Abstract
Infection with varicella zoster virus (VZV) results in chicken pox and reactivation of VZV results in herpes zoster (HZ) or what is often referred to as shingles. Patients with HZ experience decreased motivation and increased emotional distress consistent with functions of the ventral tegmental area (VTA) of the brain. In addition, activity within the ventral tegmental area is altered in patients with HZ. HZ primarily affects individuals that are older and the VTA changes with age. To begin to determine if the VTA has a role in HZ symptoms, a screen of 10,000 genes within the VTA in young and old male rats was completed after injecting the whisker pad with VZV. The two genes that had maximal change were membrane progesterone receptors PAQR8 (mPRβ) and PAQR9 (mPRε). Neurons and non-neuronal cells expressed both PAQR8 and PAQR9. PAQR8 and PAQR9 protein expression was significantly reduced after VZV injection of young males. In old rats PAQR9 protein expression was significantly increased after VZV injection and PAQR9 protein expression was reduced in aged male rats versus young rats. Consistent with previous results, pain significantly increased after VZV injection of the whisker pad and aged animals showed significantly more pain than young animals. Our data suggests that PAQR8 and PAQR9 expression is altered by VZV injection and that these changes are affected by age.
Introduction
Herpes zoster (HZ) is a viral infection that is caused by reactivation of the varicella zoster virus (VZV). After initial exposure to VZV, which causes chicken pox, the virus establishes lifelong latency within the sensory ganglia. 1 Therefore, any individual infected with VZV is at risk for developing HZ and one in three adults infected with VZV develop HZ at least once in their lifetime. 2 However, the risk for developing HZ and its complications increases with age. 3 HZ is an inflammatory response that damages neurons both peripherally and centrally, as well as damages the nerve resulting in the characteristic painful, blistering rash that manifests in a unilateral dermatomal pattern. HZ can develop along any dermatome but one of the most affected is the trigeminal, after the thoracic and cranial. 4 HZ of the trigeminal dermatome or orofacial HZ involves the trigeminal nerve and affects 20% of HZ sufferers. 5 Acute orofacial HZ patients experience interference with daily activities, social interactions, and disruption in sleep leading to increased emotional distress, reduced motivation and a reduction in quality of life.6–8 The trigeminal nerve has nociceptors that innervate the head, neck, face, and oral regions sending sensory information, such as pain, touch, and temperature, to the trigeminal ganglia, which relays the information to the central nervous system affecting various brain regions, including those involved in reward and motivation. 9
Aging is a natural process associated with a multitude of anatomical and functional changes, ranging from telomere shortening, changes in cellular morphology, alterations in gene expression, alterations in proteins, lipids, and organelles, as well as alterations in hormones and neurotransmitters. 10 Aging is also associated with alterations in pain perception as observed as an increase in pain threshold.11,12 This alteration in pain perception is unclear but may be due to alterations in brain regions involved in pain perception,13,14 alterations in functional connectivity, 15 or alterations in nociceptor sensitivity. 16 There are numerous pain conditions, such as herpes zoster, where age is a risk factor but whether pain perception for age-related pain disorders, such as HZ, is altered with aging is unclear.
The ventral tegmental area (VTA) is a dopaminergic pathway that has a role in reward processing, motivation, anxiety, aversion, and pain.17–24 Age can alter the VTA dopaminergic pathway and alter connectivity to other regions of the brain. 25 One factor that contributes to alterations within the VTA is pain. Human brain imaging studies report that the VTA of fibromyalgia patients has reduced dopamine function to anticipated and evoked pain26. Trigeminal neuralgia patients have reduced volume of the ventral diencephalon including the VTA. 27 Chronic migraine patients have altered connectivity between the extended amygdala and VTA 28 and chronic low back pain patients have impaired VTA connectivity. 29 Moreover, HZ patients, have spontaneous brain activity in emotion, reward, and affective brain regions 30 and animal models of pain report altered neuronal firing properties in the VTA. 31
Age effects gene expression in the VTA 32 and age is a risk factor for HZ associated pain. 3 To determine genes that are altered by age after HZ infection gene expression in the VTA was compared in young adult and aged male rats. Gene expression was analyzed after injection of VZV or control into the whisker pad. One week after the injection, the VTA tissue was collected and used to analyze all coding RNA within the VTA. RNA-seq analysis of gene expression in young adult and aged adult males were compared in VZV and controls. Genes selected for quantitation of protein content in the VTA from the RNA-seq analysis were gonadal hormone receptor genes that were altered in an age-dependent manner.
Methods
Animals
Long Evans male rats (Envigo, Indianapolis, IN, USA) were used in these experiments. Rats were either single-housed or double-housed in a colony room with 14:10 hr light/dark cycle (lights on at 7 a.m.). Food and water were available ad libitum. Rats were allowed 1 week to acclimate to the facility before experiments began. All studies were approved by Texas A&M University College of Dentistry’s Institutional Animal Care and Use Committee. Experiments conformed to federal guidelines and the committee for Research and Ethical Issues of the International Association for the Study of Pain. 33
Treatment groups
A total of 42 young (4- to 6-month-old) and 42 aged adult (18- to 24-month old) rats were obtained. Six of the young rats and six of the old rats had their left whisker pad injected with VZV. Six of the young rats and six of the old rats had their left whisker pad injected with control MeWo cells; resulting in four groups: young male control, young male VZV, aged male control, aged male VZV. 15 of the young rats and 15 of the old rats had their whisker pad injected with VZV bilaterally. 15 of the young rats and 15 of the old rats had their whisker pad injected with control MeWo cells bilaterally. Two weeks after injection pain behavior was tested and after testing the rats were sacrificed and perfused. The bilaterally injected rats were sacrificed and a tissue punch of the VTA was collected and frozen. RNA-Seq analysis was used to quantitate the expression of all coding RNA. RNA quality was not met in some samples and those samples were discarded. Three samples per group met the RNA quality specifications and were used in the RNA-Seq analysis. The remaining punch samples (8-12 rats per group) were used for protein (i.e., ELISA) analysis.
Behavioral testing
Behavioral pain responses were measured using place escape/avoidance paradigm (PEAP) by an experimenter blind to treatment. Seven days post-injection, animals were placed in an acrylic box measuring 30 cm × 30 cm × 30 cm, of which half was covered in black cloth. The test chamber is modeled from PEAP test performed and detailed by Fuchs lab. 34 and used in inflammatory and neuropathic models, with males and females, and various strains and ages of rats as a means to measure affective aspects of pain.34–40 Each individual rat was placed into the chamber and testing was initiated by stimulating the face with a 60 g von Frey filament every 15 sec. When the animal was on the dark side of the box, the injected side was stimulated, while their non-injected side was stimulated when they were on the clear side. Since VZV was injected into the whisker pad, the target region for poking was the area below the eye and caudal to the whisker pad. This region is innervated by sensory neurons of the second branch of the trigeminal ganglia. 41 Time spent on the dark side of the box was recorded in 5 min bins and testing was performed for a total of 30 min per rat on test day. The PEAP test assumes that if the animal escapes and/or avoids a noxious stimulus, then the stimulus is aversive to the animal. Rodents, being nocturnal in nature, preferred to stay on the dark side of the chamber when placed inside. Thus, the theory behind the test is that if the rat is experiencing VZV-induced pain when poked in the sensitive area, it will not stay on the preferred dark side but will move to the non-preferred light side to avoid the poke.
VZV infection
Rats were anesthetized with 2% isoflurane and 2 L of air flow per minute. Each rat was injected with 100 µL of MeWo cells infected with VZV or vehicle (MeWo cells without VZV) into either the left whisker pad or bilaterally. The concentration of MeWo cells without virus was the same as the concentration of MeWo cells containing virus.
VTA tissue collection
Animals were euthanized by exposure to CO2 followed by rapid decapitation. After removing the brain with rongeurs, the brain was placed in a brain slicer (Zivic, Pittsburgh, PA, USA) and cooled using dry ice. After cooling a 1 mm brain section was cut and kept on dry ice. The entire ventral tegmental area was collected bilaterally with punches 1 mm in diameter centered on coordinates 0.5 mm lateral of midline and to a depth of 8.1 mm from top of skull. Tissue samples for both RNA-Seq and ELISA’s were stored at −80° until use.
RNA-seq analysis
Sequencing libraries were generated from 20 ng/µL of purified RNA. Reference genome and gene model annotation files were downloaded directly from the genome Web site (NCBI/UCSC/Ensembl). Indexes for reference genome were built using STAR and pair-end clean reads were aligned to reference genome using STAR (v2.5). Differential expression analysis between two conditions/groups were performed using DESeq2 R package (2_1.6.3). Read counts were adjusted by edgeR program package through one scaling normalized factor. TFCat and cosmic database were used to annotate differential expressed gene. HTSeq v0.6.1 was used to count read numbers mapped for each gene. Fragments per kilobase of transcript per million fragments mapped (fpkm) was calculated based on the length of the gene and reads count mapped to this gene.
Enzyme-linked immunosorbant assay
Ventral tegmental tissue was placed in 500 µl of T-PER™ Total Protein Extraction Reagent (Thermo Scientific, Rockford, IL, USA) containing HALT protease inhibitor (Thermo Scientific, Rockford, IL, USA) to prevent proteolysis. Tissue was homogenized on ice, centrifuged at 13,000 r/min for 15 min at 4°C, and supernatant collected. Total protein concentration was determined by Pierce™ BCA assay kit (Thermo Scientific, Rockford, IL, USA). Protein quantitation of VTA samples was performed in duplicate using ELISA for progestin and adipoQ receptor family member 8 (PAQR8; MBS7243037; MyBiosource, San Diego, CA) and progestin and adipoQ receptor family member 9 (PAQR9; MBS9332395; MyBiosource, San Diego, CA) according to manufacturer’s instructions.
Immuno-fluorescent staining
Two weeks after injection, animals were sacrificed by injecting with 100 mg/kg ketamine and 10 mg/kg xylazine, then perfused with 9% sucrose followed by 4% paraformaldehyde in phosphate buffered saline (PBS) pH 7.4. Fixed tissues were stored in 25% sucrose, frozen, cryo-sectioned and the 32 µm sections placed on Histobond slides (VWR international, Radnor, PA). The tissue was post-fixed for 5 min in 4% paraformaldehyde, rinsed and then blocked for 2 h at room temperature with a PBS solution containing 5% normal goat serum (Sigma-Aldrich, St Louis, MO) and 0.3% Triton-X 100. The slides were then incubated in a primary antibody solution overnight at 4°C. The primary antibody consisted of a mixture of the mouse NeuN antibody (Millipore, Burlington, MA, catalog # MAB377) diluted to 1.6 µg/ml and rabbit PAQR8 antibody (Novus Biologicals, Littleton, CO, catalog # NBP2-56,507) diluted to 1.3 µg/mL or rabbit PAQR9 antibody (Novus Biologicals catalog # NBP2-30,402) diluted to 0.2 µg/mL. The primary antibodies were diluted with PBS, 5% BSA and 0.3% Triton X-100. After incubation in primary antibody the slides were then rinsed three times in PBS and 0.3% Triton-X 100 for a total of 45 min and placed for 2 h in secondary antibody and PBS and 0.3% Triton X-100. Secondary antibodies (1:500 dilution) included a mixture of goat anti-mouse 488 and goat anti-rabbit 568 (Invitrogen, Carlsbad, CA). After rinsing the slides three times in PBS and 0.3% Triton X-100 for a total of 45 min, the slides were treated with TrueVIEW Autofluorescence quenching kit (Vector Labs, Burlingame, CA), mounted with Fluoromount-G mounting medium containing Hoechst 33,342 stain (Electron Microscopy Sciences, Hatfield, PA). The fluorescent signal was imaged using a Leica Stellaris eight confocal microscope (Leica Microsystems, Wetzlar, Germany). Controls eliminating the primary antibody showed no signal (data not shown). Two animals in each treatment group did not have adequate perfusion, resulting in poor identification of cells after immunostaining and the data from these animals was not included in the cell counts.
Cell counts were completed by an experimenter blind to the identity of the treatment groups. Every other section was selected for staining. Typically, three sections were counted for each animal. The slides were analyzed using Image J software, the average background for the slides within a treatment group was subtracted from the image and a fluorescent signal associated with a cell nucleus was counted as a positive cell. Counts were completed for the number of NeuN/PAQR8 or NeuN/PAQR9 stained cells within a 0.04 mm2 field. To define the VTA, the Stellaris microscope would take multiple images over the entire section. Scanned images were automatically stitched together using the Leica software to produce an image of entire section. An atlas image 42 could be overlayed on the scanned image and the field to be counted was placed within the borders of the entire VTA. Cell counts from two fields on each section were then averaged. This average count for the three sections was averaged for each animal. Four animals were analyzed for each treatment group. Values were given as a mean and standard error of the mean (SEM) for the animals in each treatment group.
Statistics
Protein expression data were analyzed using two-way ANOVA with protein content as the dependent variable and whisker pad injection and age as the independent variables (Prizm 7.05, GraphPad Software, La Jolla, CA). PEAP data were analyzed by two-way ANOVA with repeated measures. PEAP data were analyzed as repeated measures ANOVA because data were collected multiple times over a 30-min period from each rat for each behavior test. The dependent variable was the time spent on the dark side of the box during 5-min intervals and the independent variables were whisker pad injection, age, and within-test time (collected in six 5-min bins i.e., at 5, 10, 15, 20, 25 and 30 min). Tukey’s post-hoc testing was completed on data with significant main effects. A significant effect was indicated when
Results
One week after injection, whisker pad injection treatment significantly affected the pain response [F (1, 20) = 27.189, Behavioral pain response in young and aged rats after control or VZV injection of the whisker pad (a) 1 week after injection and (b) 2 weeks after injection. Two-way ANOVA and Tukey’s post-hoc test was performed to compare groups and an asterisk indicates a significant difference (
RNA-Seq was completed comparing all the genes in young and old rats, two hormone receptor genes that showed a significant decrease with age were PAQR8 (Mdn = 29.3 for young rats and 7.9 for old rats,
Protein content for PAQR8 was determined by ELISA. No significant main effect in PAQR8 protein levels was observed when comparing young and aged animals [F (1, 38) = 0.02, Histograms of PAQR8 and PAQR9 protein content within the VTA for young and aged male rats. In panel (a) PAQR8 was measured in young and aged rats after injecting the whisker pad with either MeWo control or VZV. In panel (b) shows a histogram of PAQR9 protein content within the VTA for young and aged rats after injecting the whisker pad with either MeWo control or VZV. Two-way ANOVA and Tukey’s post-hoc test was performed to compare groups and an asterisk indicates a significant difference (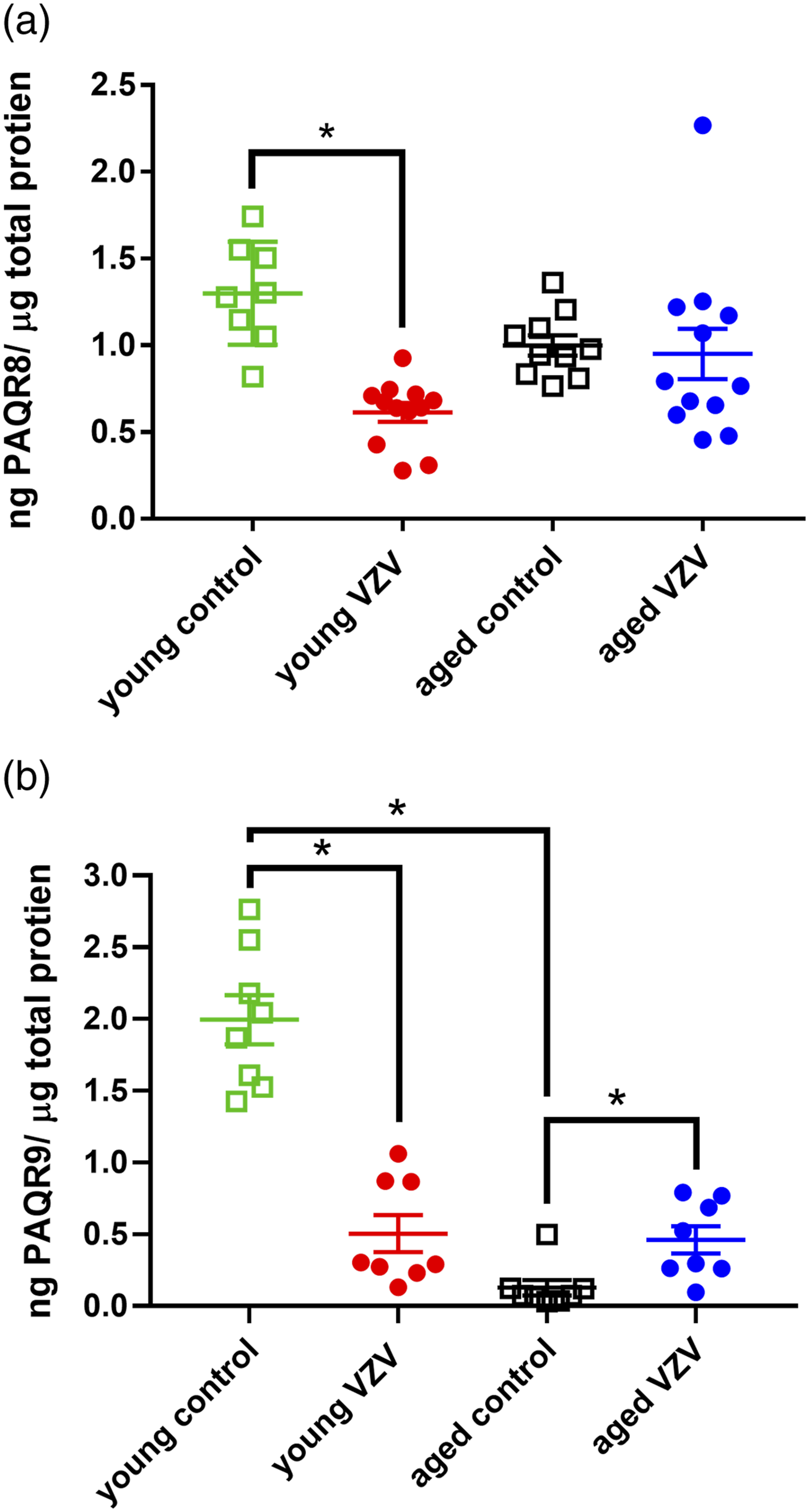
Next the protein content for PAQR9 was quantitated. Comparison of young and old rats showed a significant main effect in PAQR9 protein content [F (1, 29) = 67.7,
Immunostaining the VTA for PAQR8 and counting neurons positive for PAQR8 (Figure 3) indicated no significant effect of either age or VZV treatment (Figure 5(a)). Cell counts of PAQR9-positive neurons (Figure 4) also showed no significant change in the number of neurons staining positive for PAQR9 (Figure 5(b)). The number of neurons staining for PAQR8 (Mdn = 4, PAQR8 expression in neurons within the VTA. (a) Low magnification image of a coronal brain slice from a representative young male rat injected with MeWo control. Hoechst 33,342 nuclear stain is blue, NeuN staining is red and PAQR8 staining is green. (b) A high magnification image of cells positive for NeuN. (c) Cells staining positive for PAQR8. (d) Cells that colocalize for NeuN and PAQR8 are shown with black (arrows). (e) Staining for Hoechst 33,342, NeuN and PAQR8. Arrows point to the same cells in panels (b), (c), (d) and (e) cross section images through the z plane of the brain slice was completed at the green line in panels (b) through (e) cross sections of cell (open arrow) are shown in panels (f) through (h); NeuN and PAQR8 colocalized for the cell on the left and PAQR8 positive cell without NeuN is shown on the right. Bar = 50 µM. PAQR9 expression in neurons within the VTA. (a) Hoechst 33,342 nuclear stain (blue) from cells within the VTA from a coronal brain slice from a representative young male rat injected with VZV control. (b) Image of NeuN positive cells (red). (c) Cells staining positive for PAQR9 (green). (d) Image of overlay for Hoechst 33,342, NeuN and PAQR9 staining. Cross section images through the z plane of the brain slice were completed at the green line in panels (a) through (d) panels (e) through (h) are the cross section of the cell indicated by the open arrow. Panel (i) is the cross section through the cell indicated by the arrow. Colocalized NeuN and PAQR9 were observed in the cell in panels (e) through (h) PAQR9 was not colocalized with NeuN in the cell in panel (i) Bar = 20 µM. Histogram for cell counts colocalizing with the PAQRs positive signal and the neuronal marker NeuN in the entire VTA. For the histogram in panel (a) the counts for cells colocalizing for PAQR8 and NeuN in the VTA are shown and panel (b) shows cells colocalizing for PAQR9 and NeuN. Four animals were counted for each treatment group.


Discussion
To begin to determine if the VTA has a role in HZ symptoms a screen of 10,000 genes within the VTA in young and old male rats was completed after injecting the whisker pad with VZV. The two genes that had maximal change were membrane progesterone receptors PAQR8 (mPRβ) and PAQR9 (mPRε). Progesterone has five known membrane progesterone receptors (mPRs; mPRα, mPRβ, mPRγ, mPRδ, mPRε) which are G protein-coupled receptors (GPCRs) that belong to class II members of progesterone and adipoQ receptor (PAQR) family. Functions of mPRs are known to include sperm motility, oocyte maturation, reproductive behaviors, prolactin secretion, and neurite outgrowth43–48 but, their role in pain are unknown. Here we show that both PAQR8 (mPRβ) and PAQR9 (mPRε) were expressed on neuronal and non-neuronal cells within the VTA. Pain significantly increased after VZV injection of the whisker pad, consistent with previous results 49 and aged animals showed significantly more pain than young animals. Expression of PAQR8 and PAQR9 protein was reduced in young male rats after VZV injection. In old rats PAQR9 protein expression was increased by VZV injection. Moreover, PAQR9 protein expression was reduced in aged male rats versus young rats. We are the first to report that PAQR8 and PAQR9 expression differs in an age-dependent manner and we are the first to show that VZV alters protein expression of both PAQR8 and PAQR9 within the VTA.
Progesterone has been shown to dissociate pain and unpleasantness by regulating emotional centers within the brain. 50 In humans, the VTA is part of a pathway that alters the emotional state of humans to effect the pain response. 51 One mechanism for VTA’s control of pain is a reduced excitatory output to the dopaminergic neurons within the VTA. 52 This decrease in dopamine has been linked to an increased aversive response. 53 Targeting progesterone signaling by developing agonists/antagonists to PAQRs could alter VTA dopaminergic release and potentially reduce the pain response.
PAQR8 and PAQR9 expression was altered by VZV injection of the whisker pad in young males but only PAQR9 was altered in aged males. PAQR8 has been shown to inhibit G-proteins 54 resulting in a decrease in cyclic adenosine monophosphate (cAMP) while PAQR9 activates stimulatory G-proteins 55 resulting in an increase in cAMP. Studies show that inhibition of cAMP is associated with a decrease in neuronal activity and a reduction in pain. 56 PAQR8 expression declined after VZV injection of young rats. This decline in PAQR8 expression would increase cAMP and increase neuronal activity. Increased GABAergic activity in the VTA can negatively regulate VTA DAergic neurons and this attenuation of VTA DAergic neurons is associated with an increased aversive pain response. 52 The increase in pain after VZV injection is consistent with the idea that a decrease in PAQR8 contributes to the aversive pain response by suppression of dopamine release. To further this idea, PAQR9 is coupled to a stimulatory G protein and VZV-induced orofacial pain in aged males was greater than in young males. Thus, the increase in PAQR9 expression within aged males could increase cAMP within the VTA and increase GABA release. This increase in GABA would inhibit dopamine signaling and increase the pain response after VZV injection of aged males.
PAQRs were expressed on non-neuronal cells (Figure 4) and can be expressed on Schwann cells, where they regulate cell migration, proliferation, and differentiation of Schwann cells. 57 Schwann cells, which are glial cells in the peripheral nervous system, modulate nociception. 58 Astrocytes and oligodendrocytes, which are glial cells in the central nervous system express PAQRs 59 and have a role in nociception.60,61 These results are consistent with the idea that PAQRs modulate pain by regulating glial cells. It is of note that the pain response was affected by age and VZV treatment but the number of non-neuronal cells expressing PAQR8 and PAQR9 was not altered by age or VZV. It is still possible that PAQR8 and PAQR9 modulate pain and PAQR8 and PAQR9 protein content in non-neuronal cells of the VTA should be quantitated to determine any association between PAQR expression and pain.
The current study has several limitations. One limitation of the study was not measuring gonadal hormone levels in either young adult or aged male rats. Progesterone and allopregnanolone bind to mPRs to exert their effects but surprisingly testosterone has been shown to bind to mPRs suggesting that in males mPRs may be modulated by testosterone, as well as progesterone. 55 An age-related decline in testosterone may alter nociception in an age-dependent manner through mPR signaling. The gonadal hormone, progesterone, may play a bigger role in aging females due to their role in reproductive aging than males and future studies need to include female rats.62–66 Another potential limitation of these studies is that all injections were performed on the left side. It has been shown that chronic pain can be affected by differences between the two sides of the brain. 67 Although, preliminary studies did not observe a difference in the pain response between young rats injected with VZV on the left versus the right whisker pad it is possible the pain response is different in older animals. Moreover, the effect of PAQR8 or PAQR9 could differ in the right or left sides of the brain to alter the response.
In conclusion, we show aged males experience a greater aversive pain response after VZV injection. VZV injection decreased PAQR8 and PAQR9 expression in the VTA of young males but PAQR9 expression increased in aged male rats. These correlative data are consistent with the idea that PAQR8 and PAQR9 have an age-related role in VZV-induced orofacial pain.
Footnotes
Acknowledgements
The authors would like to thank Priscilla Hooks, Gerald Hill, and Connie Tillberg for their technical assistance.
Authors’ Contributions
RSH contributed to experimental design, conducting experiments, data analysis and interpretation, and preparation of the manuscript. PRK (Paul R. Kinchington) contributed to experimental plan, virus treatment methods, and preparation of manuscript. MU contributed to data analysis and interpretation and manuscript preparation. PRK (Phillip R. Kramer) contributed to experimental design, data analysis and interpretation, and manuscript preparation. All authors have read and agreed to published version of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by National Institute of Dental and Craniofacial Research grant (DE026749) to PRKramer, National Institute Neurological Disorders and Stroke grant (NS064022) to PRKinchington, and National Eye Institute core grant (EY08098).
