Abstract
Microglia activation and subsequent pro-inflammatory responses play a key role in the development of neuropathic pain. The process of microglia polarization towards pro-inflammatory phenotype often occurs during neuroinflammation. Recent studies have demonstrated an active role for the gut microbiota in promoting microglial full maturation and inflammatory capabilities via the production of Short-Chain Fatty Acids (SCFAs). However, it remains unclear whether SCFAs is involved in pro-inflammatory/anti-inflammatory phenotypes microglia polarization in the neuropathic pain. In the present study, chronic constriction injury (CCI) was used to induce neuropathic pain in mice, the mechanical withdrawal threshold, thermal hyperalgesia were accomplished. The levels of microglia markers including ionized calcium-binding adaptor molecule 1 (Iba1), cluster of differentiation 11b (CD11b), pro-inflammatory phenotype markers including CD68, interleukin-1β (IL-1β), tumor necrosis factor-α (TNF-α), and anti-inflammatory phenotype markers including CD206, IL-4 in the hippocampus and spinal cord were determined on day 21 after CCI. The results showed that CCI produced mechanical allodynia and thermal hyperalgesia, and also increased the expressions of microglia markers (Iba1, CD11b) and pro-inflammatory phenotype markers (CD68, IL-1β, and TNF-α), but not anti-inflammatory phenotype marker (CD206, IL-4) in the hippocampus and spinal cord, accompanied by increased SCFAs in the gut. Notably, antibiotic administration reversed these abnormalities, and its effects was also bloked by SCFAs administration. In conclusion, data from our study suggest that CCI can lead to mechanical and thermal hyperalgesia, while SCFAs play a key role in the pathogenesis of neuropathic pain by regulating microglial activation and subsequent pro-inflammatory phenotype polarization. Antibiotic administration may be a new treatment for neuropathic pain by reducing the production of SCFAs and further inhibiting the process of microglia polarization.
Introduction
Neuropathic pain is usually resulted from a direct lesion or disease affecting the somatosensory system, 1 leading to unbearable pain experience for millions of people worldwide. Despite recent advances, the pathophysiological mechanisms of neuropathic pain remain unknown and current available treatments are still unsatisfactory, causing a huge burden for patients, health care providers, as well as the society.2,3
In recent years, accumulating preclinical and clinical evidence suggest the contributing role of gut microbiota in various pain conditions such as chemotherapy induced peripheral neuropathic pain, carrageenan induced inflammatory pain, fibromyalgia, and visceral pain. 4 Gut microbiota derived various signaling molecules such as microbiota metabolites, neurotransmitters, neuromodulators, and neuropeptides prominently regulate the peripheral and central sensitization via acting on their receptors. 5 Targeting gut microbiota seems to provide us promising therapeutic potential for various types of chronic pain. However, the exact mechanisms underlying the contributing role of gut microbiota in chronic pain development remains to be clarified.
Microglia cells are broadly distributed immune cells within the central nervous system (CNS), 6 accounting for 10% of the adult glial cell population 7 and participating in CNS hemostasis maintenance via microglia-mediated phagocytosis and secretion of inflammatory molecules. 8 “Resting” microglia are high motile cells with a ramified morphology. 9 Upon stimulation, a series of morphological and functional changes termed microglial activation was followed with different activation status pro-inflammatory or anti-inflammatory phenotype depending on the environmental milieu and stimulus encountered. 10 Pro-inflammatory phenotype microglia promote pro-inflammatory responses with excess tumor necrosis factor (TNF-α), interleukin-1β (IL-1β) production, contrbuting to neural network dysfunction. 11 By contrast, anti-inflammatory phenotype microglia are associated with up-regulation of anti-inflammatory cytokines and play a vital role in restoring homeostasis, such as dampening inflammation and wound healing. 12
Various signals and modulators can regulate microglial activation including abnormal bacterial DNA/RNA and viral origin, endogenous proteins, complements, antibodies, cytokines, chemokines, neurotropic factors, plasma components, neurotransmitters, ions, and other compounds. 13 Of note, short-chain fatty acids (SCFAs), microbiota-derived bacterial fermentation products, was also found to regulate the differentiation and function of immune cells in the intestine, periphery and brain. 14 The absence of a complex host microbiota also leads to defects in microglial maturation, differentiation, and function, 15 demonstrating that the SCFAs promotes the maintenance of microglia under steady-state conditions. Further, microglial activation is known to play a critical role in neuropathic pain. 16 And hippocampus and spinal cord are key brain regions in the development of neuropathic pain.17,18 Therefore, in this study, we tried to explore the possible involvement of SCFAs in nerve injury induced neuropathic pain, as well as microglial polarization and inflammation markers expression in the hippocampus and spinal cord.
Materials and methods
Animals
Specific pathogen free (SPF) male C57 mice (8 weeks, 25–30 g) purchased from the Animal Center of Jinling Hospital, Nanjing, China were fed with food and water ad libitum, maintained on a 12-hour light-dark cycle, and used as CON mice in this study. Part of SPF mice were provided drinking water containing cocktail antibiotics containing 0.5 g/L Ampicillin, Neomycin, Metronidazole, and 0.25 g/L Vancomycin (Sigma-Aldrich) for three weeks from 8th week to 10th week to eradicate gut microbiota so as to establish ABX mice. Antibiotics water were renewed every other day and maintained during the entire experimental period. SCFAs treated mice were provided drinking water containing 67.5 mM sodium acetate, 25mM sodium propionate, and 40 mM sodium butyrate (Sigma-Aldrich) beginning at 6 weeks until 12-13 weeks of age. 19 All procedures were approved by the Ethics Committee of Jinling Hospital under the Guidelines for the Care and Use of Laboratory Animals from the National Institutes of Health.
Chronic constriction injury (CCI) model establishment
We used CCI of the sciatic nerve to induce neuropathic pain in this study. In brief, under deep anesthesia with intraperitoneal injection of sodium pentobarbital 60 mg/kg(Sigma, St Louise, MO, USA), four ligatures of 4–0 chromic gut loosely ligated around the left sciatic nerve. Sham surgery was carried out with an identical procedure but with no nerve ligature. Mechanical allodynia and thermal hyperalgesia were assessed one day before and day 3, 7, 10, 14, and 21 post injury. 20 Hippocampus and lumber spinal cord was obtained on the last day.
Mechanical hyperalgesia examination
To evaluate mechanical hyperalgesia, a series of calibrated von Frey filaments, were applicated in an incremental order of bending forces to the central region of the injured hind paw plantar surface. Each filament was applied with sufficient force to bend for 5 s, which was repeated at an interval of 6 min. The mechanical withdrawal threshold (MWT) was defined as the smallest filament size which evoked at least 3 withdrawal responses in 5 measurements. Cut off value was considered 5.0 g to prevent hurting animals. Paw withdrawal, licking, and shaking were considered as nociceptive-like responses. 21
Thermal hyperalgesia examination
A radiant heat test was performed to assess thermal hyperalgesia by using a plantar analgesia tester. Briefly, the mice were placed on the glass pane, and the plantar surface was vertically heated by a 5 mm-diameter laser radiant heat source. When the mice withdrew their paw, this movement was detected using an instrument (Infrared Hot Spur Pain Meter, RWD, China). Thermal withdrawal latency (TWL) was averaged with three consecutive tests performed at least 5 min apart. A cut-off value of 20 s was applied to avoid possible tissue damage. 22
SCFAs extraction and analysis
Intestinal contents were collected at the last day of the experiment. Each content was mixed with 1mL sterile 18 Ω de-ionized water. The pellet-water mixtures were homogenized by mixing at 3200 rpm for 5 minutes and centrifuged for 15 minutes at 13,000 rpm at 4°C. Supernatants were filtered using Acrodisc LC 13 mm sterile syringe filters with 0.2 μm PVDF membranes (Pall Life Sciences). The filtrates were used with short chain fatty acids (SCFAs) analyzed using high performance liquid chromatography (HPLC, LC-20AT, Shimadzu) technique, which equipped with a carbohydrate column (Aminex HPX-87H column, Biorad) and photodiode array detector (PDA, Shimadzu). The eluent was 5 mM H2SO4, fed at 0.6 mL/min, and column temperature set at 50°C. The run time was 60 minutes. Standard curves were generated by diluting 10 mM volatile fatty acid standard solution (acetic acid, propionic acid, isobutyric acid, butyric acid, isovaleric acid, valeric acid) from 50 nM to 5000 nM with SCFAs concentrations normalized to soluble chemical oxygen demand.
Western blot
The hippocampus and spinal cord were harvested after anesthesia with intraperitoneal injection of sodium pentobarbital on day 21 after surgery. The sample was placed on ice plate and homogenized in a lysis buffer containing 0.1% sodium deoxycholate, 1% Nonidet P-40, 0.1% SDS, 66 mM EDTA, 10 mMTris–HCl, (pH 7.4) with protease inhibitor cocktail. Homogenate was centrifuged at 4°C at 12,000 rpm for 15 min and then the supernatant was collected. Protein concentration was determined eaccording to Bradford assay. Twenty micrograms of proteins were separated on 8–12% SDS-PAGE and transferred to polyvinylidene fluoride membranes (Millipore, Billerica, MA, USA), which was then blocked with 5% non-fat milk for 1 h at room temperature. Then, the membrane was incubated with rabbit anti-CD11b (1:1,000, proteintech, USA), rabbit anti-CD68 (1:1,000, proteintech, USA), rabbit anti-CD206 (1:500, proteintech, USA), rabbit anti-TNF-α (1:1,000, CST, USA), rabbit anti-IL-1β (1:1000, Abcam, USA), rabbit anti-IL-4 (1:1000, Affinity Biosciences, USA), rabbit anti-GADPH (1:1000, Proteintech, USA) and rabbit anti-β-tubulin (1:1,000; Bioworld, St. Louis Park, MN, USA) overnight at 4°C room. After washing in TBST (Tris Buffered Saline with Tween) for three times, the horseradish peroxidase-conjugated secondary antibodies (goat anti-rabbit; Sevicebio, China) were diluted as 1:10,000 and the membranes were incubated for 1 h at room temperature. Chemiluminescence was used to detect the protein bands and Image J Software (Wayne Rasband, National Institute of Health, USA) was used to quantitate the band intensity.
Immunofluorescence examination
Mice were anesthetized with intraperitoneal injection of 2% sodium pentobarbital 60 mg/kg, perfused with saline, and perfused with 4% paraformaldehyde (PFA) in phosphate buffered saline (PBS) on day 21 after surgery. Brain and spinal cord were collected and fixed in the 4% PFA overnight and dehydrated in 30% sucrose at 4°C overnight. We collected 30 μm thick coronally sections of the hippocampus and 15 μm thick lumbar enlargement. After blocking with 10% normal goat serum for 1 h at room temperature, the slices were incubated with primary antibodies: rabbit anti-Iba1 (1:500; Wako, Japan) in 10% norm goat serum at 4°C overnight. After washing with PBST for 6 × 10 min, the slices were exposed to the secondary antibodies Alexa fluor 488/549 goat anti-rabbit, and DAPI (1:1,000; Sigma, St. Louis, MO, USA) for 1 h at room temperature. Confocal microscope (Olympus, FV1000, FV1000, Japan) was used to capture the fluorescent images with fluorescence intensity calculated by Image J Software.
Quantitative real-time PCR (qRT-PCR)
Total RNA was extracted from the hippocampus and spinal cord, and isolated with RNAeasy™ Animal RNA Isolation Kit with Spin Column (Beyotime, China). HiScriptQ RT SuperMix (Vazyme, Nanjing, China) was used to synthesize cDNA. qRT-PCR was performed by the ABI StepOne™ Real-Time PCR System (Applied Biosystems, Foster City, CA, USA) with SYBR Green PCR Master Mix (Vazyme, Nanjing, China) and analyzed using StepOne Software v2.3 (Applied Biosystems). Primers sequence used for qRT-PCR are available in Table 1. The cycling conditions were 95°C for 5 min, 40 cycles of 95°C for 10 s followed by 60°C for 30 s. GAPDH was used to normalize gene expression and relative gene expression levels were calculated by ΔΔCT method.
The primer sequences using qRT-PCR of microglia markers and the reference gene.

Statistical analysis
All analyses were performed by SPSS 22.0 software (version 22.0, IL, USA) and data are presented as mean ± SEM. Pair-wise comparisons were generated with two-tailed t tests. Mechanical allodynia was analyzed by repeated measures TWO-WAY ANOVA + ad Hoc test. Significant statistical difference was regarded as P < 0.05.
Results
CCI induces mechanical and thermal hyperalgesia, as well as up regulates SCFAs content in the gut
On day 3, 7, 14, and 21 after surgery, CCI mice presented significant mechanical (Figure 1(a)) and thermal hyperalgesia (Figure 1(b)), which phenomenon was absent in the sham mice. Meanwhile, we analyzed SCFAs concentrations in gut contents by HPLC, including acetate, propionate, butyrate, isobutyrate, valeric acid, isovaleric acid, and hexanoic acid with results shown in Figure 1(c). Quantitative analysis showed acetate, propionate, butyrate, and valeric acid contents but not the other SCFAs significantly increased in the CCI mice compared to sham mice (Figure 1(d) to (k)).
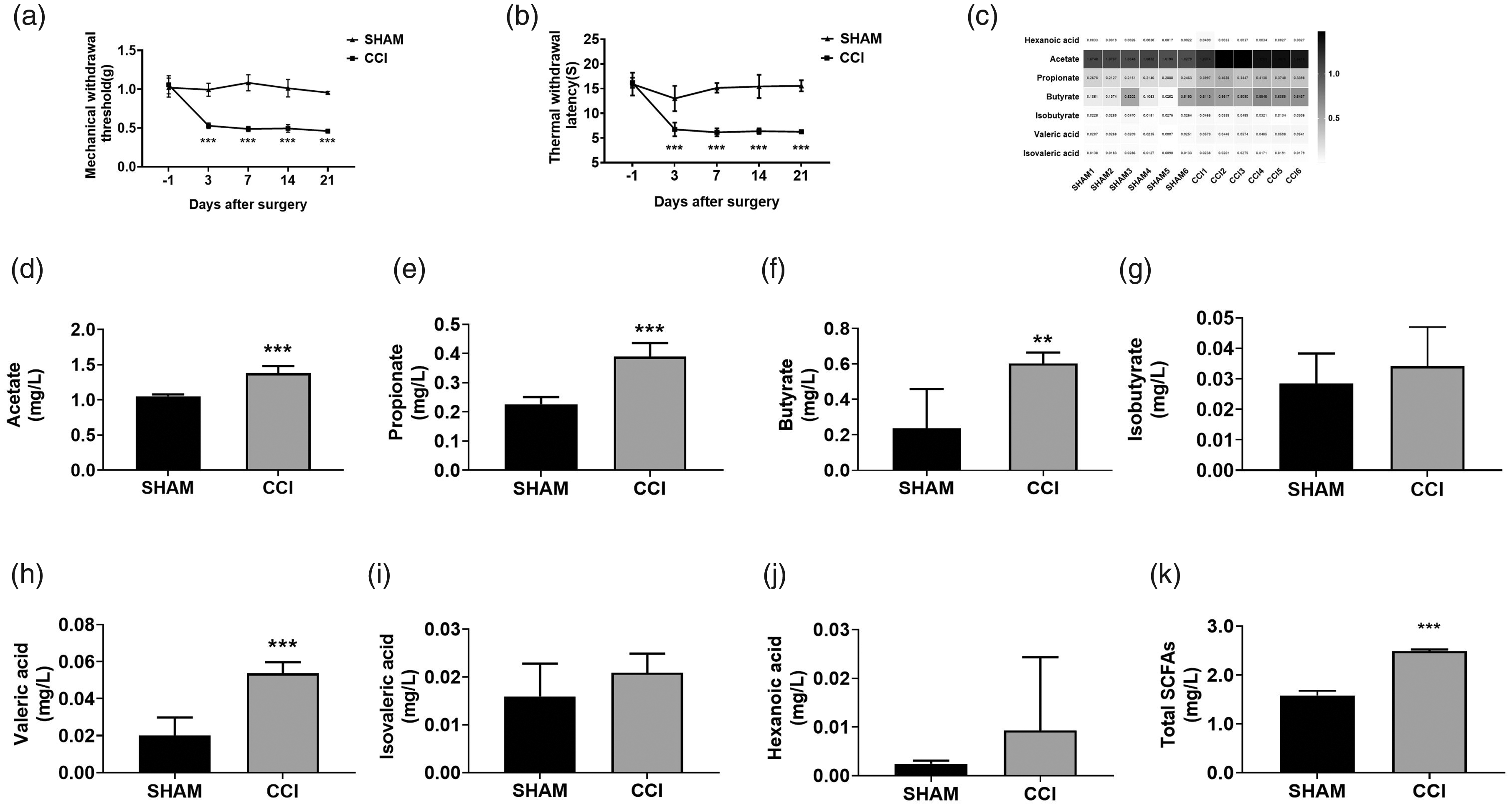
Nociceptive manifestations before and after surgery and the changes of SCFAs in fecal samples of mouse treated with surgery.Time courses show mechanical thresholds to von Frey filaments (a) and withdrawal latency to heat stimulation in the plantar test (b). Measures are shown before CCI/sham surgery (day -1), after surgery (day 3,7,14,21). Plantar test showed a persistent decrease in the mechanical thresholds and withdrawal latency to heat stimulation in paws of all CCI-operated mice(***p < 0.001 vs. sham-operated mice).(c) total SCFAs heat map, (d) acetate, (e) propionate, (f) butyrate, (g) isobutyrate, (h) valeric acid, (i) isovaleric acid, (j) hexanoic acid, and (k) total SCFAs. acetate, propionate, butyrate valeric acid and total SCFAs in CCI mouse are significantly higher than those of sham mouse. ***p < 0.001, **p < 0.01 (n = 8, mean ± SEM).
SCFAs is essential for CCI induced hyperalgesia, as well as microglia activation in the hippocampus and spinal cord
To investigate the role of increased SCFAs in CCI induced hyperalgesia, we established ABX mice with cocktail antibiotics in drinking water from the 8th week to the 10th weekto clear their gut microbiota, and continued to be given throughout the experiment. According to one previous report, this treatment regimen reduced the fecal bacterial load by more than two log folds, decreased the gut microbiota α diversity, a significant indicator of bacteria or species diversity within a community or habitat. 23 Then, we exposed ABX mice and CON mice to either CCI or SHAM surgery. ABX mice and CON mice showed comparable weight gain during entire experimental period for neuropathic pain. (Figure 2(a)), suggesting antibiotics do not affect normal physiological functions in mice. However, there are significant differences in SCFAs contents between groups (Figure 2(b)), demonstrating the effectiveness of gut microbiota eradication regimen. While ABX mice did not show changes in nociceptive thresholds in the presence of CCI or not. By contrast, mechanical and thermal hyperalgesia was presented after oral taken SCFAs in ABX + CCI mice (Figure 2(c) and (d)). To evaluate whether CCI could induce microglia activation in the hippocampus and spinal cord, microglia markers Iba-1 were detected by immunofluorescence (Figure 2(e) and (f)). Results showed CCI up regulated the expression and the number of Iba1-positive cells in the hippocampus and spinal cord (Figure 2(g) and (h)), whereas antibiotic administration alleviated these abnormalities, similarly, Iba1 high expression was also observed in SCFAs treated ABX + CCI mice (Figure 2(g) and (h)).

The changes of SCFAs in Intestinal contents of mouse treated with antibiotics and surgery.Immunofluorescence staining to Iba1 in the hippocampus and spinal cord on day 21 post-CCI. (a) There was no statistical difference in the body weight monitoring of the five groups of mice during the experiment. (b) The use of antibiotics significantly reduced the levels of total SCFAs. ***P < 0.001 compared to the Sham group. (c) The use of antibiotics improves in the mechanical thresholdsl, and exogenous SCFAs can reverse this effect. *P < 0.05 compared to the Sham group. (d) The use of antibiotics improves in the withdrawal latency, and exogenous SCFAs can reverse this effect.*P < 0.05 compared to the Sham group. (e) Representative images of Iba1 (green) in the hippocampus. (f) Representative images of Iba1 (green) in the spinal cord. (g) Antibiotics significantly attenuated CCI-induced fluorescence intensity increase of Iba1 in the hippocampus,but exogenous SCFAs can reverse this effect (Scale bar = 50 µm). (h) Antibiotics significantly attenuated CCI-induced fluorescence intensity increase of Iba1 in the spinal cord,but exogenous SCFAs can reverse this effect (Scale bar = 50 µm). The data are expressed as the mean ± SEM of 4 mouse in each group. *P < 0.05 compared to the Sham group, DAPI (4′, 6-diamidino-2-phenylindole) staining is shown in blue.
SCFAs is critical for CCI induced differential expression of microglia markers in the hippocampus and spinal cord
To ascertain the role of SCFAs in microglia activation, we measured CD11b (a microglia marker), CD68 (a pro-inflammatory phenotype marker), and CD206 (a anti-inflammatory phenotype maker) expression to quantify pro-inflammatory and anti-inflammatory phenotypes microglial with western blot. Results showed CCI increased the expression of CD11b and CD68 but not CD206 in the hippocampus and spinal cord, which was blocked by antibiotic administration. Further, CD11b and CD68 expression in SCFAs treated ABX + CCI mice is similar with that in CCI mice either in the hippocampus or spinal cord (Figure 3(c), (d), and (f) to (g)). We also noticed SCFAs increased the expression of CD206 after CCI in the spinal cord but not hippocampus (Figure 3(e) and (h)).

Western blot results of the hippocampus and spinal cord. (a) A representative blot of CD11b, CD68, CD206 in the hippocampus. (b) A representative blot of CD11b, CD68, CD206 in the spinal cord. (c to e) A quantitative analysis of CD11b, CD68, CD206 in the hippocampus. (f to h) A quantitative analysis of CD11b, CD68, CD206 in the spinal cord. β-tubulin was included as control. The data are expressed as the mean ± SEM of 4 mice in each group. *P < 0.05 compared to the sham group. **P < 0.01 compared to the sham group. ***P < 0.001 compared to the sham group.
SCFAs alters pro-inflammatory and anti-inflammatory phenotypes ratio
To evaluate the alteration of pro-inflammatory/anti-inflammatory phenotypes microglia at the gene level, this two kind of microglia markers were detected by qRT-PCR. As shown, CCI increased CD11b and CD68 but not CD206 mRNA expression (Figure 4(a) to (c) and (e) to (g)). However, antibiotics administration reversed the increase of pro-inflammatory microglia marker and not influence anti-inflammatory microglia marker expression (Figure 4(a) to (c) and (e) to (g)). But in SCFAs treated ABX mice, abnormal CD11b and CD68 expression cord was replicated as that observed in CCI mice, with no effect observed for CD206. Of note, CD68/CD206 ratio, a mark of microglia polarization, was increased following CCI, abrogated after antibiotic administration and recurred after SCFAs supplement (Figure 4).

mRNA expressions of pro-inflammatory/anti-inflammatory phenotypes microglia markers in the hippocampus and spinal cord. Fold increase of pro-inflammatory phenotype markers (CD11b, CD68) in the hippocampus (a and b); Fold increase of anti-inflammatory phenotype microglia markers (CD206) in the hippocampus (c). Fold increase of the ratio of CD68/CD206 in the hippocampus (d). Fold increase of pro-inflammatory phenotype markers (CD11b, CD68) in the spinal cord (e and f); fold increase of anti-inflammatory phrnotype microglia markers (CD206) in the spinal cord (g). Fold increase of the ratio of CD68/CD206 in the spinal cord (h). The data are expressed as mean ± SEM; n = 4 for each group; *P < 0.05 compared to the Sham group.
CCI induces hippocampus and spinal cord neuroinflammation, which is affected by antibiotics and SCFAs administration
As microglia polarization is highly associated with inflammation, thus, we used western blot to measure pro-inflammatory cytokines TNF-α, IL-1β and anti-inflammatory cytokine IL-4 expression in the hippocampus and spinal cord. Consistent with the trend observed for microglia polarization, IL-1β, TNF-α expression increased after CCI and antibiotics administration reversed such overproduction (Figure 5(a) to (d) and (f) and (g)). But in SCFAs treated ABX mice, such increase was replicated. In addition, there was no significant difference in IL-4 level among the 5 groups (Figure 5(a), (b), (e) and (h)).

Western blot results of the hippocampus and spinal cord. (a) A representative blot of IL-1β, TNF-α, IL-4 in the hippocampus. (b) A representative blot of IL-1β, TNF-α, IL-4 in the spinal cord. (c to e) A quantitative analysis of IL-1β, TNF-α, IL-4 in the hippocampus. (f to h) A quantitative analysis of IL-1β, TNF-α, IL-4 in the spinal cord. β-tubulin was included as control. The data are expressed as the mean ± SEM of 4 mice in each group. ***P < 0.001 compared to the sham group.
CCI up regulated hippocampus and spinal cord cytokines expression is also proved at mRNA level, similarly, affected by antibiotics and SCFAs administration
qRT-PCR was performed to evaluate the changes of inflammatory cytokines at mRNA level in the hippocampus and spinal cord. Results showed CCI up regulated the mRNA level of pro-inflammatory cytokines IL-1β and TNF-α, antibiotics and SCFAs administration abrogated and replicated such upregulation, respectively (Figure 6(a) to (c) and (e) to (g)). Similar trend was also observed for IL-1β/IL-4 ratio (Figure 6(d) and (h)), suggesting the essential role of gut microbiota in neuropathic pain development via microglia mediated neuroinflammation.

mRNA expressions of different inflammatory cytokines in the hippocampus and spinal cord. Fold increase of pro-inflammatory cytokines (TNF-α, IL-1β) in the hippocampus (a and b); fold increase of M2 anti-inflammatory cytokine in the hippocampus (IL-4) (c). Fold increase of the ratio of IL-1β/IL-4 in the hippocampus (d). Fold increase of pro-inflammatory cytokines (TNF-α, IL-1β) in the spinal cord (e and f); fold increase of M2 anti-inflammatory cytokine in the spinal cord(IL-4) (g). Fold increase of the ratio of IL-1β/IL-4 in the spinal cord (h). The data are expressed as mean ± SEM; n = 4 for each group; **P < 0.01 compared to the Sham group, ***P < 0.001 compared to the Sham group.
Discussion
In this study, our results showed SCFAs highly participated in the development of neuropathic pain, possibly viae hippocampal and spinal microglia polarization mediated neuroinflammation. Of note, elimination of intestinal flora by oral taken antibiotics can reverse these abnormalities, which may be mediated by promoting pro-inflammatory but not affecting anti-inflammatory microglial polarization. These findings provide new explanations for the involvement of intestinal flora in the development of neuropathic pain, and may help provide new insights for neuropathic pain treatment.
Mounting evidence have suggested the essential role of gut microbiome for human health, including gut-barrier integrity, protection from pathogens, energy regulation, immune system function, and brain development. 24 And the disturbance of gut microbiota homeostasis was shown to participate in a variety of metabolic diseases, liver, cardiovascular, and neurological diseases.25,26 Although neurological abnormalities are appreciated as brain disorders, it is now recognized that peripheral etiology, especially gut microbiota, may also contribute to the development of various neurological disorders including depression and anxiety, autism spectrum disorder, Parkinson’s disease (PD), Alzheimer’s disease, and chronic pain. 27 Various pain conditions are highly associated with gut microbiota, such as visceral or abdominal pain, inflammatory pain, opioid tolerance, headache, and chemotherapy induced peripheral neuropathic pain.
Our results firstly showed the involvement of gut microbiota in nerve injury induced neuropathic pain. One the one hand, mechanical and thermal hyperalgesia was ameliorated in antibiotics pretreated ABX mice; on the other hand, neuropathic pain phenotype was replicated after oral taken SCFAs in ABX mice. Similar contributing role of gut microbiota were also observed in other types of neuropathic pain such as chemotherapy induced peripheral neuropathic pain 28 or spared nerve injury induced pain and anhedonia-like phenotype. 29 In cehemotherapy related pain model, gut microbiota derived lipopolysaccharide, as well as intestinal mucosal structure damage collectively promote bacterial translocation, aggravate pro-inflammatory factors release, resulting in central astrocytes or microglia activation or peripheral nociceptors sensitization.28,30 Different from these studies, our study suggested the contributing role of SCFAs mediated hippocampal and spinal microglia polarization and the followed neuroinflammation in nerve injury induced hyperalgesia.
SCFAs are produced primarily form bacterial fermentation of carbohydrates and proteins, acting on their receptors, FFAR2/3 to regulate immune response, as well as recently recognized chronic pain. Four types of SCFAs were found to increase after nerve injury including acetate, propionate, butyrate, and valeric acid, however, we cannot differentiate the exact role of each component, protective or etiological, during neuropathic pain development. For example, in patients with irritable bowel syndrome or neuropathic pain rat model, butyrate was reduced and oral butyrate supplement was reported to be beneficial in relieving peripheral nerve sensitization via receptor mediated mechanisms, epigenetic regulation mechanisms, or both.31,32 We are still unknown the underlying mechanisms of such paradoxical results between ours and the available reports with regard to butyrate content, it is plausible that increased butyrate may be an inherent protective mechanism after nerve injury in our study, however, the integrated effect of increased SCFAs are detrimental or undesirable.
Of note, there is no SCFAs receptor FFAR2 expression in the CNS, instead only finding it at peripheral myeloid cells. 33 But recent work suggested that SCFAs may cross the blood brain barrier, 34 meanwhile, several cell types in the brain including microglia are able to receive input from the periphery, including from the gastric intestinal tract. 35 Previous work had shown gut microbiota significantly contribute to the regulation of microglia maturation and function via derived fermentation products SCFAs. 36 And gut microbiota participates in the pathogenesis of various neurological disorders, such as PD, via regulation of microglia by SCFAs. 37 Our study is the first one showing neuroinflammation mediated central sensitization underlying neuropathic pain can be also directly regulated by gut microbiota derived SCFAs.
Microglia polarization is implicated in mediating glia-neuron interaction, playing an essential role in central sensitization and pain hypersensitivity. SCFAs promote microglia pro-inflammatory polarization either in the hippocampus or spinal cord, however, not affect anti-inflammatory phenotype, with the final effect showing an pro-inflammatory effect, demonstrated an significantly increased IL-1β and TNF-α. Intriguingly, SCFAs, have been proven to be anti-inflammatory in several animal models, such as atherosclerosis, autoimmune diseases, and acute kidney injury. Such inconsistency suggested that SCFAs may have different roles in different diseases.
Therefore, our data showed gut microbiota derived SCFAs might induce hippocampus and spinal microglia polarization towards the pro-inflammatory phenotype, and the produced central neuroinflammation participating in the development of neuropathic pain. It seems that gut microbiota intervention by dietary and pharmabiotics might be a promising alternative for neuropathic pain treatment. Indeed, mounting clinical and preclinical studies have demonstrated the beneficial effect of probiotics, prebiotics, and symbiotics on chronic pain. Our study also showed oral taken SCFAs ameliorated hyperalgesia in microbe eradicated CCI mice. However, not every kind of probiotics could provide antinociceptive effects on nerve injury induced neuropathic pain. 38 Further investigations may be necessary for the discovery of specific gut microbiota for pain relief.
We also acknowledged there were disadvantages in our study. Firstly, other than the hippocampus and spinal cord, other pain regulatory regions such as dorsal root ganglion was not explored. Secondly, the epigenetic mechanism of certain SCFAs for pain regulation is not validated herein. Finally, the time-dependent dynamic regulation for microglial polarization needs further investigation.
In conclusion, our results suggested that SCFAs highly participated in the development of neuropathic pain, possibly via hippocampal and spinal microglia polarization mediated neuroinflammation. Our study provided additional evidence indicating the potential molecular mechanisms underlying SCFAs mediated neuropathic pain.
Footnotes
Acknowledgments
The authors thank Qingzhen Liu and Guangfen Zhang for their excellent technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by No. 81571083 and 81600960 from the National Natural Science Foundation of China.
