Abstract
Objective
Subsequent limb amputation (SLA) may be necessary due to disease progression, infection, or to aid prosthesis fit. SLA in Saskatchewan has increased 3.2% from 2006 to 2019 with minor SLA increasing 9.6% during that period. Diabetes affects a large proportion of patients who require SLA; however, the impact of additional comorbidities is not clear.
Methods
First-episode subsequent lower extremity limb amputation (SLEA) cases with the presence/absence of diabetes, other comorbidities, and demographic characteristics from 2006–2019 were retrieved from Saskatchewan's Discharge Abstract Database. Logistic regression was performed to examine the magnitude of the odds of SLEA.
Results
Among the 956 first-episode SLEA patients investigated, 78.8% were diagnosed with diabetes. Of these, 76.1% were male and 83.0% were aged 50 + years. Three comorbidities: renal failure (AOR = 1.9, 95% Cl 1.1 − 3.0), hypertension (AOR = 3.0, 95% Cl 2.0 − 4.5), and congestive heart failure (AOR = 2.0, 95% CI 1.2 − 3.2), conferred the highest odds of SLEA. The odds of SLEA is greatest for those aged 50–69 years, males, Registered Indians, and associated with a prolonged hospital stay.
Discussion
These data are important as they may help medical providers identify patients at the highest risk of SLEA and target interventions to optimize outcomes.
Introduction
The overall rate of limb amputation has remained stable over two decades in Saskatchewan despite increasing community efforts to educate and manage people with diabetes, the leading cause of lower extremity amputation. 1 Subsequent limb amputation (SLA), defined as ipsilateral revision amputation or contralateral index (single) amputation, may be performed to mitigate disease progression, reduce pain and enhance prosthesis fit.2–4 A recent meta-analysis by Liu et al. (2021) found SLA rates of 19% at 1 year and 37.1% at 5 years in people with diabetes. 5 This frequently performed procedure may be devastating, 2 along with a high rate of hospital readmission, extended post-operative hospital stay, and an increased risk of mortality when compared to index amputation.3,6–8
People with diabetes have a high prevalence of multimorbidity including, but not limited to, hypertension (HTN), heart disease, chronic kidney disease, ischemic heart disease, peripheral arterial disease (PAD), and stroke.3,9,10 These comorbidities may differentially impact SLA however, conflicting reports exist in the literature.3,11 Fard et al. found no risk factors associated with 1-year re-amputation among patients in Northern Netherlands, 11 while Norvell and Czerriecki found several risk factors, including chronic obstructive pulmonary disease, kidney failure, and diabetes among patients with limb amputation in the United States. 3 Further, Littman et al. found substantial geographical and ethnoracial variation contributing to SLA rates after minor (below the level of the ankle) amputation with greater risk among African American patients than white patients and those living in the southeast region of the United States compared to other regions. 12
In Saskatchewan, the rate of SLA (10.6 per 100,000 population) is steadily rising with a 3.2% average annual percent change from 2006−2019 13 ; this, in combination with a 45% provincial increase in overall diabetes prevalence over the last decade, 14 leads to the question of how epidemiologic correlates impact the rate of SLA in the province.
Addressing this gap may provide useful information to avoid the devastating impact of SLA, preserve limb length after index LA and enhance ambulatory function. 15 Additionally, identifying the demographic characteristics and most prevalent comorbidities may allow for targeted interventions to alter the rate of subsequent lower extremity amputation (SLEA). Therefore, the purpose of this study is to explore potential risk factors including comorbidities and demographic characteristics of SLEA among patients in Saskatchewan.
Methods
A cohort of patients with first-episode SLEA was identified and retrieved from Saskatchewan's Discharge Abstract Database (SK-DAD). The SK-DAD is part of Canada's nationally collated hospital abstract databases 16 that compiles health information from all hospital discharges in the province under the provincially managed Canadian universal single-payer healthcare system. Health information captured in the SK-DAD includes demographic and clinical data for discharges of inpatient acute care, chronic illnesses, rehabilitation, and surgical procedures. 16 The SK-DAD accommodates up to 25 diagnoses defined by the Canadian adaptation of the International Classification of Diseases codes (ICD-10-CA) and 20 interventions defined by the Canadian Classification of Health Intervention codes (CCI) per hospitalization. 1
The SK-DAD is similar to the national discharge data collated by the Diagnosis-Related Group Statistics (DRG-Statistics) in Germany, 17 which captures Germany's all acute care hospital inpatient cases, diagnoses defined by the German adaptation of the International Classification of Diseases (ICD-10-GM), German intervention procedure codes (OPS), demographic information and discharge status. 17 Furthermore, the SK-DAD is also comparable to the Swedish National Inpatient Register (INP) comprises the Hospital Discharge Register18,19 and links several national Swedish registers via a unique Swedish personal identifier number.18,19 In the Swedish INP, limb amputation cases are defined by the Nordic Classification of Surgical Procedure (NCSP), and diagnosis is defined by the International Classification of Diseases (ICD-10). 18 Sweden, like Germany and Canada, has a universal health care system, with coverage provided to all inhabitants irrespective of their income level, 20 making limb amputation data more comparable in these countries. For example, NCSP codes (NFQ09, NFQ19, NGQ09, NEQ19, NGQ 19, NHQ09, and NHQ11) are for major lower limb above-knee amputation (LL-AKA) and lower limb below-knee amputation (LL-BKA) in Sweden. 18 And this is equivalent to the Canadian CCI code (1VC 93, 1VQ93, 1VA93, 1SQ93, 1WA93, and 1VG93) for major LL- AKA and LL-BKA 1 and German OPS code (5-864).17,21 Also, the lower limb minor amputation (LLA) OPS codes 5-865 in Germany17,21 are equivalent to CCI codes (1WE93, 1WI93, 1WJ93, 1WK93, 1WL93, 1WM93, 1WN93) for minor LLA in Canada 1 and NCSP codes NHQ12, NHQ13 and NHQ14 in Sweden. 18 Likewise, the diagnosis code (ICD-10-CA) for diabetes E10-E14 in Canada is the same as the diabetes diagnosis code E10-E14 in German (ICD-10-GM)17,21 and ICD-10 codes in Sweden. 18
The present study data included all patient cases from January 1, 2006, to December 31, 2019, which were identified in the SK-DAD using the Canadian Classification of Health Interventions procedure codes that describe the various levels and specific anatomical sites of amputation (CCI: 1SQ93, 1VA93, 1VC93, 1VG93, 1VQ93, 1WA93, 1WE93, 1WI93, 1WJ93, 1WK93, 1WL93, 1WM93, 1WN93) (see Appendix 1 for the description of each code). 22 The primary outcome variable of interest was SLEA in patients diagnosed with diabetes. The diabetes diagnosis history of each patient was accessed five years prior to the index LEA intervention date using the International Classification of Disease (ICD-10-CA) codes for diabetes E10-E14 (E10.2-E10.8, E11.2-E11.8, E12.2-E12.8, E13.2-E13.8, E14.2-E14.8) 23 (see Appendix 1 for the description of each code) 24 and index into a binary indicator (‘Yes’ representing the presence of diabetes and ‘No’, absence of diabetes) via the Elixhauser comorbidity index. 25
In a similar manner, other comorbidities, including peripheral vascular disease (PVD), renal failure, HTN, arrhythmia, congestive heart failure (CHF), and cardiovascular disease (CVD) status was accessed. The present study accounted for these comorbidities due to their demonstrated direct or indirect influence on diabetes-related limb amputation research.26–28
The demographic characteristics of patients, including age (0–49 years, 50–59 years, 60–69 years, and 70 + years), sex (female/male), and location of residence, determined by postal code population (rural < 1000 and urban ≥1000)29,30 were retrieved from the Person Health Registration System (PHRS). Due to the unavailability of data on the location of residence in the years 2018−2019, the 2017 location of residence was used as a surrogate for those years. The patient's post-operative acute care length of stay (POALOS) after the SLEA procedure was also included in the present study as an explanatory variable. POALOS was calculated as patients’ discharge date minus intervention date, recorded in days, and grouped into short (≤ 7 days) and prolonged (> 7 days).31,32 The Saskatchewan administrative data repository does not capture a person's ethnicity, but the data repository can identify self-declared First Nations (FN) persons registered under the Indian Act of Canada, 33 further referred to as Registered Indians (RI), and other Saskatchewan residents (including whites, immigrants, Indigenous peoples with registered Indian status who have not self-identified in the PHRS, non-registered Indian status, Metis and Inuit peoples). For clarity, this study would refer to the other Saskatchewan residents as the General Population (GP) cohort. The 2016 Canadian census reported 106,440 people with RI status representing 60.8% of the 175,015 Indigenous people living in Saskatchewan. 34
Statistical analysis
Descriptive analyses specifically, frequencies and proportions were used to describe the study population demographic characteristics and the presence of comorbidities. A multiple logistic regression (MLR) analysis35,36 was also applied to assess potential comorbidities and other risk factors associated with SLEA in the presence of diabetes. The MLR process began by first fitting an unadjusted model between the outcome of interest and each comorbidity and demographic factor. All comorbidities and factors with p-values <0.25 35 in the unadjusted model were considered candidates for the adjusted model. The final adjusted model was arrived at by implementing the manual backward elimination method to eliminate all factors with p-values that exceed the set significant level of 0.05. 35
Change in estimate method was used to assess potential confounding factors 37 and interactions evaluated by Hosmer et al.'s model-building approach. 35 Finally, the odds ratio (OR) and its companion 95% confidence interval were reported from the study models.
Results
The demographics and comorbidities of patients are presented in Table 1. Of the 956 patients with first-episode SLEA investigated, 78.8% (753) were diagnosed with diabetes, and 21.2% (203) had no diagnosis of diabetes. Among the SLEA patients with diabetes, the majority, 59.1% (445) were 60 + years of age, whereas in the cohort without diabetes, those aged 70 + years reported the highest proportion, 41.3% (84), followed by patients aged 0–49 years, 28.1% (57). Males dominated both study groups, with 76.1% (573) of SLEA among diabetes patients reported in males and 64.0% (130) of SLEA without diabetes being males. SLEA was more common in urban dwellers with 57.7% (435) and without 62.1% (126) diabetes than their counterparts residing in the rural areas. Individuals with diabetes reported a higher proportion, 48.7% (367), of prolonged hospital stay after SLEA than the proportion of extended hospitalization witnessed in those without diabetes 36.9% (75), with median days of stay being 7 days vs. 5 days respectively. SLEA proportion reported in the general population (GP) with and without diabetes was 65.7% vs. 90.2% compared to 34.3% vs. 9.8% in Registered Indians (RI).
Demographic characteristics and comorbidities in subsequent lower extremity amputation patients with and without diabetes.
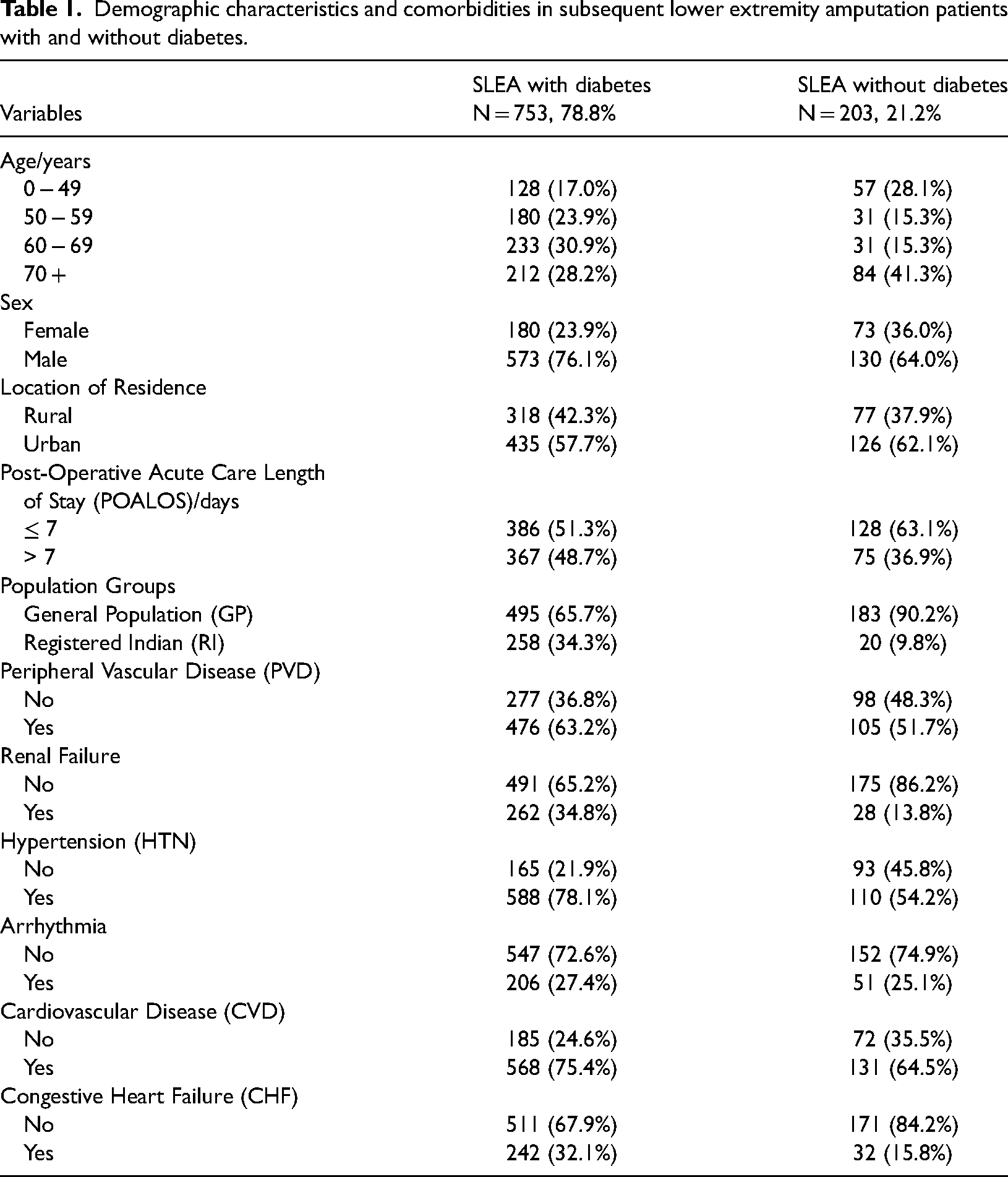
Three comorbidities for SLEA dominated in both individuals with and without diabetes. These were peripheral vascular disease (diabetes 63.2% vs. non-diabetes 51.7%), HTN (diabetes 78.1% vs. non-diabetes 54.2%) and CVD (diabetes 75.4% vs. non-diabetes 64.5%).
Table 2 summarizes the unadjusted and adjusted model results of demographic characteristics and comorbidities associated with subsequent LEA among individuals with diabetes. The unadjusted results showed significant differences in age, sex, population subgroups, renal failure, HTN, CHF (p < 0.001), POALOS, cardiovascular disease (p < 0.002), and peripheral vascular disease (p < 0.003) associated with SLEA in patients with diabetes. In contrast, location of residence (p = 0.270) and arrhythmia (p = 0.524) were not significantly associated with SLEA in patients with diabetes.
Demographic characteristics and comorbidities associated with diabetic subsequent lower extremity amputation.
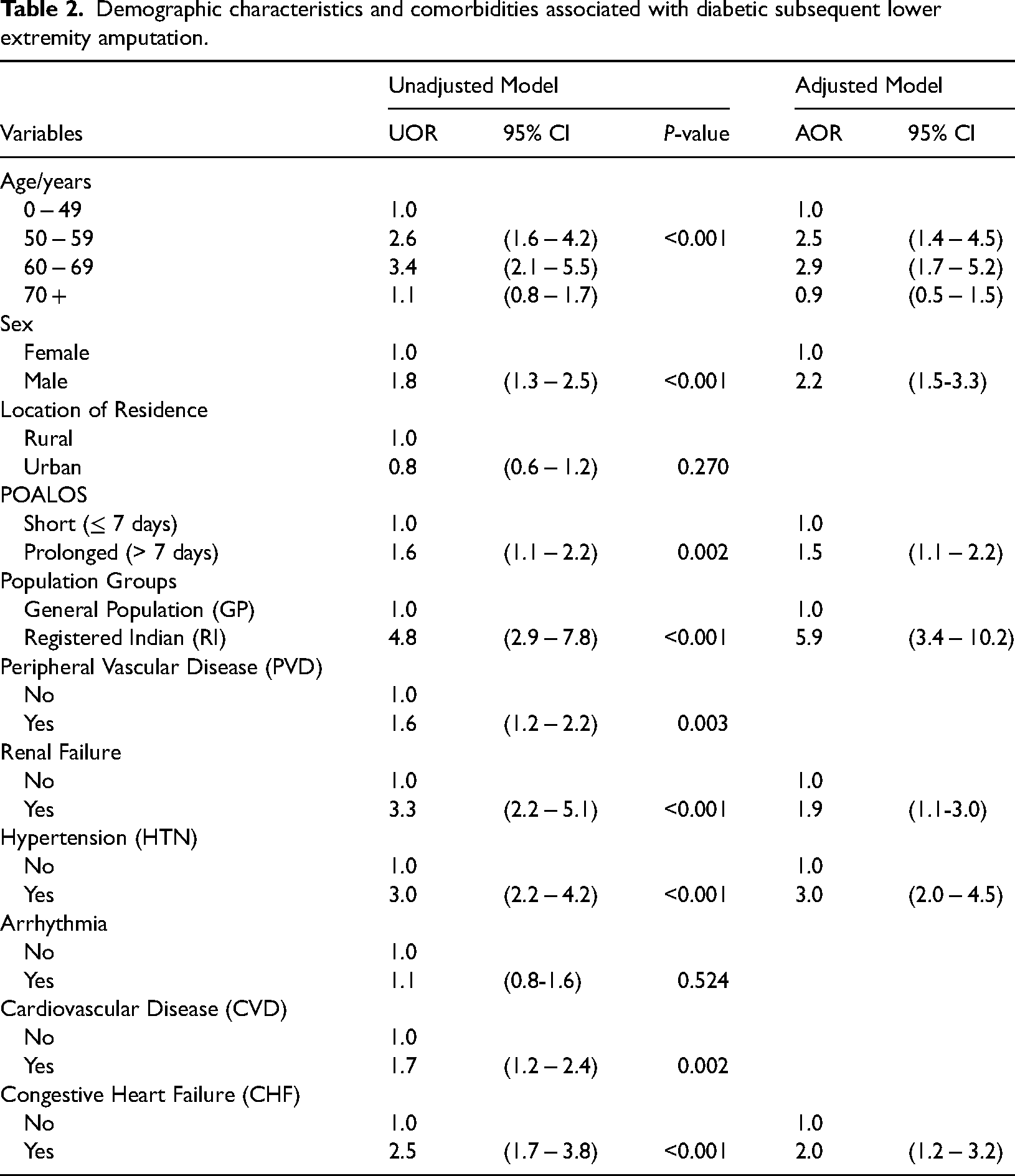
When other person-level factors and comorbidities were adjusted for, four demographic characteristics (age, sex, length of hospital stay, population subgroups) and three comorbidities (HTN, renal failure, CHF) were found to be associated with SLEA in patients with diabetes. Age 50–69 years increased the odds of SLEA in patients with diabetes by 2.5−2.9-fold (50−59 years: AOR = 2.5, 95% Cl 1.4−4.5; 60–69 years: AOR = 2.9, 95% Cl 1.7−5.2) than age 0–49 years. However, the odds of SLEA in patients with diabetes attenuated at age 70 + years (AOR = 1.1, 95% CI 0.8 −1.7). The sex of the study population also contributed significantly to SLEA in patients with diabetes, with 2.2 times higher odds of SLEA in males (AOR = 2.2, 95% Cl 1.5−3.3) than females. In addition, the odds of having pronged POALOS after SLEA with diabetes increased by 1.5-fold (AOR = 1.5, 95% Cl 1.1−2.2). Further, in the presence of diabetes, Registered Indians (RI) were 5.9 times more likely to have SLEA than the general population (AOR = 5.9. 95% Cl 3.4−10.2).
Three comorbidities conferred the strongest odds of SLEA in individuals with diabetes, with renal failure, increasing the odds by 1.9-fold (AOR = 1.9, 95% Cl 1.1−3.0), HTN by 3.0-fold (AOR = 3.0, 95% Cl 2.0−4.5), and CHF by 2.0-fold (AOR = 2.0, 95% Cl 1.2−3.2).
Discussion
The need for SLEA among patients is confounded by other comorbidities. We found that over a 14-year-study period, approximately 79% of people who had first-episode SLEA in Saskatchewan also had a history of diabetes. Further, a diagnosis of HTN increased the odds of SLEA by 3.0-fold, renal failure by 1.9-fold, and CHF by 2.0-fold in people with diabetes. People with diabetes aged 50–69 years had a 2.5–2.9-fold increased odds of SLEA than those aged 0–49 years, with lower odds observed in those aged 70 + years. Males with diabetes were 2.2 times more likely than females to have SLEA, and Registered Indians (RI) were 5.9 times more likely to have SLEA than the general population (GP). In addition, people with diabetes were 1.5 times more likely to have prolonged POALOS after SLEA compared to people without diabetes. When other person-level factors and comorbidities were adjusted for, location of residence, peripheral vascular disease (PVD), arrhythmia, and cardiovascular disease (CVD) were not significantly associated with SLEA in individuals with diabetes.
Our finding that HTN increased the odds of SLEA by 3.0-fold in people with diabetes is supported by the literature.27,38–40 Alshallwi (2019) found that HTN increased the odds of SLEA by 3.7-fold in patients with diabetes. 38 A similar trend was observed in Seçkin et al.'s study where HTN increased the odds of subsequent amputation in patients with diabetes by 2.2-fold. Our findings that CHF increases the odds of SLEA by 2.0-fold in patients with diabetes are consistent with the findings of Zhang et al., who identified a 1.5-fold odds of 1-year re-amputation in patients with CHF and diabetes. 41
Although the association between renal failure and LEA among diabetes patients has been broadly studied,42,43 few studies have attempted to specifically examine the association of SLEA6,39,44,45 in patients also diagnosed with diabetes.3,46 These studies, similar to ours, suggest that having renal failure in addition to diabetes increases the risk of SLEA.3,46 Acar and Kacira (2017) found that patients with diabetic nephropathy had 2.8 times higher odds of re-amputation, 46 but Seçkin et al. found chronic renal failure was not a risk factor for re-amputation among patients with diabetes. 40
Our finding in the unadjusted model that PVD and CVD were associated with SLEA in individuals with diabetes did not persist in the adjusted model. These latter findings are in line with those of Seçkin et al., in which peripheral arterial disease (PAD) did not significantly increase the risk of re-amputation in patients with diabetes. 40 However, other studies found a significant association between SLEA and PVD/PAD.27,38,41 The inconsistencies in the association between SLEA and PVD/PAD and renal failure warrant further studies explicitly focusing on these factors.
We found that ethnicity, specifically First Nations Saskatchewanians registered under the Indian Act of Canada, with diabetes were 5.9 times more likely to undergo SLEA than people in the general population with diabetes. This finding was the most divergent of all variables studied and questions why such a large disparity exists. Although disparities in LEA rates have been recently identified in the literature, including our previously reported findings that the overall rate of limb amputation for RI was 1.7 to 4.2 times higher than that of GP, we did not anticipate an even greater disparity in SLEA rates.12,47,48 Littman et al. found higher SLEA rates in African Americans than whites in the 90 days after initial toe amputation (202 versus 149 per 1000 patients, respectively) in military veterans with diabetes. 12 Gandjian et al. found higher rates of overall (1.16) and primary (1.34) major amputation in nonwhites than whites living in California after diagnosis of acute limb ischemia, 47 and Tan reported African Americans were at a 1.9% greater risk and Native Americans were at a 1.8% greater risk of major amputation when compared with whites after diagnosis of diabetic foot ulceration or infection. 48 The root of these disparities is multifactorial, with sociodemographic factors (economic, racism, health care accessibility, health behaviours, and stresses associated with systemic racism) and biologic factors, including diabetic phenotypes and risks of diabetic complications that may differ among ethnic groups and contribute to the imbalance in SLEA, and LEA, identified between First Nations and non-First Nations Saskatchewanians.49–51 This highlights the need to explore diabetes phenotypes among ethnic groups to tailor treatment needs.
Our findings also identified that advancing age, specifically age 50−69 years increased the risk of SLEA in patients with diabetes. Interestingly, the risk attenuated with age 70 + years. These findings are partly supported by Zambetti et al. and Erşen et al., who reported an association of ages 68.8 ± 10.8 years and 60.8 ± 17.1 years, respectively, were associated with re-amputation.6,52
The differential impact of sex on SLEA among patients with diabetes in the present study is reflective of the differences in the prevalence of males diagnosed with diabetes (76.1%) compared to 23.9% of females. We identified that males diagnosed with diabetes were 2.2 times more likely than females to undergo SLEA. These findings are supported by Johannesson et al. who found a higher incidence rate of re-amputation in men with diabetes than in females (21 per 100 amputee-years vs. 16 per 100 amputee-years). 53 And further re-echoed by Acar and Kacira (2017), who found adult males with diabetes had 4.1 times increased odds of re-amputation. 46
In the present study, patients with diabetes had a 1.5-fold higher risk of prolonged POALOS after SLEA (7 days vs. 5 days) when compared to patients without diabetes. Although Zambetti et al. did not limit their study to diabetes patients, their findings were similar to increased length of hospital stay found when re-amputation occurred within 30 days of index amputation (12.9 days vs. 7.3 days). 6 Likewise, Yaghi et al.'s who broadly explored the length of stay among diabetes and non-diabetic patients with limb amputation but not limited to re-amputation cohorts found the risk of prolonged stay of LA in diabetes patients was 4.6 times higher than in non-diabetic patients. 54 As an explanation, Ceyhan et al. observed that re-amputated patients do experience prolonged lengths of hospital stay because they are prone to more postoperative complications than their counterparts. 55 These postoperative complications include renal disease, a history of sepsis, and contaminated/infected wounds, especially in diabetes patients.56,57
Strengths and limitations
Subsequent LEA is an area that has not been well explored in literature as in the case of index amputation, hence it was a strength of this study to explicitly focus on this neglected area of research. Also, since the incidence of SLEA could be impacted by many personal-level factors and comorbidities, it was a strength that the present study accounted for several of these factors. Due to limited samples, we could not stratify the data for subsequent levels of amputation (minor/major) and explore potential risk factors by these levels. This is because further stratification by amputation level would cause the study to lack statistical power. The limitation on using the 2017 location of residence as a proxy for the years 2018 and 2019 and its possible impact on the study variable is reported elsewhere. 58
Conclusion
Among this first-episode SLEA cohort diagnosed with diabetes, three comorbidities, renal failure, CHF, and HTN conferred the highest odds of SLEA. Also, the risk of SLEA is greatest for those aged 50–69 years, males, Registered Indians (RI), and is associated with prolonged POALOS. Although not all SLEA is avoidable, risk factors associated with diabetes may be independently treated thus may mitigate the need for SLEA. Our most dispirit finding, that First Nations Saskatchewanians registered under the Indian Act of Canada with diabetes were 5.9 times more likely to undergo SLEA than the general population, is among the areas in need of further in-depth study to target the root causes of the disparity.
Footnotes
Disclaimer
This study is based in part on de-identified data provided by the Saskatchewan Ministry of Health and eHealth Saskatchewan. The interpretation and conclusions contained herein do not necessarily represent those of the Government of Saskatchewan, the Saskatchewan Ministry of Health, or eHealth Saskatchewan.
Acknowledgements
We thank the Saskatchewan Amputee Patient-Oriented Research Team (PORT) and the Saskatchewan Centre for Patient-Oriented Research for their support throughout this study.
Contributorship
SKE and AZL conducted the literature reviews. SKE and AZL conceptualized and designed the study. SKE and AZL secured the study data, performed the analyses, interpreted the results, and drafted the manuscript. All authors have read and approved the final submitted copy.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study received ethical approval from the Biomedical Ethics Board (Approval number # Bio 1590).
Informed Consent
Informed consent was waived by the Biomedical Ethics committee, as the study presents no risk of harm to the study subjects.
Guarantor
* SKE
Appendix 1
1SQ93 = Amputation at pelvis 1VA93 = Amputation at hip joint 1VC93 = Amputation at femur 1VG93 = Amputation at knee joint 1VQ93 = Amputation at tibia and fibula 1WA93 = Amputation at ankle joint 1WE93 = Amputation at tarsal bones and intertarsal joints (hindfoot, midfoot) IWI93 = Amputation at first metatarsal bone and first metatarsophalangeal joint 1WJ93 = Amputation at tarsometatarsal joints, other metatarsal bones and other metatarsophalangeal joints (i.e., forefoot) 1WK93 = Amputation at first phalanx of foot 1WL93 = Amputation at phalanx of other foot 1WM93 = Amputation at other interphalangeal joints of toe 1WN93 = Amputation at first interphalangeal joint of toe
