the outpatient clinic of the Neurology Department at Shanghai Ninth People’s Hospital, affiliated with Shanghai Jiao Tong University School of Medicine. To ensure diagnostic accuracy, two seasoned neurologists validated and categorized the diagnoses of all patients according to the International Classification of Headache Disorders (3rd edition, ICHD-3) guidelines. 23
Patients suffering from other mental illnesses or taking prophylactic medications were excluded from the study. The patients included in this study did not experience headache episodes 2 days before the MRI scan, and there were no episodes the day after the scan as well. Additionally, during the scanning process, they did not report any noticeable discomfort or aches.
The healthy group participants presented with no past or present bodily or psychological issues, nor did they have any first-degree relatives who had ever experienced migraines or other types of headaches. In addition, we excluded participants who were left-handed or had a history of neurological or metabolic conditions such as diabetes or heart diseases, substance misuse, and mental health disorders, as determined through medical assessments and comprehensive interviews. The medical and demographic details of the subjects are presented in Table 1.
Demographic information.

MRI scanning
At the School of Psychology at Shanghai University of Sport, the participants were scanned utilizing the 3.0 Tesla Siemens Trio Prisma MRI system (Siemens Healthcare, Erlangen, Germany), which was equipped with a 20-channel head coil. We required the participants to keep their eyes closed during the scanning process, not to fall asleep, and to keep their heads stationary at the same time. From each individual, two collections of data were acquired. A T2*-weighted gradient-echo echo-planar imaging (EPI) pulse sequence was employed to obtain the resting-state fMRI data, with the following parameter values: repetition time (TR) = 2000 ms; flip angle (FA) = 90 degrees; 3.5 mm slice thickness with 20% distance factor; echo time (TE) = 30 ms; field of view (FOV) = 220 mm × 220 mm; matrix = 64 × 64; and time points = 210. The following parameter values were used to acquire high-resolution structural images, which were obtained through a 3D magnetization-prepared rapid-acquisition gradient-echo pulse sequence.: TR = 2530 ms; inversion time (TI) = 1100 ms; TE = 2.34 ms; FA = 7 degrees; matrix size = 256 × 256; FOV = 256 mm × 256 mm; number of slices = 192; sagittal orientation; and each slice had a thickness of 1 mm and a 50% gap.
Pre-processing of data
Utilizing the Data Processing Assistant for Resting-State fMRI package (DPARSF, obtained from http://rfmri.org/DPARSF), which is constructed upon the Statistical Parametric Mapping software (SPM-12, obtained from http://www.fil.ion.ucl.ac.uk/spm), the fMRI data underwent preprocessing. The data preprocessing workflow consisted of six steps in total: (1) from the time-series fMRI images of each subject, data from the first 10 time points, which were presumably collected prior to reaching steady-state magnetization, were excluded; (2) slice acquisition times were corrected using the slice timing; (3) to correct for head motion, the images were spatially realigned. Any images with a displacement greater than 2.0 mm or a rotation exceeding 2.0 degrees were subsequently discarded; (4) in order to eliminate the linear trend. The time-series signals of each voxel were detrended and then filtered using a band-pass filter with a passband ranging from 0.01 to 0.08 Hz; (5) following processing, the images were standardized by applying EPI templates to align with the Montreal Neurological Institute (MNI) standard brain space, and subsequently resampled to a resolution of 3 × 3 × 3 mm³; and (6) using a regression algorithm, the preprocessed data was utilized to compute nuisance covariates, which included cerebrospinal fluid signal, global mean signal, white matter signal, and the 24 head motion parameters. 24
ALFF, ReHo, and DC analyses
Resting-state fMRI has proven to be an effective technique for exploring the intrinsic neural activities of the brain. Various metrics, such as ALFF, ReHo, and DC, have been devised to assess different facets of spontaneous brain activity. We generated ALFF, ReHo, and DC maps utilizing the DPARSF software package. We employed the fast Fourier transform to analyze the frequency spectra of each voxel’s time-series signals to create the ALFF maps. Before calculating ALLF, we applied spatial smoothing to the functional images using a 3D Gaussian filter with a 6 mm full-width at half-maximum (FWHM). ALFF was then determined by calculating the square root of the average power spectrum density across the frequency range of 0.01 to 0.08 Hz. Finally, we applied the Fisher transformation to convert the ALFF maps into Z-score maps to improve normality.
To assess ReHo, we employed Kendall’s coefficient of concordance (KCC), comparing the time-series signals of every voxel with its adjacent 26 voxels in the gray matter. The derived ReHo maps were normalized for standardization purposes by dividing them by the average KCC value across the participant’s entire brain. The ultimate images underwent smoothing with a 3D Gaussian kernel featuring a 6 mm isotropic full-width at half-maximum (FWHM).
Independent component analysis
Independent Component Analysis (ICA) is one of the most commonly used data-driven methods for processing fMRI imaging data. ICA plays a significant role in fMRI imaging analysis due to its strong robustness to artifacts, minimal assumptions about the shape of time courses or spatial patterns, and ease of operation. One of the notable advantages of ICA is its ability to extract components representing large-scale neural networks from complex fMRI signals. We utilize the GIFT fMRI toolbox specifically designed for group ICA to achieve this. We concatenate the data from each subject temporally and apply the Infomax algorithm, based on the maximum information transfer principle, to calculate 17 subject-specific Independent Components (ICs). Finally, we compare these Independent Components with the YeoRSN 25 to select the most suitable ones.
ROI analysis based on ICA results
Using ICA-derived components as ROIs, we extracted key metrics, including ALFF and ReHo, from subjects across experimental groups. Data were then analyzed via one-way ANOVA in SPSS. Statistical significance was set at p < 0.05.
Functional connectivity
FC is defined as the temporal correlation that exists between two or more anatomically distinct time series. As mentioned earlier, the five seed regions were derived from the brain networks identified through ICA. For each pair of regions, the corresponding time series were extracted. The Pearson correlation coefficient was then used to determine the FC between the brain regions’ extracted time courses, yielding a 5 × 5 FC weight matrix. Fisher’s r-to-z transformation was subsequently employed to enhance normality.
Statistical analysis
We utilized SPSS statistical software (version 24.0) to compare demographic data and compute significance levels. We employed skewness and kurtosis tests to examine the normality of continuous variables. Subsequently, we used one-way ANOVA to assess statistical significance levels. Additionally, we analyzed gender composition using the Chi-Square test.
For the extracted ALFF and ReHo values, we also used SPSS statistical software (version 24.0) to compare the differences between groups for each ROI extraction value and the FC among ROIs. We employed one-way ANOVA to evaluate the statistical significance level, with a statistical significance threshold set at p < 0.05.
Results
Demographic and clinical characteristics
Table 1 displays demographic and clinical information related to chronic migraine, episodic migraine, trigeminal neuralgia, and healthy groups. There were no significant differences in age and gender distribution among the four groups.
ICA results
By comparing with the YeoRSN template, we selected five independent components from seventeen as regions of interest. These five independent components represent the dorsal attentional network, somatomotor network, default mode network (DMN), frontoparietal network, and visual network. Refer to Figure 1 for details.
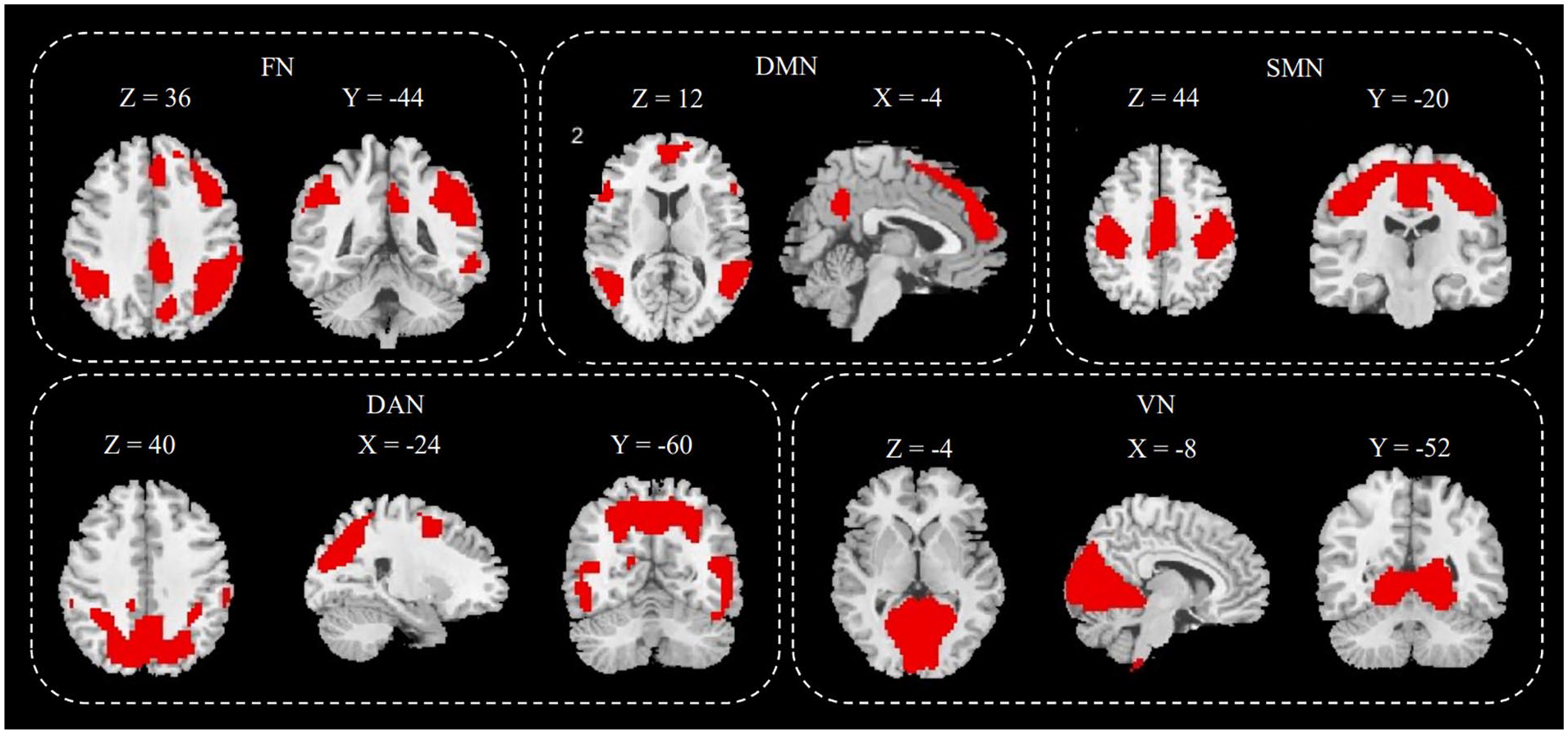
After performing independent component analysis, five components were selected from the 17 independent components as regions of interest.
Between-group comparison of ALFF and ReHo
Our research findings indicate significant differences in ALFF and ReHo values among the four participant groups within the dorsal attentional network. Detailed posthoc analyses unveil that patients suffering from trigeminal neuralgia manifest notably reduced ALFF values within the dorsal attentional network in comparison to both migraines without aura patients and healthy counterparts. Moreover, a similar trend is observed in the ReHo values of trigeminal neuralgia patients, which are considerably lower than those of the episodic migraine cohort and the healthy controls. Furthermore, our findings indicate that individuals with chronic migraine exhibit distinctly lower ReHo values relative to the healthy controls. For detailed information, please see Tables 2 and 3 and Figure 2.
Among-group comparison of amplitudes of low-frequency fluctuation (ALFF).

“*” represents a significant difference among the four groups.
Among-group comparison of regional homogeneity (ReHo).

“*” represents a significant difference among the four groups.
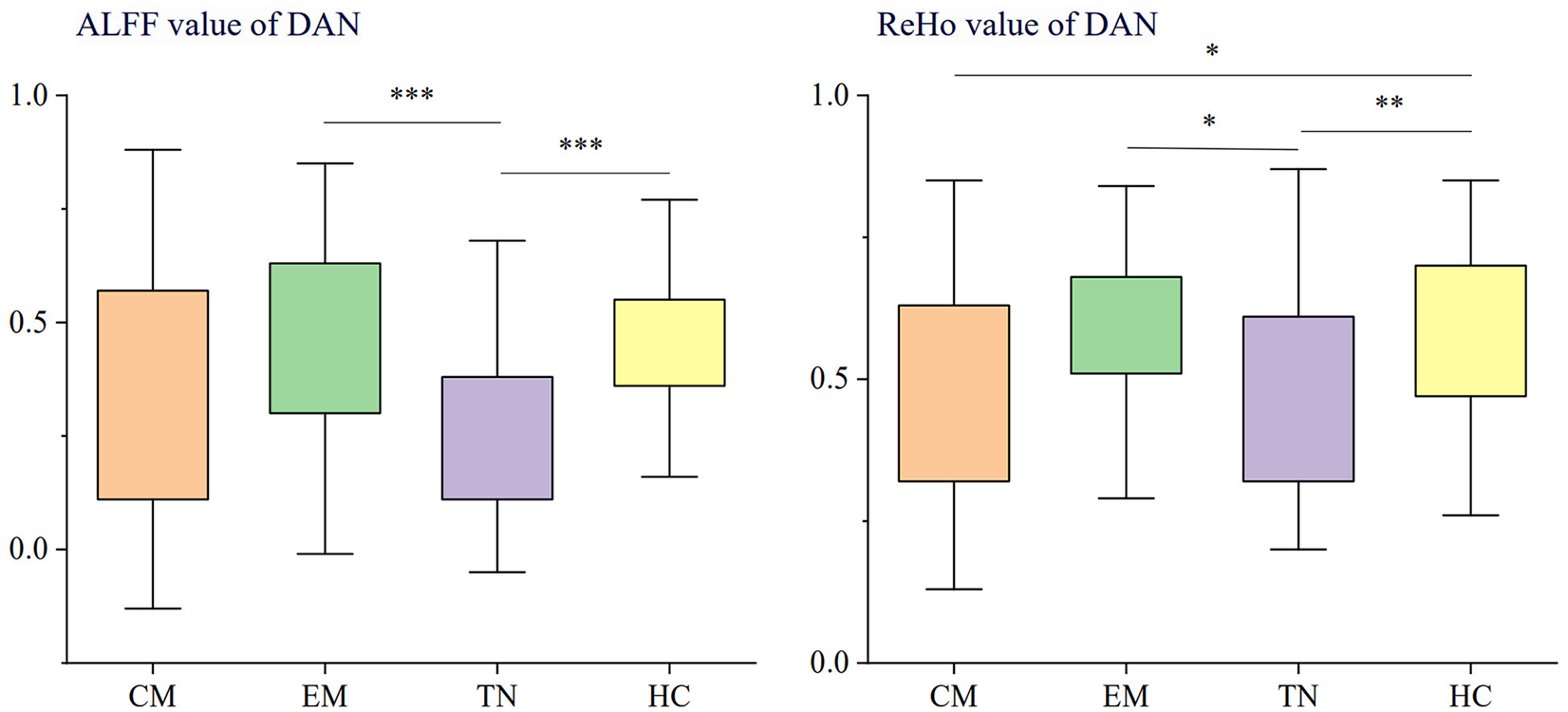
Contrast in ALFF and Reho within the dorsal attention network between groups of chronic migraine, episodic migraine, trigeminal neuralgia, and healthy controls.
Between-group comparison of FC
After conducting a detailed comparison of functional connectivity strength among ROIs, we observed significant differences in functional connectivity among multiple brain networks across the four participant groups. Post-hoc analysis revealed that, compared to the healthy control group, the chronic migraine and episodic migraine groups exhibited significantly reduced functional connectivity strength between the default mode network and the visual network. Simultaneously, we found that the connectivity strength between the somatomotor and dorsal attentional networks in patients with trigeminal neuralgia was also significantly higher than that in healthy individuals. Additionally, patients with episodic migraine showed notably lower connectivity strength between the visual network and the dorsal attentional network compared to patients with trigeminal neuralgia and chronic migraine. For more details, refer to Table 4 and Figure 3.
Among-group comparison of functional connectivity (FC) results.

DAN: dorsal attentional network; DMN: default mode network; FN: frontoparietal network; SMN: somatomotor network; VN: visual network.
“*” represents a significant difference among the four groups.

There are significant differences in functional connectivity between multiple brain networks among the four participant groups.
Discussion
This study utilizes a combination of ICA and FC to investigate the characteristics of different brain functional activities in migraine, chronic migraine, and trigeminal neuralgia with fMRI during the resting state. The results reveal that when compared to healthy controls and patients with episodic migraine, the ALFF and ReHo values of the dorsal attention network in patients with trigeminal neuralgia decreased. Similarly, patients with chronic migraine exhibit a comparable downward trend in ReHo values within the dorsal attention network. However, no similar changes were observed in episodic migraine patients. These findings suggest that the activity of the dorsal attention network in patients with trigeminal neuralgia and chronic migraine is reduced, which provides a new perspective for understanding different subtypes of headache.
Based on numerous behavioral studies on migraine and trigeminal neuralgia, these painful conditions frequently result in attentional function decline. 18 Additionally, reports indicate a frequent reduction in metacognitive abilities due to deactivating the dorsal attention network.26,27 This study hypothesizes that the deactivation of the dorsal attention network could be a significant contributor to cognitive decline in patients with chronic migraine and trigeminal neuralgia. This result is consistent with previous research inferences. Some reports have indicated that patients with trigeminal neuralgia have thinner cortical thickness in brain regions related to the dorsal attention network compared to healthy controls,28,29 and this phenomenon is also observed in studies of chronic migraine. 30 In this study, patients with migraines without aura did not demonstrate significant deactivation of the dorsal attention network. This could be explained by the fact that episodic migraine is typically not associated with long-term chronic pain, contrasting sharply with chronic migraine and trigeminal neuralgia. Therefore, it is inferred that the reduced activity in the dorsal attention network among patients with chronic migraine and trigeminal neuralgia could be a consequence, rather than a cause, of long-term chronic pain.
In this study, a comparison of functional connectivity between different networks was conducted. The results indicated that patients with chronic migraine and trigeminal neuralgia exhibited significantly stronger functional connectivity between the dorsal attention network and the visual network than migraine patients without aura. Previous research on patients with chronic migraine has reported abnormalities in the functional connectivity between these two networks.16,31 The visual network, as a component of the sensory system, is responsible for processing external stimuli, including pain information. 32 Conversely, the dorsal attention network serves as a sophisticated cognitive network that recognizes external stimuli, among which pain information holds significance, along with relevant tasks.33–35 It is worth noting that previous studies on the visual network of patients with chronic migraine and episodic migraine have shown that there are abnormalities in the functional connectivity of the visual network in patients with chronic migraine, 36 while the visual network of patients with episodic migraine does not significantly differ from that of the normal control group. 37 The results of this study are consistent with previous findings and further reveal that patients with trigeminal neuralgia also have abnormalities in the visual and dorsal attention networks. Based on these findings and previous studies, it is hypothesized in this study that chronic pain associated with chronic migraine and trigeminal neuralgia may result in an additional cognitive load, strengthening the connection between the visual and dorsal attention networks.
Compared to the dorsal attention network, which primarily processes external stimuli and tasks, the default mode network is a negative network supporting internal psychological exploration. 38 Specifically, the default mode network activation decreases during task states and increases during resting states. Previous studies on migraine have shown that compared with healthy controls, patients with episodic migraine exhibit significantly reduced connection strength between the default mode network and the visual network. 39 This finding is consistent with the results of the present study, which further confirms that the same phenomenon is also observed in patients with chronic migraine. This may indicate that the decreased connection strength between the default mode network and the visual network is a common pathological feature among patients with various types of migraine. However, it is worth noting that the difference between patients with trigeminal neuralgia and the control group is not significant in our research findings. We believe that this may be due to the fact that there is not a close correlation between trigeminal neuralgia and visual sensation.
Our study revealed that the linkage between the somatomotor and dorsal attention networks in patients suffering from trigeminal neuralgia is notably stronger compared to the healthy control group, corroborating previous research on individuals with chronic pain. 40 The somatomotor network is pivotal in the sensory-discriminative dimension of pain processing,41,42 and its impact on pain threshold is also evident across different populations.43,44 It is worth noting that trigeminal neuralgia and migraine manifest differently pathologically. In particular, trigeminal neuralgia patients may experience discomfort upon contact with specific zones, referred to as “trigger points.” 45 However, migraine patients do not have this pain trigger mechanism. Correspondingly, in this study, neither episodic nor chronic migraine patients showed abnormal connections between the somatomotor and dorsal attention networks. As a result, we postulate that the abnormal connectivity between the somatomotor network and the dorsal attention network in trigeminal neuralgia cases might be closely associated with the existence of these “trigger points.”
In summary, there are certain degrees of connections and distinctions between the intra-network activity intensity and inter-network connection intensity in the three headache subtypes. Specifically, we found that patients with trigeminal neuralgia exhibited significantly reduced activity in the dorsal attention network, manifested by decreased values of ALFF and ReHo. This finding is similar to the situation in patients with chronic migraine, although the changes in patients with chronic migraine are mainly reflected in ReHo values. In contrast, patients with episodic migraine did not show significant differences in these two indicators, suggesting that the reduction in dorsal attention network activity may be more closely related to the state of long-term chronic pain. Further analysis revealed that patients with chronic migraine and trigeminal neuralgia not only exhibited abnormalities within the dorsal attention network but also showed significantly enhanced functional connectivity between this network and the visual network. This finding may reflect an increased demand for pain information processing in the brain under chronic pain conditions, thereby enhancing the interaction between the dorsal attention network responsible for external stimulus processing and the visual network responsible for visual information processing. On the other hand, we noted the performance of the default mode network in migraine patients. Compared with the healthy control group, patients with episodic migraine showed significantly reduced connection intensity between the default mode network and the visual network, a finding also validated in patients with chronic migraine. However, this difference was not significant in patients with trigeminal neuralgia, which may be related to the weaker direct correlation between trigeminal neuralgia and visual perception. Furthermore, this study also found significantly enhanced connectivity between the sensorimotor network and the dorsal attention network in patients with trigeminal neuralgia, which may be related to the unique “trigger point” phenomenon in trigeminal neuralgia. In contrast, neither patients with episodic migraine nor patients with chronic migraine showed abnormalities in the connectivity between these two networks, further emphasizing the differences in pathological mechanisms among different headache subtypes.
Limitation
The current study has several limitations. Firstly, our study adopted a cross-sectional design, which limits the ability to infer causality from the observed results. Secondly, the sample sizes of both the trigeminal neuralgia and chronic migraine groups are not large enough, and under strict statistical thresholds, we can only identify the main differences. Large cohort studies in the future can help discover more interesting results. Lastly, more clinical data is needed to validate imaging findings and establish causal relationships between imaging results and behavior.
Conclusion
Overall, this study utilized the ICA method to identify common brain networks among patients with various headache subtypes and compared the activation and connectivity of these brain networks among the patients. The research findings indicate that the additional attentional load caused by long-term chronic headaches may contribute to abnormalities in the activation and connectivity of the dorsal attention network. Meanwhile, compared to migraine patients, patients with trigeminal neuralgia exhibit abnormal brain network connectivity, especially within the somatomotor network, which may be the primary reason for the presence of significant “trigger points.” These discoveries corroborate current research on chronic pain and provide a new perspective for understanding the characteristics of patients with trigeminal neuralgia, thereby facilitating a deeper understanding of the brain function features of different headache subtypes.
Footnotes
Acknowledgements
We thank the patients who participated in the study for their support.
Availability of data and materials
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by grants from the Social Sciences and Humanities project of the Ministry of Education (23A10277004 to X. X. Du), the Shanghai Municipal Education Commission-Gaofeng Clinical Medicine Grant Support (grant number 20161422 to J. R. Liu), and the Natural Science Foundation Project from the Shanghai Municipal Science and Technology Commission (grant number 22ZR1436900 to J. R. Liu).
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
