Abstract
Effective prevention and treatment options for bone cancer-related pain (BCP) are lacking. In recent years, numerous studies have investigated the association between m6A epigenetic modifications and pain, revealing their significant role in pain initiation and maintenance. This study aimed to provide theoretical support for the treatment of BCP and to identify target drugs for future development. Specifically, we investigated the involvement of fat mass and obesity-related protein (FTO) in rat models of BCP by administering varying doses (1/5/10 mg/kg) of the FTO inhibitor meclofenamic acid (MA) and assessing changes in mechanical sensitivity through domain analysis, gait analysis, and open-field experiments. After successfully establishing the BCP model, we verified it by performing mechanical sensitivity assessments. We observed significantly increased expression levels of the demethylase FTO within the spinal dorsal horn accompanied by decreased m6A methylation levels in the model. Compared with untreated BCP rats, remarkably improved behavioral responses indicative of reduced pain were observed in the model rats after administration of 10 mg/kg MA, concomitant with decreased expression levels of FTO and increased m6A methylation levels. Compared with untreated BCP rats, the expression levels of p-ERK and pro-inflammatory cytokines were also significantly decreased after MA administration. Taken together, FTO can downregulate m6A methylation level and activate ERK/inflammatory cytokines signaling pathway to maintain BCP in rats.
Keywords
Introduction
Bone cancer pain (BCP) refers to the pain caused by primary or metastatic bone tumors, with breast cancer, prostate cancer, and lung cancer being the most common causes of metastasis. Pain induced by metastatic tumors is the most prevalent form of BCP. 1 When cancer cells spread to the skeletal system, they cause bone destruction, acidosis, ischemia and other symptoms that result in persistent severe pain. This type of pain is typically dull and worsens as the disease progresses. 2 Clinical studies have shown that approximately 80% of patients with advanced cancer in United States experience severe pain, particularly those caused by metastatic bone cancers. These symptoms significantly reduce patients’ quality of life and survival rates.3,4 To date, there are no highly effective prevention or treatment methods for BCP. Therefore exploring its pathogenesis and treatment options is crucial.
In recent years, a growing body of research has focused on the relationship between epigenetic post-transcriptional m6A modification of mRNA and pain. Numerous studies have demonstrated its crucial role in regulating the occurrence and maintenance of pain. 5 Additionally, m6A modification has been found to be significant for various neurophysiological processes, including neuronal axon guidance 6 and, learning and memory, 7 as well as central nervous system-related diseases. 8 The involvement of m6A methylation in post-transcriptional RNA modification has been confirmed in relation to pain occurrence and maintenance. 9 In a mouse model of CFA-induced inflammatory pain, the RNA methyltransferase METTL3 was shown to influence pain sensitivity by regulating the promoter region of m6A-dependent microRNA-365-3p. 10 However, the regulatory function of m6A methylation in the occurrence of other forms of pain, particularly BCP, remains unclear.
Notably, m6A methylation is a reversible biological process regulated by both “writer protein” methyltransferases (e.g., METTL3 11 ) and “eraser protein” demethylases (e.g., fat mass and obesity-related protein, 12 FTO). FTO is an α-ketoglutaric acid-dependent dioxygenase responsible for removing m6A methylation modifications from mRNA. FTO plays a scavenging role in reversible m6A methylation both in vitro and in vivo. 13 Based on our previous animal experiments conducted on rat BCP models, we observed a significant down-regulation of m6A methylation levels within spinal cord tissue. Subsequently, we detected no significant changes in expression levels of “write proteins” (METTL3/METTL14/WTAP) or “eraser protein” ALKBH5; however, there was a notable up-regulation in FTO protein levels.
Currently, despite the significant analgesic effects of certain drugs such as morphine in alleviating chronic pain, their routine clinical application is limited because of a range of side effects including addiction. 14 Consequently, effective treatments for pain, particularly inflammatory and neuropathic pain associated with bone cancer, are lacking. Meclofenamic acid (MA), a selective inhibitor targeting FTO, has demonstrated considerable potential to reduce FTO expression levels and promote the up-regulation of m6A methylation. Clinically approved as a non-steroidal anti-inflammatory drug, 15 MA holds promise as a potential treatment for BCP. Our study aims to provide a theoretical foundation for identifying target drugs for future development in this field.
Materials and methods
Materials
MA was purchased from Shanghai Selleck Chemicals (Shanghai, China). MADB106 breast cancer cells were purchased from Wuhan Procell Life Technology Co., Ltd. The primary antibodies used in this study recognized: FTO (abcam, ab92821), ALKBH5 (ProteinTech, 16,837-1-AP), WTAP (ProteinTech, 60,188-1-Ig), METTL3 (abcam, ab195352), METTL14 (LSBio, LS-C107729-100), YTHDF2 (ProteinTech, 24,744-1-AP), and GAPDH (abcam, ab8245). Horseradish peroxidase (HRP)-conjugated secondary antibodies were purchased from Beyotime (Shanghai, China). Fetal bovine serum was purchased from Gibco (St Louis, United States). Dulbecco’s modified Eagle’s medium (DMEM) was purchased from Corning (Manassas, United States). All chemicals and reagents used were of standard biochemical quality.
Animals
Female Sprague-Dawley (SD) rats weighing 180-200 g, obtained from Charles River Laboratories (Zhejiang Pinghu), were utilized for this study. The animal experimental protocol was approved by the Animal Ethics Committee of the Medical College of Jiaxing University (approval no.: JUMC 2021-163). The animals were individually housed in ventilated cages in a room with a controlled temperature of 22 ± 2°C and 12 h light-dark cycle, allowed ad libitum access to food and water. All experimental procedures strictly adhered to the ethical guidelines outlined by the International Association for the Study of Pain (IASP) regarding animal usage in pain research, with utmost consideration given to implementing the principles of replacement, reduction, and refinement (3 Rs).
Bone cancer pain model
The BCP rat model was established according to our previous method. 16 A liquots of MADB106 breast cancer cells stored in liquid nitrogen were thawed at 37°C. Subsequently, 0.5 mL (2 × 107) of MADB106 cells were implanted into the peritoneal cavity of SD rats, resulting in ascites formation after 3-4 days. The ascites was then extracted and washed three times with PBS for subsequent cell counting. To induce bone metastasis, SD rats were anesthetized with pentobarbital sodium (50 mg/kg), and a longitudinal incision was made on the lower third of the left tibia. Using a micro syringe, 10 μL of MADB106 breast cancer cells (1 × 106) were slowly injected into the bone marrow cavity from the metaphyseal end towards the bone itself. In the control group, inactive cancer cells were injected and removed after 1 minute; subsequently, medical glue was immediately applied to seal the pinhole.
Mechanical pain tests
The paw withdraw threshold (PWT) was measured using von Frey electronic equipment (Zhongshi, China)on days 1, 3, 7, and 14 post-modeling to assess pain sensitivity in the left hind limbs of SD rats. This measurement was conducted by individuals not involved in this study. Rats were placed inside a plexiglass box (20 cm × 20 cm×25 cm) with a bottom surface area for adaptation purposes within a quiet environment for 30 minutes prior to testing initiation. Using von Frey electronic equipment , painful mechanical stimuli were delivered to middle part of the plantar region of the left hindlimb. The stimulation intensity was gradually increased until the foot withdrawal reaction occurred. Observations of the foot withdrawal behavior were made while recording the corresponding readings as von Frey pain thresholds. Intervals of approximately 5 minutes separated each measurement session.
Computed tomography (CT) imaging
14 days after MADB106 cell injection, rats were deeply anesthetized by intraperitoneal injection of pentobarbital sodium (100 mg/kg), and CT imaging was performed the left lower limb. Three-dimensional (3D) reconstruction of the rat tibiae using CT was performed to observe bone destruction. The CT scanning parameters were set based on previous studies as follow 17 : spiral scanning, tube voltage of 120 kVp, layer thickness and spacing both at 1 mm, and core: U30u medium smooth. High-resolution CT images (visual field of 100 mm) were acquired for analysis using Siemens PACS system software.
Hematoxylin and eosin staining
14 days after MADB106 cell injection, hematoxylin and eosin (HE) staining was performed on the left tibiae of rats. After deeply anesthetizing the rats with an intraperitoneal injection of pentobarbital sodium (100 mg/kg), the tibiae were collected and fixed in 4% paraformaldehyde solution followed by decalcified, paraffin embedded, and sectioned for HE staining. Tumor growth and bone destruction were imaged using an optical microscope (Olympus) and analyzed.
Gait analysis
The CatWalk gait analysis system, which has been validated as an effective approach for quantifying pain-related behaviors, was used to assess the gait of the rats. Fluorescent light was emitted from a fluorescent tube placed under glass plate. Upon contact with the glass plate, the rat footprints were reflected downwards and captured by a high-speed camera positioned below, enabling the collection of footprint parameters throughout the entire process. Prior to data acquisition, training sessions were conducted to establish uniform and uninterrupted passage through the channel as a standard protocol. The CatWalk system commonly employs footprint contact area measurement, 18 thus, the maximum contact area was employed to evaluate relevant behaviors before and after bone cancer-induced pain. To minimize the influence of confounding factors, the left hindlimb/right hindlimb ratio was calculated with data presented as percentages.
Open-field tests
In a quiet environment, the rats were placed in the central area of an open-field test box (50 cm × 50 cm×50 cm). Video recording was automatically initiated to capture the trajectory of the rat’s autonomous activities of the arts for 15 min, and the time spent in the central area during the last 10 min was logged. Before observing each animal, the experimental box was thoroughly cleaned using 75% ethanol.
Western blotting
After deeply anesthetizing the rats by intraperitoneal administration of pentobarbital sodium (100 mg/kg) for deep anesthesia, the lumbar segments of the spinal cord (L3-L5) were promptly dissected and placed on ice. The proteins were subsequently extracted for SDS-PAGE gel electrophoresis, with 40 μg protein sample added to each well. Wet transfer to nitrocellulose e membranes was performed, followed by blocking with a protein-free rapid blocking solution (Epizyme Shanghai China) at room temperature for 1 h. The membranes were incubated with primary antibodies overnight at 4°C, washed, and incubated with secondary antibodies at room temperature for 2 h. Enhanced chemiluminescence (ECL) reagent was added to visualize the proteins liquid. AlphaEaseFC software was used to analyze the images and obtain the net optical density of the target bands. The relative expression levels of FTO and other proteins were determined by calculating the ratio of their gray values to those of the corresponding GAPDH bands.
Dot blot
The isolated total RNA was denatured at 65°C for 5 min and subsequently combined with sodium citrate (SSC) buffer. Subsequently, the RNA samples were transferred to the Amersham Hybond-N+ membrane (Solarbio, PRN303 B). The membrane was dried at 60°C for 30 min, then stained with 0.05% methylene blue (sigma, 319,112) and scanned as a loading control. Alternatively, the UV crosslinked membrane was blocked with a protein-free rapid blocking solution (Epizyme Shanghai China) at room temperature for 1 h and then treated with primary antibody against m6a (abcam, ab284130) at 4°C overnight. Finally, the membranes were incubated with secondary antibody at room temperature for 2 h before development using ECL reagent. 19
Quantitative PCR analysis
The dorsal horn of the rat spinal cord was dissected in the same manner as the Western blot protocol, and total RNA was extracted using the TRIzol. Subsequently, 1000 ng of RNA was reverse transcribed into cDNA using the revertAid cDNA synthesis kit (Thermo Scientific, USA) after concentration determination. Quantitative PCR (qPCR) was conducted using PowerUp SYBR Green Master Mix (Applied Biosystems) in a real-time fluorescent qPCR thermocycler. Primer design and synthesis were performed by Shanghai Sangon Biotech. The specific primer sequences used were as follows: FTO, forward 5′-GCTGTGGAAGAAGATGGAGAGTGTG-3′, reverse 5′-ATCAGGACGGCAGACAGAATTTCAC-3′; ALKBH5, forward 5′-GGCGGTCATCATTCTCAGGAAGAC-3′, reverse 5′-CATAGCTGGGTGGCAATGTGGAG-3′; WTAP 5′-GAGAACATCCTTGTCATGCGGCTAG-3′; METTL3 forward 5′-CGTAACCTATGCTGACCACTCCAAG-3′, reverse: 5′- TGTGATAGTCCCTGCTGCCTCTC-3′; METTL14 forward 5′-TTCGGGAGGGACAGCACTATCAG-3′, reverse 5′-GCCTCTCAATTTCCTCGGTACATCC-3′; YTHDF2 forward 5′-TTTGGGTGAAGCTGCTTGGTCTAC-3′, reverse 5′-GGCTCTCCGTTGCTCAGTTGTC-3′; GAPDH forward 5′-AGTTCAACGGCACAGTCAAGGC-3′, reverse 5′-CGACATACTCAGCACCAGCATCAC-3′. The 2-∆ ∆ Ct method was used to calculate relative expression levels normalized to GAPDH.
Immunofluorescence staining
On the 14th day after BCP modeling, the rats were deeply anesthetized with pentobarbital sodium (50 mg/kg). A vein needle was inserted through the apex into the left ventricle, followed by perfusion with 500 mL of PBS and fixation with 4% paraformaldehyde (250 mL). The spinal cord was then carefully removed and postfixed in 4% paraformaldehyde for 6 h before undergoing gradient dehydration in increasing concentrations of sucrose (15% and 30%) over a period of 48 h. After removing excess moisture, OCT embedding medium (Sakura, Japan) was applied and the samples were sectioned into 16 μm thick slices, which were placed on slides and stored at −80°C for future use. These sections were washed with PBS, blocked with 5% BSA at room temperature for 1 hour, incubated overnight at 4°C in primary antibodies recognizing FTO (abcam, ab92821), NeuN (ProteinTech, CL594-26,975), GFAP (abcam, ab201732), Iba-1 (ProteinTech, CL594-10,904), followed by a thirty-minute incubation at room temperature on the next day. The sections were washes with PBS at room temperature with the corresponding secondary antibodies (Abcam; Alexa Fluor®488 ab150073; Alexa Fluor®594 ab150080) for 60 minutes followed by another round of PBS washing, and then sealed with Vectashield-DAPI mounting medium. Images were collected using a fluorescence confocal microscope (Zeiss, Germany).
ELISA analysis
Samples of spinal cord were collected on the 14th day after BCP modeling, following the same timeline as the Western blotting steps. The lysates were used for protein extraction and subsequently diluted 10 folds. Tumor necrosis factor (TNF)-α and interleukin (IL)-1β levels in the samples were determined as described previously, 20 and an enzyme-linked immunosorbent assay (ELISA; abcam) was performed according to the manufacturer’s instructions. Samples were added, enriched, and incubated; followed by plate washing, addition of an antibody, another round of plate washing, substrate staining, and reaction termination, and the sample OD value at 450 nm was measured plate reader. A standard curve was fitted to calculate the sample concentration.
Statistical analysis
All measurement data were analyzed using GraphPad software and expressed as mean ± SEM Behavioral results underwent two-way repeated measures ANOVA analysis, and qPCR and western blot results underwent t test or one-way ANOVA analysis. Statistical significance was set at p < .05.
Results
Establishment of BCP model
After inducing BCP in the rats, we continuously monitored changes in the mechanical pain threshold using the von Frey method to confirm the success of our model. The results demonstrated a significant decrease in the mechanical pain threshold in the bone cancer pain (BCP) group compared to that in the control (Con) group, indicating pronounced hypersensitivity to pain and the successful establishment of our rat model of BCP (Figure 1(a)). CT images obtained 14 days post modeling revealed severe tibial damage in the BCP group, whereas an intact bone structure was observed in the Con group (Figure 1(b)). Establishment of the bone cancer pain (BCP) model. (a) The paw withdraw threshold (PWT) of rats was reduced in a time-dependent manner for 14 days after MADB106 cell injection. Results are shown as mean ± SEM. (n = 10). **p < 0.01 vs. Con group. (b) Computed tomography (CT) images showing severe bone destruction in the tibia of BCP group (white arrow indicates the location where the cells were injected) compared to Con group at 14 days after injection. (c) HE staining showing extensive damage to trabecular tissue structure and presence of dark cancer cell particles within tibial sections from rats in the BCP group, whereas intact bone structure was observed in rats from the Con group. Scale bar = 50 μm. (d, e and f) Gait analysis using a specialized system indicated a significantly reduced print area of footprints and abnormal step order in rats from the BCP group compared to controls. RH is Right Hind, RF is Right Front, LH is Left Hind and LF is Left Front. Results are shown as mean ± SEM. (n = 10). *p < 0.05 vs. Con group.
To further validate our rat model of BCP, we performed HE staining of paraffin sections of tibia samples from both the Con and BCP groups after 14 days of modeling. The results showed extensive damage to the trabecular tissue structure and the presence of dark cancer cell particles within the tibial sections from rats in the BCP group, whereas an intact bone structure was observed in rats from the Con group (Figure 1(c)). Gait analysis using a specialized system indicated a significantly reduced print area of footprints and an abnormal step order in rats from the BCP group compared to controls (Figure 1(d), (e), (f)). Collectively, these findings provide strong evidence supporting the successful establishment of a rat model for studying BCP.
The expression of m6A demethylase FTO was significantly upregulated in the BCP model
We observed a significant decrease in m6A methylation levels in the SDH tissue (Figure 2(a) to (c)) of the BCP model rats. Interestingly, there were no significant alterations in the protein and mRNA expression levels of m6A methylation “writing proteins” (METTL3/METTL14/WTAP) ,”erasing protein” (ALKBH5) and “reader protein” (YTHDF2). However, the expression of the demethylase FTO was significantly increased (Figure 2(d), (e) and (f)), but not METTL3, METTL14, WTAP, ALKBH5 and YTHDF2, suggesting that FTO plays a major regulatory role in m6A methylation in the rat model of BCP. The expression of m6A demethylase FTO was significantly upregulated in the BCP model. Spinal cord tissues were harvested at day 14 after MADB106 cell injection. (a) Dot blot analysis was performed to assess the level of total RNA m6A in spinal cord. (b) The amount of RNA detected by methylene blue staining was equal between groups. (c) The level of m6A in the BCP group was significantly lower than that in the Con group. (d, e and f) Western blotting and qPCR detection of m6A related regulatory proteins showed that only FTO expression was significantly upregulated compared with the Con group, but not METTL3, METTL14, WTAP, ALKBH5 and YTHDF2. The results are exhibited as mean ± SEM. (n = 5). *p < 0.05 vs. Con group.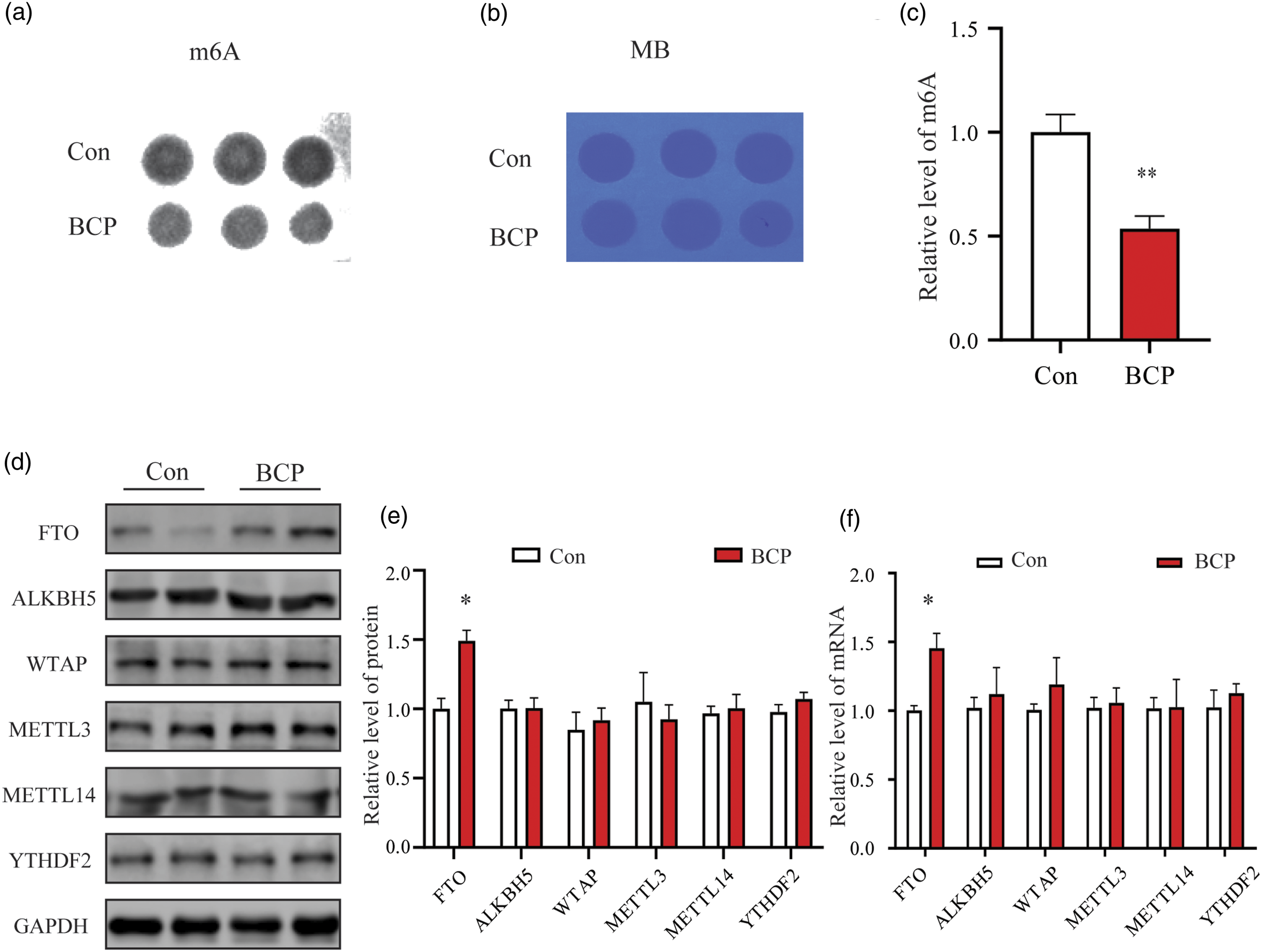
FTO expression increased over time in BCP and localized to neurons
We further examined the expression level of FTO in the spinal cord tissue of BCP rats at different time points and found that it progressively increased with time. Western blotting and qPCR results showed a significant increase in FTO expression at 7 and 14 days after BCP modeling (Figure 3(a) to (c)). Immunofluorescence staining of whole spinal cord slices revealed higher FTO fluorescence intensity on the ipsilateral compared to the contralateral side (Figure 3(d)). The number of FTO-positive cells in the ipsilateral L5 spinal cord on day 14 after BCP increased by 1.39 fold compared with the corresponding Con rats (Figure 3(e)). To determine which type of nerve cells primarily expressed FTO, we performed co-immunofluorescence labeling experiments for NeuN (neuron marker), GFAP (astrocyte marker), and Iba-1 (microglia marker). We observed significant colocalization between FTO and NeuN, but no obvious colocalization with GFAP/Iba-1 (Figure 3(f) and (g)). FTO expression increased over time in BCP and was localized to neurons. (a to c) Results of western blotting and qPCR showing a significant increase in FTO expression at 7 and 14 days after BCP modeling. The results are exhibited as mean ± SEM. (n = 6). *p < 0.05, **p < 0.01 vs. Con group. (d) Immunofluorescence staining of FTO (green) showing that the fluorescence intensity of the ipsilateral dorsal horn is greater than that of the contralateral side. Scale bar = 200 μm. (e) The number of FTO-positive cells in the ipsilateral L5 spinal cord on day 14 after BCP increased by 1.39 fold compared with the corresponding Con rats. The results are exhibited as mean ± SEM. (n = 3). **p < 0.01 vs. Con group. Scale bar = 50 μm. (f and g) Immunodouble staining showing that FTO is co-localized with NeuN (90%), but not with Iba-1 and GFAP. NeuN (neuron marker), GFAP (astrocyte marker), Iba-1 (microglia marker). Scale bar = 50 μm.
Meclofenamic acid abolished pain-related behaviors in the BCP rat model
Von Frey detection showed that the mechanical pain threshold of rats in the BCP + MA group was significantly increased in a dose-dependent manner compared with that in the BCP + NS group after the intraperitoneal injection of MA, while there was no significant difference between the Con + MA group and the Con + NS group (Figure 4(a)). Additionally, an open-field test (OFT) demonstrated that MA administration for 7 days at a dose of 10 mg/kg significantly increased the activity time of rats in the central area (Figure 4(b) and (c)), and gait analysis revealed a significant increase in the print area of the footprints after MA administration for 7 days at a dose of 10 mg/kg (Figure 4(d), (e) and (f)). OFT and gait analysis showed that there was no significant difference in behavioral changes between Con + MA group and Con + NS group. These behavioral experiments suggest that FTO plays an important role in BCP and that MA administration can effectively alleviate the related symptoms. Meclofenamic acid abolished pain-related behaviors in the BCP rat model. (a) The Von Frey detection method showed that the mechanical pain threshold of BCP rats increased significantly in a dose-dependent manner after intraperitoneal injection of MA. (b and c) The open-field test demonstrated that MA administration for 7 days at a dose of 10 mg/kg significantly increased the activity time of rats in the central area. (d) Print area. (e) Timing view. (f) CatWalk analysis revealed a significant increase in ratio of left/right of print area after MA administration for 7 days at a dose of 10 mg/kg. RH is Right Hind, RF is Right Front, LH is Left Hind and LF is Left Front. Results are shown as mean ± SEM. (n = 10). *p < 0.05, **p < 0.01, ***p < 0.001 vs. Con + NS group. #p < 0.05, ##p < 0.01 vs. BCP + NS group.
Treatment with MA inhibited FTO expression and rescued changes in m6A modification levels of rats with BCP
In our previous behavioral experiment, we found that administration of MA at a dose of 10 mg/kg resulted in the most significant improvement in pain behavior in rats. Therefore, we examined FTO expression levels and m6A modifications in the spinal cord tissues of rats treated with this dose. Our results indicated that compared to the BCP + NS group, those administered MA (BCP + MA group) had significantly lower levels of FTO expression (Figure 5(a) and (b)) and exhibited restored m6A modification levels within their spinal cords (Figure 5(c) and (d)). WB and DB detection showed that there was no significant difference in FTO and m6A modification levels between Con + MA group and Con + NS group. MA inhibited FTO expression and rescued m6A modification levels of rats with BCP. (a and b) Western blot results showing a significant decrease in FTO expression at 14 days after administration of MA (10 mg/kg). (c and d) After MA administration, the level of m6A in spinal cord tissue was significantly restored. Results are shown as mean ± SEM. (n = 6). **p < .01 vs. Con + NS group. #p < 0.05 vs. BCP + NS group.
MA alleviated pain by inhibiting ERK and proinflammatory cytokines
Previous studies have demonstrated that activation of the ERK signaling pathway contributes to BCP by influencing the downstream expression of proinflammatory cytokines. We observed a significant increase in p-ERK expression in the BCP + NS group compared to the Con + NS group, which was significantly reduced following MA (10 mg/kg) administration (Figure 6(a) and (b)). We detected the levels of proinflammatory cytokines IL-1β and TNF-α by ELISA and found that the expression of related factors was significantly decreased after MA administration (Figure 6(c), (d)). While there was no significant difference in p-ERK expression and proinflammatory cytokines IL-1β and TNF-α between the Con + MA group and the Con + NS group. These findings indicate that MA can ameliorate BCP in rats by suppressing both the ERK signaling pathway and the inflammatory response. MA inhibited ERK and proinflammatory cytokines. (a and b) Western blot results showing a significant increase in p-ERK expression in the BCP group compared to the control group, which was significantly reduced following administration of MA (10 mg/kg). (c and d) Levels of related proinflammatory cytokines IL-1β and TNF-α of spinal cord exhibited a similar trend. Results are shown as mean ± SEM. (n = 6). *p < 0.05, **p < 0.01 vs. Con + NS group. #p < 0.05 vs. BCP + NS group.
Discussion
In this study, we found a significant decrease in m6A methylation levels in BCP rats, along with the upregulation of protein expression of the demethylase FTO. This suggests that FTO plays a crucial regulatory role in modulating m6A methylation in the rat BCP model. In behavioral experiments related to pain perception, selective inhibition of FTO using MA significantly improved pain-related behaviors in rats. It has been established that activation of the ERK signaling pathway occurs during BCP development and subsequently affects downstream expression of related proinflammatory cytokines. Consequently, our investigation revealed a substantial reduction in both the ERK signaling pathway activity and proinflammatory cytokine levels following MA administration. Collectively, these findings suggest that MA exerts analgesic effects on BCP through the inhibition of FTO expression as well as the suppression of both the ERK signaling pathway and associated inflammation.
Current research indicates that BCP exhibits unique characteristics involving a combination of inflammatory and neuropathic pain.21,22 Traditionally, inflammation at the tumor site and the release of products from cancer and immune cells have been considered the primary contributors to pain. Previous studies have identified significant activity of astrocytes and microglia in the spinal cord during BCP.23,24 These astrocytes and microglia, derived from immune system macrophages, can release various pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6, leading to pain sensitization and hypersensitivity.25–27 To sum up, in the case of bone cancer, tumors and stromal cells within the bone marrow release various chemical signals, including inflammatory factors, colony-stimulating factors, chemokines, and hydrogen ions, which induce inflammatory pain. 28 At the same time, the growth of tumors in the bone marrow cavity will also lead to the destruction of bone, and will compress the nerve endings in the bone marrow cavity, resulting in neuropathic pain.
In recent years, research on the mechanism of BCP has focused on epigenetic modifications, with particular emphasis on m6A methylation in post-transcriptional RNA modification, which has been implicated in pain initiation and maintenance. 9 In a mouse model of CFA-induced inflammatory pain, METTL3 regulated pain sensitivity by modulating the promoter region of the m6A-dependent MicroRNA-365-3p. 10 M6A methylation is a reversible biological process that is regulated by methyltransferases along with demethylases. Our animal experiments demonstrated a significant decrease in m6A methylation levels in spinal cord tissue of a rat model of BCP, along with a substantial increase in FTO protein expression. However, no significant alterations were observed in the expression levels of “writer proteins” (METTL3/METTL14/WTAP) and “eraser proteins” (ALKBH5).
ERK, a member of the MAPK-mediated signaling pathways, transmits signals through a highly conserved cascade kinase complex. Upon phosphorylation by upstream kinases, ERK undergoes a transformation from its non-phosphorylated form and acts as a kinase to phosphorylate downstream proteins, translocates from the cytoplasm into the nucleus, regulates various transcription factors, and ultimately activates downstream biological functions.29,30 In this study, we observed significant activation of the ERK signaling pathway in spinal cord tissues associated with BCP. Importantly, this elevation was effectively reversed by MA treatment, leading to improved responses to pain. Collectively, these findings provide compelling evidence of the involvement of ERK in the regulation of BCP. m6A methylation has the potential to affect various biological processes, including cellular stress response. 31 ERK signaling is a crucial intracellular kinase signaling pathway. Following MA administration, there is an increase in the m6a methylation modification of downstream target genes regulated by FTO, resulting in reduced gene expression levels and potentially contributing to inflammation reduction. Therefore, in future studies, we will investigate the downstream target genes of FTO and their roles in pain using transcriptome sequencing and other methodologies.
Meclofenamic acid, a specific inhibitor of FTO, has been clinically approved as a non-steroidal anti-inflammatory drug 15 because of its ability to significantly reduce FTO expression levels and promote the upregulation of m6A methylation. As a specific inhibitor of FTO, MA can significantly relieve pain and injury by inhibiting FTO in animal models of hemorrhagic thalamic pain. 32 Previous reports have indicated significant upregulation of Kv1.2 protein in the dorsal root ganglia (DRG) of rats with pathological pain after MA administration. 33 We found that FTO expression was significantly increased in the BCP rat model, and hyperalgesia was significantly relieved after MA administration. According to previous reports, continuous folic acid treatment significantly improved BCP and reduced the expression of P2X2 and P2X3 receptors in rat DRG and SDH. 34 As a methylation donor, folic acid effectively increased the methylation levels of P2X2 and P2X3 receptor genes, thereby inhibiting their protein upregulation and exerting analgesic effects. This study revealed that the inhibition of FTO expression elevated m6a methylation levels in rat spinal cord tissue, further contributing to analgesia. Collectively, these findings highlight the crucial role of m6A methylation in BCP.
Although certain analgesic drugs such as morphine can effectively alleviate chronic pain, their routine clinical application is limited due to various side effects, including addiction. 35 Consequently, there remains an unmet need for effective treatments targeting inflammatory and neuropathic pain associated with bone cancer. In recent years, the identification and exploration of novel therapeutic targets, along with the development of new analgesic drugs, has become a prominent research area worldwide. As an FTO inhibitor, MA is a potential drug candidate for the treatment of BCP. Our study provides valuable theoretical evidence and identifies a target drug for the future development of novel therapeutics for bone cancer-induced pain.
Conclusion
In conclusion, the m6A demethylase FTO is significantly upregulated in a rat model of BCP, thereby activating downstream ERK and proinflammatory cytokine-related signaling pathways, which contributed to the initiation and progression of BCP. Administration of a specific dose of the FTO-selective inhibitor MA inhibited ERK and pro-inflammatory cytokine-related signaling pathways, leading to an improvement in pain behavior in rats.
Supplemental Material
Supplemental Material - Fat mass and obesity-related protein contributes to the development and maintenance of bone cancer pain in rats by abrogating m6A methylation of RNA
Supplemental Material for Fat mass and obesity-related protein contributes to the development and maintenance of bone cancer pain in rats by abrogating m6A methylation of RNA by Beibei Liu, Danyang Meng, Man Luo, Longsheng Xu and Ming Yao in Molecular Pain
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Health Science and Technology Program of Zhejiang Province (2022RC267); Science and Technology Project of Jiaxing City (2021AD30137); Zhejiang Provincial Clinical Key Specialties-Anesthesiology (2023-ZJZK-001); Key Discipline of Endocrinology in Jiaxing City (2023-FC-003).
Data availability statement
The raw data supporting the conclusions of this manuscript will be made available by the authors, without undue reservation, to any qualified researcher.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
