Abstract
Background/Aims:
The stepped-wedge cluster randomised trial design has received substantial attention in recent years. Although various extensions to the original design have been proposed, no guidance is available on the design of stepped-wedge cluster randomised trials with interim analyses. In an individually randomised trial setting, group sequential methods can provide notable efficiency gains and ethical benefits. We address this by discussing how established group sequential methodology can be adapted for stepped-wedge designs.
Methods:
Utilising the error spending approach to group sequential trial design, we detail the assumptions required for the determination of stepped-wedge cluster randomised trials with interim analyses. We consider early stopping for efficacy, futility, or efficacy and futility. We describe first how this can be done for any specified linear mixed model for data analysis. We then focus on one particular commonly utilised model and, using a recently completed stepped-wedge cluster randomised trial, compare the performance of several designs with interim analyses to the classical stepped-wedge design. Finally, the performance of a quantile substitution procedure for dealing with the case of unknown variance is explored.
Results:
We demonstrate that the incorporation of early stopping in stepped-wedge cluster randomised trial designs could reduce the expected sample size under the null and alternative hypotheses by up to 31% and 22%, respectively, with no cost to the trial’s type-I and type-II error rates. The use of restricted error maximum likelihood estimation was found to be more important than quantile substitution for controlling the type-I error rate.
Conclusion:
The addition of interim analyses into stepped-wedge cluster randomised trials could help guard against time-consuming trials conducted on poor performing treatments and also help expedite the implementation of efficacious treatments. In future, trialists should consider incorporating early stopping of some kind into stepped-wedge cluster randomised trials according to the needs of the particular trial.
Introduction
In a stepped-wedge (SW) cluster randomised trial (CRT), an intervention is introduced across several time periods, with the time period in which a cluster begins receiving the experimental intervention assigned at random. Although the SW-CRT design was actually first proposed over 30 years ago, 1 it has only been in recent years that it has gained substantial attention in the trials community.
Numerous papers have now been published containing new research on the design. Methodology2–5 and software 6 now exist to determine required sample sizes, and several results on optimal SW-CRT designs have been established,7,8 while extensions to the standard design to allow for multiple levels of clustering have also been presented. 9
However, as has been noted, little is known about the design of SW-CRTs with interim analyses. 10 In an individually randomised trial setting, it has been well established that group sequential methods can bring substantial ethical benefits and efficiency gains to a trial. 11 Explicitly, allowing the early stopping of a trial for either efficacy or futility can reduce the number of patients administered an inferior intervention and allow efficacious interventions to either move to later phase testing or to be rolled out across a population with greater speed. Given that SW-CRTs can be highly expensive because of the large number of time periods and measurements they can require, it would be advantageous to be able to incorporate interim analyses into the design.
In this article, we present methodology for establishing such designs. We then conclude with a discussion of the practical and methodological considerations associated with the use of interim analyses.
Methods
Notation, hypotheses, and analysis
We assume that a SW-CRT is to be carried out on
We next assume that the accrued data from our SW-CRT trial will be normally distributed, and a linear mixed model has been specified for analysis as

where
Note that, in particular, it is the prescribed
We assume the final element of
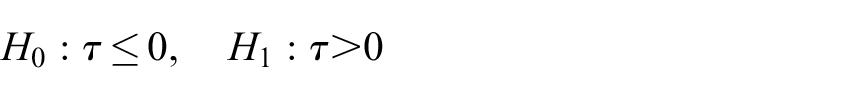
Moreover, we assume it is desired to control the type-I error rate of this test to some level
We specify a set of integers
With the above,
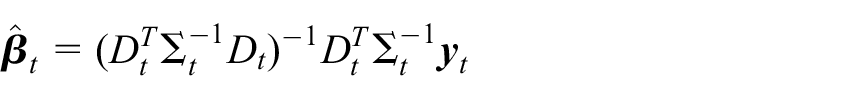
Here, the subscript
Following the notation of Jennison and Turnbull,
11
we acquire
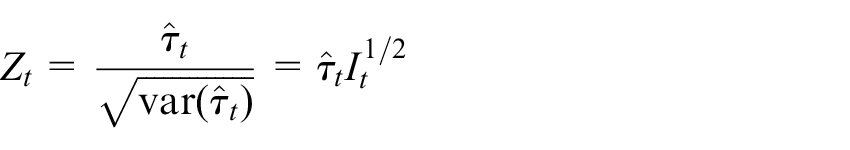
where
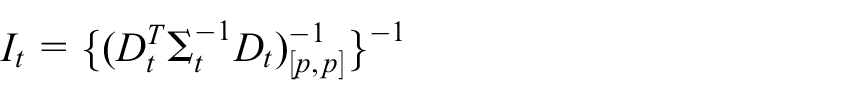
is the information for
Note that all of
While group sequential methodology is typically associated with designs with an independent increment structure, the important results hold for the more general scenario utilising linear mixed models considered here. In particular, we have that 11
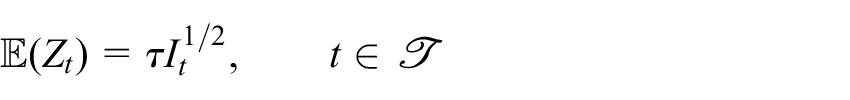
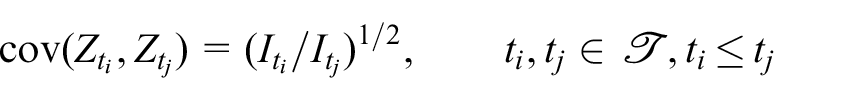
Finally, given futility and efficacy bounds,
For - If - If * if * if * if
For - if - if
We denote by
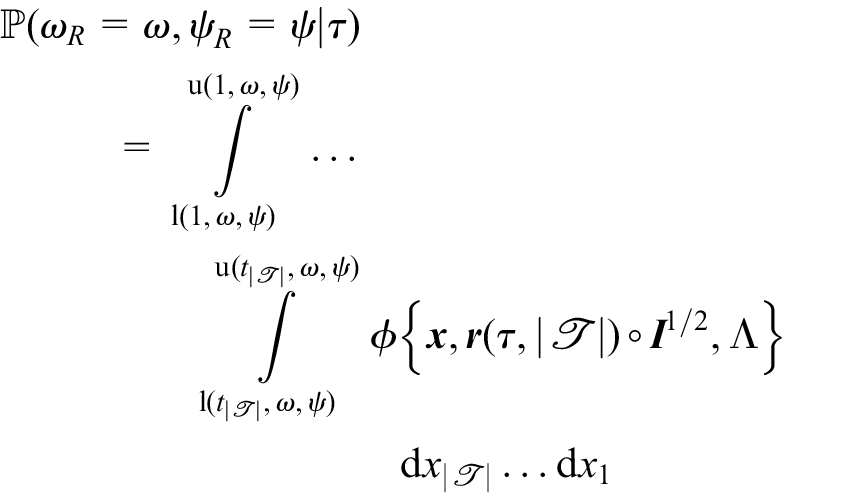
where
The probability that
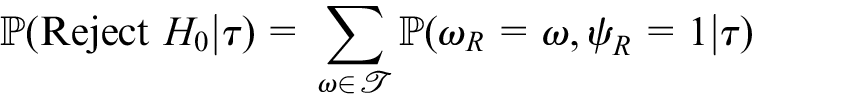
Moreover, we can determine the expected number of measurements that would be required by a design for any
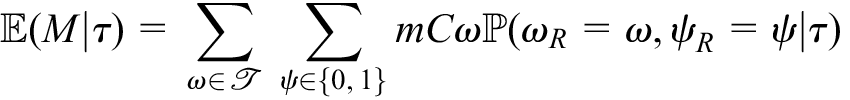
Here,
Error spending
Numerous procedures have today been proposed for the determination of group sequential trial designs. One of the earliest and most flexible such methods is the error spending approach.
12
In this case, functions


We define
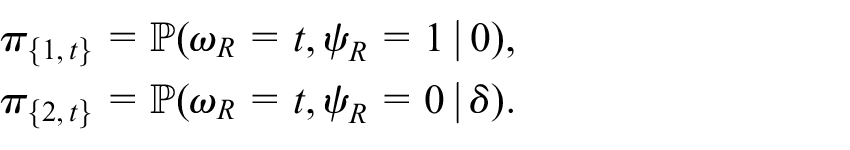
Thus,
Then, for given choices of


and
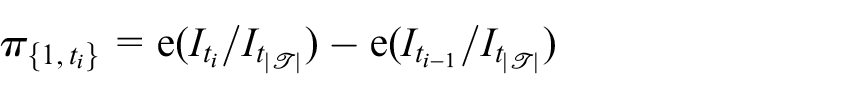
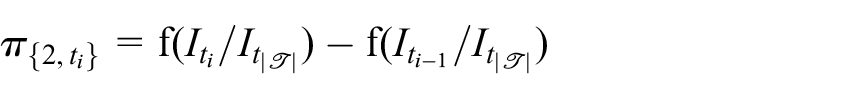
for
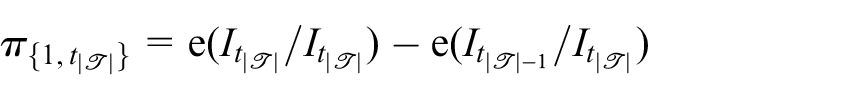
Note that one can prevent early stopping for futility or efficacy by setting
Now, all that remains is to be able to identify the
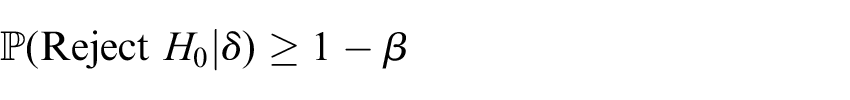
For individually randomised trials, this search is usually done assuming the relevant parameter is continuous, with it then rounded up to the nearest allowable integer to ensure the desired power is met. The ability to do this here depends upon having an explicit closed-form expression for
Hussey and Hughes model
In this section, and for the majority of the remainder of the article, we focus on cross-sectional SW-CRTs since the majority of research into the design has been set in this domain. This means that our value of
In addition, for all considered examples, we utilise the following model which has been proposed for the analysis of cross-sectional SW-CRTs 2
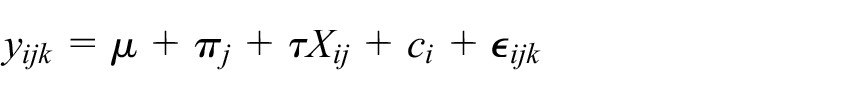
where
Note that specification of the matrices
Our reasons for focusing on this model are twofold. First, as a commonly studied and utilised model, it is a sensible choice to consider when determining and exploring the performance of example sequential SW-CRT designs. Additionally, in this case, if
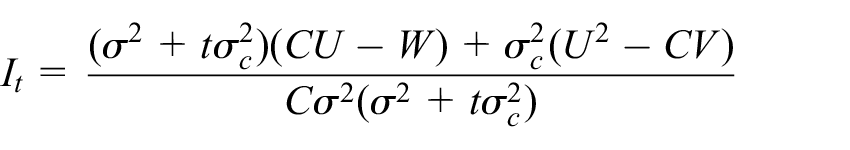
where



Software for determining designs in this scenario is available from https://sites.google.com/site/jmswason/supplementary-material.
To summarise the above, a design in this scenario can now be determined given values for
Unknown variance
In the above, we required all variance parameters to be fully specified. In practice, the key variance parameters of any analysis model will not be known precisely. Instead pre-trial estimates are provided, which we denote for the Hussey and Hughes model by
Even in the case where there is strong confidence in their values, it would often still be preferred to utilise the values for the variance parameters estimated from a trial’s accrued data in the formation of the test statistic at each interim analysis, rather than the specified pre-trial estimates

where
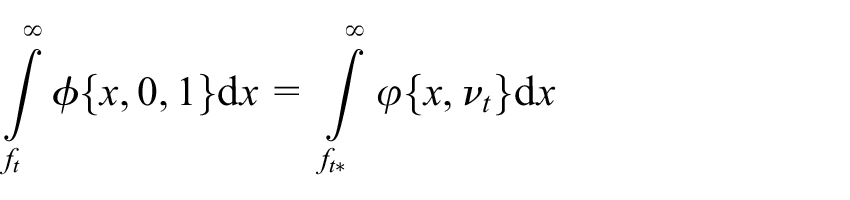
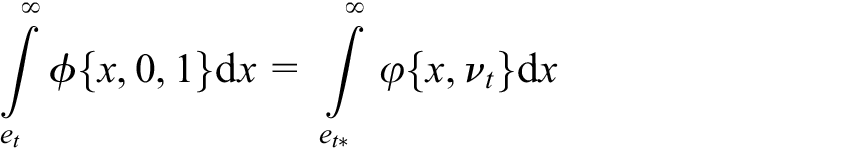
for

In this instance, it is also necessary to decide whether to utilise ML, or restricted error maximum likelihood (REML) estimation, when fitting the chosen linear mixed model at each interim analysis. Here, we consider the performance of both options.
Thus, in total, the performance of each of four possible analysis procedures was explored: ML or REML estimation, with or without boundary adjustment through quantile substitution. To estimate empirical rejection rates, 100,000 trials were simulated for each considered parameter set.
Note that for simplicity, when generating data
Example SW-CRT design scenarios
A SW-CRT on the effect of training doctors in communication skills on women’s satisfaction with doctor–woman relationship during labour and delivery was recently conducted.
16
The trial included four hospitals
We motivate our second example design scenario (Scenario 2) based on the average design characteristics of completed SW-CRTs according to a recently completed review.
17
Explicitly, we set the number of clusters to be 20 (
For both scenarios, we then consider the effect of different choices for the remaining design parameters:
Results
Example sequential SW-CRT designs
The performance of several example sequential SW-CRT designs with differing choices for
The performance of several sequential SW-CRT designs (Designs 1–6), along with that of the corresponding classical SW-CRT design (Design 7), is summarised, for Scenarios 1 and 2.
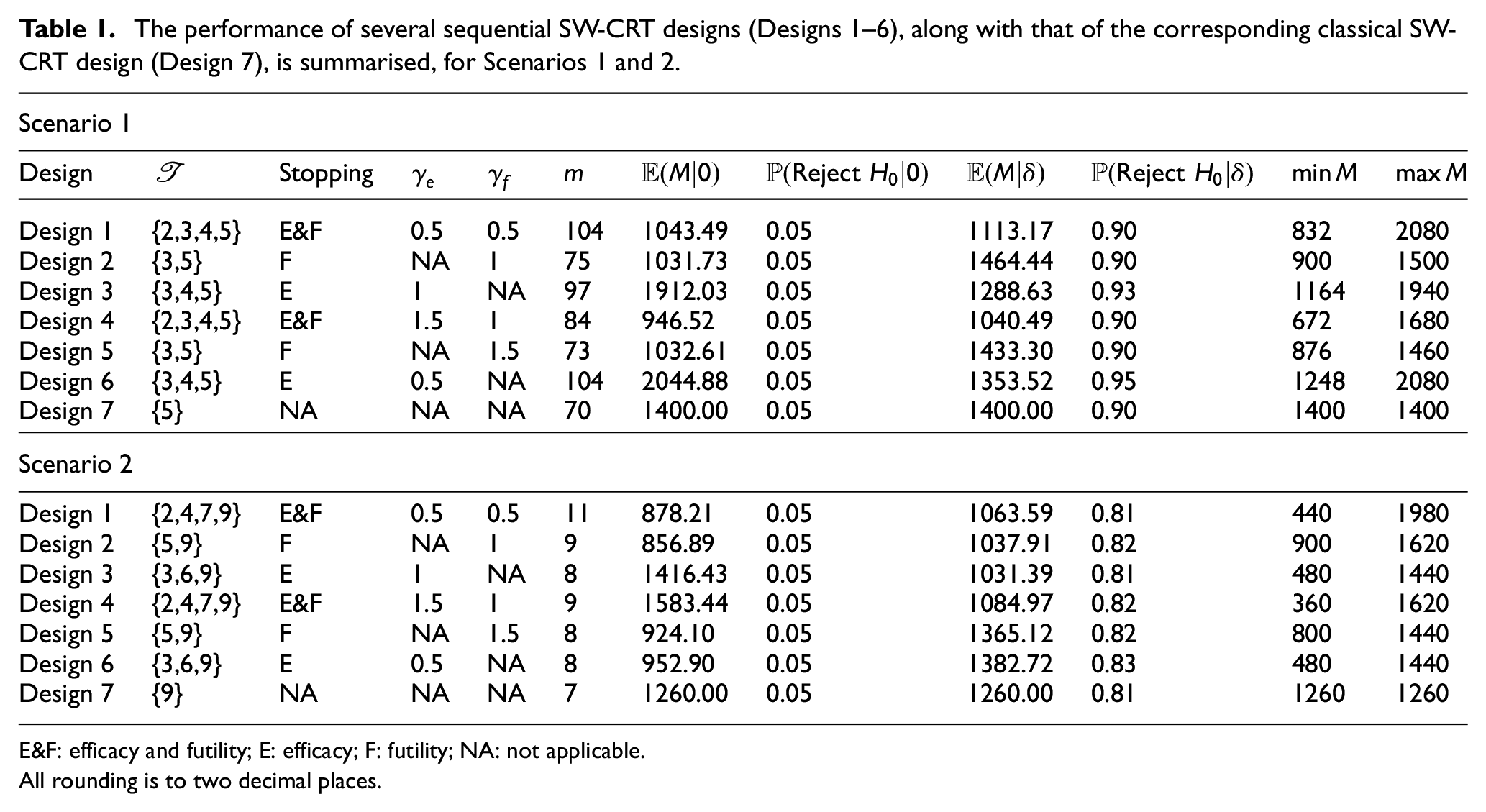
E&F: efficacy and futility; E: efficacy; F: futility; NA: not applicable.
All rounding is to two decimal places.
However, as would be expected, the maximal sample size that could be required by the sequential designs is larger than that of the corresponding fixed sample design. Furthermore, the sample size required by the sequential designs can be subject to substantial variability. In Figure 1, this variability is displayed for the sequential designs with early stopping for efficacy and futility (
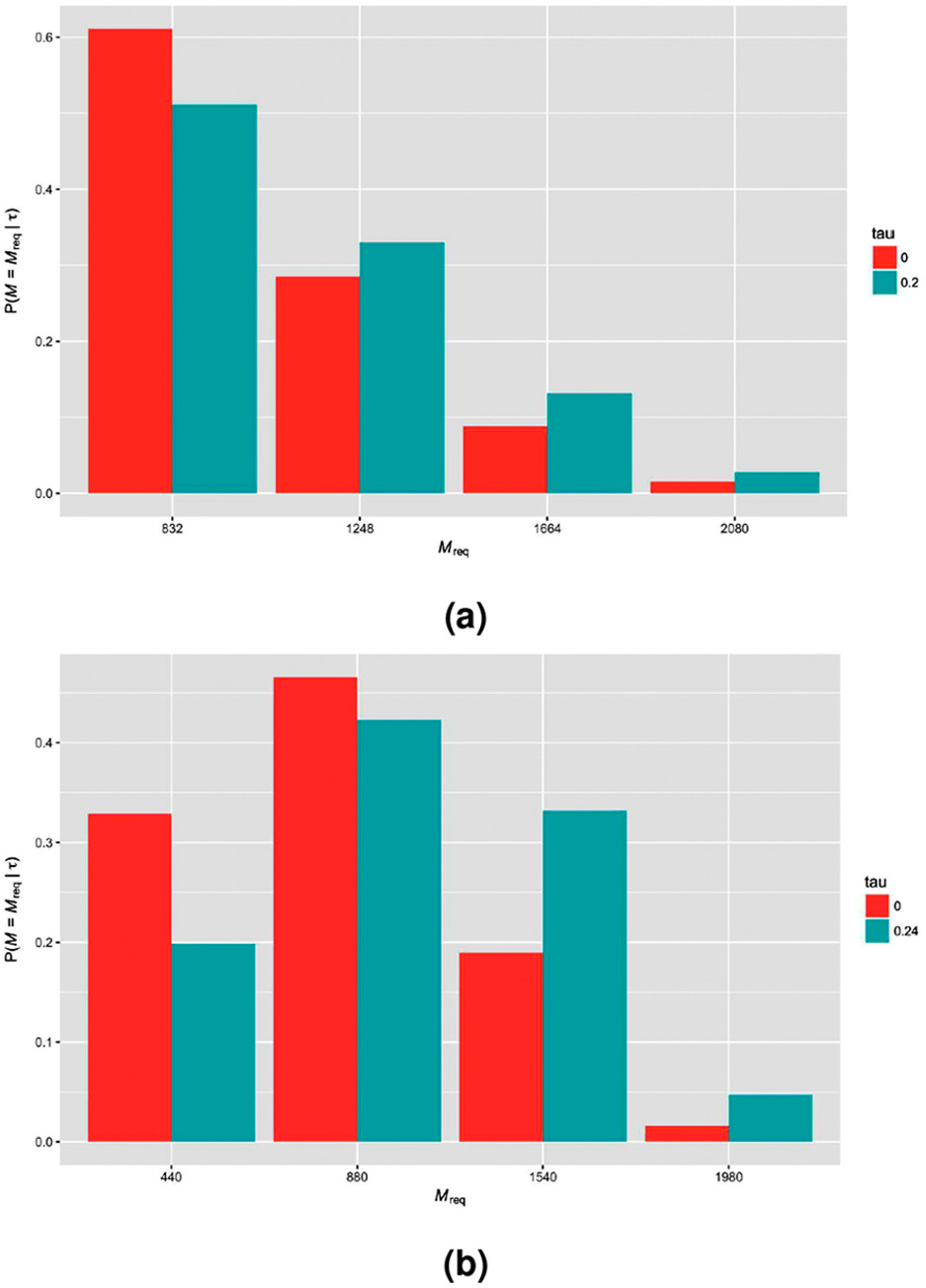
The probability distribution of the sample size required by example sequential designs (early stopping for efficacy and futility with
Considerations on
,
,
, and the allowed reasons for early stopping
In Figure 2, we demonstrate the effect of different choices for
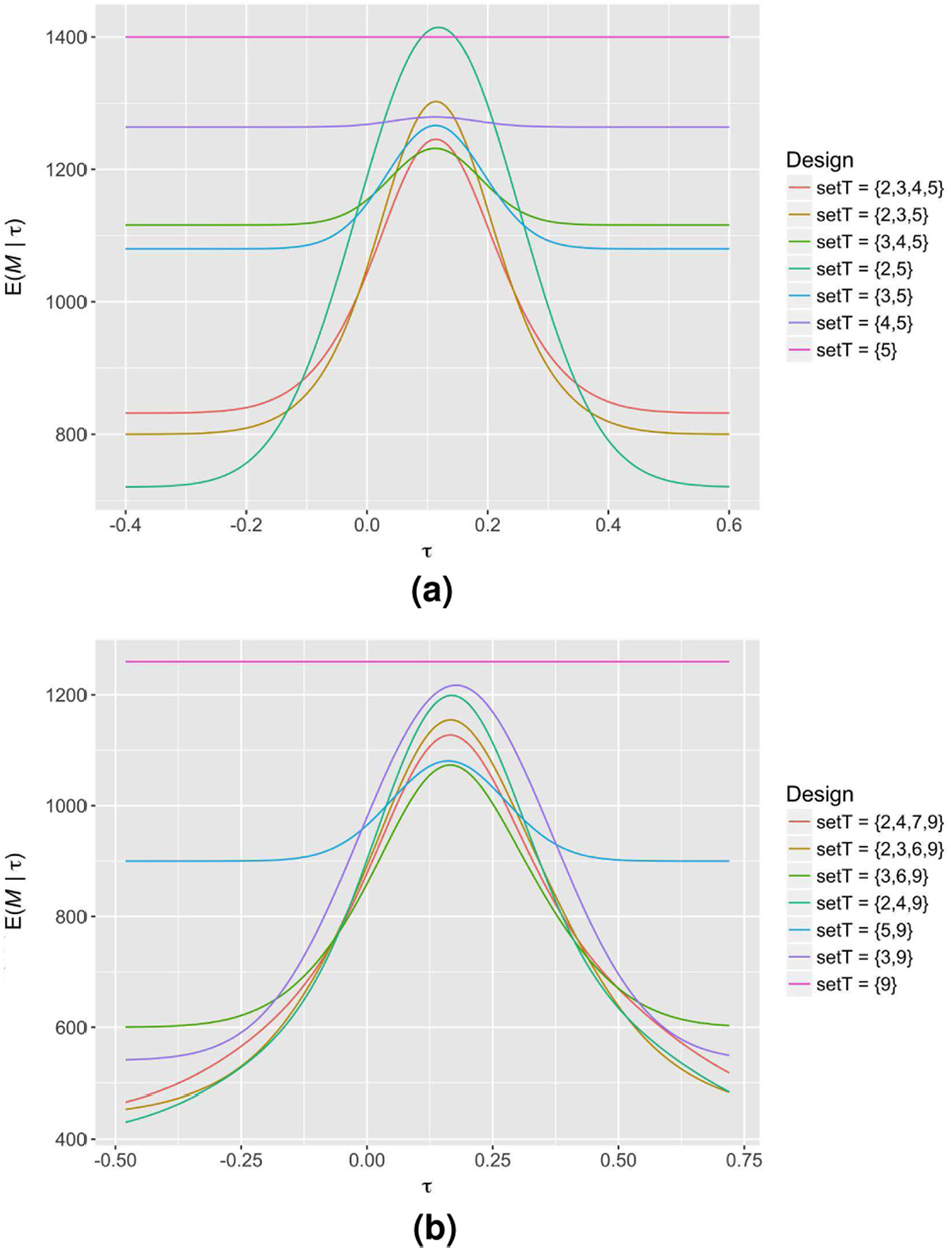
The expected sample size curves of several sequential SW-CRT designs with different possible choices for
The performance of several sequential SW-CRT designs with different possible choices for
Early stopping is allowed for efficacy and futility, with
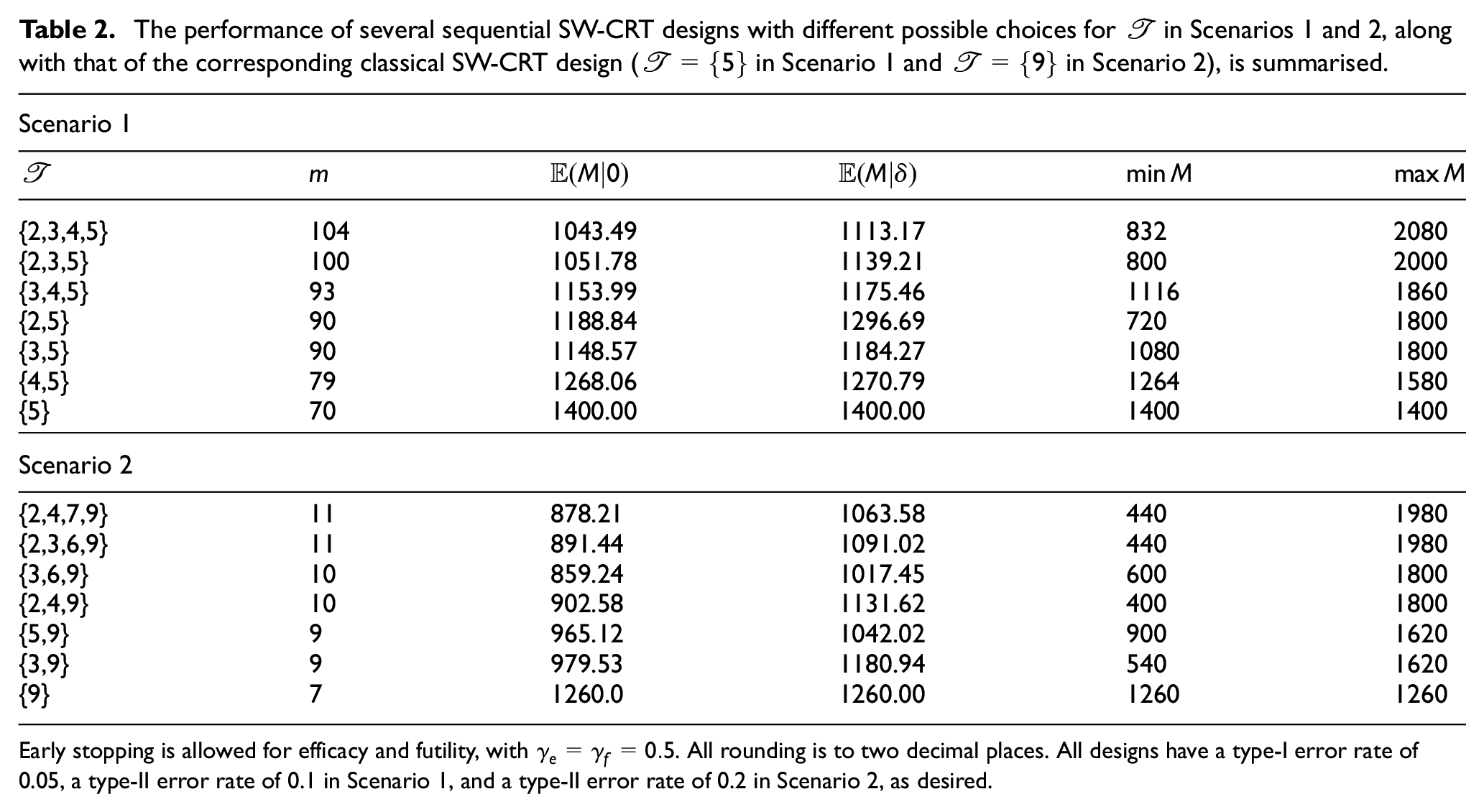
Similarly, Figure 3 displays the impact of altering
The expected sample size curves of several sequential SW-CRT designs with different possible choices for
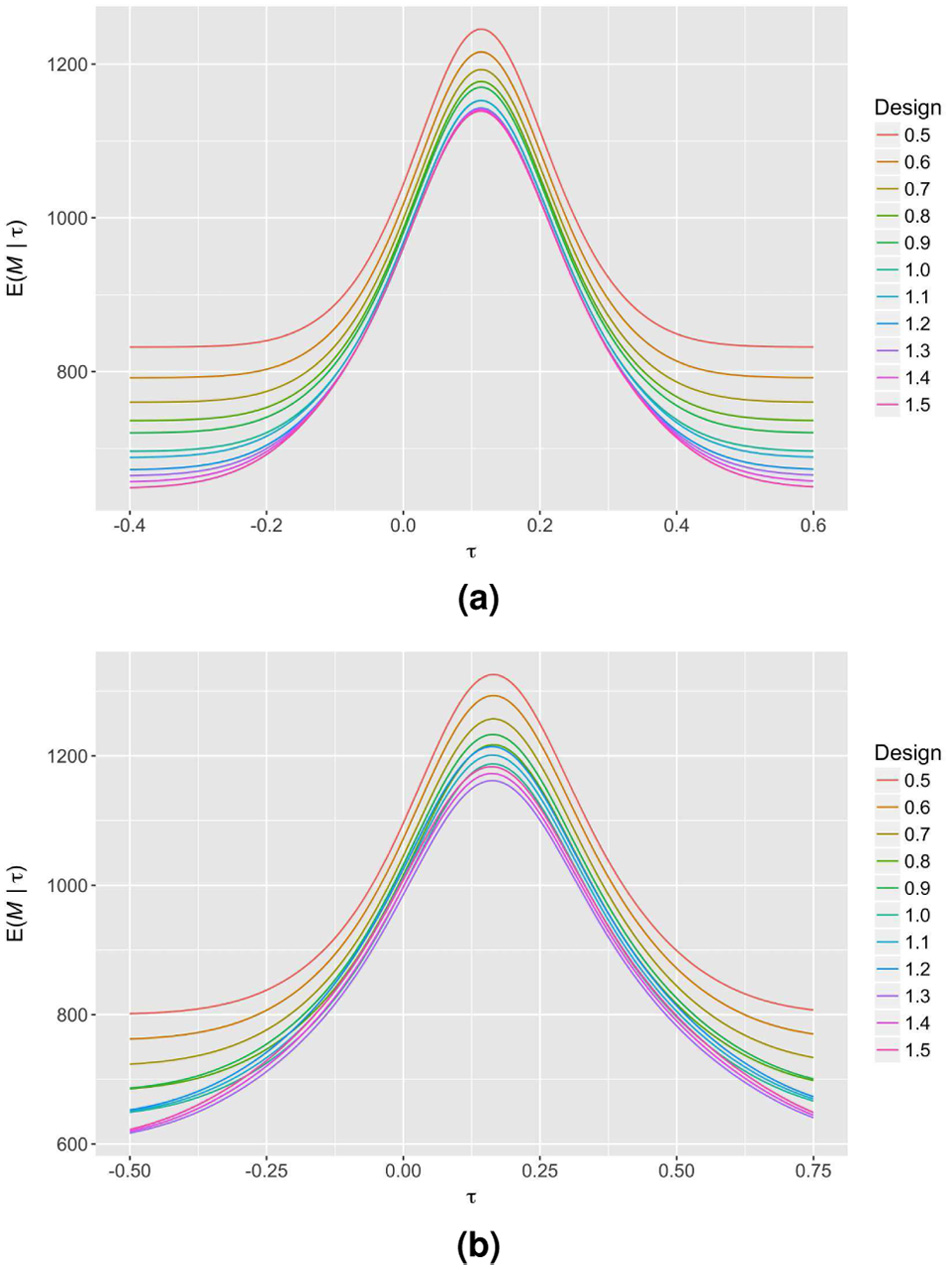
Finally, in Figure 4, we observe the effect of the choice of allowed reasons for early stopping (Scenario 1 with
The expected sample size curves of several sequential SW-CRT designs with different possible allowed reasons for early stopping are displayed for Scenario 1. Each design has
Quantile substitution
In Table 3, the empirical rejection rate of the sequential SW-CRT designs for Scenario 1 with
The empirical rejection rate using the four considered analysis procedures (ML or REML estimation, with or without boundary adjustment (BA) through quantile substitution) is displayed, for several possible values of the assumed variance parameters, true treatment effect, and the designs with
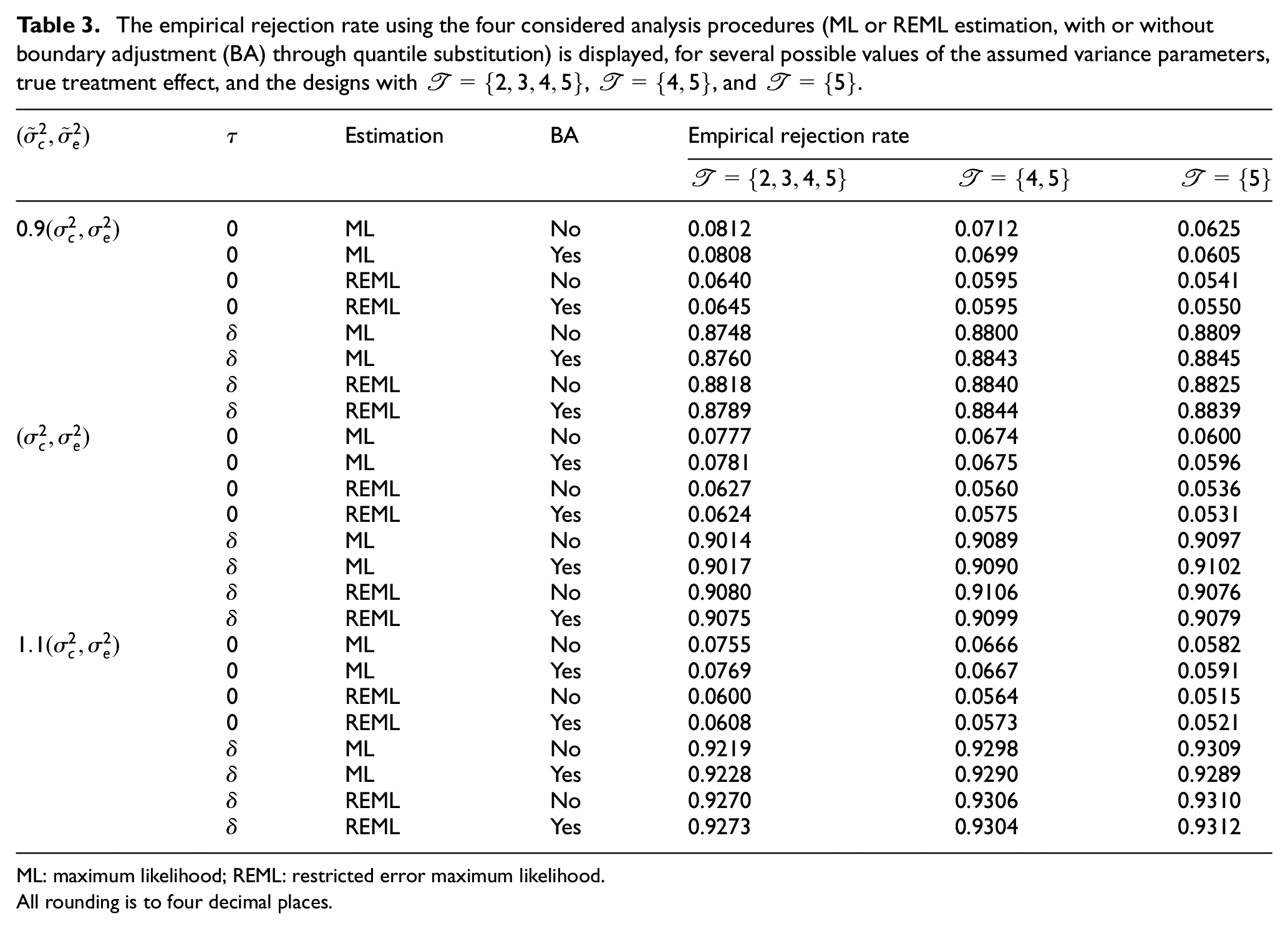
ML: maximum likelihood; REML: restricted error maximum likelihood.
All rounding is to four decimal places.
It is clear that when ML estimation is utilised in the sequential designs there can be substantial inflation in the empirical type-I error rate (up to 0.0812 for
The small number of clusters also results in inflation of the type-I error rate in the fixed sample designs. It is clear that this inflation is typically less than that for the corresponding sequential design and analysis procedure, though the difference is smaller when REML estimation and quantile substitution is utilised. Moreover, the inflation is smaller for the sequential designs with
Discussion
In this article, we demonstrated how established group sequential trial methodology can be adapted to determine SW-CRT with interim analyses. It was clear from our examples that the incorporation of interim analyses into the SW-CRT design could bring substantial reductions in the expected sample size under both
Although the inherent time period structure of SW-CRTs lends itself well to sequential methods, this does rely upon the efficient collection and storage of data for analysis. Putting measures in place to prevent operational issues would therefore be essential. In reality, a small delay between time periods may be necessary to allow for an interim analysis to be conducted. Without this, the clusters will have already begun data accrual for the following time period, which would bring a loss of efficiency to the required number of measurements. In addition, the more interim analyses a trialist includes theoretically reduces the expected sample size; however, this too comes with a larger burden in terms of the cost of analysis. In practice, trading off some loss in efficiency to reduce this burden may be wise.
Furthermore, the increase in sample size required per cluster per period in the sequential designs may mean the length of each time period needs to be increased. This would specifically be true when the length of a time period is chosen based on the supposed achievable recruitment rate. In this instance, however, the possibility to stop the trial early in the sequential designs means the average length of a trial could often still be reduced.
Moreover, the methodology presented here requires data to be unblinded at each interim analysis. Although many SW-CRTs are performed in an unblinded manner, 18 it would be important to ensure even then that the results of the data analysis at interim are kept hidden from all but those on the Data Monitoring Committee.
There is also much to consider in terms of the choice of allowed reasons for early stopping. It was previously noted that stopping early for futility would be unlikely in a SW-CRT because of the often held a priori belief the intervention will be effective. 10 However, a recent literature review established that 31% of SW-CRTs completed to date did not find a significant effect of their intervention on any primary outcome measure. 17 For this reason, incorporation of futility stopping does in fact seem warranted. Nonetheless, there are additional factors to consider. Primarily, the plan to eventually implement the intervention in all clusters, as is often the case in SW-CRTs, could be decided upon as an incentive for cluster participation in the trial. If this is the case, one must be careful to acknowledge to enrolled clusters that they may in fact not receive the intervention if the trial is stopped early for futility. Furthermore, some SW-CRTs are planned roll-outs of a programme, in which case there may not be a desire to stop the roll-out for futility if the study is part of a larger programme implementation. If this is the case, it may be likely that a SW-CRT design with early stopping would not be appropriate.
Moreover, the stopping of a trial for efficacy would typically imply the immediate deployment of an intervention to all clusters will then follow. However, with SW-CRTs often used when there are logistic constraints, this may not be possible. It could be that an intervention is rolled out as quickly as is possible, but this fact should be considered before early stopping for efficacy is included in a design. Finally, in some instances, there may be a desire to study the development of an intervention within the clusters over time. Stopping a trial early for efficacy or futility may prevent this possibility. In this case, it could be wise to only include stopping for futility to guard solely against harmful interventions.
There are several methodological considerations that should be recognised. First, the approach used to sequential SW-CRT design here assumes the trial’s nuisance parameters to be known. We demonstrated that REML estimation can help deal with this problem in the case where there is only small uncertainty in their values, and the number of clusters is small. As was noted, a sample size re-estimation procedure would be required if this was not the case. Moreover, even in this instance, there was still some inflation to the empirical type-I error rate. This is common, however, to both the classical fixed sample design and our proposed sequential designs. Nonetheless, smaller inflation was observed in a sequential design with fewer interim analyses, placed later into a trial. Therefore, similar to the burden introduced from introducing additional analyses discussed above, this should be factored in when choosing an appropriate sequential design.
Additionally, as with any trial design scenario, if the model assumed at the design stage does not hold, the trial’s operating characteristics will not be reliable. For a sequential design, depending on the violation, the degree to which the type-I and type-II error rates depart from their planned values could be larger than that of a fixed sample SW-CRT design. It would be important therefore when choosing an appropriate sequential SW-CRT, as for classical SW-CRT designs, to assess the sensitivity of the design to deviations in the underlying distribution of the data.
Finally, we have here only considered the design of SW-CRTs with interim analyses. It is well known that if naive estimators are used after a sequential trial, then acquired treatment effects will be biased. The development of methodology to account for this would be required. Fortunately, there is a breadth of literature on this for individually randomised trials upon which such work could be based (see, for example, Bretz et al. 19 ).
In conclusion, although there are several factors that must be considered by a trialist before deciding to incorporate early stopping in to a SW-CRT design, they should certainly consider whether the methodology is appropriate. With the inclusion of interim analyses, they can more suitably guard against much investment being spent on an inferior intervention or indeed help hasten the roll-out of an efficacious treatment.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Wellcome Trust (grant number 099770/Z/12/Z to M.J.G.), the National Institute for Health Research Cambridge Biomedical Research Centre (grant number MC_UP_1302/4 to J.M.S.W.), and the Medical Research Council (grant number MC_UP_1302/6 to A.P.M.).
