Abstract
Short-chain free fatty acids (SCFAs) are generated by gut microbiota through anaerobic fermentation of dietary fibers. Although gut microbiota-derived SCFAs modulate voltage-gated Ca2+ channels via G-protein-coupled receptor 41 (GPR41) in isolated sympathetic ganglion neurons, the influence of SCFAs, specifically propionic acid (PA), on the excitability of nociceptive neurons under in vivo conditions has yet to be ascertained. In the current study we assessed whether systemic PA administration diminishes the excitability of nociceptive trigeminal spinal nucleus caudalis (SpVc) wide-dynamic range neurons responding to mechanical stimulation. Extracellular single-unit recordings from SpVc wide-dynamic range neurons were performed in anesthetized rats after mechanical stimulation of the orofacial region. PA significantly and reversibly inhibited the mean firing frequency of SpVc neurons in response to both non-noxious and noxious mechanical stimuli in a dose-dependent manner. Simultaneous administration of a GPR41 inhibitor abolished the PA-induced inhibited firing rate of SpVc neurons, indicating that systemic PA decreased the excitability of nociceptive secondary trigeminal neurons by activating GPR41 signaling-mediated inhibition of voltage-gated Ca2+ channels in the central terminals of the SpVc. Modulation of trigeminal nociception by systemic SCFA administration indicates that gut microbiota-derived SCFAs could be effective analgesic agents for relieving trigeminal pain, creating a new therapeutic strategy for the management of trigeminal pain, including clinical pain.
Keywords
Introduction
Information from the orofacial region, carried by small-diameter Aδ-fibers and unmyelinated C-fibers, is conveyed through the trigeminal ganglion (TG) neurons to second-order neurons in the spinal trigeminal nucleus caudalis (SpVc).1–3 The SpVc acts as a crucial junction for neural trigeminal nociceptive signals after inflammation and tissue damage.1–3 SpVc nociceptive neurons are designated as nociceptive-specific or wide-dynamic range (WDR) according to their sensitivity to mechanical stimulation, such as facial skin, with WDR neurons responsive to both noxious and non-noxious stimulation. 3 When graded noxious stimuli are applied to receptive fields, the firing frequency of SpVc WDR neurons increases in relation to the intensity of the stimulus; thus, it is plausible that WDR neurons are essential for encoding the intensity of stimuli. SpVc neurons are also involved in the process of hyperalgesia and/or referred pain linked to dental discomfort.4,5
It has been shown recently that gut microbiota influence the maintenance of biological functions through bidirectional communication between the intestinal tract and the central nervous system (CNS), in addition to their role in the digestive system, and studies have revealed their connection to the occurrence of various diseases. 6 Gut microbiota employ metabolites, neurotransmitters, cytokines, and hormones generated as humoral factors, along with neural components like the vagus nerve, to support energy metabolism and CNS functions.6–8 Attracting particular attention are the short-chain free fatty acids (SCFAs), including acetic, propionic, and butyric acids, which are generated by gut microbiota through anaerobic fermentation of dietary fibers.7,8 Previous research indicates that SCFAs bind to G-protein-coupled receptor 41 (GPR41)/FFAR3 receptors, which are present on the cell membrane, and exert physiological functions through various intracellular signal transduction systems. In addition, SCFAs influence the permeability of the blood-brain barrier as well as the function of neurons and glial cells. 6 Since there have been reports showing that SCFAs may be involved in alleviating inflammatory pain and neuropathic pain in animal models,9,10 SCFAs may modulate pain via nociceptive neuronal activity. Recent reports suggest that the periodontal disease bacteria, Porphyromonas gingivalis, triggers the butyric acid-GPR41 signaling pathway and significantly contributes to the progression of periodontitis without causing periodontal inflammatory pain. 11 Consequently, the butyric acid-GPR41 signaling induced by Porphyromonas gingivalis might be a crucial pathway in controlling periodontal pain perception in periodontal disease. 11
To date, there have been no reports that SCFAs derived from gut microbiota enter the systemic circulation and modify the excitability of neurons in the CNS. 12 Recently though, it has been reported that voltage-gated N-type Ca (N-type Cav) channel currents in isolated sympathetic ganglion neurons under in vitro conditions are suppressed via GPR41 by administration of the SCFA, propionic acid (PA). 13 N-type Cav channels are widely distributed in the central axon terminals of peripheral nerves and central nerves, and are involved in synaptic transmission by releasing neurotransmitters. 14 Nøhr et al. 15 also reported that GPR41 expression is not only distributed in the autonomic nervous system, but also in TG neurons. Collectively, these findings suggest that systemic SCFA administration alters nociceptive neuronal transmission in the SpVc by GPR41 signaling pathway-mediated inhibition of Cav channels in the nerve terminal of TG neurons. There is limited knowledge suggesting that there is an acute influence of SCFAs on nociceptive transmission in in vivo environments. Therefore, the aim of the current study was to assess whether systemic administration of PA diminishes the excitability of nociceptive SpVc WDR neurons in response to mechanical stimulation. In addition, we also examined whether SCFA modulation of nociceptive neuronal transmission in the SpVc occurs via the GPR41 signaling pathway.
Materials and methods
Approval for the experiments described herein was granted by the Animal Use and Care Committee at Azabu University (No. 200529-3) and we adhered to the ethical guidelines set by the International Association for the Study of Pain. 16 Extensive actions were undertaken to decrease the use of animals and ease their suffering.
Recording extracellular single-unit activity of WDR neurons in the SpVc
Adult male Wistar rats (weighing 215–255 g) were kept under a fixed lighting schedule (lights on: 07:00–19:00). Temperature in the room was controlled to be 23°C ± 1°C. Food and water were available at all times. Recordings of electrophysiological activity were taken from 14 rats. Each rat was sedated with 3% isoflurane and maintained with additional doses of a mixture of anesthetics (0.3 mg/kg of medetomidine, 4.0 mg/kg of midazolam, and 5.0 mg/kg of butorphanol) at 2–3 mg/kg/h as required, through a cannula into the jugular vein. During the recording session, the absence of a reaction to paw pinching confirmed the level of anesthesia. A homeothermic blanket maintained the rectal temperature at 37.0°C ± 0.5°C (Temperature Controller, 40-90-8D; FHC, TX, USA) during recording. Throughout the experiments, the edges of all wounds were consistently covered with a local anesthetic, 2% lidocaine (Xylocaine). The animals were subsequently situated in a stereotaxic device (SR-50; Narishige, Tokyo, Japan) and the neck muscles were divided along the animal’s midline. The atlanto-occipital ligament and underlying dura mater were incised to access the medullary brain stem. A tungsten microelectrode (impedance 3–5 MΩ) was used to perform extracellular recording of single-unit activity from the SpVc region in the ipsilateral medulla. The microelectrode was moved forward or backward in 10 μm increments using a micromanipulator (SM-11 and MO-10; Narishige), according to the stereotaxic coordinates of the rat brain atlas of Paxinos and Watson. 17 Neuronal signals were amplified (DAM80; World Precision Instruments, Sarasota, FL, USA), filtered (0.3–10 KHz), and observed using an oscilloscope (SS-7672; Iwatsu, Tokyo, Japan), with data recorded for future analysis using PowerLab and Chart 5 software (ADI Instruments, Oxford, UK) as described previously. 18
Electrophysiological recordings
The process for analyzing extracellular single-unit SpVc WDR activity in response to whisker pad mechanical stimulation involved the following steps. To prevent sensitization of peripheral mechanoreceptors, the approximate receptor area of the receptive field on the left side of the whisker pad was identified using a paint brush swiftly as a search stimulus. Subsequently, the left side of the whisker pad was examined for individual units that reacted to a series of von Frey hairs (Semmes-Weinstein Monofilaments, North Coast Medical, USA) with non-noxious (2, 6, 10 g) and noxious (15, 26, 60 g) mechanical stimulation for 5 s at intervals of 5 s. 18 The criteria for WDR neurons were previously identified as follows: graded mechanical stimulation, whether non-noxious or noxious, applied to the receptive field results in an increased firing frequency proportional to the stimulus strength. Following the identification of whisker pad-responsive nociceptive SpVc WDR neurons, we established the mechanical stimulation threshold and the size of the receptive field. The mapping of the neurons’ mechanical receptive fields was accomplished by applying von Frey hairs to the face and subsequently outlining them on a life-sized facial illustration. 18 The quantification of WDR neuronal discharges, due to mechanical stimulation, was achieved by removing the background activity from the evoked activity. Spontaneous discharge frequencies were determined over 2–5 min. Previous research has shown that WDR neurons in the SpVc region play a crucial role in the mechanisms behind hyperalgesia and referred pain.2,3 Our study focused on the influence of SCFA on nociceptive SpVc WDR neuronal activity, omitting the examination of nociceptive-specific neurons. Post-stimulus histograms (bin = 100 ms) were generated in response to each stimulus. As a previous in vitro study in sympathetic cervical ganglion neurons expressing rat GPR41 showed that propionate (C3) was more potent in inhibiting Cav channel currents than acetate (C2), we used PA as the SCFA in this study. 13 The impact of systemic administration of PA (1 mM and 10 mM; Fujifilm-Wako, Tokyo, Japan) was assessed at 5, 10, 15, 20, 25, and 30 min following administration due to the peak effect being observed within that timeframe.
PA was dissolved in 0.9% NaCL to create a stock solution of 10 mM. The stock solution was stored at –20°C until use and then diluted to the desired concentrations using saline immediately before use. The GPR41 antagonist, (R)-(-)-3-hydroxybutyric acid (BHB; Sigma-Aldrich, Milan, Italy), was dissolved in 0.9% NaCL to create a stock solution. The concentration of the GPR41 antagonist (50 mg/kg, i.p,) used in this study was based on previous evidence showing that this dose of GPR41 antagonist significantly blocked SCFA-induced increases in sympathetic cervical ganglion neuronal activity and heart rate.19,20 Heart rate was analyzed by electrocardiograms recorded with an amplifier (DAM80; World Precision Instruments) and PowerLab and Chart
Data analysis
Values are presented as means ± SEMs. A one-way repeated-measures analysis of variance was used for statistical evaluation, with Tukey–Kramer or Dunnett’s tests administered as post-hoc analyses and t-tests for electrophysiological data. A two-sided p-value < 0.05 was considered to indicate a significant difference.
Results
General characteristics of SpVc WDR neurons innervating the orofacial skin
The activity of extracellular single units was recorded from 14 neurons in the SpVc region. In response to non-noxious and noxious mechanical stimulation, the SpVc neurons showed a somatic receptive field in the orofacial area (mainly whisker pad; Figure 1(a)), as described previously. 18 Additionally, each of these neurons was activated by mechanical stimulation of the receptive field associated with maxillary branches. Two of the 14 units displayed spontaneous discharges. The recording sites were located in Layers I–II (n = 4; 28.6%) and III-IV (n = 10; 71.4%) in the SpVc (obex –0.5 to 2 mm; Figure 1(b)). Figure 1(c) depicts representative examples of action potential waveforms and SpVc WDR neuronal unit responses. A gradual mechanical stimulation was applied to the most reactive part of the receptive field, enhancing the firing frequency of SpVc WDR neurons in proportion to stimulus intensity. The mean threshold for spikes triggered by mechanical stimulation was 2.2 ± 3.3 g (n = 14). Figure 1(d) illustrates the association between the intensity of mechanical stimulation and the average discharge frequency (stimulus-response curve) for WDR neurons. All recorded neurons were classified as WDR neurons. 18

Fundamental features of spinal trigeminal nucleus caudalis (SpVc) wide-dynamic range (WDR) neuron activity induced by mechanical stimulation of the facial skin. (a) Blackened area represents the whisker pad’s receptive field on the facial skin. (b) Spatial distribution of SpVc WDR neurons reacting to mechanical stimulation (non-noxious and noxious) of the facial skin (n = 14). D, dorsal; V, ventral; M, medial; L, lateral. (c) Representative SpVc WDR neuronal responses caused by non-noxious (2, 6, and 10 g) and noxious (15, 26, and 60 g) mechanical stimulation applied to the orofacial area. Inset: wave form of action potential. (d) Response curve of SpVc WDR neurons to stimuli (n = 14).
Effects of PA on the excitability of SpVc WDR neurons in response to non-noxious stimuli
Figure 2(a) shows a representative case of the impact of PA (10 mM, i.v.) on SpVc WDR neuron excitability in reaction to non-noxious mechanical stimulation. Ten minutes following PA injection, non-noxious (2, 6, and 10 g) mechanical stimulation-induced SpVc WDR neuronal activity was suppressed, but this inhibition disappeared, with activity returning to control levels, within approximately 20 min. There were no noticeable differences in the size of the receptive field or mechanical threshold post-PA administration.
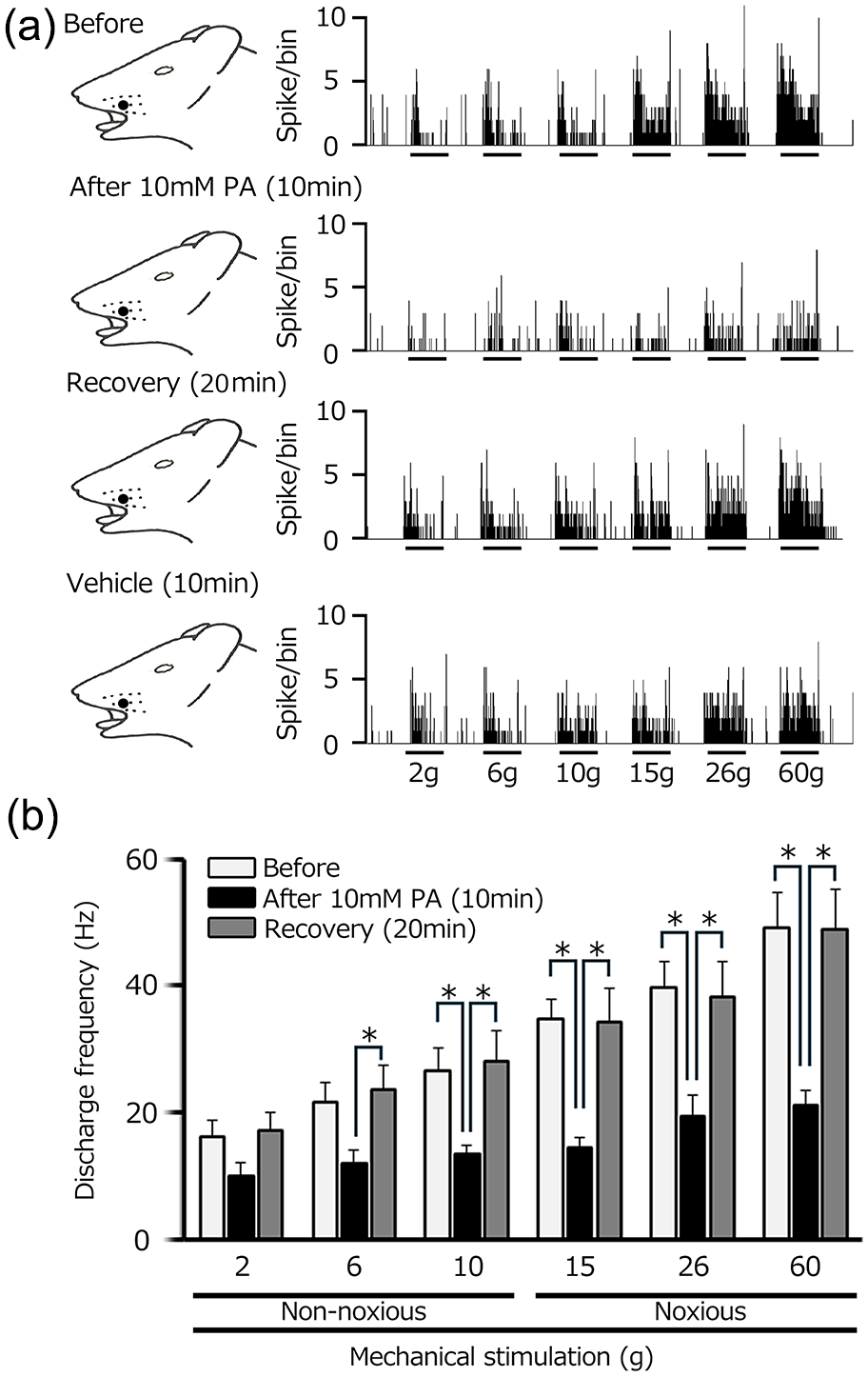
Effect of intravenous administration of 10 mM propionic acid (PA) on the SpVc WDR neuronal response induced by non-noxious and noxious mechanical stimulation. (a) Representative examples of SpVc WDR neuronal responses triggered by non-noxious and noxious mechanical stimuli before, and 10 and 20 min after, administration of 10 mM PA. Effect of intravenous administration of vehicle (0.9% NaCL) on the SpVc WDR neuronal activity is also shown. (b) The timeline of intravenous PA application and its impact on the average firing rate of SpVc WDR neurons responding to non-noxious, noxious, and noxious mechanical stimulation.
SpVc WDR neuronal activity evoked by non-noxious mechanical stimulation is summarized in Figure 2(b). After intravenous PA injection, there was a significant decrease in the mean firing rate of non-noxious mechanical stimulation-evoked SpVc WDR neuronal activity, which eventually returned to control levels. Mean firing rates before and after PA were 21.8 ± 2.9 and 11.9 ± 2.2 Hz, respectively, in response to the 6-g stimulus, and 26.5 ± 3.7 and 13.4 ± 1.4 Hz, respectively, in response to the 10-g stimulus (6 and 10 g; n = 10).
Effects of PA on excitability of SpVc WDR neurons in response to noxious stimuli
Figure 2(a) shows a representative case of the impact of PA (10 mM, i.v.) on SpVc WDR neuron excitability in reaction to noxious mechanical stimulation. Ten minutes following PA injection, noxious (15, 26, and 60 g) mechanical stimulation-induced SpVc WDR neuronal activity was suppressed, but this inhibition disappeared, with activity returning to control levels, within approximately 20 min. There were no noticeable differences in the size of the receptive field or mechanical threshold post-PA administration. Some SpVc WDR neurons (3/14) showed spontaneous discharges after PA administration, and there was a tendency for a decrease in mean spontaneous discharges after PA.
SpVc WDR neuronal activity evoked by noxious mechanical stimulation is summarized in Figure 2(b). After intravenous PA injection, there was a significant decrease in the mean firing rate of noxious mechanical stimulation-evoked SpVc WDR neuronal activity, which eventually returned to control levels. Mean firing rates before and after PA were 36.0 ± 3.5 and 14.3 ± 1.8 Hz, respectively, in response to the 15-g stimulus and 46.6 ± 9.7 and 22.8 ± 3.0 Hz, respectively, in response to the 60-g stimulus (15 and 60 g; n = 10, *, p < 0.05 for both).
Intravenous injection of 10 mM PA resulted in a greater suppression of SpVc WDR neuronal firing compared to 1 mM PA (22.7% ± 6.8% (n = 4) vs 52.8% ± 3.7% (n = 10); p < 0.05) for noxious stimuli (Figure 3). Intravenous administration of vehicle (0.9%NaCL) had no significant effect on spontaneous, non-noxious, and noxious mechanical stimulation-evoked SpVc WDR neuronal activity (n = 3), as described previously (Figure 2). 18

PA exhibited a dose-dependent inhibition on the average firing frequency of SpVc neurons in response to both non-noxious and noxious mechanical stimuli.
SpVc WDR neuronal activity in response to noxious versus non-noxious stimuli after PA
We assessed the relative suppressive effect of PA (10 mM i.v.) on responses to non-noxious and noxious stimuli. There was a significant difference in the mean inhibition by SCFA on SpVc WDR neuronal discharge between noxious and non-noxious stimuli (non-noxious vs. noxious, 38.0% ± 5.6% vs 52.8% ± 3.7%; Figure 4)

Comparison of the decrease in SpVc neuron discharge frequency induced by PA between non-noxious and noxious stimuli.
Effect of GPR41 antagonist on the PA-induced reversible inhibition of SpVc neurons in response to noxious and non-noxious stimuli after PA
Next, we examined whether systemic injection of PA suppresses the excitability of nociceptive SpVc WDR neurons in response to non-noxious and noxious mechanical stimulation via activation of GPR41. We investigated whether administration of a GPR41 inhibitor, BHB (50 mg/kg, i.p.), abolished the suppression in discharge frequency in five SpVc neurons whose discharge frequency was reversibly suppressed by PA. The obvious inhibition produced by PA administration was restored 20 min later. The PA-induced inhibition in discharge frequency of this neuron was abolished by simultaneous administration of BHB (Figure 5(a)). The presence of the GPR41 antagonist, BHB, significantly abolished the inhibitory effect of PA on the firing frequency of SpVc WDR neurons (n = 4, p < 0.05; Figure 5(b)).

Effect of the GPR41 antagonist, 3-hydroxybutyric acid (BHB), on the PA-induced reversible inhibition of SpVc neurons in response to noxious and non-noxious stimuli. (a) A typical example of obvious inhibition caused by PA administration. The PA-induced discharge frequency of this neuron was abolished by simultaneous administration of the GPR41 inhibitor, BHB (50 mg/kg, i.p.). (b) Summary of the effect of a GPR41 antagonist on the PA-induced reversible inhibition of SpVc neurons in response to noxious and non-noxious stimuli (n = 4).
Lack of effect of SCFA on heart rate
Previous administration of PA has been shown to stimulate sympathetic ganglion neurons via GPR41. 19 Therefore, we tested whether intravenous administration of PA activated the sympathetic nerves and increased heart rate at the same range of PA concentration (1–10 mM) that induced inhibition of SpVc WDR neuronal activity in response to mechanical stimulation of the orofacial skin. We found no significant changes occurred in heart rate before or 5–20 min after intravenous administration of 10 mM PA (330.2 ± 56.6 vs 311.6 ± 50.8 beat/min, respectively, n = 3).
Discussion
Systemic application of SCFA suppresses the excitability of nociceptive secondary trigeminal neurons
The objective of the current study was to investigate whether systemic administration of the SCFA, PA, reduces the excitability of nociceptive SpVc WDR neurons in response to mechanical stimulation. In vitro studies have shown that 0.1 mM SCFA administration inhibits N-type Cav channel currents in isolated sympathetic ganglion neurons. 13 Therefore, it is reasonable to conclude that following systemic administration of 10 mM SCFA, SCFA enters the general circulation and is diluted in the bloodstream to a calculated concentration of approximately 0.1 mM. In this research, we investigated whether systemic administration of 10 mM SCFA reduces the excitability of SpVc WDR neurons. The following results were observed in this study: (i) the mean SpVc WDR neuronal firing rate in response to both non-noxious and noxious mechanical stimuli was significantly and dose-dependently inhibited by PA (1–10 mM i.v.); (ii) the inhibition of the discharge frequency in response to both non-noxious and noxious mechanical stimuli was reversible and occurred within approximately 20 min; (iii) the mean magnitude of inhibition by SCFA of SpVc WDR neuronal discharge frequency was significantly greater for noxious compared to non-noxious stimuli; (iv) co-administration of the GPR41 antagonist, BHB, abolished the inhibitory effect of PA on the firing frequency of SpVc; and (v) injection of vehicle (saline) had no significant effect on SpVc WDR neuronal activity in response to either non-noxious or noxious mechanical or pinch stimuli. Taken together, these findings suggest that under in vivo conditions, acute intravenous PA administration suppresses trigeminal nociceptive neuronal excitability via activation of GPR41 signaling-mediated inhibition of Cav channel currents, probably in primary sensory neurons.
Alternatively, a previous study indicated that systemic administration of PA significantly increased heart rate via increased sympathetic neuronal activity. 19 Hence, it is plausible that the rise in serum noradrenaline activates presynaptic nociceptive transmission via α2-adrenoceptor pathways.3,21 Many previous studies have shown that noradrenaline signaling exerts a potent antinociceptive effect by the activation of α2-adrenoreceptors 21 involving the descending inhibitory system. 22 Clonidine, an α2-adrenoreceptor agonist, has an antinociceptive effect23–28 and is used for the treatment of migraine.26–28 Evidence also suggests that iontophoretic administration of clonidine suppresses N-methyl-aspartate-induced responses in SpVc nociceptive neurons. 29 According to prior findings by Takeda et al., 25 clonidine, acting as an α2-adrenoreceptor agonist, diminished the postsynaptic excitability of C1 neurons which have convergent inputs from tooth pulp and sagittal sinus afferents, and that activation of α2-adrenoreceptors in C1 dorsal horn neurons may contribute to a useful therapeutic target for the trigeminal referred pain associated with migraine and tooth pain. In the present study, we observed that systemic administration of 10 mM SCFA produced no significant change in heart rate, suggesting that PA administration suppresses trigeminal nociceptive neuronal excitability via Cav channels, and not the noradrenaline-activated local α2-adrenoreceptor inhibitory system.
Mechanism underlying suppression of SpVc neuronal excitability by systemic administration of SCFA
Many previous studies indicated that SCFA binds to GPR41 receptors on the cell membrane, affecting physiological processes via diverse intracellular signaling pathways, and SCFAs have been found to regulate the permeability of the brain-blood barrier. 12 The molecular target for the activation of GPR41 signaling that mediates the inhibition of Cav channels has been studied previously. 13 To our knowledge, currently, expression of GPR41 has not been confirmed in SpVc neurons, but it has been confirmed in TG neurons. 15 In this experiment, acute administration of PA suppressed the activity of nociceptive SpVc neurons, and this effect was suppressed by a GPR41 inhibitor. Therefore, it is speculated that PA inhibits Cav channels via presynaptic GPR41, suppressing excitatory neurotransmitter release, resulting in the suppression of nociceptive SpVc neuron activity.
Cav channels are recognized to open upon membrane depolarization, enabling the influx of Ca2+ ions. This influx results in two key consequences: (i) since Ca2+ ions possess two positive charges, the membrane undergoes further depolarization, which results in the activation of additional voltage-gated ion channels (e.g. Na+ and K+ channels) and (ii) Ca2+ serves as a second messenger that triggers the activation of a range of cellular processes, such as contraction, secretion, and gene transcription. 30 There are two categories of Cav channels: low voltage-activated, also known as T-type, and high voltage-activated, for example, L, P/Q, N, and R-types. Although, to our knowledge, SCFAs have been previously shown to act only on N-type Cav channels, 13 T-type Cav channels in the primary afferents involved in the pain pathway are crucial for maintaining neuronal firing and seem to play a role in neurotransmitter release at afferent terminals in the spinal dorsal horn.31,32 This increased neuronal excitability leads to amplification of sensory transmission resulting in enhanced neuronal excitability, intensified sensory processing, and, in turn, pathological pain perception. 33 Therefore, blocking T-type Cav/Cav3.2 channels mediate analgesia. 34 Gadotti et al. 34 noted that the flavonoid, gossypetin, a molecule structurally very similar to quercetin, suppressed inflammatory and neuropathic peripheral tissues, in part via an action on Cav3.2 channels. Gambeta et al.33,35 demonstrated that T-type calcium channels are crucial controllers of neuronal functions within the trigeminal system and significantly influence trigeminal pain. Taken together, these findings indicate that acute intravenous SCFA administration reduces trigeminal nociceptive neuronal excitability via T-type Cav channels, leading to inhibition of Na+ and opening K+ channels in the TG neurons. Nonetheless, further verification studies, such as in vitro patch-clamp analysis of dissociated TG neurons with fluorescent labeling from the receptive field of the whisker pad, are needed.
Previous studies have demonstrated that activation of vagal afferents stimulate the endogenous descending inhibitory system.36–38 Several physiological factors also trigger the descending inhibitory system; for example, the jaw-opening reflex activated by tooth-pulp electrical stimulation-evoked spinal trigeminal nucleus oralis (SpVo) neuronal activity inhibits conditioning stimulation of vagal afferents. 37 Further, we provided evidence that the physiological stimulation of vagal afferents induced by volume expansion induced nociceptive transmission in the SpVo neurons related to the jaw-opening reflex, and that this inhibitory effect appeared via the endogenous opioid system (the periaquaductal gray – nucleus raphe magnus – trigeminal pathway). 38 Previous studies have reported that nociceptive stimulation-evoked SpVc neuron activity is suppressed by conditioning peripheral nerve stimulation via 5-HT3 receptor-mediated GABAergic inhibition. 39 These findings indicate that the low-pressure cardiopulmonary baroreceptor, whose afferent travels in the vagal nerve, inhibits trigeminal nociceptive transmission. 38 Since Goswami et al. 40 previously reported that intraperitoneal injection of SCFA activates vagal afferent neurons to suppresss food intake, it is possible that systemic injection of SCFA inhibits nociceptive SpVc WDR neurons via activation of vagal afferents inducing the descending inhibitory system. Therefore, these findings suggest that PA administration suppresses trigeminal nociceptive neuronal excitability, not only via Cav channels, but also by activation of the vagal afferent-induced descending opioid inhibitory system. However, to further investigate this possibility, future experiments using multi-barrel electrodes in the SpVc region will be necessary to clarify the relationship between local administration of opioids, GABAA, 5HT3 receptor antagonists, and systemic administration of SCFAs, using the methods described in previous studies.25,29
Functional significance of SCFA suppression of nociceptive stimulation-induced hyperexcitability of SpVc neurons
Recently, our investigations revealed that administering natural dietary components, such as resveratrol, intravenously and acutely reduced nociceptive SpVc WDR neuronal activity in a concentration-dependent manner via inhibition of Cav channels and glutaminergic noxious synaptic transmission in the SpVc. This indicates that a dietary component may serve as a potential complementary and alternative therapeutic agent for the treatment of trigeminal nociceptive pain. 18 According to a recent study, food intake manages energy balance, and its misregulation causes metabolic issues, such as obesity and diabetes. Free fatty acids are not only essential nutrients, but also act as signaling molecules in various cellular processes. 19 There are several metabolites from gut microbiota, for example, SCFAs, that regulate pain sensation via multiple mechanisms. 12 SCFAs are derived primarily from bacterial fermentation of carbohydrates and protein, producing a plasma SCFA concentration of between 0.1 and 10 mM.41,42 Frost et al. 43 reported that systemic administration of SCFA reduced food intake via the brain, crossing over the blood-brain barrier and directly activating hypothalamic neurons. The present study showed that acute intravenous administration of a physiological concentration of SCFA (1–10 mM) significantly inhibited trigeminal nociceptive neuronal excitability via activation of GPR41 signaling-mediated inhibition of Cav channels. These findings suggest that systemic administration of gut microbiota-derived SCFAs modulate trigeminal nociception, indicating that SCFAs may act as intravenous analgesic agents to provide relief from trigeminal pain, producing a new therapeutic strategy for the management of trigeminal pain, such as orofacial clinical pain, and also visceral pain.
Interestingly, Murakami et al. 11 recently reported that butyric acid-GPR41 signaling triggered by Porphyromonas gingivalis plays a critical role in the development of periodontitis without periodontal inflammatory pain in a mouse model. They also reported that (i) the number of GPR41-immunoreactive TG neurons innervating the inflamed periodontal tissue after treatment of Porphyromonas gingivalis was larger than for Complete Freund’s Adjuvant (CFA)-treated mice and control mice and (ii) SCFA administration into the gingival tissue in CFA-treated mice recovered the decrease in head withdrawal threshold. Ma et al. 10 also reported that resveratrol significantly inhibited CFA-induced inflammation innervating the temporomandibular joint, and reversed the CFA-induced reduction of SCFAs and the relevant gut bacteria. Therefore, since these findings suggest that SCFA administration alleviates CFA inflammatory pain, such as trigeminal mechanical hyperalgesia, it can be assumed that SCFAs play an important role for alleviating pain caused by the small daily inflammatory responses in internal organs. Therefore, further studies are needed to examine the possibility that gut microbiota-derived SCFAs are associated with the intake of food phytochemicals, such as resveratrol, lutein and quercetin.
Conclusions
The present study provides the first evidence that systemic administration of the SCFA, PA, suppresses the excitability of nociceptive secondary trigeminal neurons via activation of GPR41 signaling-mediated inhibition of Cav channels in the central terminals of the SpVc (Figure 6). Therefore, systemic administration of gut microbiota-derived SCFAs modulate trigeminal nociception, and the use of SCFAs as intravenous analgesic agents may provide relief from trigeminal pain, creating a new therapeutic strategy for the management of trigeminal pain, including clinical pain.

A possible mechanism underlying SCFA-induced inhibition of SpVc WDR neuronal discharge responding to nociceptive mechanical stimulation. When noxious mechanical stimulation is applied to the skin, mechanosensitive ion channels open, activating generator potentials. This depolarization further opens voltage-gated Na+ (Nav) and K+ (Kv) channels, generating action potentials, which are in turn discharged through primary afferent fibers to the central terminal of nociceptive neurons in the SpVc. Once the action potential reaches the central end of the nerve terminal, voltage-gated Ca2+ (Cav) channels at this location open, causing the nerve terminal to depolarize and permit the entry of Ca2+ ions. When the intracellular concentration of Ca2+ rises, it prompts the discharge of excitatory neurotransmitters, such as glutamate (Glu), from the presynaptic neuron into the synaptic cleft, allowing cations to flow into the cell by activating ionotropic glutamate receptors on the secondary sensory neurons. When glutamate receptors are activated, causing cations to flow into the cell, an excitatory postsynaptic potential (EPSP) is produced. Once this EPSP reaches a specific membrane potential threshold, an action potential is initiated. Intravenous administration of the gut microbiota-derived SCFA, PA, suppresses SpVc WDR neuronal excitability via activation of GPR41 signaling-mediated inhibition of Cav channels in the presynaptic terminal of trigeminal ganglion (TG) neurons,13,14 decreasing the discharge rate of action potential firing of SpVc WDR neurons propagating to higher centers of pain.
Footnotes
Author contributions
YS performed all experiments, analyzed data, and prepared figures. MT participated in the design of the experiments and wrote the manuscript. All the authors have read and approved the paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by a Grant-in-Aid for Scientific Research (C) from the Japanese Society for the Promotion of Science (No.22K10232).
