Abstract
Background:
Hepatocellular carcinoma prognosis depends on both liver and tumor determinants, especially on maximum tumor diameter, multifocality, and presence of portal vein thrombosis, despite apparently complete tumor removal by resection or liver transplantation.
Aims:
To examine parameters of hepatocellular carcinoma aggressiveness as tumor size increases.
Methods:
A large hepatocellular carcinoma database was examined for trends in serum alpha-fetoprotein and the percentage of patients with macroscopic portal vein thrombosis or tumor multifocality.
Results:
A total of 13,016 hepatocellular carcinoma patients were identified having full tumor and survival data. Of these, 76.56% were male and 23.44% were female, with a median age of 64.4 years. We found that as the maximum tumor diameter increased, there was a significant trend for increased alpha-fetoprotein levels (P<0.001) and an increased percentage of patients with either portal vein thrombosis or tumor multifocality, each P<0.0001. Furthermore, the increases of both alpha-fetoprotein and portal vein thrombosis were proportionately greater than the related maximum tumor diameter increases. These trends of increased alpha-fetoprotein, portal vein thrombosis, and multifocality with increasing maximum tumor diameter had non-linear patterns. Within alpha-fetoprotein and multifocality trends, there were identifiable sub-trends associated with specific maximum tumor diameter ranges.
Conclusions:
The greater fold-increases in alpha-fetoprotein and portal vein thrombosis compared with increases in maximum tumor diameter imply that hepatocellular carcinoma characteristics may change with increasing size to a more aggressive phenotype, suggesting that follow-up tumor sampling might be useful, in addition to baseline tumor sampling, for optimal therapeutic choices to be made.
Introduction
The concept that a 5 cm maximum tumor diameter (MTD) might represent a watershed for liver transplant for single hepatocellular carcinoma (HCC) lesions, after which recurrence rates increased, was introduced with the Milan criteria 1 and has generally withstood the test of time. Subsequently, small MTD increases were also shown to be compatible with prolonged post-transplant survival.2-4 However, it has never been clear how these unchallenged size guidelines could be explained by tumor biology, since at transplant both the tumor and non-tumor underlying liver are removed. Furthermore, although increase in tumor size has been known to be an adverse prognostic factor, it has recently been suggested that size alone may not be so important, but rather other factors may be more important and that these factors might change with increase in MTD, which could explain the relevance of increasing MTD to worse prognosis.2,4 The current study was motivated by the desire to know whether HCC biology, as expressed in tumor factors alpha-fetoprotein (AFP), macroscopic portal vein invasion (PVT), or tumor multi-nodularity/multifocality in addition to MTD, which are all known HCC adverse prognosis factors,5-8 might change with increase in MTD.
Methods
Clinical data collection
We analyzed prospectively collected data in the Italian Liver Cancer (ITA.LI.CA) study group database of 13,016 HCC patients accrued until 2018 at collaborating Italian centers, with full baseline tumor parameter data, including computed tomography scan information on MTD, the number of tumor nodules, the presence or absence of PVT and serum AFP levels; plus complete blood counts; routine blood liver function tests, (total bilirubin, gamma glutamyltranspeptidase (GGTP), alkaline phosphatase (ALKP), albumin, aspartate aminotransaminase (AST), alanine transaminase (ALT); demographics and survival information, as previously reported. 9
Ethics
The ITA.LI.CA database management is compliant with the current Italian legislation on privacy, and the study conforms to the ethical guidelines of the Declaration of Helsinki. All patients provided informed consent to having their data entered into the ITA.LI.CA database with an anonymized identification number. The study design was approved (protocol n. 99/2012/O/Oss) by the Independent Ethic Committee of S. Orsola-Malpighi hospital of Bologna, that operates as coordinating center of the ITA.LI.CA network. In all the remaining centers, data inclusion into ITA.LI.CA registry was approved by the local ethics committees.
Statistical analysis
Patient characteristics were reported as mean ± SD (M ± SD) or as median for continuous variables, and as frequencies and percentages for categorical variables.
Normal distributions of quantitative variables were tested using the Kolmogorov-Smirnov test.
For testing the associations between groups, the Chi-square test for categorical variables was used; when the variables were not distributed normally, the Kruskal-Wallis rank test was used for continuous variables.
The χ2 method for trend was performed to evaluate the trend between categorical levels for PVT positive (PVT(+)) and multifocality (>2). For continuous variable AFP (median), the non-parametric test for trend across ordered groups was used.
The proportion test was applied to evaluate the statistical differences between the parameters as category for each level of ordered MTD (cm).
To evaluate the variation in the increase for the median of AFP or the percentage of PVT(+) and multifocality, the equation of the interpolating line for each modification of variation of the increase was used.
The variation of medians of AFP, percentage of PVT(+) and percentage of multifocality (n>2) in relation to the increase of MTD was calculated both as a percentage variation from the previous value of parameters analyzed as increase in tumor size compared to the reference band. This proportionality factor represents how many times the value of the single factor increased with increasing MTD compared to the first category of MTD.
When testing the null hypothesis of no association, the probability level of error at two tails was 0.05. All the statistical computations were made using STATA, StataCorp. 2019. Stata Statistical Software: Release 16. College Station, TX: StataCorp LLC.
Results
Clinical characteristics of increasing MTD groups
Patient clinical characteristics were grouped according to 3 cm increases in MTD, with a central point of ⩽5 cm or >5 cm (Table 1a). The tumor characteristics of the percentage of patients with PVT, the percentage of patients with tumor multifocality, and the median serum AFP levels all increased with the increase in MTD categories. The increase was most profound for the transition from MTD 2.1–5.0 cm to MTD 5.1–8 cm, being 13.09% of patients with PVT versus 33.44% of patients with (2.55-fold increase); and was 11.42% of patients with multifocality versus 27.67% of patients with PVT (2.42-fold increase); and was a median serum AFP of 17.04 versus 90.0 IU/mL (5.28-fold increase).
Clinical and tumor parameter values subdivided by MTD categories.
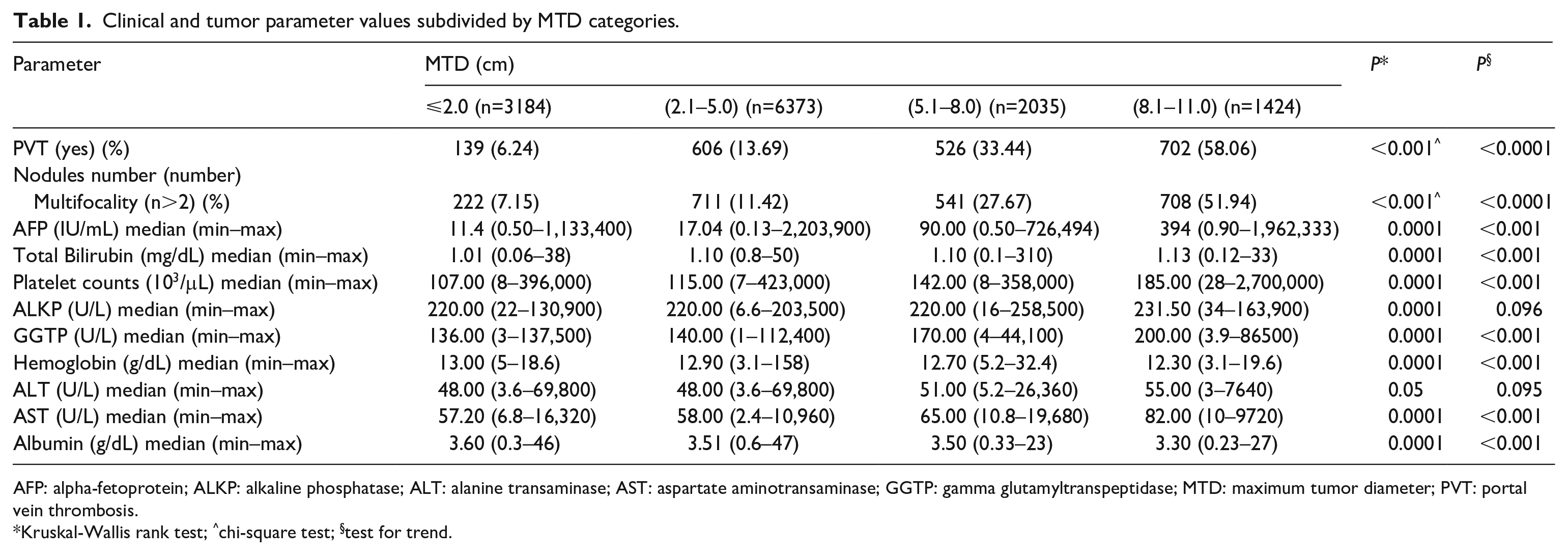
AFP: alpha-fetoprotein; ALKP: alkaline phosphatase; ALT: alanine transaminase; AST: aspartate aminotransaminase; GGTP: gamma glutamyltranspeptidase; MTD: maximum tumor diameter; PVT: portal vein thrombosis.
Kruskal-Wallis rank test; ^chi-square test; §test for trend.
The increases of all the blood parameters were significant except for ALKP and ALT, although white blood cell count, hemoglobin (Hb) and albumin had decreases with increasing MTD (for albumin, this reflected a worsening in inflammatory response.10-12 The maximum increase in parameter from smallest to largest MTD was for GGTP, which did not reach a doubling (a two-fold increase).
Serum AFP trends in relation to increases in MTD
To examine the tumor parameters in relation to MTD in more detail, the levels of serum AFP were plotted according to 1 cm MTD increments, and significant increases were seen in AFP levels as MTD increased (Figure 1(a)). Examination of the results in Figure 1(a) suggested three components to the graph, which were then analyzed separately as shown in Figure 1(b), together with the equations for their interpolation lines. From MTD ⩽2 cm to 4.1–5.0 cm there was a very weak AFP increase, followed by a moderate increase or variation from MTD 4.1–5.0 cm to 6.1–7.0 cm, and then a strong increase from MTD 6.1–7.0 cm to 7.1–8 cm. We attempted to quantitate the changes for each point in Figure 1(a) with respect to each previous point, as shown in the histogram in Figure 1(c), demonstrating the percentage variation of each point in comparison with its predecessor point.
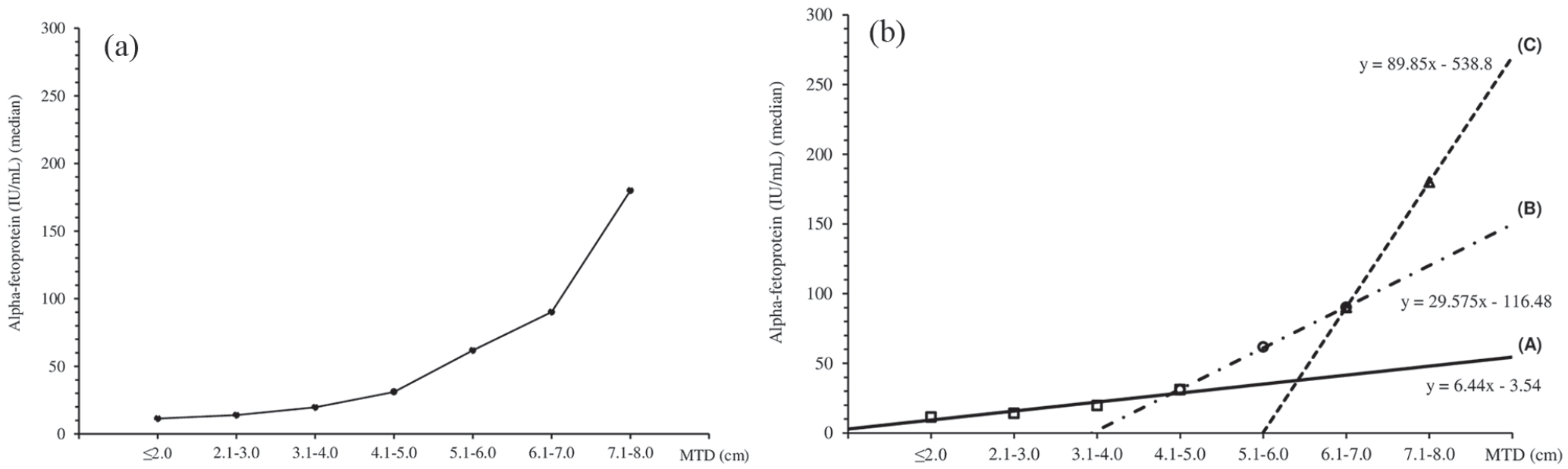
Trends in serum AFP levels in relationship to MTD.
Trends in percentage of patients with tumor multifocality or PVT in relation to MTD increases
The percentage of patients with tumor multifocality were then plotted according to 1 cm MTD increments, and a significant increase was found in the percentage of patients with multifocality as MTD increased (Figure 2(a)). The graph was also non-linear as found for AFP, and sub-trends were identified as shown in Figure 2(b), with the equation of the interpolation line each being depicted next to each sub-trend. A similar approach was taken with respect to the percentage of patients with PVT (Figure 2(c)), which were also significantly associated with increases in MTD. Furthermore, a significant trend was also found for the percentage of PVT in patients with low serum AFP (<100 IU/mL) levels, as shown in Figure 2(c1) (inset to Figure 2(c)).
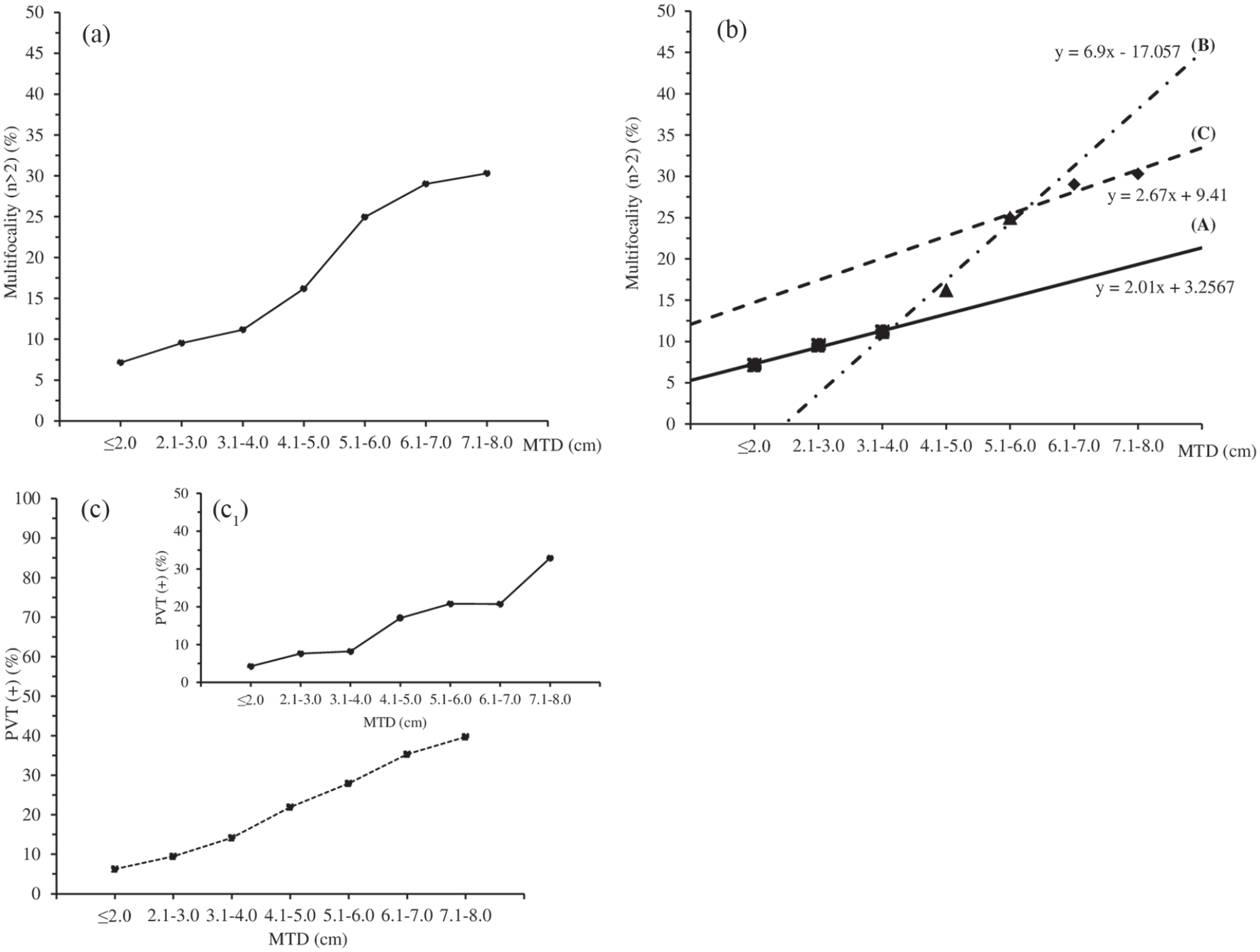
Trends in tumor multifocality (>2) (% patients) in relation to MTD.
Variation of AFP, percentage of patients with PVT or multifocality in relation to MTD changes
We then approached the extent of the changes in each of the three tumor parameters, namely AFP levels, rate of PVT (%) or rate of multifocality (%) in relation to the extent of the MTD changes (Table 2), as we tried to determine if the increase in each parameter was just proportional to the increase in MTD and thus reflective of the tumor amount, or more than proportional and thus indicative of a change in tumor biology with an increase in tumor size.
Variation of AFP, PVT(+), and multifocality (n>2) in relationship to maximum tumor diameter in categories in HCC patients.
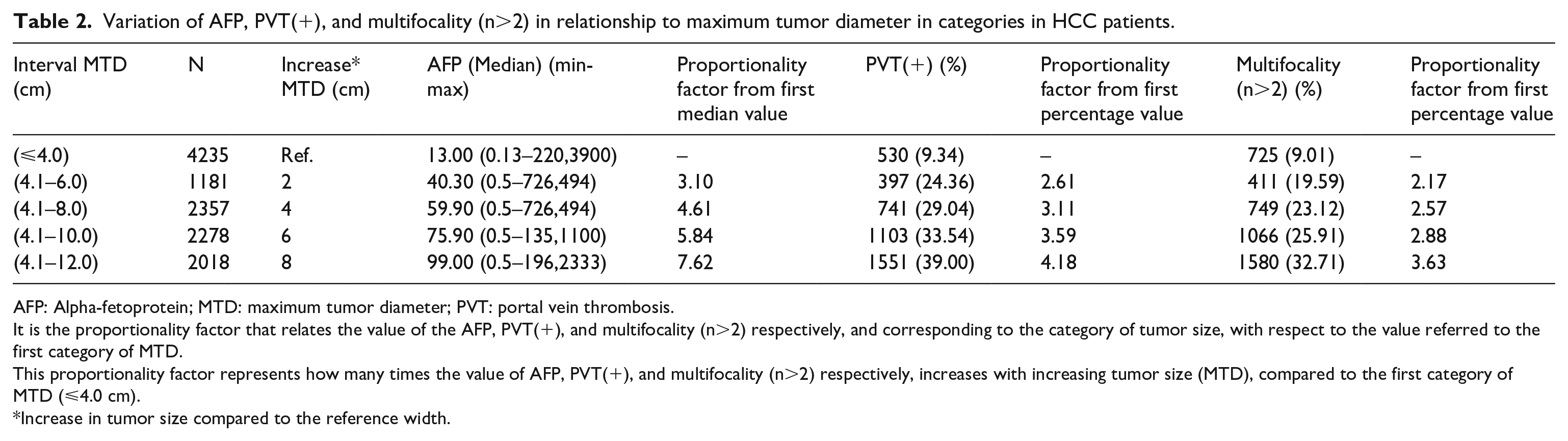
AFP: Alpha-fetoprotein; MTD: maximum tumor diameter; PVT: portal vein thrombosis.
It is the proportionality factor that relates the value of the AFP, PVT(+), and multifocality (n>2) respectively, and corresponding to the category of tumor size, with respect to the value referred to the first category of MTD.
This proportionality factor represents how many times the value of AFP, PVT(+), and multifocality (n>2) respectively, increases with increasing tumor size (MTD), compared to the first category of MTD (⩽4.0 cm).
Increase in tumor size compared to the reference width.
We found that for median AFP levels and the rate of PVT, the increase in parameter value was greater than the MTD increase. However, the rate of multifocality increased in proportion to the MTD increases. These findings support the idea that as the tumor size increased, the tumor parameter increased by an even greater amount (for AFP and PVT, but not for multifocality), supporting the hypothesis that the HCC biology became more aggressive with the increase in MTD.
Discussion
The motivation for this study was the apparent “barrier” around 5 cm for long survival post liver transplant for HCC. HCC recurrences post resection or ablation can be explained as due to local invasion or second malignancies when the predisposing cause (inflammation, cirrhosis, hepatitis, alcohol, etc.) continues. However, after transplant, the predisposing liver (but not the systemic) milieu has been removed. An obvious explanation for post-transplant recurrences is that, like other malignancies, there may be circulating micro-metastases that can seed the new liver after removal of the primary tumor (cf. breast, lung, colorectal cancers). Doubtless this could occur for HCC, especially under the influence of residual circulating hepatitis virions plus immunosuppression. This work was not intended to study liver transplant, but to enquire whether there might be a general change in HCC phenotype as the tumor grows, which could explain the high recurrences post ablative or transplant therapies, when performed for larger HCCs.
When this large cohort of HCC patients was ordered by increase in MTD, we found that there was an increase in the three parameters of AFP, the rate of PVT and the rate of multifocality with increasing MTD (itself an HCC aggressiveness parameter 13 ). This may be unsurprising, as with each doubling in MTD, there is a doubling or more (tumor volume is 4/3 π r3) in tumor mass. However, inspection of the graphs for AFP and the rate of multifocality (Figures 1(a) and 2(a)) suggest that there are two striking findings. First, there is more than a single component to each graph, as analyzed in Figures 1(b) and 2(b) (although this possibility is less clear for % PVT, in Figure 2(c)). Second, when the fold-increases (proportionality) were then calculated for each parameter with respect to the MTD fold-increases (Table 2), we found that for AFP and the rate of PVT (but less so for % multifocality), the parameter fold-increases exceeded the MTD fold-increases.
These results suggest that the parameter increases were not linear or in proportion to the MTD increases, but were greater, and might be explained by a change in HCC phenotype with the increase in tumor size. This hypothesis could be more formally tested in the future by examination of the baseline tumor biopsy in comparison with the explanted HCC material, for tumor mutation burden, microsatellite instability, or other measures of change in HCC biology, including growth factor receptors and programmed cell death-1-receptors. Of course, the data from Figures 1(a), 2(a), and 2(c) also show that the HCC patients are heterogeneous, since there are still many patients with low serum AFP (median values are shown) and with both a low rate of multifocality and a low rate of PVT.
Interestingly, the examination of trends for several blood count and liver parameters (Table 1), suggests changing trends for more than one parameter. Thus, the Hb trend downwards with the increase in MTD is possibly associated with systemic inflammation, yet blood platelets trend upwards, as has been documented previously for larger sized HCCs.9,14 AST trends upwards with increasing MTD, consistent with increased inflammation, as well as GGTP, which may also reflect hepatic inflammation, but is additionally reported as a candidate HCC biomarker.15,16 Similarly, albumin trends downwards with increasing MTD, consistent with it being an inflammation biomarker.10,12 Major increases in serum AFP, the rate of PVT and the rate of multifocality were found around a 5 cm MTD pivot point.
Changes in solid human tumors during disease management have been reported for decades, especially in breast and colon cancer, when resistance can develop to cancer chemotherapy treatments over time. However, the changes reported here, which are associated with the increase in MTD, do not seem to be induced by a clear cause. However, there is considerable literature in rodent hepatocarcinogenesis studies on the induction by multiple hepatotoxic chemicals and hepatocarcinogens of resistance to subsequent challenge by a variety of unrelated hepatotoxins, as well as to the chemotherapeutic agent doxorubicin.17-21 Thus it is possible that the well-described pleiotropic drug resistance of experimental chemical hepatocarcinogenesis may have its clinical counterpart in the predominantly cancer chemotherapeutic resistance of most human HCC.
HCC aggressiveness factors of PVT, AFP, or multifocality have been previously shown to correlate with the increase in tumor size or MTD.22-27 Whether this is causal or mere association has not been clarified. The two most likely hypotheses to explain the relation of size to other aggressiveness factors are the probability of aggressiveness factors per increased unit of tumor size versus an increased tumor size producing more factors that stimulate tumor invasiveness.
Therefore, a partial answer to the initial enigma of a 5 cm threshold for HCCs in liver transplant may be that other aggressiveness factors—such as AFP, PVT, and multifocality—increase in tandem with the increase in MTD. Thus, it is not the MTD alone that is increasing the probability of post-transplant recurrence, but rather the total aggressiveness profile that is a part of the increasing MTD.
An implication of these findings is an apparent change in HCC tumor biology with the increase in MTD. If correct, then a single baseline tumor biopsy at diagnosis might be insufficient for rational therapeutic decision making during the months of disease progression. Additional biopsies over the disease course might then be reasonable. However, since they are invasive procedures, liquid biopsy through repeated blood sampling might have a major advantage in this setting, especially when uniformity of the various liquid biopsy platforms has been established. Liquid biopsy for analysis of the analytes circulating tumor cells, circulating tumor RNA, or for genetic alterations in cell free tumor DNA in the bloodstream, is moving from experimental to practical clinical application in many solid tumors, including HCC, both for tumor typing,28-30 for early detection in patients at risk, and with small tumors 31 ; for the prediction of drug responsiveness, such as for the detection of the epidermal growth factor receptor and P13K3CA mutations. 32 Plasma cell-free DNA-based epigenomics tests, such as for DNA methylation, 33 are starting to head to market, by companies such as Helio, Guardant (FDA-approved Guardant360 CDx test), NeoGenomics, Ivygene Dx Liver Cancer test, as well as several others.
Furthermore, some clinically approved tests, such as positron emission tomography imaging, based on the uptake of 18fluorodeoxyglucose into metabolically active HCCs, has begun to attract interest due to its ability to predict survival, portal venous invasion, and degree of HCC differentiation.34-36
The findings reported here suggest a change in HCC biology with tumor growth and imply the need for sequential tumor sampling during the months of patient care and treatments.
Conclusions
We found that as MTD increased, so did median serum AFP levels, as well as the percentage of patients with both PVT and multifocality, but in a non-linear manner and to a greater extent than the increase in MTD. These findings suggest that HCC aggressiveness might worsen or evolve with tumor growth. Multiple—perhaps liquid—biopsies might thus better inform treatment decisions as the tumor grows, rather than a single biopsy at baseline.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported in part by NIH grant CA 82723 (B.I.C).
