Abstract
Ultraviolet B (UVB) radiation induces cutaneous inflammation, leading to thermal and mechanical hypersensitivity. Here, we examine the mechanical properties and profile of tactile and nociceptive peripheral afferents functionally disrupted by this injury and the role of oxytocin (OXT) as a modulator of this disruption. We recorded intracellularly from L4 afferents innervating the irradiated area (5.1 J/cm2) in 4-6 old week male mice (C57BL/6J) after administering OXT intraperitoneally, 6 mg/Kg. The distribution of recorded neurons was shifted by UVB radiation to a pattern observed after acute and chronic injuries and reduced mechanical thresholds of A and C- high threshold mechanoreceptors while reducing tactile sensitivity. UVB radiation did not change somatic membrane electrical properties or fiber conduction velocity. OXT systemic administration rapidly reversed these peripheral changes toward normal in both low and high-threshold mechanoreceptors and shifted recorded neuron distribution toward normal. OXT and V1aR receptors were present on the terminals of myelinated and unmyelinated afferents innervating the skin. We conclude that UVB radiation, similar to local tissue surgical injury, cancer metastasis, and peripheral nerve injury, alters the distribution of low and high threshold mechanoreceptors afferents and sensitizes nociceptors while desensitizing tactile units. Acute systemic OXT administration partially returns all of those effects to normal.
Introduction
Ultraviolet radiation in the B spectrum (UVB, wavelength 280–315 nm) is one of the three ultraviolet components of solar radiation.1,2 UVB is absorbed by the epidermis, which induces several intracellular changes that include DNA damage, changes in gene expression, increased levels of reactive oxygen species, and inflammation. This inflammatory response is primarily mediated by the production and release of pro-inflammatory cytokines3–7 and is both dose and time-dependent, peaking at 24 to 48 h after UVB exposure.8,9 These effects are accompanied by local erythema and increased cutaneous hypersensitivity to mechanical and thermal stimuli.8–10 Importantly, UVB radiation produces very similar effects on the skin of humans, rats, and mice, which led to its use as a translation model in the study of acute inflammatory pain. 11
Mechanical hypersensitivity occurs in pain states in humans and is also present in rodent models of preclinical models. It is commonly assumed that hypersensitivity after nerve injury reflects only the sensitization of nociceptive afferents, even though neuropathic pain in humans is as often accompanied by hyposensitivity as hypersensitivity to mechanical stimuli. 12
We recently demonstrated that surgical injury nerve damage) 12,13 or exposure to expanding cancer produces a comprehensive peripheral effect modulating tactile and nociceptive sensibilities.14,15 In both cases, injury and disease reduce the sensibility of tactile afferents (desensitization) while increasing the sensibility and responsiveness of nociceptive afferents (sensitization). Notably, the return to normal behavior after surgical nerve injury parallels the recovery of tactile and nociceptive afferent sensibilities towards normal.
Oxytocin (OXT) produces analgesia in humans16–20 and rodents, 21 and increased OXT signaling during and after childbirth may explain the quicker recovery from pain after injury in humans22,23 and rodents.24–26 These OXT effects likely involve central and peripheral sites. Peripherally, OXT directly modulates sensory afferent excitability and membrane potential, returning afferent sensitivity towards normal in both tactile and nociceptive afferents following injury. 27
Unlike other preclinical models and human pain conditions, the UVB burn is unique in its restriction of hyperalgesia and allodynia to only the irradiated area. As such, the primary goal of this study is to expand previous observations of afferent plasticity after acute and chronic injury to the primary hyperalgesia setting of a UVB-induced peripheral injury. A secondary goal is to evaluate the effects of systemic OXT on reversing these effects on afferent sensibility after injury. Based on our previous studies using mild peripheral injury 28 and somatic exposure to OXT, 27 we hypothesized that UVB-radiation would induce a more profound effect than skin incision on afferent mechanical sensibility, 28 corroborating the integrative function of this peripheral plasticity. We also expected that, due to its effect on membrane and action potentials, OXT would return the pathological activation pattern (tactile desensitization and nociceptive sensitization) towards normality. We also assessed the presence of receptors upon which OXT directly acts (OXT and vasopressin V1a receptors) in afferents innervating the skin.
Methods
One hundred and 43 male mice, 4–6 weeks of age, of two breeds: (C57BL/6J termed wild type (WT) [Jackson Laboratory, Bar Harbord, ME. USA] and Oxtr
tm1.1Knis
termed OXTR knockout (KO) [courtesy of Dr Elizabeth Hammock. Florida State University) were used to study two aims: (1) effect of UVB radiation on peripheral afferent sensibility and (2) OXT modulation of UVB-induced injury. Figure 1 shows the number of animals across experimental groups. Animals were housed in pairs under a 12-h light/dark cycle in a climate-controlled room. The use and handling of animals were under guidelines provided by the National Institutes of Health and the International Association for the Study of Pain, and the Institutional Animal Care and Use Committee of Wake Forest University Health Sciences approved the procedures and experiments. Experimental method and design. (a) Schematic diagram of the lateral aspect of the mouse hind leg and area of radiation by UVB (5.1 J/cm2). Area of L4 sensory distribution G = glaborous skin H = hairy skin. (b) Sequence of electrophysiological assessment (CBP: cellular basal properties, SEP: somatic electrical properties, CR: intracellular current response, MT: mechanical threshold, CV: conduction velocity). (c) Animals and peripheral afferents included in the study and profile of the neurons recorded including in the 30 min before (white boxes) and after (grey boxes), IP OXT (6 mg/Kg). LTMR: low threshold mechano receptors, AHTMR: A-fiber high threshold mechano receptors, CHTMR: C-fiber high threshold mechano receptors, MS: muscle spindle, UNEX: unexcitable, F-type: cell with no mechanical RF and narrow APs (fast), S-type: cell with no mechanical RF and wide APs (slow).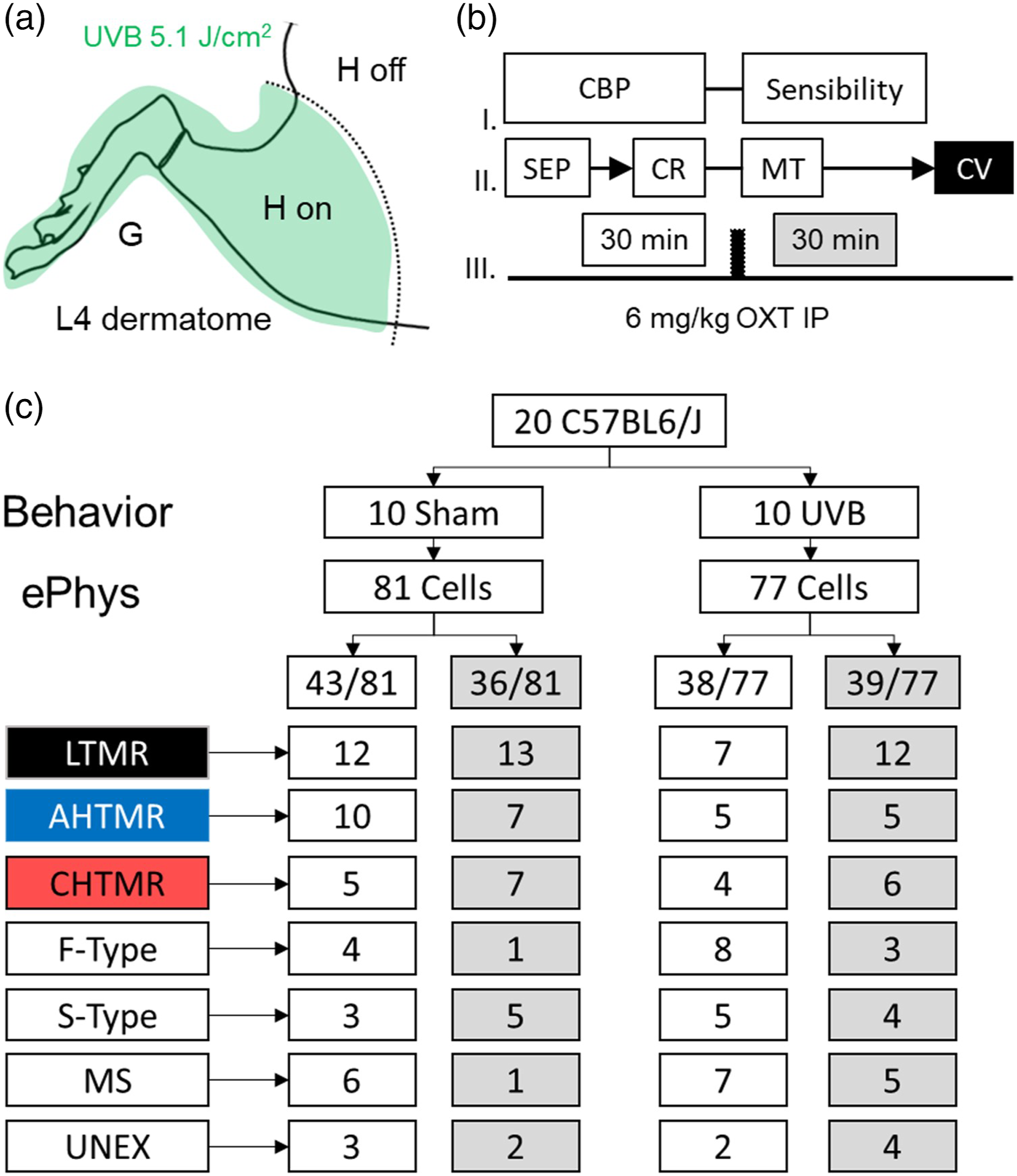
Ultraviolet B radiation
This model has been described elsewhere 29 and parallels processes used to produce the same injury as in humans. It allows the characterization of the UVB radiation effects at an intensity causing a moderate redness (first 6 h) of the irradiated skin area that resolves in less than 24 h without skin lesions, scarring inflammatory reaction, or blisters. Briefly, animals were anesthetized with isoflurane 3% (Teva Pharmaceuticals, North Wales, PA). Mice were randomized to Sham or UVB. The mice were covered with an opaque material fabric with only the relevant surface (Glabrous [G] and hairy skin [H on] below the right knee) exposed through a small opening (∼3 cm2) (Figure 1(a)). Sham group (n = 56) were not exposed to anesthesia or UVB previous to electrophysiology. For the UVB group (n = 56), a stationary UVB lamp narrowband 311 nm Philips Bulb 9W (UV Phototherapy SKU 1651, KN-4006BL Kernel, Leana Inc, NY, US) was positioned perpendicularly 5 cm above the exposed area and activated for 10 min to reach 5.1 J/cm2 of radiation per treatment. Because of the inherent fluctuation of UVB lamps, the UVB lamp was allowed to stabilize prior to the radiation session. UVB intensity was measured using a digital UVA/UVB Meter, 280–400 nm (General tools & instruments, #UV513AB, NJ, USA) before each treatment.
Behavioral testing
Twenty-four hours after UVB radiation, mice were placed on a mesh surface in a plastic cage and were acclimated for 20 min before testing. The paw withdrawal threshold (PWT) was assessed by application through the mesh flooring of calibrated von Frey filaments to the footpad until the filaments bent. This was done by a person blinded to the surgical treatment. The von Frey filaments used were 3.84, 4.08, 4.31, 4.56, 4.74, 4.93, 5.18, 5.46, and 5.88, corresponding to 0.5, 0.9, 1.7, 3.7, 5.5, 8.0, 12.4, 21.5, and 53.0 g, respectively. A filament was applied three times, separated by 5 s, with a response considered positive if it resulted in a brisk withdrawal of the paw on any of the three applications. The force resulting in a 50% probability of withdrawal was determined using the up-down method as previously described. 30 PWTs were determined before UVB radiation or sham and 24 h after radiation.
A group of animals (48 WT) were used to establish the effects of UVB radiation on PWT. From this group, 20 mice were then anesthetized for electrophysiologic recordings (see below). A second group (64 WT) was injected with 6 mg/Kg OXT IP or placebo (aCSF), 31 and their PWT was observed at 30 min after injection. All animals were included in the data analysis and no animal in the study had skin lesions or infection during the study.
Electrophysiology (ePhys)
Twenty animals were anesthetized with isoflurane 3%. The trachea was intubated, and the lungs were ventilated using pressure-controlled ventilation (Inspira PCV, Harvard Apparatus, Holliston, MA, USA) with isoflurane in humidified oxygen. Heart rate and noninvasive blood pressure were monitored throughout as a guide to the depth of anesthesia. Anesthetized animals were immobilized with pancuronium bromide (2 mg/kg), and end-tidal isoflurane concentration was maintained at 2% throughout the study. A dorsal incision was made in the lumbar midline, and the L4 dorsal root ganglion (DRG) and adjacent spinal cord were exposed by laminectomy, as previously described. 32 The exposed nervous tissue was continuously superfused with oxygenated artificial cerebrospinal fluid [aCSF (in mM): 127.0 NaCl, 1.9 KCl, 1.2 KH2PO4, 1.3 MgSO4, 2.4 CaCl2, 26.0 NaHCO3, and 10.0 D-glucose]. The spinal column was secured using custom clamps, and the anesthetized animal was transferred to a preheated (32 °C–34 °C) recording chamber where the superfusate was slowly raised to 37°C (MPRE8, Cell MicroControls, Norfolk, VA USA), and its flow rate kept at 1 mL per minute (bath exchange time of ∼5 s). The pool temperature adjacent to the DRG was monitored with a thermocouple (IT-23, Physitemp, Clifton, NJ, USA). Rectal temperature (RET-3, Physitemp) was maintained at 36 ± 1°C with radiant heat.
The electrophysiological recording session from each animal was limited to a maximum duration of 60 min (30 min before and after OXT IP 6 mg/kg) (Figure 1(b)) to diminish the likelihood that surgery and experimental manipulation would result in primary sensory afferent sensitization. DRG neuronal somata were impaled with borosilicate micropipettes (80–250 MΩ) containing 1 M potassium acetate. DC output from an Axoclamp 2B amplifier (Axon Instruments/Molecular Devices, Sunnyvale, CA, USA) was digitized and analyzed offline using Spike2 (CED, Cambridge, UK). The sampling rate for intracellular recordings was 21 kHz throughout (MicroPower1401, CED).
Cellular classification protocol
The general procedure to classify primary sensory afferents was applied as described elsewhere for DRG neurons.
33
The receptive field (RF) was searched, starting on the animal’s paw in a longitudinal pattern, first covering the digit and then progressing proximally to the hairy skin and ending 5 mm distal to the surgical incision. Neurons with receptive field (RF) located near the skin incision were excluded because of the potential sensitization by surgery of those afferents. RFs were located with the aid of a stereomicroscope using increasing mechanical stimulation; the latter progressed from light touch with a fine sable hair paintbrush to searching with a blunt probe (back of the paintbrush) and ultimately gentle to intense pinching with fine-tipped forceps. Based on their mechanical RF (or absence thereof) and their capability to generate a somatic action potential (AP), these cells were classified as follows: (a) Cells with RF: Based on the combination of their mechanical threshold, conduction velocity (CV), response to vibration (tunning fork 256 Hz) and dynamic response (phasic; on-off; tonic) to suprathreshold stimulation, neurons were classified into three groups: LTMRs (low-threshold mechanoreceptors, rapidly adapting [RA] and slow adapting [SA] tactile afferents), muscular spindles (MS), AHTMRs (A fiber high-threshold mechanoreceptors), and CHTMRs (C fiber high-threshold mechanoreceptors) innervating skin. (b) Cells without RF: Cells that were electrically excitable but without mechanical RF were separated into two different populations based on the shape of the action potential (AP)34–36 as neurons with inflection in the repolarizing phase (slow or S-type neurons) and neurons without this inflection (fast, or F-type neurons). To more clearly determine the presence of this inflection, the first derivative records of the AP were used (presence or absence of a second additional negative component in the time course of the AP first derivative). Cells without RF or electrical excitability were classified as unexcitable (UNEX). Since RF properties, especially response characteristics, were used to define differences in the fast-conducting afferents (those without inflected APs), the ability to define and categorize these three populations further was not possible. After electrical characterization, S-type, F-type, and UNEX cells were included in the description of distribution but not otherwise examined.
All included cells satisfied the following requirements: Resting membrane potential (Em) more negative than −40 mV, AP amplitude ≥30 mV, and the presence of afterhyperpolarization (AHP). Passive membrane properties indicative of poor impalement (extremely low input resistance [Ri], extremely short time constant [tau, τ]), and unstable impalement were also reasons for exclusion. CV was measured at the end of the recording.
Mechanical sensitivity and cellular excitability
Peripheral and somatic cellular excitability were measured at two stages (Figure 1(b)). All neurons were characterized by cellular basal properties (CBP) and, if a RF was present, by mechanical sensibility. (1) Cellular basal properties (CBP): (a) RF area mapping: After identifying the cellular RF area of responsiveness to search stimuli, the area was marked using a red fine point marker. This initial procedure was performed gently to avoid damaging the skin (as assessed visually by lack of development of erythema, edema, glossiness, etc.). (b) Somatic Active Electrical Properties (SEP): Active membrane properties of all excitable neurons were analyzed in APs obtained during RF characterization. These parameters included amplitude and duration of the AP and after-hyperpolarization (AHP) of the AP, along with the maximum spike depolarization rate (MDR) and repolarization rate (MRR). AP and AHP durations were measured at half amplitude (D50 and AHP50, respectively) to minimize hyperpolarization-related artifacts. (2) Sensibility: Resting mechanical threshold (MT) was determined in each afferent with calibrated von Frey filaments (Stoelting, Wood Dale, IL) as the minimum force to generate at least one AP, testing the most sensitive area of the cellular RF. The presence or absence of post-discharge hyperpolarization (PDH)
37
was determined during this procedure.
Conduction velocity
Spike latency was obtained by stimulating the RF at the skin surface using a bipolar electrode (0.5 Hz, current range: 0.1–1.2 mA) and a stimulus isolator (A360LA, WPI, Sarasota, FL, USA). This was performed following mechanical stimulation to prevent potential alterations in RF properties by electrical stimulation (Figure 1(b)). All measurements were obtained using the absolute minimum intensity required to excite neurons consistently without jitter. This variability (jitter) in the AP generation latency, particularly at significantly shorter latencies seen at two-to three-fold threshold intensity, has been presumed to reflect spread to more proximal sites along axons. Stimuli ranged in duration from 50 to 100 µs; utilization time was not taken into account. The distance was measured for each afferent on the termination of the experiment by inserting a pin through the RF (marked with ink at the time of recording) and carefully measuring the distance to the L4 DRG along the closest nerve.
Immunohistochemistry (IHC)
To test the specificity of the OXR antibody, olfactory bulb (OB) and hind paw skin were collected and the tissue was analyzed with IHC and Western blot (WB). OXTR KO and WT mice, n = 14 each, were anesthetized with pentobarbital, a tail sample was taken for genotyping, and the mice were transcardially perfused with 20 mL of 0.1 M phosphate buffer (PB, ph 7.4). Whole bodies were postfixed overnight submerged in 4% paraformaldehyde in PB, then transferred after 24 h into PB and stored at 4°C until dissection. Hind paw skin was removed and immersed in fixative (post-fixation) for 2 h at 4°C. Afterward, the tissue was washed and immersed in 30% sucrose at 4°C for cryoprotection until sectioned on a cryostat. Hind paw sections (20 μm) were collected on slides and stored at −80°C until processed. Sections were processed simultaneously with antibodies for OXT receptor and NF-200 antibodies. NF200 positive immunoreactivity in OXTR KO skin samples helped to establish that sections contained immunoreactive nerve fibers.
Sections were washed with 0.01 M phosphate buffer saline (PBS) with 0.1% Triton X-100 (PBST), incubated for 1 hour in blocking solution (3% normal donkey serum (NDS) (# 017-000-121, Jackson Immuno Research Labs, West Grove, PA, USA) in PBST and overnight at 4°C in 1.5% NDS with rabbit anti-OXT receptor antibody (1:1000, #ab181077, Abcam, Cambridge, USA) and chicken anti-Nf200 (1:1000, #CH22104, Neuromics, Edina, MN, USA). Afterward, sections were washed three times for 10 minutes with PBS and incubated 2 h at room temperature with donkey anti-rabbit Cyanine three and donkey anti-chicken Cyanine 5 (1:400, Jackson Immuno Research Labs, West Grove, PA, USA). Finally, sections were incubated for 5 min with 4′,6-diamidino-2-phenylindole (DAPI), 1:10,000, D21490, Invitrogen, Molecular Probes, Eugene, OR, USA) washed thoroughly in PBS, mounted on plus-slides, air-dried, dehydrated in ethanol, cleared in xylene, and coverslipped with DPX mounting media. The experimenter performing image analysis was blinded to the group for the images.
Sections were examined, and images of the hind paw skin were acquired at 20x objective lens on a Nikon Eclipse Ni fluorescent microscope system equipped with a Nikon DS-Qi1Mc digital camera (Nikon, Japan). In addition, a higher magnification of selected sections from WT mice was acquired at 40X using an Olympus FV1200 confocal microscope.
Western blots (WB)
OXTR specificity study: OXTR KO and WT mice, n = 7 each, were euthanized by carbon dioxide. A tail sample was taken for genotyping, and brains were removed and frozen in isopentane on dry ice and stored at −80°C.
UVB study: Hind paw skin and L4-L5 DRG were collected from 17 WT processed for ePhys. All tissue was resuspended in tissue extraction reagent (#FNN0071, Invitrogen, Thermo Fisher, Scientific, Waltham, MA, USA) containing a protease inhibitor cocktail (#05892970001, Roche, Mannheim, Germany) and centrifuged. Afterward, supernatants were collected for immunoblotting. Protein concentration was determined by a protein assay (#Pierce BCA Protease assay kit, #232225 and 23,227, Thermo Fisher Scientific, Waltham, MA, US). 50 µg of total protein were combined with gel loading buffer, heated to 95°C for 5 min, and separated on 10% Tris–HCl gels (#5671034, Bio-Rad Laboratories, Hercules,CA, USA). Groups were loaded next to their corresponding controls at each time point in each gel. The proteins were transferred to PVDF membranes (#162-0177, Bio-Rad Laboratories, Hercules, CA, USA), blocked in 5% dry milk in PBS, and incubated overnight at 4°C with the primary antibodies: Anti-OXR or anti-Vasopressin 1a Receptor (1:500, #A53272, Epigentek, Farmingdale, NY, USA, and anti-GAPDH (1: 2000, #MAB374, EMD Millipore, Burlington, MA, US). After washing, the membranes incubated with horseradish peroxidase-conjugated anti-rabbit or anti-mouse secondary antibodies (1:5000, #AP182P Millipore, Burlington, Massachusetts, USA or 1:2000 #ab97030, 1:2000, Abcam Cambridge, USA, respectively). The signal was visualized using SuperSignal (West Pico or Femto Chemiluminescence Substrate, (#34080 and #34095, respectively, Thermo Scientific, Hanover, IL, USA) and quantified using an imager (GE HealthCare Amersham Imager 600, Chicago, IL, USA). The ratio of OXTR and V1aR to GAPDH was calculated for each lane, and the values of these ratios were normalized to the control group.
Oxytocin enzyme-linked immunosorbent assay assay
Blood was collected by cardiac puncture from a separate group of 16 C57BL/6J WT male mice deeply anesthetized with isoflurane 3% (Teva Pharmaceuticals, North Wales, PA) after OXT or aCSF was injected IP (as described above). Approximately ∼30 min after the injections, a thoracotomy was performed, and 0.5–1 ml of blood was obtained from the left ventricle by withdrawing it slowly to prevent the heart from collapsing. The serum was collected and OXT was extracted from the samples. A commercially available OXT ELISA kit (#ADI-900-153A, ELISA kit, Enzo Life Sciences, Farmingdale, NY, USA) was then used to measure the concentration of OXT in the extracted samples following the manufacturer’s instructions. Briefly, blood was collected in tubes containing 1 mg/mL EDTA (#324503, EMD Millipore, Burlington, MA, US) and 500 KIU of aprotinin/ml of blood, #A6279, Sigma, St Louis, MO, USA). Blood samples were then centrifuged (1600 g/15 min/4°C), and the plasma was stored at −80°C. C18 Sep-pak columns (#39002, Honeywell Burdick & Jackson, Muskegon, MI, USA), trifluoroacetic acid (Acros organics-Thermo Fisher, Scientific, Waltham, MA, USA), and acetonitrile (Thermo Fisher, Scientific, Waltham, MA, USA) were used for the OXT extraction (detailed protocol in ELISA kit product manual). After the eluted samples evaporated to dryness with a centrifuge concentrator under vacuum (Savant instruments, ISS100, Integrated Speedvac System, New York, NY, USA), samples were reconstituted in ELISA assay buffer followed by the OXT assay. Duplicates of OXT standards and samples were mixed with the antibody and incubated at 4°C for 18–24 h. After washing, all the samples were then reacted with the substrate for 1 h at room temperature, the reaction terminated, and the absorbance was read at 405 nm using a spectrophotometer (Epoch Microplate Spectrophotometer, BioTek Instruments Inc, Winooski, Vermont, USA). The limit of sensitivity of the assay was 15 pg/mL, and the intra-assay coefficient of variation was 11.6%.
Statistical analysis
Before analysis, parametric assumptions were evaluated for all variables using histograms, identifying outliers with boxplots, descriptive statistics, and the Shapiro–Wilk test for normality. Data are reported as median (range or quartiles) if not normally distributed or mean (standard error) if normally distributed. Student’s t-test and repeated measures analysis of variance (ANOVA) were used for normally distributed data, and Friedman test and Mann Whitney U-test were used for not normally distributed data. Changes in Em in AHTMR over time were analyzed using repeated-measures ANOVA with Greenhouse & Geisser sphericity correction as distributions at each time point proved to be parametric, and there were no significant outliers. Friedman tests were run on the number of APs per stimuli and duration data as the distributions were non-parametric at one or more time points in each dependent variable. For all analyses, p was set at 0.05 for statistical significance. All post hoc analyses were Bonferroni adjusted. Analyses were carried out using SPSS Statistics for Windows, version 22 (IBM Corp, Armonk, NY), and OriginPro 9.5 (Northampton, MA).
Results
Effects of ultraviolet B radiation on paw withdrawal threshold and peripheral afferent mechanical sensibility
Figure 1(c) depicts the animal disposition and cell types recorded during our study. 20 WT male animals were used 24 hours after UVB (or sham) radiation. After the behavioral assessment, the animals were immediately anesthetized for ePhys. The two columns correspond to the cells recorded during the first 30 min of every experiment before OXT IP administration.
During this initial experimental phase (30 min), it was possible to impale and study an average of four cells per animal. This average was upheld in both parts of the study for a total of 158 cells recorded (81 in sham animals [43/81, 53% prior OXT] and 77 in UVB irradiated animals [38/77, 49% prior OXT]).
As observed in Figure 2(a) (black boxes), UVB-irradiated animals were significantly (p < .001) more sensitive to mechanical stimulation (PWT) than the sham group. Effect of injury and OXT on physiologically characterized afferents. (a) Effects of UVB radiation on reflexive paw withdrawal threshold (PWT). Data are presented on Log10 scale at baseline, 24 h after UVB radiation, and 30 min after IP OXT (6 mg/Kg) or vehicle (artificial cerebrospinal fluid (aCSF). Values are mean ± SE. ** = p < .01; *** = p < .001. n = 8 (per group). (b
This increase in sensibility is associated with a numerical reduction in the proportion of myelinated fibers (AHTMRs: 23% to 13% and LTMR: 28% to 18%), and an increase in cells without RF (S-Type: 7% to 13% and F-Type: 12% to 21%) (Figure 2(b)). Of the mechanosensitive afferents, mechanical threshold was numerically reduced in A and C HTMRs and increased in LTMRs (Figure 2(c)).
In addition to UVB-induced reduction in the number of mechanosensitive afferents, those still active were not normal. Approximately 50% of the sensitized nociceptive afferents (3/5 AHTMR and 2/4 CHTMR) responded to brushing (in contrast to Sham, with 0% of nociceptive afferents sensitive to brushing). All LTMRs in the UVB group (100%, 7/7) failed to follow a 256 Hz vibratory stimulus with 1:1 fidelity.
No effect was observed in the CBP (active or passive) of these afferents in any group (Sham vs UVB groups) (Data not shown). Although UVB radiation did not significantly affect CV regardless of their modality, no afferent (with A fiber) above 11 m/s or below 3.9 m/s was recorded in the UVB-irradiated group.
Oxytocin-mediated modulation of the effects of ultraviolet B radiation in animals’ reflexive behavior, peripheral mechanical sensibility, and conduction velocity
The administration of OXT significantly (p < .01) increased the PWT of UVB irradiated animals by 75% (from 0.2 to 0.35 g) after 30 min (Figure 2(a)).This increase correlated with an increase in the numerical proportion of cells identified as LTMRs from 28 to 36% in Sham group and from 18 to 31% in UVB group and a significant increase (Sham: p < .05, UVB: p < .01) in the threshold of CHTMRs (Sham: from median: 588 mN [min: 13.7, max: 588 mN to 588 mN [min: 588 mN to max: 980 mN]) (UVB: from median: 1.53 mN [min: 0.7 to max: 13.7] to 424 mN [min: 80 to max: 588) (Figure 2(b) and (c)). OXT did not significantly change MT of LTMRs, but did increase from 0% to 38% (5/13) the percentage of afferents which followed the 256 Hz vibratory stimulus with 1:1 fidelity. OXT did not significantly affect CV of tactile or nociceptive afferents. Although it failed to recover any slow-conducting LTMR (below 3.9 m/s), 3/13 afferents showed CV above 11 m/s with a maximal speed of 19 m/s (Figure 2(d)).
Oxytocin receptors and vasopressin receptor expression
Oxytocin receptor antibody specificity
WB showed a band with a molecular weight ∼37–50 kDa, corresponding with the native form of the OXTR in samples from WT, but this band of immunoreactdivity was absent in OB tissue from OXTR-KO (Data not shown). IHC showed OXTR-positive immunoreactivity in the hind paw skin of WT mice confined mainly in cutaneous fibers and hair follicles that were absent in OXTR-KO tissue. OXTR immunoreactivity was not observed in neuronal soma of L4-L5 DRGs.
Oxytocin receptor and V1Ra distribution and expression in the skin
OXTR-positive fibers were closely aligned with those NF200-positive fibers innervating the skin. Additional confocal microscopy of hairy skin confirmed co-localization. As exemplified in Figure 3(a), OXTR positive cutaneous afferents displayed a punctuated pattern co-localized with NF-200 in most of these fibers. In contrast, their distribution appeared to diverge among the circumferential endings around hair follicles. WB showed an OXTR band (∼37–50 kDa) (Figure 3(g)) where was significantly (p < .05) decreased in tissue 24 h after UVB irradiation but with similar expression in the skin of both ipsi- and contra-lateral hind paw. (Figure 3(g–H)). V1aR showed no difference between groups (Sham vs UVB irradiated) but a significant (p < .01) increase in the ipsilateral hind paw 24 h after UVB injury (Figure 3 (i–J)). OXT receptors (OXTR and V1aR) immunoreactivity and expression in the skin innervated by L4 ganglia (sham and after UVB radiation). (a, c, and e) Representative OXTR receptor immunoreactivity in sections from hairy skin (hair follicle) innervated by L4 dorsal root ganglia (DRGs) afferents and visualized by fluorescence microscopy (DAPI in blue, OXT receptors in red, and NF200 in green). Calibration bar: 20 µm. (b
Oxytocin and V1Ra expression in L4-L5 dorsal root ganglions
WB was used to evaluate further the V1aR and OXR content in L4-L5 DRGs (n = 16) 24 h after UVB radiation. WB showed bands of ∼50–75 kDa or ∼37–50 kDa corresponding with the molecular weight for V1aR (n = 6) and OXR (n = 10), respectively (Figure 4(a) and (c)). Although the OXTR DRG content remained unchanged (Figure 4(b)), a significant increase in the V1aR content was observed in the UVB irradiated group (UVB, p < .05), both ipsilateral and contralateral to irradiation (p < .001 and p < .01) (Figure 4(d)). OXT receptors (OXTR and V1aR) expression in L4 DRGs (sham and after UVB radiation). (a and c) UVB-induced changes in the expression of OXTR and V1aR on the skin using Western blot analysis. (b and d) Data represents OXTR and V1aR/GAPDH ratio (left panel) and normalized ipsilateral/contralateral ratio (right panel). Data are presented as median (numbers and horizontal lines) with boxes representing the 25 and 75 percentiles. * = p < .05, ** = p < .01, *** = p < .001.
Discussion
In this study, UVB radiation induced moderate peripheral sensitization with increased mechanical threshold in tactile and decreased mechanical threshold in nociceptive afferents, effects which were partially reversed by systemic administration of OXT. The presence of OXTR and V1aR on the terminals and cellular bodies of both types of afferents suggests that OXT directly targeted sensory afferents rather than acting through an intermediary. These results largely agree with our observations on the effects of other acute and chronic injuries to peripheral nerves and tissue on peripheral afferent neurophysiology and acute, partial reversal of this pathophysiology by OXT.
Effects of ultraviolet B radiation on peripheral sensibility and excitability
Acute exposure of skin to UVB produces significant thermal and mechanical hyperalgesia in humans38–40 and hypersensitivity in rats.41–43 Several symptomatic parallels between human and rats exposed to UVB suggest that this model is appropiate for the translational study of experimental hyperalgesia.38,41–43
Despite these inter-species similarities in UVB effects and the extended use of this model for the study of nonsteroidal anti-inflammatory drugs (NSAIDs),5,44,45 very few studies have focused on the effects of UVB radiation on peripheral sensory afferents. This lack of information is surprising given the documented nature of UVB-induced injury, restricted entirely to the areas of radiation and the simplicity of the model.
As a notable exception, Bishop and Cols (9) described the effects of radiation using an ex vivo skin-nerve preparation.9,41,46 Their observations broadly concluded that low UVB-radiation intensity (1 J/cm2) induces a net shift in the peripheral nociceptive response properties but no sensitization (mechanical and thermal). Interestingly, C-HTMRs increased responsiveness, whereas A-HTMRs decreased responsiveness. Curiously, they did not report tactile afferents in their study despite the 4-fold greater density of tactile than nociceptive afferents (31).
In contrast, we detected a significant sensitization in both A- and C- C-nociceptive mechanosensitive afferents. This difference may reflect the greater UVB-radiation intensity (6 J/cm2), in our study. Notably, neither Bishop and Cols (9), nor this study observed spontaneous activity in nociceptors after UVB radiation.
On the other hand, our observations also show a shift in afferent availably (detectable presence) with different conduction velocities and sensibility. Our results suggest that UV-radiation degrades the availability of A-fiber afferents (tactile and nociceptive) without affecting the C-fiber population. This result is consistent with the observation that UVB radiation decreases the density of nerve endings in the skin of human patients, 47 although it does not explain its selectivity.
In many pathological conditions associated with behavioral hypersensitivity in rodents, there is a shift in the population distribution of sensory afferents encountered for recording, as well as their sensibility.12,14,48 These changes suggest that input to the spinal cord changes dramatically from the damaged area. One may speculate that this disrupted peripheral input may significantly affect the function and integration of converging circuitry in the spinal cord. In support of this speculation, it has been shown that the mechanically evoked firing of wide dynamic range neurons in the deep dorsal horn is enhanced by UVB radiation in rats. 29 Precise modality-specific studies on different subsets of spinal cord neurons are required to establish the impact of this disrupted peripheral signal on central processing, and UVB radiation seems to be an appropriate, valuable, and simple model to examine this disruption.
Ultraviolet B on oxytocin receptors and V1aR expression in the skin and the dorsal root ganglions
Systemic OXT produces analgesia in some human studies18,19,49 and reduces hypersensitivity in rodents.21,24,25 Importantly, these effects seem to reflect, in part, the modulation of the responsiveness of tactile and nociceptive sensory neurons. Our work 27 has corroborated31,50,51 the presence of both OXT receptors (OXTR and V1aR) on the membrane of the cellular body of sensory neurons with different diameters, indicating that OXT can affect cells with both modalities.
In the current study, we extended these observations in rats by verifying the presence and expression of OXTR and V1aR31,50,51 on peripheral nerve endings in mice and their opposing actions of OXT on tactile and nociceptive afferents. As expected, both myelinated and unmyelinated fibers express these receptors. 27
Effects of oxytocin on the sensibility of normal and sensitized peripheral afferents
Several lines of evidence point to the peripheral antinociceptive effects of OXT in multiple pain states.26,31,51,52 In humans, a lower-than-anticipated incidence of persistent pain has been observed after cesarean delivery. 23 Rats recover more rapidly after neuropathic injury when this occurs at the time of delivery, an effect which is reversed by intrathecal injection of antagonists of OXT signaling, 24 suggesting a role for OXT in speeding recovery from pain after injury. 25
In addition to these reports, Ito and Cols 53 speculate on the putative role of OXT in the modulation of complex human states and behaviors. Particularly relevant to the current study are the effects of physical stimulation of the somatosensory organs to regulate local OXT released in the skin. Seminal observations indicate that physical stimulation of hairy and glabrous skin relieves joint and muscle pain. 54 Furthermore, the systematic touching to soften skin tissues, including the back, neck, arms, and legs, promotes the release of OXT.53,55,56
We previously demonstrated that OXT acutely hyperpolarizes sensitized nociceptors while simultaneously depolarizing desensitized tactile afferents. 27 That study, however did not examine the impact of OXT on the mechanical sensibility of tactile and nociceptive afferents.This study addressed this gap, demonstrating an effect of OXT on peripheral afferent sensibility. Under normal conditions, systemic OXT increases the likehood of recording from tactile afferents. Although we did not explore the effect of OXT on RF size, depolarization of the terminal will likely render tactile afferents more reactive to mechanical stimulation and, therefore, easier to locate. Further studies are planned to determine OXT effects on afferent activation patterns in response to vibratory stimulation at different frequencies, as well as examine effects on RFs.
Conclusions
These data are consistent with existing behavioral studies in rodents demonstrating an anti-hypersensitivity effect of peripheral OXT after injury and some psychophysical studies of peripherally administered OXT in normal humans and those with injury-induced pain.18,19 Electrophysiological data corroborate our observations on OXT-mediated modulation of cellular excitability and extend these observations to the modulation of the system’s overall response to acute injury by recovering the peripheral mechanoreceptive system toward normal. OXTR and V1aR expression indicate that both receptors may be involved in the peripheral effects of OXT.57,58
Footnotes
Acknowledgements
In loving memory of Juan Bernardo Boada Bustos.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grant 1P01NS119159-01A1 to MDB and JCE from the National Institutes of Health, Bethesda, MD, USA.
