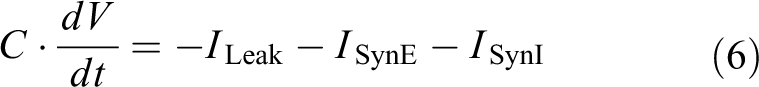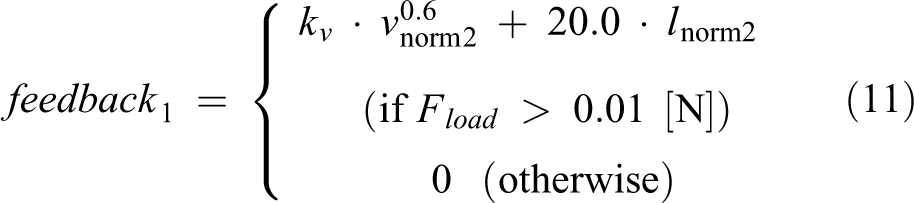Abstract
We aim to design a neuromorphic controller for the locomotion of a quadruped robot with muscle-driven leg mechanisms. To this end, we use a simulated cat model; each leg of the model is equipped with three joints driven by six muscle models incorporating two-joint muscles. For each leg, we use a two-level central pattern generator consisting of a rhythm generation part to produce basic rhythms and a pattern formation part to synergistically activate a different set of muscles in each of the four sequential phases (swing, touchdown, stance, and liftoff). Conventionally, it was difficult for a quadruped model with such realistic neural systems and muscle-driven leg mechanisms to walk even on flat terrain, but because of our improved neural and mechanical components, our quadruped model succeeds in reproducing motoneuron activations and leg trajectories similar to those in cats and achieves stable three-dimensional locomotion at a variety of speeds. Moreover, the quadruped is capable of walking upslope and over irregular terrains and adapting to perturbations, even without adjusting the parameters.
Keywords
Introduction
The existence of central pattern generators (CPGs) in an animal’s spinal cord and their contribution to generating basic locomotion rhythms, even without afferent sensory feedback and control signals from the brain, are well documented. 1 Based on the findings, a variety of mathematical CPG models have been proposed for quadrupedal locomotion. 2 –8 A CPG, which is a nonlinear oscillator, can autonomously adjust the phase differences between the four legs of a quadruped robot and its duty factor by receiving feedback from sensory information. Therefore, a quadruped robot with a CPG can safely locomote over irregular terrain without explicitly changing its leg trajectory, 9 which is a common method for quadruped robots without CPGs. Many mammalian quadruped simulated models 10 –13 and robots 9,13 –22 with CPG models have thus successfully achieved quadrupedal locomotion. They made significant contributions to recognizing the value of CPG for quadrupedal locomotion; however, the mechanisms and nervous systems of the quadrupeds were fairly simplified to allow them to safely locomote. For examples, each joint was driven not by plural muscles as for animals but by a single mechanical actuator, such as a motor or a bidirectional linear actuator, leading to fewer total actuators. The relatively simple CPG models were therefore sufficient to control all the actuators.
Since muscles have elasticity, which typical mechanical actuators do not have, when a practical artificial muscle actuator is developed in the future, we can expect to improve the energy efficiency and running speed of quadruped robots by exploiting this elasticity. Moreover, if the robot has two-joint muscles, it can further improve its energy efficiency. 23 However, to allow a quadruped robot with the realistic legged mechanisms driven by plural muscle actuators to achieve safe locomotion in the future, we should design more realistic neural controller capable of synergistically driving its plural muscles by using more realistic CPGs. To do this, we build a simulated cat model by equipping each leg with three joints driven by six muscle models incorporating two-joint muscles (24 muscles in total) controlled by neuromorphic two-level CPGs and allow it to safely locomote in three-dimensional (3-D) space. We model it on a cat because cats are often used for animal testing in locomotion, such as in decerebrated cat walking, 24 and there are many relevant published experimental data available. 24 –32
In legged locomotion of an animal, the phases of the leg movement in a cycle are typically divided into four phases: swing, touchdown, stance, and liftoff, which are sequentially repeated (Figure 1). The leg form in each sequential phase is generated when the different set of multiple muscles are synergistically activated. 25 In particular, there are durations when flexors, extensors, and two-joint muscles are simultaneously activated in each sequential phase, forming a realistic leg trajectory in a cycle. To achieve this animal-like kinematic movement in each leg driven by multiple muscles, the following two conditions should be satisfied:

A diagram of the four sequential phases of a leg in a cycle. The bold blue lines represent activated muscles, and the thin lines represent relaxing muscles. The different set of multiple muscles are synergistically activated in each sequential phase.
Phase transition: The four sequential phases must be switched with appropriate timing.
Synergistic activation of multiple muscles: In each sequential phase, the different set of multiple muscles must be synergistically activated.
In current neuroscience literature, CPG is divided into multiple layers, and the two-level CPG model is proposed with two hierarchical levels of a rhythm generation (RG) part and a pattern formation (PF) part. 33 RG and PF parts are mainly responsible, respectively, for conditions (A): phase transition and (B): synergistic activation of multiple muscles. Since the CPGs used in the simple quadruped models and robots 9 –22 are not explicitly split in the two layers of RG and PF, they have difficulty with being simultaneously responsible for both (A) and (B). We believe a more realistic CPG model is required to satisfy the two conditions and achieve 3-D locomotion. Thus, we propose a cat-like neuromuscular model incorporating two-level CPG enabling 3-D quadrupedal walking.
Some representative studies have achieved realistic animal-like legged locomotion by synergistically driving multiple muscles in each leg in simulation 34 –40 and in robots, 41,42 some of which 36,37,39,40,42 use two-level CPGs designed with neural networks consisting of realistic neuron models, similar to our work. However, all of the related works were limited to biped walking on either forelimbs 37 or hind limbs, 34,36,38 –42 and their motions were constrained to the sagittal plane, except for Ekeberg and Pearson’s 35 3-D biped model with two immovable forelegs sliding on the ground surface without friction.
Our aim in this article is to suggest a minimal nervous system to enable a cat-like muscle-driven quadruped model to three-dimensionally walk even in uneven terrain, where stability is crucial. This article’s main contribution is the improvement of existing components and their appropriate integration to enable 3-D locomotion of a muscle-driven quadruped model, which has not yet been achieved even in simulation, let alone in any robot. The improvement and integration in our quadruped model are specified as follows: We reproduce the synergistic patterns of muscle activity in each sequential phase by assembling the PF part in unique and simple ways to satisfy condition (B) and form the appropriate motions in the four phases. We set the passive elastic element Fp
of each muscle so that it can both roughly control each leg’s trajectory and self-stabilize each support leg without any particular control. We stabilize rolling motion with a non-preprogrammed walk gait (i.e. the phase differences between the four legs are not explicitly determined by our program, but the gait results in a walk) that is autonomously generated when the phase differences between the four legs are adjusted via leg loading feedback to the RG part, despite the CPG network being hard-wired. We facilitate the tuning of a large number of parameters through three procedures, including a genetic algorithm (GA).
Each of these features is discussed in the “Discussion” section, but here we briefly explain how the four features are different from the features of existing models. Regarding No. 1, we expand the PF part of a two-level CPG originally proposed by Markin et al., 33 which was designed to move a single-joint limb. Markin et al. 40 also expanded the same original model 33 in a different way to achieve hind leg locomotion in simulation, but their expanded PF part was so complex that the CPG of each leg had 32 neurons, whereas our CPG has only 12 neurons, because the expanded PF part is simpler. Despite its simplicity, our CPG is capable of robustly adapting to 3-D quadruped locomotion with perturbations (“Robustness test when changing Fp ” section) and over uneven terrain (“Irregular terrain walking” section).
Regarding No. 2, Fp is also used in related works, 34 –40 but they never discuss effective setting of the equilibrium point and stiffness in Fp . We demonstrate their importance for stable 3-D quadruped locomotion in simulation, especially in perturbed locomotion (“Robustness test when changing Fp ” and “Irregular terrain walking” sections).
Regarding No. 3, leg loading feedback to the RG part is applied in related works, 34 –42 but it is novel that we find that this element causes a non-preprogrammed walk gait in a 3-D environment (“Flat terrain locomotion with afferent feedback” section) and that this gait results in stable locomotion even over uneven terrain (“Irregular terrain walking” section).
Regarding No. 4, our CPG model is simpler than other closely related two-level CPG models, 36,37,40 but it still has a large number of parameters. Therefore, we propose three procedures, including a GA, which is not used in related works, 36,37,40 to facilitate the tuning (“Parameter tuning” section). In addition, we test the parameter values on an artificial muscle-driven 3-D quadruped robot and demonstrate that they work relatively well (see section S1 in Supplementary text for the results).
In this article, we present simulation results of successfully achieving stable 3-D quadrupedal walking, in which recorded motoneuron activations and produced leg trajectories are similar to those of a cat walking. Moreover, even without adjusting the parameters, the simulated quadruped is capable of walking upslope and over irregular terrains and adapting to perturbations. It can also safely walk at a variety of speeds by changing a tonic descending input to CPG and a couple of parameters to change muscle strengths. We thus design a biomorphic neural controller for the locomotion of a quadruped model with realistic muscle-driven leg mechanism. We believe the results contribute to proposition of the control method for the locomotion of a biomorphic quadruped robot with animal-like joint configuration and artificial muscle actuators.
Related works with neuromorphic two-level CPG models
We here describe the simulated models and the robot that used two-level CPGs in the aforementioned related works, 36,37,39,40,42 which succeeded in producing animal-like realistic stepping motions by controlling the muscle-driven leg mechanism, and explain the advantages of our two-level CPG model.
Several studies (simulations by Hunt et al., 39 Maufroy et al. 36,37 and Markin et al. 40 and a robot by Hunt et al. 42 ) have thus focused on the neuromorphic two-level CPG, which was designed with neural networks consisting of realistic neuron models, capable of simultaneously satisfying conditions (A) and (B) to reproduce a realistic leg movement. Maufroy et al.’s 37 simulated model only had forelegs and the others only 36,39,40,42 had hind legs, and all performed planar locomotion.
Hunt et al.’s 42 robot called Puppy had hip, knee, and ankle joints in each leg, and each joint had an antagonistic pair of artificial muscle actuators (without two-joint muscles). Puppy’s CPG models were simplified versions of two-level CPG models proposed by Zhong et al. 43 Muscle length and loading feedback to the CPGs were used. The CPG synergistically activated and drove a different set of artificial muscle actuators in each of the four sequential phases. However, the single CPG was put on each joint and there was no CPG to activate would-be two-joint muscles, which are important for fast and energy-efficient locomotion, as mentioned in the “Introduction” section. The researchers also developed a rat simulation model using the same method to perform hind leg walking. 39
Maufroy et al. 36,37 and Markin et al. 40 used a two-level CPG in a more realistic way for each leg. Maufroy et al. 36 demonstrated the planar walking of cat-like hind legs in simulation. Each leg had three joints driven by seven muscle models. The neural phase generator (NPG) proposed by Wadden and Ekeberg 44 and a motor output shaping stage (MOSS) were used as RG and PF parts, respectively. The NPG and MOSS received a variety of sensory feedback of muscle length and loading. However, since their model was very complex (i.e. each leg’s CPG consists of 73 neurons, whereas our CPG has 12 neurons), parameter tuning was a problem, even for hind leg walking. It may be difficult to use their model for a quadruped robot. The researchers also succeeded in biped walking of the forelegs, using the same method. 37
The most similar two-level CPG to ours was Markin et al.’s model. 40 Each CPG manipulated three joints driven by nine muscle models in each leg, allowing the simulated hind legs of a cat to walk in the sagittal plane. Similar to our CPG, this two-level CPG was an extended version of a CPG for driving a single-joint limb that was proposed by Markin et al., 33 as mentioned in the “Introduction” section (see “Design concept of two-level CPG” section for the differences). Each CPG received a variety of sensory feedback on muscle length and loading. They built a finely designed hind leg model based on biological findings because of their stated aim from a physiological standpoint that “The model can be used as a testbed to study spinal control of locomotion in various normal and pathological conditions.” 40 Therefore, similarly to Maufroy et al.’s model, 36,37 Markin et al.’s model 40 may be too complex to be used as a locomotion controller for a quadruped robot (i.e. each leg’s CPG consists of 32 neurons, whereas our CPG has 12 neurons).
Methods
Mechanism of a simulated quadruped model
We used a dynamic simulator called Webots,
45
which has been used in many locomotion studies. Figure 2 shows the simulated quadruped model, Nyanko, and its diagram. The head, torso, and tail are united in a single rigid body. The lengths of the leg segments were determined by being modeled on a cat in Reighard and Jennings’s work.
26
The model weighs 6.0 kg. Each foreleg has scapula, shoulder, and elbow joints, and each hind leg has hip, knee, and ankle joints. All the joints turn around the pitch axis. Each leg is driven by six muscles as shown in Figure 2(b). The length and fitted position of each muscle were approximately determined based on those of each main muscle in the anatomical figures of cats.
25,26
Nyanko has neither joints around the roll nor the yaw axis. We found in our test that the six muscles in each leg at least enabled the leg to reproduce the kinematic movements for the four sequential phases. We also plan to apply our method to a quadruped robot with artificial muscle actuators in future studies, so with its mechanical design in mind, we limited the number of muscles so that we could equip the robot with actuators that do not rub against one another. We used a muscle model proposed by Brown et al.,
27
similar to related works.
33
–38,46
The model was constructed based on soleus muscles, but it could be used for other muscles, according to their findings.
47
The output force is

A quadruped model “Nyanko” built in a simulator (a) and its diagram (b).
where
where
We used the following equation of passive force Fp
proposed by Brown et al.
27
as used in the related works
33,35,46
except for using the adjustable parameters of
Each muscle has different values of
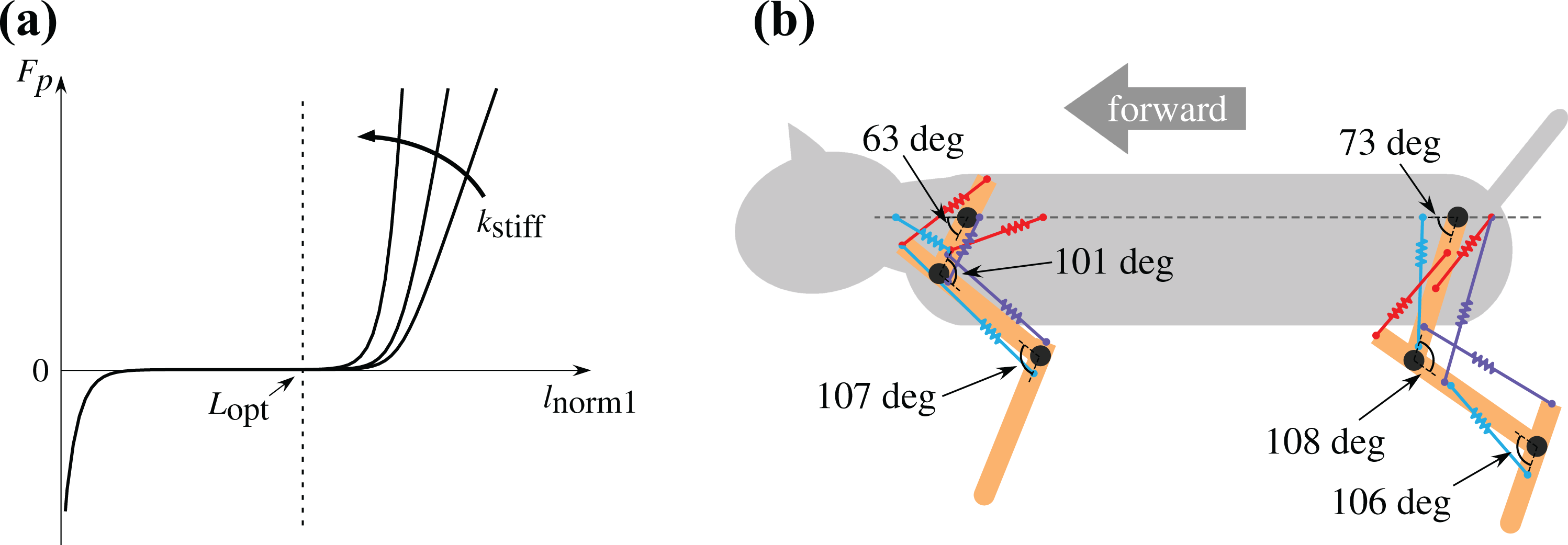
Each muscle’s passive elastic element and its effect on Nyanko. (a) Graph of the passive elastic element Fp (equation (4)) of each muscle. l norm1 indicates a normalized muscle length given by l norm1 = l/L opt (l is a muscle length, and L opt is the optimal length of the muscle when Fl = 1). The stiffness of the muscle is adjusted by k stiff. (b) An example of the equilibrium leg position of Nyanko during locomotion. Each spring shows the passive elastic property of the corresponding muscle.
Neural system
Design concept of two-level CPG
A key problem is the construction of a two-level CPG model for each leg allowing for 3-D quadruped locomotion. Our CPG model extends the two-level CPG model proposed by Markin et al., 33 which was divided into the RG and PF parts. We call this basic model the Rybak CPG model. Since the Rybak CPG model was designed to move a single-joint limb, as shown in Figure 4(a).

Three kinds of two-level CPG models. (a) The Rybak CPG model proposed by Markin et al. 33 to move a single-joint limb. (b) Expanded version of (a) by Markin et al. 40 that is designed to move a simulated hind leg model with multiple joints modeled on a cat. (c) Expanded version of (a) by the current authors that is designed to move a simulated cat quadruped model whose leg consists of multiple joints. E, F, and M denote neurons for extension, flexion, and motoneurons (neural populations, to be exact), respectively. PF-{hip, knee, ankle, two-joint} in (b) represent PF circuits for the hip, knee, ankle joint muscles, and the two-joint muscles, respectively. PF{sw, td, st, lo} in (c) represent PF neurons for the swing, touchdown, stance, and liftoff phases, respectively. Excitatory and inhibitory synaptic connections are represented by arrows and small black circles, respectively. For simplicity, we omitted some details from these figures (e.g. feedback and interneurons through which E and F in each model are mutually inhibited). CPG: central pattern generator.
The organization of the PF part to synergistically activate a different set of multiple motoneurons is still unrevealed in biology. To address this issue, Markin et al. 40 expanded the PF part of the Rybak CPG model 33 as shown in Figure 4(b) to drive the hind leg models with multiple joints modeled on a cat. Their improved CPG model has three PF circuits for the three hip, knee, and ankle joints, each of which activates the motoneurons of the muscles to drive the corresponding joint. The model has another PF circuit that activates the two motoneurons to drive the two-joint muscles of a biceps femoris posterior and a rectus femoris, which are partially active during both flexor and extensor phases. Thus, the model has four PF circuits. Each PF circuit has a half-center form consisting of an extensor neuron and a flexor neuron inhibiting each other.
On the other hand, as shown in Figure 4(c), our proposed two-level CPG model uses the four sequential PF neurons PFsw, PFtd, PFst, PFlo to make the phase patterns of swing, touchdown, stance, and liftoff, respectively. Each PF neuron does not form a half-center, but a single neuron, resulting in the four simple PF neurons in the PF part. The PF part in Figure 4(c) has only 20 parameters, whereas that in Figure 4(b) has 74. However, our CPG model was capable of generating realistic motoneuron activation to reproduce the leg movements in the four sequential phases. Other advantages are discussed in the “Discussion” section.
Neuron models and their connection in two-level CPG
Figure 5 shows a detailed diagram of our CPG model. The model of each neuron itself used in our CPG model is similar to the neuron models in the Rybak CPG model.
33
However, the number of neurons and the neural connections are completely different, as shown in Figure 4(a) and (c). The membrane potential of the extensor and flexor neurons in the RG and PF parts (RG-E, RG-F, PFsw, PFtd, PFst, PFlo in Figure 5) and the motoneurons (Mn1–Mn6 in Figure 5) is expressed as

Detailed diagram of the proposed two-level CPG for each leg. This CPG is divided into the two layers of RG and PF parts. The RG part consists of half-center rhythm generators, including RG-E (RG neuron for extension) and RG-F (RG neuron for flexion). The PF part consists of the four PF neurons, where PF{sw, td, st, lo} are PF neurons for the swing, touchdown, stance, and liftoff phases, respectively. Mn{1−6} show motoneurons to activate the corresponding muscles. Inab-E shows an excitatory interneuron, and each In shows an inhibitory interneuron. All neurons and interneurons are represented by large and small spheres, respectively. Excitatory and inhibitory synaptic connections are represented by arrows and small circles, respectively. CPG: central pattern generator; RG: rhythm generation; PF: pattern formation.
and the membrane potential of the interneurons (In and Inab-E in Figure 5) is expressed as
This activity-based neuron model was also used in the aforementioned related works of Hunt et al.
39,42
and Markin et al.
40
In these equations,
where
In equation (7), the voltage-dependent activation variables for the potassium delayed rectifier and persistent sodium channels are represented as
respectively. The voltage-dependent inactivation variables
in which
where
where

Outline of the CPG network. Each dashed line oval indicates a CPG. RG-E and RG-F represent the RG neurons for the extension and flexion, respectively. The small black circles indicate inhibitory synaptic connections. The weights α and β represent the connection strengths between the contralateral neurons and ipsilateral neurons, respectively.
Tables S4, S5, and S6 (in Supplementary tables) show the parameters in equations (5
) to (10) used for our simulation. The weights (
Sensory feedback
Ia afferent feedback to extensor motoneurons as a stretch reflex is effective to support weight; 34,40 thus, we applied the Ia afferent feedback used in Yakovenko et al.’s simulation. 34 Specifically, the following feedback signals from the scapula and hip extensors are input to the corresponding motoneurons when the leg is loaded
where
where l is a muscle length, and
Studies have noted the importance of leg loading feedback to CPG. 24,28 In our CPG model, the following feedback was input to the RG flexor to make it inhibited (Figure 5), based on Pearson’s reports 24
where
Parameter tuning
As described in the “Related works with neuromorphic two-level CPG models” section, our CPG model for each leg is simpler, with far fewer parameters than in the most closely related works,
36,37,40
but it still has a large number of parameters. However, we found that the parameters in each part of our CPG had a particular feature. This could help tune all the parameters. Specifically, the connection parameters between the RG and PF neurons were responsible for making a basic rhythm. The connection parameters between the PF neurons and motoneurons mainly took care of creating the leg trajectories in the four sequential phases. The stiffness parameter We suspended Nyanko with its legs swinging in the air and implemented a rough manual tuning of the parameters of the CPGs and the muscle models so that each leg could reproduce a loose trajectory in each of the four sequential phases and steadily oscillate. Specifically, we first determined the connections between the RG and PF neurons to reproduce the basic rhythm. Secondly, we focused on exploring the connections between the PF neurons and motoneurons to roughly reproduce the synergistic patterns of muscle activity in each of the four sequential phases. We put Nyanko on the ground and tuned a variety of parameters using a GA so that Nyanko could walk on flat terrain. We manually modified the parameters so that Nyanko’s motoneuron activation patterns and leg trajectories were close to those of cats.
To alleviate the complexity of tuning so many parameters, we left the majority of the tuning to GA. Specifically, we trained 76 parameters altogether, including the muscle stiffnesses
We used real-coded GAs with BLX-α. 49 The initial population in this GA comprised 300 individuals, which was randomly generated near the values of the parameters tuned in the aforementioned (I). For the population of the next generation, the best two individuals in the current population were kept as elite individuals, and the others were renewed by BLX-α with a roulette wheel selection with a 100% crossover rate between the individuals, a 30% crossover rate of the loci, and a 200% expansion rate of the hyperrectangle. The fitness function is given by
where t in seconds is a continuous locomotion duration at speeds of no less than 0.05 m/s,
Results
Suspended locomotion
We allowed suspended Nyanko to locomote in the air to roughly create basic kinematic leg movements for locomotion. We did not use the afferent feedback of equations (11) and (13) in this suspended locomotion.
Initially, to examine whether our two-level CPG could reproduce the four sequential leg motions with only the muscles’ force generated by the CPG outputs, we excluded the muscle passive elastic element Fp
in equation (1) and manually tuned the parameters of the CPG (
Then, applying Fp
along with the initial manual tuning of its parameters (
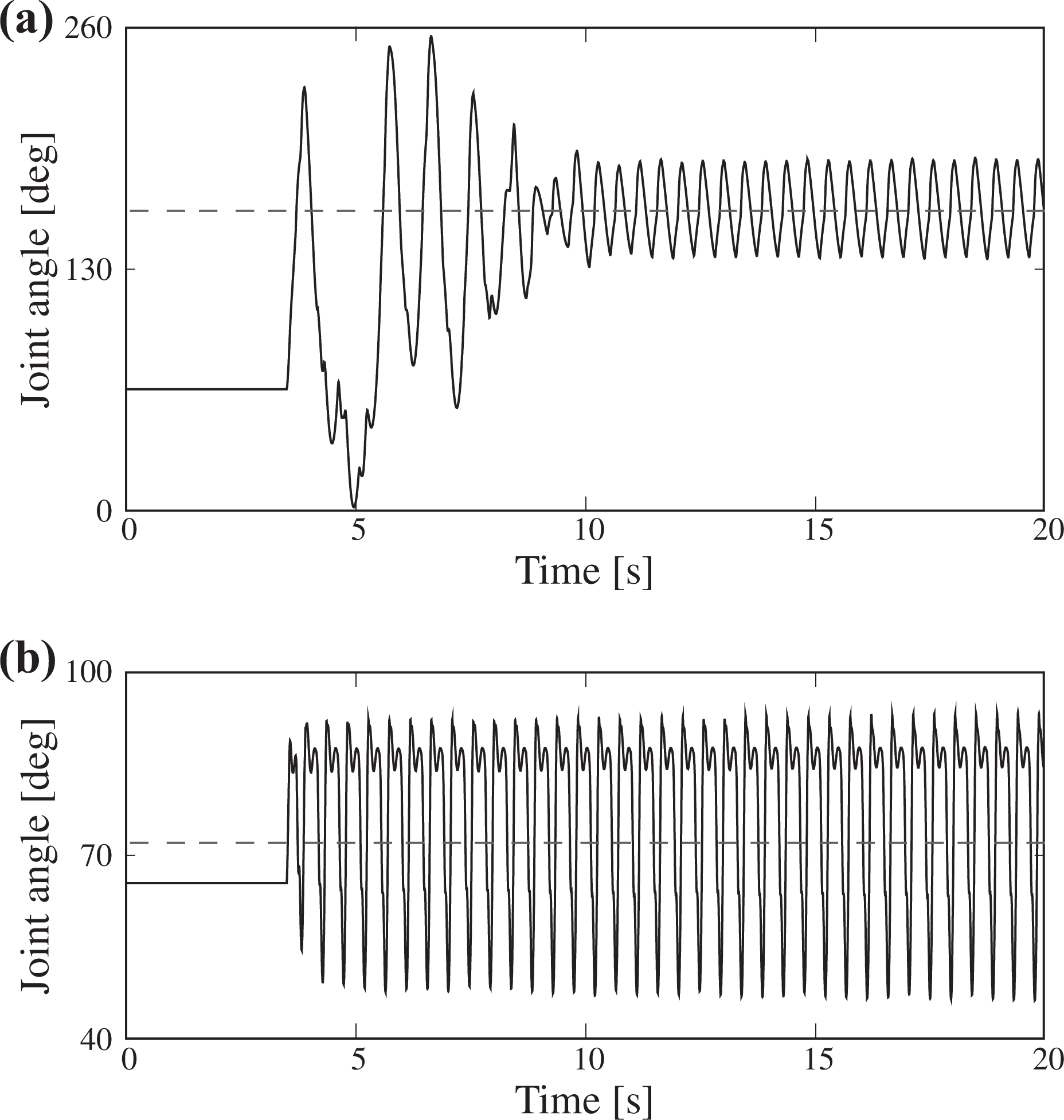
Graphs of the angles of the left hip joint while Nyanko used in the “Flat terrain locomotion with afferent feedback” section was suspended (a) without and (b) with the muscle passive element Fp for swinging the four legs. The origin and orientation of the joint angles are shown in Figure 2. The dashed line shows the center of the oscillation.
This suspended locomotion helped us to roughly design the CPG and muscle models of each leg, especially with respect to the PF part reproducing the leg trajectories in the four sequential phases and Fp
making possible the steady leg oscillations. At this stage, since we loosely targeted the steady rhythmic leg movements through the four sequential phases, we did not focus on quality (e.g. the magnitude of leg oscillation, the ratio of time between the four sequential phases, or the optimal value of each
In terms of Nyanko’s gait, a default trot gait emerged in the suspended locomotion via the hard-wired CPG network, which was the mutual inhibitory connections between the lateral neighboring CPGs.
Flat terrain locomotion without afferent feedback
After the suspended locomotion (“Suspended locomotion” section), we allowed Nyanko to locomote on flat terrain by using the controller designed in the suspended locomotion, which did not incorporate the afferent feedback of equations (11) and (13). Then we used real-coded GAs with BLX-α
49
to find parameter values with which Nyanko could locomote steadily. We tuned 76 parameters (
Next, we allowed Nyanko to have longer strides, but after taking several steps, Nyanko collapsed because Nyanko failed the timing of the transition between the support phase and the nonsupport phase. Specifically, as shown in Figure 8(a), even while the foot touched the ground, RG-F started bursting (A) and forced the support leg to lift, and the foot touched the ground early even though RG-F was still bursting (B). In addition, each support leg did not always trace the same trajectory in every cycle and wobbled. Note that at this stage, neither afferent feedback of equations (11) nor (13) were used.

Graphs of the RG-F outputs (black solid lines) and footfalls (bold line segments at the bottom) of left hind leg without any afferent feedback (a) and with leg loading feedback to CPG (b). CPG: central pattern generator; RG: rhythm generation.
Flat terrain locomotion with afferent feedback
We applied the leg loading feedback (equation (13)) to the RG part to solve the abovementioned problem in terms of the timing of transition between the support and nonsupport phases. As a result, as shown in Figure 8(b), the feedback prevented RG-F from being activated while the foot touched the ground, and the loaded leg was therefore kept in the support phase for the right amount of time.
In addition, the proper use of each muscle’s passive elastic element Fp
was capable of letting the stance leg to be controlled to the equilibrium position shown in Figure 3(b) to some extent as self-stabilization. In particular, properly setting the equilibrium position (
We observed that adding the Ia afferent feedback of equation (11) allowed the body oscillation to be more stable.
Finally, we manually modified Nyanko’s parameters (d,

Outputs from the RG and PF neurons and the motoneurons (Mn) in (a) the left foreleg and (b) the left hind leg while Nyanko walked safely on flat terrain. The enclosed dashed rectangle in the Mn signals indicates one cycle duration, and the corresponding muscles’ EMG activations of a cat in the same duration are shown on the right. The cat data in (a) and (b) are from Cabelguen et al. 29 and Goslow et al. 30 , respectively. RG-F and RG-E represent RG for flexion and RG for extension, respectively. PFsw, PFtd, PFst, and PFlo represent PF for the swing, touchdown, stance, and liftoff phases, respectively. RG: rhythm generation; PF: pattern formation.

Joint data while walking on flat terrain. Each joint angle of (a) the left foreleg and (b) the left hind leg in one cycle while Nyanko walked safely on flat terrain, compared with those of cats (Meché 31 for forelegs, Rasmussen et al. 32 for hind legs) at almost the same speeds. The origin and orientation of each joint angle are shown in Figure 2. The bold lines at the bottom represent the footfalls. (c) A phase plane trajectory of the left hip joint while Nyanko walked safely on flat terrain. The horizontal and vertical axes show, respectively, the angle and angular velocity of the left hip joint. The origin and orientation of the joint angle are shown in Figure 2.

Load of each leg while Nyanko was safely walking on flat terrain. The four legs lift in the order the left foreleg (A), the right hind leg (B), the right foreleg (C), and the left hind leg (D), which was a lateral sequence walk.
It can be seen in Figure 9(a) and (b) that PFsw, PFtd, PFst, and PFlo properly activated in the swing, touchdown, stance, and liftoff phases, respectively, in both the fore and hind legs. Therefore, the motoneurons of the six muscles synergistically activated in each sequential phase. In particular, there are periods when the flexors, extensors, and two-joint muscles are simultaneously activated in each sequential phase. For example, in the touchdown phase, which is an intermediate phase between the swing and stance phases, while the scapula and elbow flexors were still bursting (A in Figure 9) (i.e. before the scapula extensor started bursting (B)), the shoulder extensor and the two joint muscles for the shoulder and elbow were strongly burst (C). By simultaneously activating the flexors, extensors, and two-joint muscles in each leg, Nyanko enabled the motion of the foot touching forward to the ground while extending the leg. Similarly, in the liftoff phase, while the scapula extensor was still activating (D) (i.e. before the scapula flexor started bursting (E)), the two-joint muscles for the scapula and shoulder and the elbow flexor were strongly activated (F), thus enabling the leg to retract up and backward, and the tip of the leg was prevented from catching the ground at the following swing phase. The same tendencies of the activations can be seen in the hind leg (Figure 9(b)). In Figures 9(a) and (b), the enclosed dashed rectangle in the left graph indicates one cycle duration, and the corresponding muscles’ activations of a cat in the same duration are shown on the right. The formats of the cat data of (a) and (b) are different because we were unable to find data of a cat’s muscles corresponding to all the muscles used in Nyanko from biological references. There is a certain amount of variation in the data for cats in biological papers, especially in the magnitude of EMG activations, because of measurement error and individual differences. Therefore, we only compare the timing of the EMG activations between the data for Nyanko and cats. It can be observed in Figures 9(a) and (b) that each muscle in Nyanko and a cat is activated and deactivated at similar times in one cycle. The features of Nyanko’s and the cat data are similar in each leg, even though Nyanko did not copy all the muscles in a real cat.
The leg trajectories in Figures 10(a) and (b) are fairly close to those of cats. There was a slight difference between the data of Nyanko’s and the cats. The speeds of the cats and Nyanko were almost the same, but the cyclic duration of Nyanko (0.46 s) was a little shorter than that of the cats (0.64 s). In other words, Nyanko lacked speed in relation to the short cycle compared to real cats. We suggest that one of the reasons is that there are fewer degrees of freedom in Nyanko than there are in a cat because Nyanko does not have hand, toe, and pelvis segments, the spine is rigid and does not bend, and the scapulae do not move linearly. Specifically, in cats, the feet touch the ground before the end of the cycle (A in Figure 10(a) and (b)) because the hand and toe segments continue touching the ground until the end of the cycle. However, Nyanko’s legs without the hand and toe segments left the ground earlier (B), and Nyanko must have slightly lost propulsion.
For Nyanko without leg loading feedback in the “Suspended locomotion” and “Flat terrain locomotion without afferent feedback” sections, a trot gait was originally produced by the hard-wired CPG network, which is the inherent connections between CPGs. In the simulation in this section, leg loading feedback differentiated the timing of the foot lifting from the ground between the two legs of each diagonal pair, which would be in-phase while trotting. This differentiation led to the phase differences between the two legs of each diagonal pair, autonomously resulting in a non-preprogrammed walk gait, as shown in Figure 11. Specifically, the left foreleg (A) lifted earlier than the right hind leg (B) in the first diagonal pair, and the right foreleg (C) lifted earlier than the left hind leg (D) in the second diagonal pair. This was because the body swayed sideways at low speeds, due to longer stance durations, than during trotting at high speeds. This ended up autonomously causing a lateral sequence walk, in which the four legs lifted in the order left foreleg (A), right hind leg (B), right foreleg (C), and left hind leg (D), as shown in Figure 11. In other words, the autonomous gait generation of a walk caused by leg loading feedback seems to have eventually stabilized the locomotion with long stride.
Tables S2–8 (in Supplementary tables) show the values of the optimally tuned parameters for the stable walking. λ in equation (1) was 1.0 in this section’s locomotion.
Adaptation to speed variation
The tonic descending signal to the CPG (d in equation (7)) was constant while locomoting at the constant speed in the “Flat terrain locomotion with afferent feedback” section, but it is known that changing the signal allows an animal to vary the speed. 1 We tested at what range of speeds Nyanko can safely locomote either by only changing the descending signal d or by changing it along with other parameters.
Both results are shown in Figure 12. First, we changed the descending signal d around a value of 2.05, which was the value used in the “Flat terrain locomotion with afferent feedback” section, at a gradient of 0.083 per second, as shown in the dashed line in Figure 12(a). In the trial, Nyanko was capable of walking at speeds of between 0.55 and 0.72 m/s, as shown by the solid line. The figure indicates that the descending signal is constant at the beginning and at the end because Nyanko was unable to stand with the wider variation of the signal.

Speeds when Nyanko successfully walked (a) by only changing the descending signal to CPG (d in equation (7)) and (b) by adjusting the parameters (λ in equation (1) and k stiff in equation (4)) determining muscle strength in addition to the descending signal. The dashed lines represent the descending signals d. The solid lines represent walking speeds. CPG: central pattern generator.
Next, we empirically adjusted the parameters (λ in equation (1) to adjust the level of the output
Robustness test when changing Fp
Since we found in the “Flat terrain locomotion with afferent feedback” section that the passive elastic element Fp
of each muscle played an important role in self-stabilizing the support leg, we decided to perform a simulation comparison to specify the effect. We prepared two Nyanko’s with low and high levels of Fp
(i.e. in each muscle,
In the upslope walking simulations, Nyanko with low Fp could only walk up a slope of up to 1°; whereas, steady walking was observed in Nyanko with high Fp up to 9°. When Nyanko with a high Fp almost fell backward, the elastic force of Fp in the scapula and hip extensors mechanically and passively returned the support legs to the equilibrium positions. Although all the parameters were the same as when Nyanko walked on flat terrain, while locomoting upslope, the locomotion cycle was somewhat longer, and the gait was a trot at steep slope angles as observed in the supplementary video. The trot was observed because there were few differences of leg loads between the two legs of each diagonal pair through less body oscillation while locomoting upslope, and eventually each diagonal pair of legs became in-phase. The autonomous variation of Nyanko’s gait according to its locomotion states because of leg loading feedback is a main feature of our quadruped model.
For robustness tests against perturbations, we observed Nyanko’s stability when an object hit either the front end or the right side of its body. Specifically, using a pendulum with the object suspended by a 0.5 m massless string, we naturally released the object when the string was horizontal and hit Nyanko with the object. While Nyanko with a low Fp fell over when we hit a 0.5 kg object to its front end and a 0.3 kg object to its side, Nyanko with a high Fp could adapt to up to a 2.0 kg object hitting its front end and up to a 1.0 kg hitting its side.
Based on these results, Fp managed the stable locomotion well because the support legs self-stabilized around the equilibrium position (Figure 3(b)) and were capable of propping the body up in a sturdy way.
Irregular terrain walking
Walking over a step
Figure 13 shows the phase plane trajectories of the left hip joint when Nyanko walked over a step 30 mm high, 500 mm wide, and 600 mm deep. The time was 0 s at the moment the first front leg landed on the step. It took Nyanko approximately 2 s to walk over the step. The model wobbled somewhat 5–10 s after passing the step, but gradually became stable, as shown in the trajectory for 10–15 s. Nyanko was not able to walk over a step higher than 35 mm because of its foot tripping over the side of the step during the swing phase. No matter how wide and deep the step was, however, Nyanko succeeded.

Phase plane trajectories of the left hip joint after Nyanko walked over a step (30 mm high, 500 mm wide, and 600 mm deep). The horizontal and vertical axes show the angle and angular velocity of the left hip joint, respectively. The origin and orientation of the joint angle are shown in Figure 2. The red, green and blue lines show trajectories in 0–5 s, 5–10 s, and 10–15 s, respectively. The trajectory was disturbed while walking over the step (the red line), but it immediately returned close to the stable track (the green) and finally returned much closer to the stable track (the blue line).
Walking with the left feet landing on ground of a higher level
Figure 14(A) shows how the body tilted around the roll axis when Nyanko walked with its left feet landing on a step 10 mm high (the body was tilted rightward (positive) after 3 s) after walking on the flat ground for the first 3 s. Figure 14(B) and (C) show its footfalls while it was walking on the flat terrain and then with the left feet landing the step, respectively. Each of (a), (b), (c), and (d) in Figure 14(B) and (C) represents the length of the period of each footfall in (B), for the purposes of comparing (C) with (B). While Nyanko was walking with its left feet landing on the step, as shown in Figure 14(C), the stance durations of both right legs were longer and those of both left legs were shorter than when it was walking on the flat ground (Figure 14(B)). This result showed that the stance duration of each leg on the side toward which the body tilted in the lateral plane was extended, and the reverse adaptation happened on the other side, because of the leg loading feedback, and the model eventually successfully avoided falling rightward. In addition, the gait was close to a pace while walking with the left feet were landing the step, as shown in Figure 14(C). Thus the gait was autonomously adjusted according to the walking terrain.

Graphs when Nyanko walked with its left feet landing on a step 10 mm high. (A) How the body tilted around the roll axis (positive when tilting rightward). (B) and (C) Its footfalls it was walking on the flat terrain and then with the left feet landing the step, respectively.
We conducted the uneven-terrain locomotion because we wanted to show that Nyanko had an autonomous adaptability to a certain level of unevenness in the terrain without changing any parameters. However, note that our proposed controller is not sufficient to adapt to rougher terrains. For example, when Nyanko walks over terrain where there are a number of randomly placed relatively high steps, we will need to add a control that can adjust the trajectory of the tip height of each leg to maintain the body height, so that the loading will not be biased toward any particular leg.
Discussion
The following four key features enable the quadruped model to locomote in a 3-D environment. A simple configuration of the PF part enabled the proper leg trajectories in the four sequential phases of each leg
Markin et al. 40 developed the PF part divided into four half-center PF circuits (i.e. three respective PF circuits to activate the muscles of hip, knee, and ankle joints and another PF circuit to activate the two-joint muscles) as shown in Figure 4(b) and achieved realistic leg movements on each leg. On the other hand, as shown in Figure 4(c), we designed a PF part that has four single PF neurons (neural populations, to be exact) to make the four sequential phase patterns of swing, touchdown, stance, and liftoff, respectively. Despite the simple configuration, precise leg movements in the four sequential phases were performed.
In addition, since each PF neuron is responsible for the individual sequential phase unlike each PF circuit of Markin et al.’s, 40 we expect that it would be easy to induce transition to each phase by adding feedback to the corresponding neuron in the future. For example, if the length-dependent Ia feedback from the scapula/hip flexor excites the PFlo neuron when the scapula/hip is extended too far backward, the feedback could directly and quickly induce the transition from the stance phase to the liftoff phase. This could be a big advantage if we want to add sensory feedback in the future.
Moreover, since each PF neuron inhibits the other PF neurons via the corresponding interneurons in our PF configuration, as shown in Figure 5 (e.g. PFlo inhibits PFst, PFtd inhibits PFst, PFsw inhibits PFlo and PFst, and PFst inhibits PFlo, PFsw, and PFtd), we can avoid erratic simultaneous activations of the PF neurons such as PFsw activating in the middle of the stance phase. This could contribute particularly to irregular terrain locomotion, because random and irregular disturbances tend to be fed back to the CPG via sensory feedback, and the CPG is easily disturbed.
The loose trajectory control of each leg using the preferable setting of the passive elastic element of Fp
The related works of bipedal locomotion
34
–42
mainly suggested the importance of weight bearing and the timing of transition between extensor and flexor activities through afferent feedback, but the passive elastic elements of muscles such as Fp
were arbitrarily determined and their importance was hardly noted. By contrast, we found that since more stability was necessary in 3-D quadrupedal locomotion, leg trajectory control to some extent was vital. Specifically, the control was made possible by appropriately setting the equilibrium length of each muscle
Stabilization of rolling motion by a non-preprogrammed walk gait autonomously generated via leg loading feedback
The related works 34 –42 demonstrated that leg loading feedback played a key role in adjusting the stance duration by contributing to stability in the sagittal plane. In addition, the leg loading feedback to CPG played a pivotal role in stabilizing the balance in the lateral plane in our 3-D locomotion. Specifically, the feedback extended the stance duration of each leg on the side toward which the body tilted in the lateral plane and prevented Nyanko from falling toward that side even during irregular terrain walking, as described in the “Walking with the left feet landing on ground of a higher level” section, even though Nyanko has no joints around the roll axis. In addition, the feedback of different loads between the legs to each CPG modified the default trot, which was the basic gait originally generated by the hard-wired CPG network, and therefore, a walk gait which was not preprogrammed spontaneously emerged (i.e. the phase differences between the four legs in this gait were not explicitly determined by our program, but emerged on their own). In other words, the autonomous gait generation of a walk-through leg loading feedback caused stability in the lateral plane as well as in the sagittal plane, resulting in steady 3-D quadrupedal locomotion. Similarly, in our previous simulation work, 12 a walk gait was autonomously generated because of the foot loading feedback, but the quadruped model’s nervous system and mechanism were too simple and its motion was limited in the sagittal plane. In this article, we demonstrated in the 3-D quadruped model with more realistic nervous system and mechanism that the autonomous generation of a walk gait stabilized locomotion.
Facilitating the tuning of a large number of parameters through three procedures including GA.
We may be able to synergistically drive multiple muscles in the four sequential phases for walking and running by using a simpler controller with fewer parameters. However, if we want to allow our quadruped to achieve a higher level of locomotion and behaviors that real animals can perform in future studies, it is reasonable to build a realistic controller. However, the more realistically we design our model, the more parameters the model will have and the more painstaking the parameter tuning will be. The three closely related works 36,37,40 even manually tuned the parameters. Maufroy et al. 36 pointed out the difficulty in the parameter tuning of their two-level CPGs, and Markin et al. 40 mentioned that special care was taken to tune the model parameters. Our CPG model was simpler than the closely related two-level CPG models, 36,37,40 but it still had a large number of parameters. However, we proposed three procedures that included a GA, to facilitate the tuning (“Parameter tuning” section).
Conclusion
In this article, we aimed to demonstrate 3-D quadruped locomotion in simulation and designed each leg’s two-level CPG model consisting of the RG and PF parts, which can synergistically drive the multiple muscles of each leg. It was generally difficult for a quadruped model with realistic neural systems and muscle-driven leg mechanisms to walk even on flat terrain, but we successfully demonstrated stable 3-D quadruped locomotion, and based on our results, we suggested four important factors for stable 3-D quadruped locomotion: (1) design the simple PF part to reproduce the realistic trajectory of each leg; (2) set the passive elastic element of each muscle to roughly control each leg’s trajectory and to stabilize each stance leg as self-stabilization; (3) stabilize the rolling motion with a non-preprogrammed walk gait; and (4) facilitate the tuning of a large number of parameters through three procedures including GA. In our simulation results with 3-D four-legged locomotion, the leg trajectories and the motoneuron activities were very close to those recorded in cats even compared to the biped locomotion in related works. In addition, our quadruped model was able to change speed and showed robustness when it walked upslope and over irregular terrains and received perturbations.
We are testing whether our proposed controller works on a prototype cat-like muscle driven quadruped robot (see in the section S1 in Supplementary text). We demonstrate that the robot is capable of safely trotting at speeds of 0.91–1.25 m/s on the treadmill. Therefore, we consider that our proposed controller is promising enough for a muscle-driven quadruped robot.
Supplemental material
Supplemental Material, Supplementary_tables - Three-dimensional walking of a simulated muscle-driven quadruped robot with neuromorphic two-level central pattern generators
Supplemental Material, Supplementary_tables for Three-dimensional walking of a simulated muscle-driven quadruped robot with neuromorphic two-level central pattern generators by Yasushi Habu, Keiichiro Uta and Yasuhiro Fukuoka in International Journal of Advanced Robotic Systems
Supplemental material
Supplemental Material, Supplementary_text - Three-dimensional walking of a simulated muscle-driven quadruped robot with neuromorphic two-level central pattern generators
Supplemental Material, Supplementary_text for Three-dimensional walking of a simulated muscle-driven quadruped robot with neuromorphic two-level central pattern generators by Yasushi Habu, Keiichiro Uta and Yasuhiro Fukuoka in International Journal of Advanced Robotic Systems
Supplemental material
Supplemental Material, Title_and_description_for_supplemental_materials - Three-dimensional walking of a simulated muscle-driven quadruped robot with neuromorphic two-level central pattern generators
Supplemental Material, Title_and_description_for_supplemental_materials for Three-dimensional walking of a simulated muscle-driven quadruped robot with neuromorphic two-level central pattern generators by Yasushi Habu, Keiichiro Uta and Yasuhiro Fukuoka in International Journal of Advanced Robotic Systems
Footnotes
Acknowledgments
The authors would like to thank Yoshikazu Mori, Naoji Shiroma, and Kosuke Inoue for their valuable comments and advice. The authors also would like to thank the editors and the anonymous reviewers for comments on earlier version of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by Japan Society for the Promotion of Science (Grant-in-Aid for Scientific Research (C), Grant Number: 18K11489) and Ibaraki University (Life Support Project).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.