Abstract
Bone cancer pain is a complex pain state involving ongoing pain and movement-related pain, which are thought to be caused by different mechanisms. Transient receptor potential vanilloid subfamily 1 (TRPV1) is involved in ongoing pain but not movement-related pain. The purpose of this study was to investigate the role of transient receptor potential vanilloid subfamily 2 (TRPV2) in bone cancer pain.
Proportions of TRPV1- and TRPV2-immunoreactive neurons in lumbar dorsal root ganglia innervating the femurs of male mice were examined by using Fluoro-Gold. Mice were intrathecally injected with small interfering RNA (siRNA) against TRPV2 or scrambled siRNA for three consecutive days from day 14 after sarcoma injection into the left femur. In the mice with bone cancer, the number of spontaneous flinches was quantified for assessment of ongoing pain, and limb use and weight bearing were assessed as indications of movement-related pain. Changes in TRPV2 protein levels in dorsal root ganglion were evaluated by Western blotting. We also examined the effects of intrathecal administration of siRNA against TRPV2 or scrambled siRNA on thermal and mechanical sensitivities in normal mice without tumors.
The proportions of TRPV1-immunoreactive and TRPV2-immunoreactive neurons were 21% and 22% of neurons in dorsal root ganglia innervating the femur, respectively. Tumor-bearing mice exhibited an increased number of spontaneous flinches and impaired limb use and weight bearing at day 13 after sarcoma injection. TRPV2 protein level in dorsal root ganglia at day 13 was comparable to that at baseline. siRNA against TRPV2 significantly improved limb use and weight bearing but did not affect the number of spontaneous flinches compared to those in the group treated with scrambled siRNA. siRNA against TRPV2 did not affect thermal or mechanical sensitivity in normal mice.
The results suggest that TRPV2 is involved in movement-related pain but not ongoing pain in mice with bone cancer.
Keywords
Background
Cancer-induced bone pain is a major clinical problem with up to 85% of patients with bony metastases having pain. 1 Common cancers such as breast, lung, and prostate cancers frequently metastasize to bones of the body including the vertebrae, hip, and tibia. Metastatic bone cancer pain becomes worse with progression of the disease.2–4 Treatment of pain from bone metastases involves the use of a variety of complementary approaches including nonsteroidal anti-inflammatory drugs and opioids, known as the “World Health Organization analgesic ladder.” 5 However, bone cancer pain is one of the most difficult of all chronic pains to fully control because the pain becomes resistant to opioid analgesics. 4
One reason for the challenging problem of treatment of bone cancer-induced pain is that it is a complex pain state that includes ongoing pain and movement-related pain. 1 The initial pain is usually ongoing pain that appears as a dull ache or throbbing pain.3,6 Movement-related pain refers to physical activities such as weight bearing in a patient with bone metastases or dressing changes that can be predicted to worsen pain. The results of our previous study showed that intraosseous balloon inflation-elicited spontaneous pain-related behaviors and induced cutaneous mechanical hyperalgesia and allodynia. 7 These results suggest that an increase in intraosseous pressure caused by tumor growth contributes to bone cancer-induced ongoing pain and movement-related pain. In addition, we recently reported that transient receptor potential vanilloid subfamily 1 (TRPV1) activation is involved in ongoing pain in bone cancer. 8 Havelin et al. reported that ablation of isolectin B4 (IB4)-binding, but not TRPV1-positive, sensory afferents caused a profound reduction of movement-related pain in rats with bone cancer. 9 Colocalization of TRPV1 and IB4 is rare in mice. 10 These observations suggest that ongoing pain and movement-related pain in patients with bone cancer are mechanistically distinct. Although there has been a gradual accumulation of information on bone pain, information about the mechanisms by which bone cancer-induced pain is generated and maintained is still not sufficient.
TRPV1, which is the most extensively studied member of the transient receptor potential vanilloid subfamily, can be activated by capsaicin and other stimuli such as noxious heat and low pH, and TRPV1 has been shown to be localized to unmyelinated C-fibers or thinly myelinated Aδ-fibers in dorsal root ganglia (DRGs). 11 The transient receptor potential vanilloid subfamily 2 (TRPV2), discovered as a structural homologue of TRPV1 with 50% amino acid identity, is also thought to be a target for pain relief. 12 TRPV2 was found to most frequently present in mechanically sensitive myelinated fibers including both low-threshold and high-threshold cutaneous mechanoreceptors. 13 Thus, TRPV1 and TRPV2 are expressed by distinct subpopulations of primary afferents. Both TRPV1 and TRPV2 have been found in a relatively large subset of primary sensory neurons.13,14 TRPV2 contributes to peripheral sensitization during cutaneous inflammation. 14 However, the presence of TRPV2 in bone tissue and the role of TRPV2 in bone cancer pain have not been investigated.
Movement-related pain is more difficult to control because it has a profile of rapid onset, high peak, and fast resolution. 1 The mechanisms underlying movement-related pain remain unknown. A better understanding of the mechanisms that drive bone cancer pain is necessary to develop new treatment options. We hypothesized that TRPV2 is involved in movement-related pain in bone cancer. To prove the hypothesis, we investigated the presence of TRPV2-immunoreactive (ir) neurons in DRGs innervating the femur and the effects of selective inhibition of TRPV2 protein on bone cancer-induced pain by using small interfering RNA (siRNA) against TRPV2.
Materials and methods
Animals
All of the protocols of this study were approved by the Animal Care and Use Committee of Shinshu University School of Medicine, Matsumoto, Japan (reference No. 240050). All efforts were made to minimize animal suffering and to reduce the number of animals used. Mice were treated in accordance with the Ethics Guidelines for Investigations of Experimental Pain in Conscious Animals issued by the International Association for the Study of Pain. Experiments were conducted in adult male C3H/HeJ mice weighing 20 to 25 g (Japan SLC, Hamamatsu, Japan). The mice were housed in a temperature-controlled (21 ± 1°C) room with a 12-h light/dark cycle and given free access to food and water.
Retrograde Fluoro-Gold labeling for detection of TRPV1-ir and TRPV2-ir DRG neurons innervating bone marrow of the femur
Mice were anesthetized with 3% sevoflurane in 100% oxygen via a nose cone and treated aseptically throughout the experiments. For each mouse, a 5-mm-long longitudinal incision was made on the medial aspect of the left knee, and the left femur was exposed under a microscope. Five microliters of 2% Fluoro-Gold (FG) solution (Fluorochrome, Denver, CO, USA) were injected into the inside of the left femur (bone marrow) using a 26-gauge needle to label DRG neurons innervating bone marrow of the left femur (n = 8). The incision was immediately sealed with cyanoacrylate adhesive to prevent leakage of FG, and the skin was closed. Forty-eight hours after injection of FG, mice were anesthetized with sevoflurane and transcardially perfused with 4% paraformaldehyde (PFA). Left L2 to L4 DRGs were removed and immersed in 4% PFA in phosphate-buffered saline (PBS) for 2 h for postfixation. The samples were placed in Tissue-Tek embedding medium (Sakura, Tokyo, Japan) and rapidly frozen. Longitudinal DRG sections at 16 µm in thickness were cut using a cryostat (CM 3050S; LEICA, Tokyo, Japan) and processed for immunohistochemistry. Two DRG sections were obtained from the central part of each DRG. One was used for investigating the expression profile of TRPV1, and the other was used for TRPV2. The tissue sections were washed in PBS and incubated for 1 h at room temperature (RT) in a blocking solution consisting of 10% normal goat serum and 0.2% Triton X-100 (Sigma-Aldrich, St. Louis, MO, USA) in PBS (PBS-t). The sections for TRPV1 were incubated with rabbit anti-TRPV1 antibody (1:1000; Trans Genic Inc., Kobe, Japan) in 1% bovine serum albumin (BSA; Sigma-Aldrich) and 0.3% Triton-X 100 (Sigma-Aldrich) in 0.1 M PBS overnight at 4°C. The sections for TRPV2 were incubated with rabbit anti-TRPV2 antibody (1:2000; Trans Genic Inc.) in 1% BSA and 0.3% Triton-X 100 in 0.1 M PBS overnight at 4°C. After rinses with PBS-t, each section was incubated with Alexa Fluor 488-labeled antirabbit secondary antibody (1:200; Molecular Probes, Eugene, OR, USA) in PBS-t for 2 h at RT. The omission of primary or secondary antibodies resulted in almost no staining.
Slides were examined by blinded investigators under a fluorescence microscope (IX73; OLYMPUS, Tokyo, Japan). FG-labeled DRG neurons were detected through an ultraviolet filter. TRPV1-ir or TRPV2-ir DRG neurons were examined using a fluorescein isothiocyanate filter (wavelength of 470 nm for excitation and 510 nm for emission). Profiles of the DRG neurons were divided into small (<30 µm), medium (30–40 µm), and large (>40 µm) cells 15 by measuring the diameters from images on a computer. FG-labeled DRG neurons, TRPV1-ir FG-labeled DRG neurons, or TRPV2-ir FG-labeled DRG neurons were counted in each section. Then, the total numbers of neurons in the 24 sections derived from eight mice (L2–L4 DRGs) were estimated.
Bone cancer model
Murine sarcoma cells (NCTC 2472; American Type Culture Collection (ATCC), Rockville, MD, USA) were maintained in NCTC 135 medium containing 10% horse serum (HyClone, Logan, UT, USA) and passaged weekly according to ATCC® recommendations. Injection of sarcoma cells was performed according to a previously described method. 16 Briefly, mice were anesthetized with sevoflurane (3% in 100% oxygen). Twenty microliters of medium containing 1 × 105 sarcoma cells were injected into the intramedullary space of the left femur using a 29-gauge needle and a Hamilton syringe (Hamilton Co., NV, USA). It has been shown that cancer pain-related behaviors in this model are maximally exhibited at 14 days after implantation and maintained up to day 21.17,18 Each animal was used in only one experiment.
Assessment of bone cancer pain-related behaviors
Mice were placed in a clear plastic box (30 × 20 × 15 cm) and allowed to habituate to the cage for 30 min. Behavioral assessments were then performed. Ongoing and movement-related pain behaviors were analyzed according to previously described methods.10,16,19 Briefly, quantification of spontaneous flinches during a 2-min observation period was used for assessment of the degree of ongoing pain. Limb use during spontaneous ambulation and weight bearing during spontaneous standing were used as indications of movement-related pain. 20 Limb use was assessed according to a previously described method. 21 Each mouse was placed in an empty mouse pan and observed while walking across the pan in a continuous motion. Limping and/or guarding behavior of the left (sarcoma-implanted) hindlimb was rated on the following scale: 0 = complete lack of use, 1 = partial nonuse, 2 = limping and guarding, 3 = limping, and 4 = normal use. Weight bearing during standing was scored on a scale of 0 to 3: 0 = no weight bearing (the hindpaw on the injected side always being lifted and never touching the floor), 1 = touch weight bearing (the hindpaw on the injected side being lifted from the floor but occasionally touching the floor), 2 = partial weight bearing (partial support of body weight with the hindpaw on the injected side), and 3 = full weight bearing (full support of 100% of body weight with both hindpaws). All animals were euthanized at 21 days after sarcoma injection by inhalational sevoflurane overdose after behavioral analyses were complete.
Assessment of pain-related behavior in normal mice
Paw withdrawal latency to noxious heat stimuli was assessed by applying a focused radiant heat source (model number 37370; Ugo Basil, Comerio, Italy) to unrestrained mice. The hindpaw response was defined as either a foot shake or a paw lick. A cutoff period of 20 s was imposed to avoid tissue damage to the paws. The von Frey test was used to detect the mechanical threshold in the hindpaw region. Paw withdrawal thresholds in response to punctate mechanical stimuli were measured using von Frey filaments consisting of calibrated filaments. Mice were placed on a stainless mesh floor covered with a clear plastic cage top. For testing mechanical responses, calibrated von Frey filaments were applied to the hindpaw of unrestrained mice. We determined the 50% mechanical withdrawal threshold by the “up-down method” according to a previous report. 22 A series of seven von Frey filaments (0.07, 0.16, 0.4, 0.6, 1.0, 1.4, and 2.0 g-forces) were used. Limb use during spontaneous ambulation and weight bearing during spontaneous standing were also assessed.
Preparation of siRNA against TRPV2 and scrambled siRNA
siRNA against mouse TRPV2 was synthesized by Ambion (Thermo Fisher Scientific Inc., Waltham, MA, USA). Sense and antisense siRNA sequences were as follows: 5′-GAACCUGCUUUAUUAUACATT-3′ and 5′-UGUAUAAUAAAGCAGGUUCAG. A scrambled control siRNA without significant homology to any known mouse gene sequence (Silencer Negative Control #1) was obtained from Ambion. The hemagglutinating virus of the Japan (HVJ) envelope vector system (HVJ Envelope Vector Kit GenomONE; Ishihara Sangyo Kaisha, Ltd., Osaka, Japan) was used for in vivo siRNA transfer. siRNA was incorporated into the HVJ-envelope vector according to the manufacturer’s instructions. In brief, after mixing 40 µl (1 assay unit) of the HVJ-envelope vector with 4 µl of the enclosing factor, the mixture was centrifuged (10,000 g, 10 min, 4°C), and the pellet was suspended in 10 µl of buffer solution. Then, 10 µl siRNA solution was added, and the mixture was kept on ice for 5 min. The final siRNA concentration was 5 µg/10 µl.
Insertion of an intrathecal catheter for siRNA administration
Under general anesthesia with 3% sevoflurane in 100% oxygen via a nose cone, a polyurethane intrathecal (i.t.) catheter with an inner diameter of 0.005″ and an outer diameter of 0.010″ (R-ITC; Neuroscience Inc., Tokyo, Japan) was inserted 5 mm cephalad into the mouse lumbar subarachnoid space at the L4/L5 intervertebral space, with the tip of the catheter located near the lumbar enlargement of the spinal cord. The catheter was tunneled subcutaneously and externalized through the skin in the neck region. One day after implantation of the catheter, the effects of i.t. lidocaine (2%, 2 µl) were examined. Only animals that had shown complete paralysis of the tail and bilateral hind legs after i.t. lidocaine were used in the following experiments. To prevent infection, 10,000 units/kg of penicillin was injected subcutaneously every day for three days after surgery. Mice showing paralysis or impaired motor function following the surgeries were excluded from this study. The catheter implantation procedure was successfully performed in 32 of the 37 mice.
Knockdown of TRPV2 in DRGs
In the normal mice experiment and bone cancer model experiment, i.t. siRNA administration was accomplished using a microinjection syringe (Hamilton Co.) connected to an i.t. catheter under brief general anesthesia (sevoflurane in oxygen). siRNA against TRPV2 or scrambled siRNA was administered manually over a 10-s period with a single injection volume of 10 µl (5 µg) followed by a flush of physiological saline (3 µl). After discontinuance of general anesthesia, mice fully recovered within 2 min. Injections were given daily for three consecutive days.
Western blot analysis
Mice with or without bone cancer were deeply anesthetized with sevoflurane. The DRGs were rapidly excised (L1–L5, n = 4, each group), immediately frozen in liquid nitrogen, and stored at −80°C until use. The segments were homogenized in SDS sample buffer that contained a protease inhibitor (Sigma-Aldrich) on ice. Homogenates were centrifuged at 5000 g for 5 min at 4°C. EDTA was added to the supernatant, and the supernatant was centrifuged at 5000 g for 1 h at 4°C. The pellet was resuspended in a lysis buffer containing Tris buffer. Proteins in the sample were separated, subjected to 4% to 15% SDS-PAGE (BIO-RAD Laboratories, Hercules, CA, USA), and electrophoretically transferred to a polyvinylidene difluoride membrane (Millipore Immobilon-p Transfer Membrane, Germany) using Power Pac High Current (BIO-RAD Laboratories). The membrane was blocked with a blocking solution containing 2% skim milk, 0.1% Tween 20, and Tris-buffered saline. After washing, the membrane was incubated with an anti-TRPV2 polyclonal antibody (1:2000 dilution; Trans Genic Inc.) and an anti-beta actin rabbit polyclonal antibody (1:5000 dilution; Sigma-Aldrich) overnight at 4°C. After washing again, the membrane was incubated with a secondary antibody, antirabbit IgG antibody, for detection of TRPV2 or beta-actin. Positive bands for TRPV2 or beta-actin were identified around 80 kDa and 40 kDa, respectively, by the ECL plus chemiluminescence detection system (Amersham Biosciences, Piscataway, NJ, USA) and normalized to beta-actin for a loading control.
Timeline of experimental protocols
The protocol for investigating effects of selective inhibition of TRPV2 protein on bone cancer-induced pain was as follows (Figure 1(a)). A catheter was inserted into the i.t. space of each mouse at least two days before sarcoma injection. A behavioral test was performed at baseline. Then, murine sarcoma cells were implanted into the left femur of each mouse. Mice were placed in the home cages. After establishment of the bone cancer model, behavioral tests were performed on days 13, 14, 15, 16, 17, 19, and 21 thereafter. On days 14, 15, and 16, siRNA against TRPV2 or scrambled siRNA was injected into the i.t. space via the implanted catheter. To minimize the possibility of selection bias, mice were randomly divided by computer-generated randomization into two treatment groups: siRNA against TRPV2 or scrambled siRNA. Assessments of pain-related behaviors were conducted by independent investigators who were blinded to the treatment received. In a separate group of mice, L1 to L5 DRGs were removed on days 0, 13, 17, and 19, and expression of TRPV2 was evaluated using Western blot analysis.

Timeline of experimental protocols for investigating effects of selective inhibition of TRPV2 protein on bone cancer pain (a) or tactile sensory thresholds and thermal nociception in normal mice (b). (a) Two days after insertion of an i.t. catheter, behavioral tests including tests for limb use, weight bearing, and spontaneous flinches were performed as baseline controls. Then, murine sarcoma cells were implanted in the left femurs of mice. Each behavioral test was conducted on days 13, 14, 15, 16, 17, 19, and 21 thereafter. In a separate group of mice, Western blot analysis for expression of TRPV2 protein in dorsal root ganglion neurons was performed at baseline and on days 13, 17, and 19. siRNA against TRPV2 or scrambled siRNA was injected into the i.t. space via the implanted catheter on days 14, 15, and 16 (total dose, 15 µg). (b) Two days after insertion of the i.t. catheter, behavioral tests including tests for tactile sensory thresholds and paw withdrawal latency to noxious heat stimuli were performed at baseline. siRNA against TRPV2 or scrambled siRNA was injected into the i.t. space via the implanted catheter for three consecutive days (total dose, 15 µg). Each behavioral test was performed at baseline and on days 2 and 4 from the last siRNA injection. In another separate group of mice, Western blot analysis for expression of TRPV2 protein in dorsal root ganglion neurons was performed at baseline and on days 2 and 4. siRNA: small interfering RNA; i.t.: intrathecal.
The protocol for investigating effects of selective inhibition of TRPV2 protein on tactile sensory thresholds and thermal nociception of normal mice without cancer was as follows (Figure 1(b)). A catheter was inserted into the i.t. space of each mouse. Two days later, behavioral tests including tests for tactile sensory thresholds and paw withdrawal latency to noxious heat stimuli were performed as baseline assessments. Mice were placed in the home cages. siRNA against TRPV2 or scrambled siRNA was injected into the i.t. space via the implanted catheter for three consecutive days (total dose, 15 µg). Each behavioral test was performed at baseline and on days 2 and 4 from the last siRNA injection. In another separate group of mice, the expression level of TRPV2 protein in DRG neurons was determined by using Western blot analysis at baseline and on days 2 and 4.
Statistical analysis
All statistical analyses were performed using GraphPad Prism software (GraphPad, San Diego, CA, USA) and BellCurve for Excel (Social Survey Research Information, Tokyo, Japan). Number of flinches, withdrawal latency to noxious heat, and relative expression ratio of TRPV2 to beta-actin were expressed as means ± standard deviations. Number of flinches and withdrawal latency were compared among the groups by two-way analysis of variance for repeated measures with the Tukey post hoc test. Withdrawal threshold to mechanical stimulation, the score of limb use and the score of weight bearing were expressed as medians with 5 and 95 percentiles. Data for withdrawal threshold were analyzed using the Friedman test for within-group analysis and the Kruskal–Wallis test for between-group comparisons followed by Dunnett’s post hoc test. The scores of limb use and weight bearing were compared to the basal value by using the Freidman test followed by Dunn’s test and were compared between the groups by using the Mann–Whitney U test. A P value less than 0.05 was considered statistically significant.
Results
TRPV1-ir and TRPV2-ir DRG neurons innervating bone marrow of the femur
FG-labeled neurons, indicating where FG was transported from the femur, were present in lumbar DRG neurons (Figure 2). TRPV1-ir expression was observed in 21% of all FG-labeled DRG neurons (119 of the 572 FG-labeled neurons), and TRPV2-ir was observed in 22% of all FG-labeled DRG neurons (118 out of 532 FG-labeled neurons). TRPV1 expression was restricted to small-diameter neurons innervating bone marrow of the femur (Figure 2(e)). In contrast, TRPV2 expression was distributed within all sizes of DRG neurons, but the majority of TRPV2 expression was found within medium-sized and large-sized DRG neurons (Figure 2(f)).
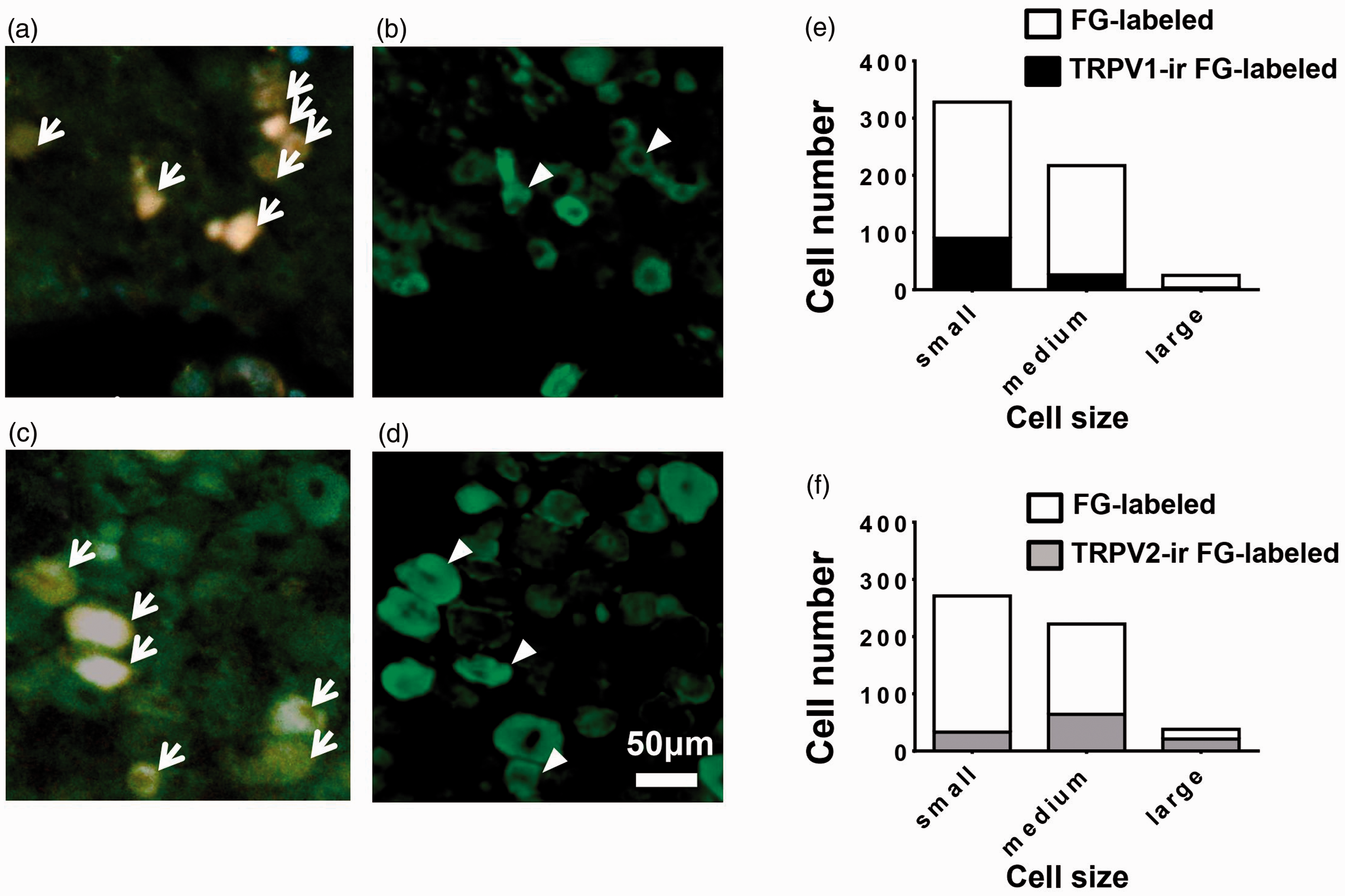
TRPV1 and TRPV2 expression in dorsal root ganglion (DRG) neurons innervating bone marrow of the femur. We injected the retrograde neurotracer FG into bone marrow of the left femurs of normal mice (5 µl of 2% FG solution). Forty-eight hours after the FG injection, lumbar DRGs were removed for TRPV1 and TRPV2 immunohistochemistry. FG-labeled DRG (a and c), TRPV1-ir (b), and TRPV2-ir DRG neurons (d) are also shown. The sections in (a) and (b) and those in (c) and (d) are the same. The section of TRPV1-ir (b) is different from that of TRPV2-ir (d). Arrows indicate FG-labeled DRG neurons (a and c), and arrowheads indicate double-labeled DRG neurons (b and d). Size distribution of TRPV1-ir FG-labeled DRG neurons (e). Size distribution of TRPV2-ir FG-labeled DRG neurons (f). Profiles of the DRG neurons were divided into small (<30 µm), medium (30–40 µm), and large (>40 µm) cells by measuring the diameters. FG-labeled DRG neurons, TRPV1-ir FG-labeled DRG neurons or TRPV2-ir FG-labeled DRG neurons were counted in each section. Then, the total numbers of neurons in the 24 sections derived from eight mice (L2–L4 DRGs) were estimated. TRPV1: transient receptor potential vanilloid subfamily 1; TRPV2: transient receptor potential vanilloid subfamily 2; ir: immunoreactive; FG: Fluoro-Gold.
Effects of i.t. siRNA against TRPV2 on bone cancer-related pain behaviors
At 13 days after sarcoma injection, the limb use score during spontaneous ambulation and the weight-bearing score during spontaneous standing were significantly decreased, and the number of spontaneous flinches was significantly increased compared with those at baseline (Figure 3(a) to (c), n = 8, in each group). I.t. administration of siRNA against TRPV2 for three days significantly improved the limb use and weight-bearing scores, and the effects were sustained for at least five days (P < 0.05, siRNA against TRPV2 vs. scrambled siRNA). On the other hand, i.t. administration of siRNA against TRPV2 did not change the number of flinches.

Effects of intrathecal administration of siRNA against TRPV2 (TRPV2-siRNA) and scr-siRNA on bone cancer pain-related behaviors. Murine sarcoma cells were injected into mouse femurs. From day 14, mice were treated with TRPV2-siRNA or scr-siRNA for three consecutive days (arrows). Scores of limb use during spontaneous ambulation (a) and weight bearing during spontaneous standing (b) in the TRPV2-siRNA group after siRNA administration were significantly higher than those in the scr-siRNA group. In contrast, there was no significant difference in the number of spontaneous flinches between the two groups (c). n = 8, in each group. *P < 0.05 versus scr-siRNA. BL: baseline; TRPV2: transient receptor potential vanilloid subfamily 2; siRNA: small interfering RNA; scr: scrambled control.
Changes in expression of TRPV2 during progression of bone cancer and effects of siRNA against TRPV2 on protein levels
We examined changes in TRPV2 protein levels in lumbar DRG neurons on the ipsilateral side to sarcoma injection and confirmed the effect of siRNA against TRPV2 used in this study. Western blot analysis showed that TRPV2 protein levels were not changed at day 13 after sarcoma implantation and that i.t. administration of siRNA against TRPV2 for three consecutive days significantly reduced TRPV2 protein levels in DRG neurons compared with the levels at baseline and day 13 (Figure 4) (n = 4, P < 0.05 vs. baseline, P < 0.05 vs. day 13).

Changes in TRPV2 protein expression in lumbar dorsal root ganglion neurons ipsilateral to the femur injected with sarcoma cells. Small interfering RNA (siRNA) against TRPV2 was injected into the intrathecal space on days 14, 15, and 16. (a) Representative results of Western blot analysis of TRPV2 (upper) and beta-actin (lower). (b) Quantitative results of TRPV2 protein expression. TRPV2 protein expression was normalized to beta-actin levels. TRPV2 protein expression did not change until day 13. TRPV2 protein expression was significantly downregulated by siRNA against TRPV2. Data are expressed as means ± standard deviations (n = 4). *P < 0.05 versus BL. #P < 0.05 versus day 13. BL: baseline; TRPV2: transient receptor potential vanilloid subfamily 2.
Effects of i.t. siRNA against TRPV2 on thermal sensitivity and tactile sensory thresholds in normal mice
We investigated the involvement of TRPV2 in thermal sensitivity and tactile sensory thresholds in normal mice without tumors by using siRNA against TRPV2. Neither siRNA against TRPV2 nor scrambled siRNA (n = 8, in each group), which were administered intrathecally for three consecutive days, affected the mechanical withdrawal threshold (Figure 5(a)) and withdrawal latency to thermal stimulation (Figure 5(b)) in the paws of normal mice. In both groups, limb use and weight-bearing activities were not impaired (data not shown). A reduction of TRPV2 protein expression was observed by Western blot analysis in DRG neurons in normal mice at two and four days after i.t. injection of siRNA against TRPV2 (Figure 6).
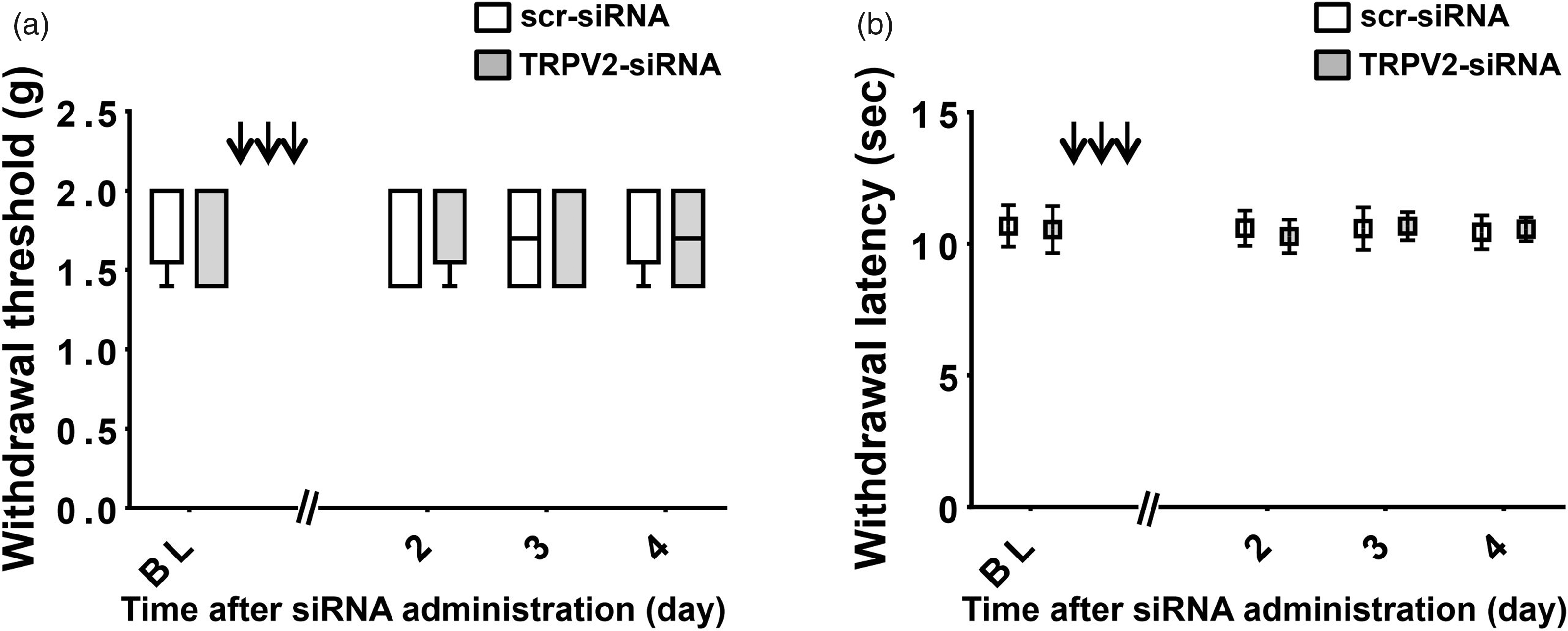
Effects of intrathecal administration of siRNA against TRPV2 (TRPV2-siRNA) and scr-siRNA on thermal sensitivity and tactile sensory thresholds of normal mice without cancer. TRPV2-siRNA or scr-siRNA was administered for three consecutive days (arrows). On the second, third, and fourth days after completion of siRNA administration, we investigated the thermal sensitivity and tactile sensory thresholds in normal mice. TRPV2-siRNA did not affect the mechanical withdrawal threshold (a) or withdrawal latency to thermal stimulation (b) in the paws of normal mice, as compared with those treated with scr-siRNA. n = 8, in each group. BL: baseline; TRPV2: transient receptor potential vanilloid subfamily 2; siRNA: small interfering RNA; scr: scrambled control.

Changes in TRPV2 protein expression in lumbar dorsal root ganglion neurons in normal mice. siRNA against TRPV2 was injected into the intrathecal space via the implanted catheter for three days. (a) Representative results of Western blot analysis of TRPV2 (upper) and beta-actin (lower). (b) Quantitative results of TRPV2 protein expression. TRPV2 protein expression was normalized to beta-actin levels. TRPV2 protein expression was decreased at two and four days after intrathecal injection of siRNA against TRPV2. Data are expressed as means ± standard deviations (n = 4). *P < 0.05 versus BL. BL: baseline; TRPV2: transient receptor potential vanilloid subfamily 2; siRNA: small interfering RNA.
Discussion
The major findings of this study were as follows: (1) One-fifth of neurons in DRGs innervating the femurs in mice expressed TRPV2 channels, (2) siRNA-mediated knockdown of TRPV2 did not affect the number of flinches but improved limb use and weight-bearing scores in mice with bone cancer pain, and (3) Western blot analysis showed that TRPV2 protein expression of DRG neurons was not increased when pain-related behaviors had developed at 13 days after sarcoma injection. These results suggest that TRPV2 is involved in movement-related pain but not ongoing pain in mice with progression of bone cancer.
TRPV2 is widely expressed in neuronal and nonneuronal cells.23–26 It is known that some of TRPV1-ir DRG neurons innervate the femur. 27 However, TRPV2 expression in bone has not been investigated. We first demonstrated the presence of TRPV2-ir FG-labeled DRG neurons innervating bone marrow of the femur. Twenty-one percent of DRG neurons innervating bone marrow of the femur expressed TRPV1, consistent with the results of a previous study showing that approximately 20% of retrograde-labeled DRG neurons expressed TRPV1 in rats. 28 Twenty-two percent of DRG neurons innervating bone marrow of the femur expressed TRPV2. Our study showed that TRPV2 expression was present in a considerable percentage of DRG neurons innervating bone marrow of the femur, being comparable with TRPV1. It was evident that TRPV2 was preferentially expressed in medium- to large-sized DRG neurons innervating the femur. In contrast, TRPV1 was exclusively expressed in small-sized DRG neurons labeled with FG. These results are consistent with results of previous studies for DRG neurons innervating the lower extremity.13,14
We used siRNA against TRPV2 to clarify the role of TRPV2 in bone cancer pain because of the absence of high-affinity selective activators and inhibitors of TRPV2. 29 I.t. injection of siRNA against TRPV2 for three consecutive days, the effectiveness of which was validated by Western blot analysis, did not affect withdrawal latency to radiant heat or tactile sensory thresholds in normal mice compared with scrambled siRNA, indicating that TRPV2 is not involved in thermal nociception and tactile sensory thresholds in the skin of the hindpaw in normal conditions without bone cancer. These results are in line with a previous study in which TRPV2 knockout mice showed normal behavioral responses to noxious heat and punctate mechanical stimuli in the basal state. 30 siRNA against TRPV2 significantly improved the limb use score of the tumor-bearing limb (Figure 3(a)) and did not affect the withdrawal thresholds of the hindpaw in response to thermal and mechanical stimuli (Figure 5(a) and (b)). These pain-related behaviors were assessed by evaluating movement of the hindpaw. Taken together, the results indicated that knockdown of TRPV2 does not have a significant impact on motor function of the hindpaw.
At 13 days after injection of sarcoma cells into the femur, the number of flinches as a measure of ongoing pain (Figure 3(c)) was significantly increased compared with that at baseline. I.t. injection of siRNA against TRPV2 for three days significantly decreased the TRPV2 protein level in DRG neurons (Figure 4) but did not affect the number of flinches, indicating that TRPV2 is not involved in ongoing bone cancer pain (Figure 3(c)). On the other hand, i.t. injection of siRNA against TRPV2 resulted in recovery of reduced limb use and weight-bearing activities as indicators of movement-related pain (Figure 3(a) and (b)). Taken together, our results suggest that TRPV2 has a crucial role in movement-related pain caused by bone cancer but not in ongoing pain.
Cancer-induced bone pain is a complex pain state and thus produces various pain-related behaviors. Pain-related behaviors in bone cancer pain models are classified as stimulus-evoked pain behaviors, ongoing pain behaviors, and movement-related pain behaviors. In tumor-bearing animals, there are significantly increased pain-related behaviors. 20 In the present study, siRNA against TRPV2 improved limb use and weight bearing but did not affect spontaneous flinching in a mouse model of bone cancer. The findings indicate important differences in mechanisms mediating components of bone cancer pain and are similar to results of previous studies showing that p38, a member of the mitogen-activated protein kinase family, was involved in specific components of cancer-induced bone pain.9,31,32 Components of cancer-induced bone pain that may reflect ongoing pain, movement-related pain, or stimulus-evoked pain are probably driven by independent mechanisms. Therefore, therapy targeting specific molecules may affect only limited pain-related behaviors. In the future, the mechanisms driving multiple components of bone cancer pain must be clarified to develop a more complete therapeutic treatment strategy.
Since sarcoma cells are restricted to the bone marrow in this bone cancer model of the mouse, the two types of pain, ongoing pain and movement-related pain, are likely to be caused by the activation of primary afferents within the bone marrow. We previously reported that selective inhibition of TRPV1-expressing afferents alleviates ongoing pain but not movement-related pain in a bone cancer model similar to that used in the current study. 8 In the current study, we showed that TRPV2 is involved in movement-related pain in a bone cancer model, although TRPV2 did not play a major role in thermal sensitivity and tactile sensory thresholds in the skin of the hindpaw in a normal state. At present, it is not clear how TRPV2 participates in movement-related pain caused by bone cancer. One possible explanation is that central sensitization caused by proliferation of sarcoma cells in the femur contributes to movement-related pain assessed by limb use and weight bearing. 18 TRPV2 is present in medium- to large-diameter Aδ- and Aβ-type primary sensory neurons.12,14,23,33 TRPV2 is expressed in mechanosensitive primary afferent neurons including both low-threshold and high-threshold mechanoreceptors. 13 There was a substantial number of TRPV2-expressing primary afferents innervating bone tissues as shown Figure 2. Therefore, it is possible that innocuous stimulation to the bone such as stimulation by limb use and weight bearing recruits electrophysiological activity in nociceptive neurons in the dorsal horn under central sensitization through TRPV2-expressing primary afferents innervating the femur.
It is not clear from the results of the current study which primary afferents convey nociceptive information for movement-related pain. Our laboratory has shown that mechanical stimulation to bone marrow produces nociception, concomitantly producing its referred pain in the corresponding skin fields. 7 siRNA against TRPV2 administered intrathecally probably mediated TRPV2 in DRG neurons innervating not only bone tissues but also other tissues including the skin and muscles. Hence, we cannot exclude the possibility that movement-related pain observed in this study might be in part due to bone cancer-induced referred pain or referred allodynia of the plantar skin or muscles on the ipsilateral hindpaw.
Another possibility is that peripheral sensitization contributes to movement-related pain in bone cancer. Bone cancer causes inflammation that activates peripheral bone marrow nociceptors through the release of inflammatory mediators, 34 which may cause a decreased threshold and increased responsiveness of nociceptors in bone marrow. TRPV2 is activated by local insulin-like growth factor I produced by activated osteoblasts in bone cancer progression.35,36 Bone cancer-induced upregulation of TRPV2 protein in lumbar DRG neurons was not observed at day 13 by our Western blot analysis (Figure 4). However, TRPV2 may be activated in a pathological state such as bone cancer-induced inflammation. Bone cancer pain is a complex pain state in which peripheral sensitization and central sensitization of the nervous system are present. Further studies are required to clarify the detailed mechanisms of bone cancer pain.
One of the weaknesses of this study is that effect of siRNA against TRPV2 on sarcoma progression in bone marrow was not investigated. It is known that loss or gain of the TRPV2 gene is associated with cancer growth and progression. The cancerogenic effects of TRPV2 are cell type-dependent. 37 We do not know how TRPV2 affects proliferation of sarcoma cells used in this study. The targeting of the TRPV2 channel was through siRNA delivered spinally and therefore likely did not impact tumor or other bone marrow cells. There was no significant difference between the number of spontaneous flinches, as a measure of ongoing pain, with and without downregulation of TRPV2. Therefore, it is unlikely that downregulation of TRPV2 by siRNA against TRPV2 suppressed sarcoma cell growth, resulting in alleviation of movement-related behaviors.
In conclusion, siRNA-mediated knockdown of TRPV2 did not affect the number of spontaneous flinches but improved limb use and weight bearing in mice with bone cancer. Our results suggest that TRPV2 is involved in movement-related pain but not ongoing pain in mice with progression of sarcoma in the femur. The development of therapeutic strategies for movement-related pain that are distinct from those for ongoing pain is required.
Footnotes
Author Contributions
KY and ST conducted experiments and data analysis and wrote the draft of the manuscript. KY and HZ performed behavioral assays. ST, TK, and MK oversaw experimentation and experimental design and edited the manuscript. ST, SF, and TI helped behavioral assays and edited the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Japan Society for the Promotion of Science Grand-in-Aid for Scientific Research (Grant Numbers 23791695 (KY) and 23390374 (TK)).
