Abstract
BACKGROUND AND AIMS:
A striking difference has been observed in structure and functional properties between plasma and platelet von Willebrand factor (VWF). While the existing evidence has revealed a clinical relevance of plasma VWF-Ag in liver regeneration (LR) and different cancers, this study was designed to explore the properties of intra-platelet (IP) and serum VWF-Ag in patients with hepatocellular carcinoma (HCC) undergoing partial hepatectomy.
METHODS:
A total of 40 patients undergoing partial hepatectomy were prospectively recruited from 3 institutions. VWF-Ag concentrations were evaluated mainly in serum and platelet extracts. Patients were followed-up for postoperative liver dysfunction and HCC recurrence.
RESULTS:
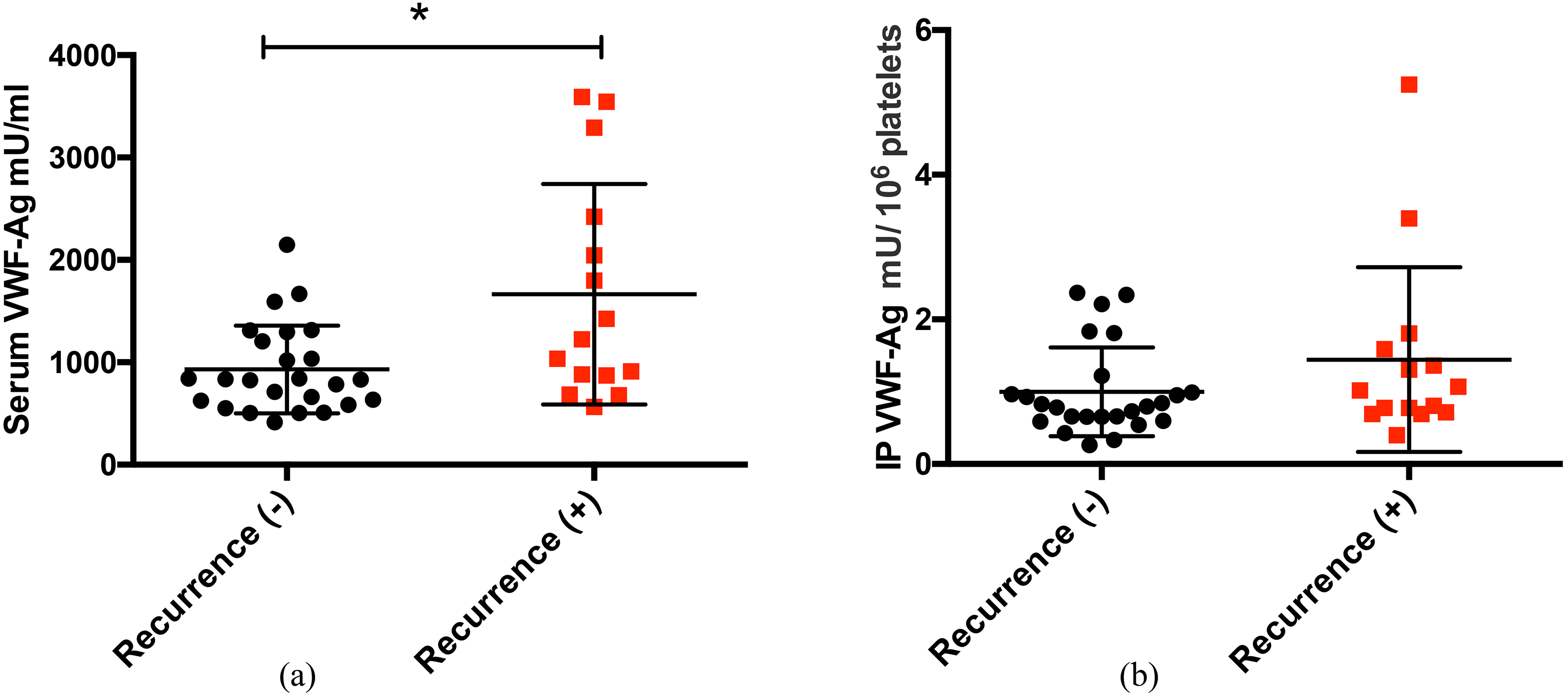
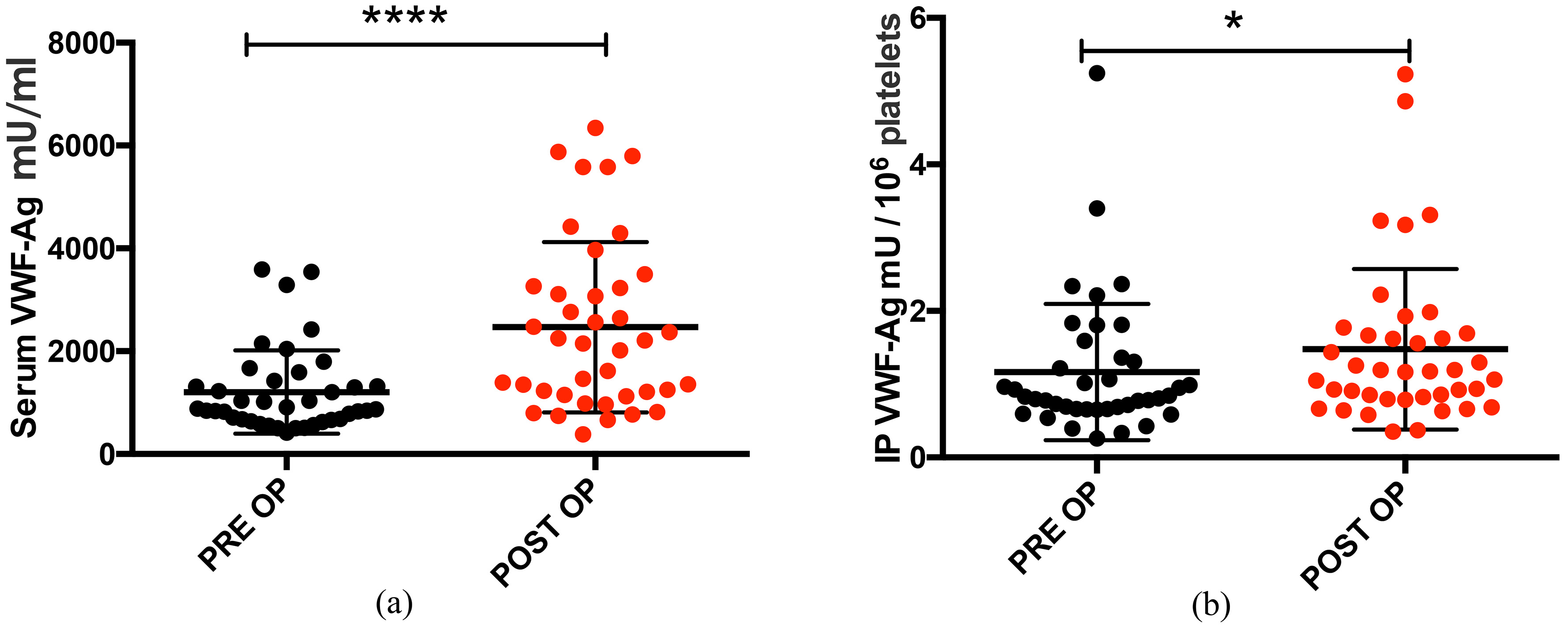
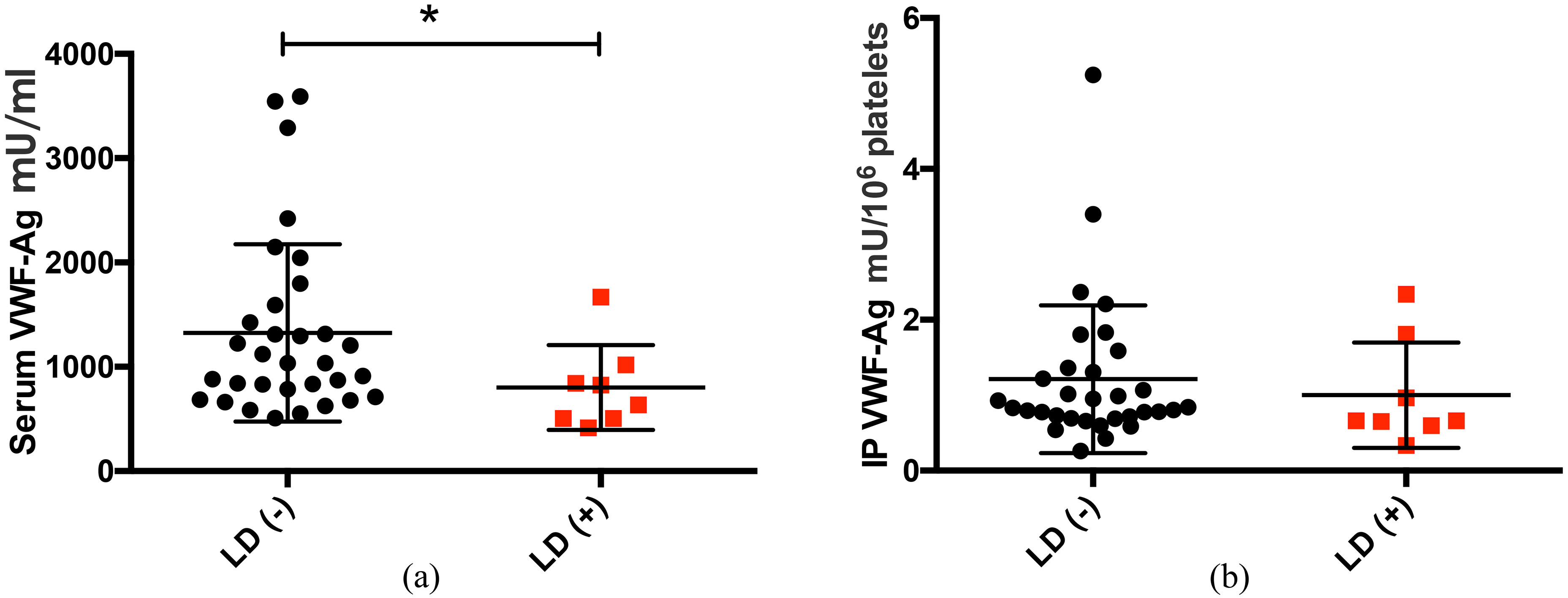
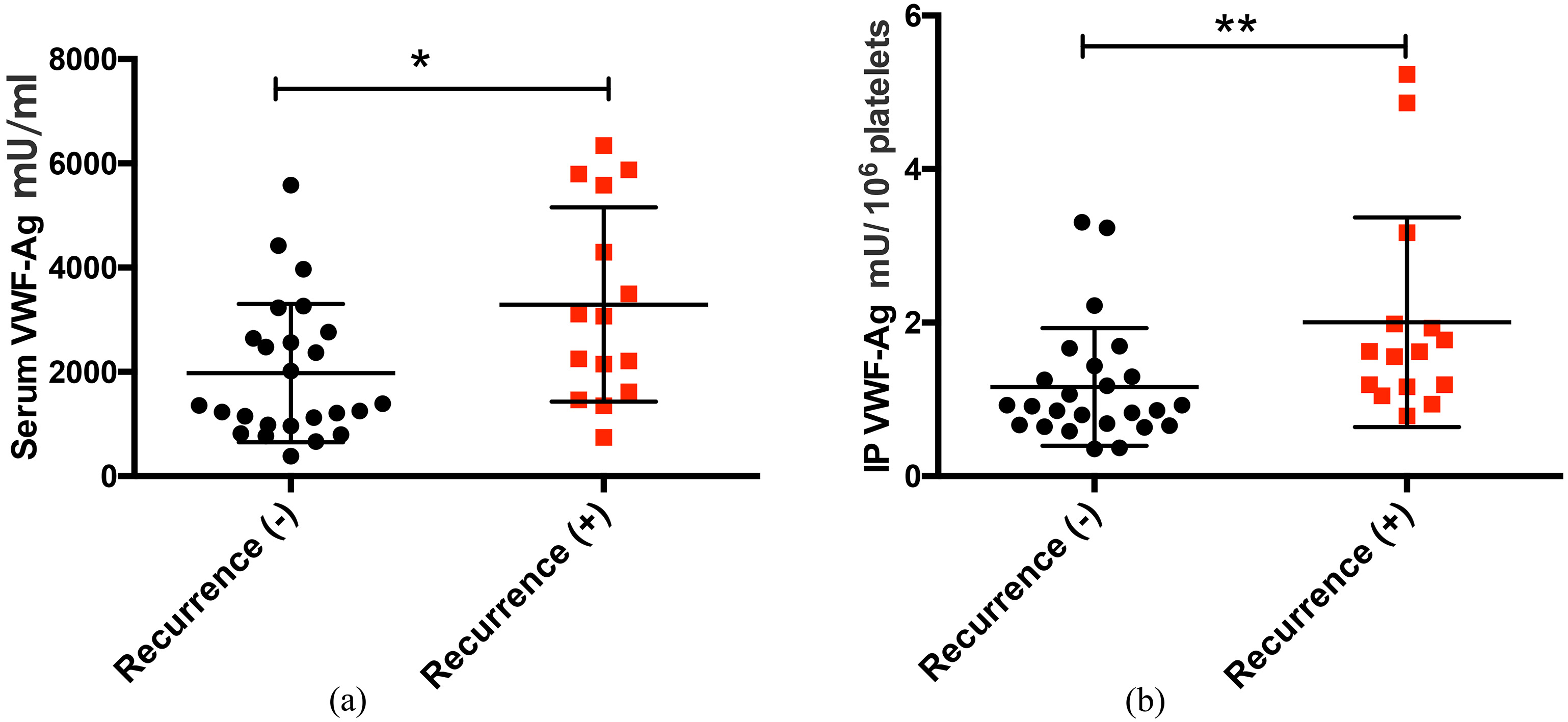
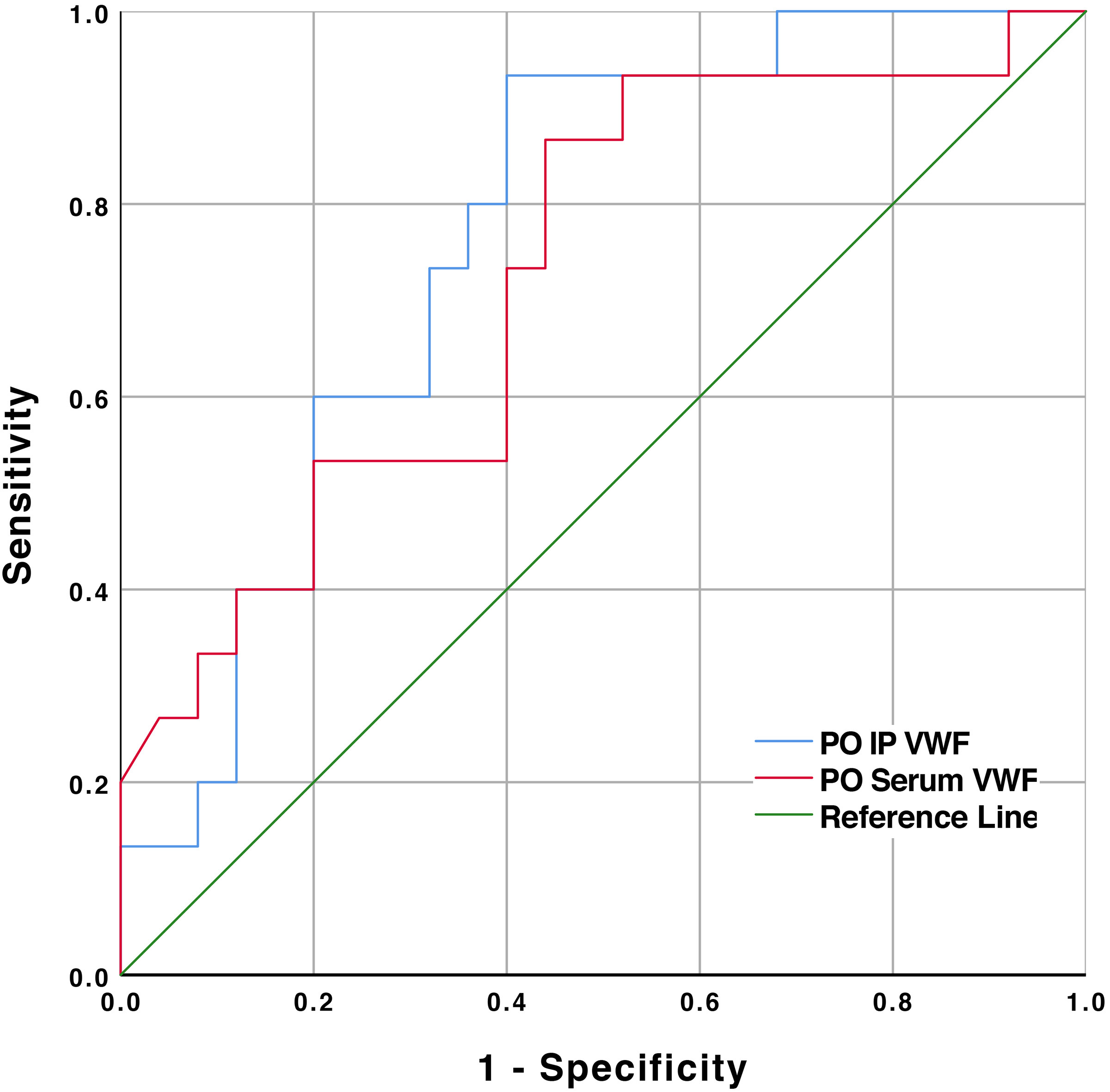
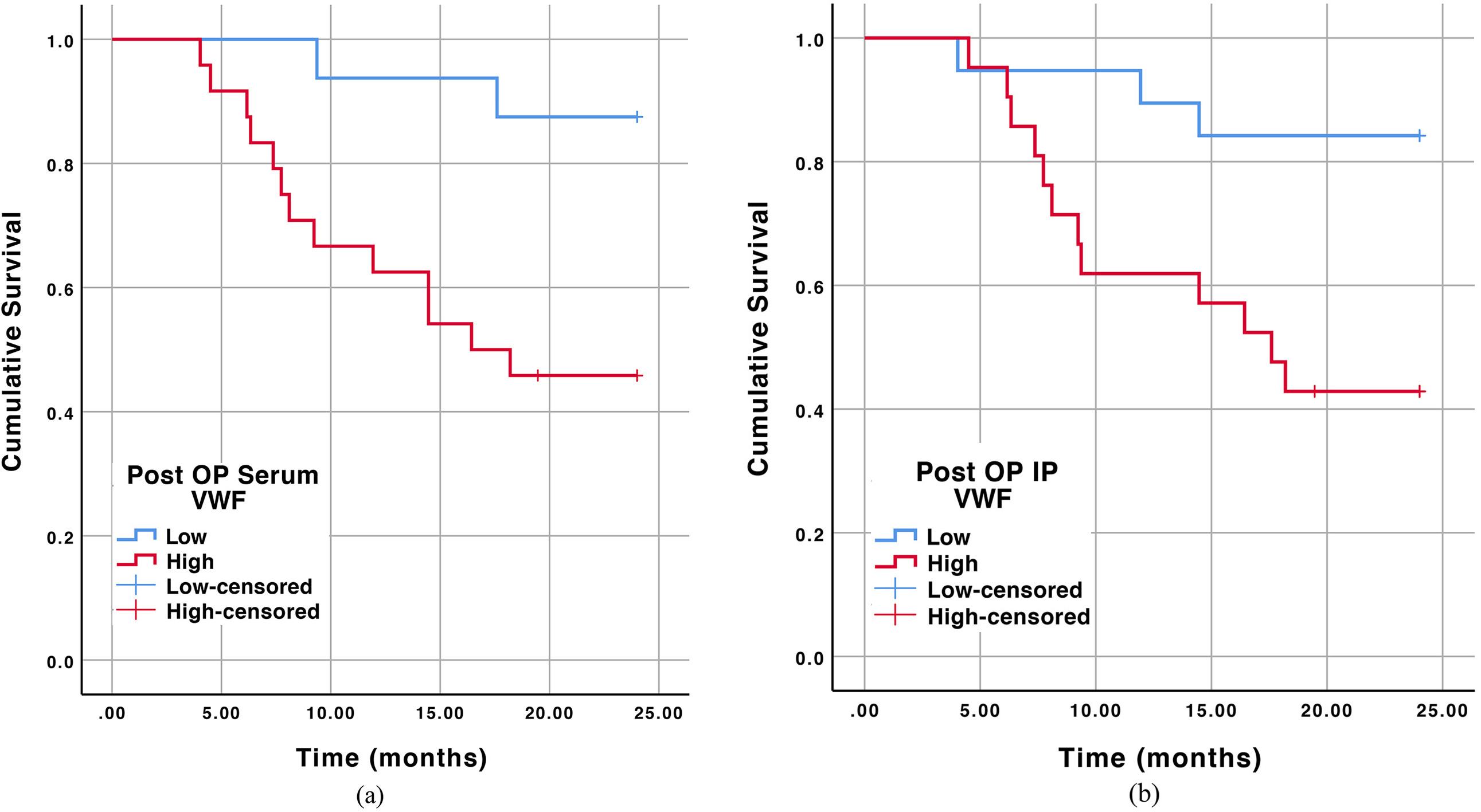
We observed a post-resection increase in the concentration of VWF-Ag in serum and platelet. Patients with postoperative liver dysfunction had substantially reduced serum and IP VWF-Ag concentrations. After a 2-year follow-up, patients with higher post-resection serum and IP VWF-Ag concentrations were found to develop early HCC recurrence. Likewise, IP VWF-Ag was able to independently predict post-resection early HCC recurrence.
CONCLUSION:
This multicenter, prospective, pilot study demonstrates a bivalent property of IP VWF in LR and oncological outcome; low preoperative VWF appeared to have a negative association on post-resection liver dysfunction, whereas, patients with higher post-resection VWF-Ag concentrations were found to have early HCC recurrence.
Introduction
Von Willebrand factor (VWF), a multimeric, multidomain, multifunctional adhesive glycoprotein, predominantly operates its functions in primary hemostasis by anchoring platelet adhesion and aggregation to exposed subendothelium in damaged blood vessels. The biosynthesis of VWF is confined to endothelial cells (EC), where it is stored in organelles known as Weibel-Palade (WP) bodies, and megakaryocytes or platelets, where it is localized in the alpha-granules. Although the major volume of VWF is secreted by EC, platelets also contain a substantial quantity of VWF, accounting for 10–25% of the total amount of VWF-antigen (concentration) present in normal platelet-rich plasma [13, 23, 37]. Interestingly, the functional properties of intra-platelet (IP) VWF markedly differs from that of the circulating (plasma) VWF [20].
Liver regeneration (LR) after partial hepatectomy (PH) triggers a hypercoagulable state associated with a reduction of gene expression of the many coagulation cascade proteins and elevation in the plasma concentrations of factor VIII and VWF [31]. In rodent models, VWF knockout negatively affected the LR [16]. It has been suggested that platelet accumulation and LR both are largely dependent on VWF [16]. Recently, Starlinger et al. were able to demonstrate that an early VWF-Ag burst after PH is crucial for adequate platelet accumulation and concomitant LR. With this first-in-human evidence they have also stressed on the diagnostic potential of preoperative plasma VWF-Ag concentrations to predict poor postoperative outcome after PH [30].
Tumor cells activate coagulation, through the release of tissue factor, microparticles or P-selectin ligands and expression of thrombin that further augment coagulation. Likewise, tumor cells achieve attachments to endothelium via e-selectin [10]. Platelet coating of tumor cells provides a mechanical shield by protecting the circulating tumor cells from immune attacks [9]. The alliance between platelet, endothelial and tumor-cell membrane integrins is now known to facilitate the hematogenous metastasis of tumor [9]. Studies have demonstrated the presence of fibrin deposition and platelet aggregation in and around different tumor specimens, indicating local activation of coagulation [7]. Furthermore, hemostatic abnormalities are found in 60–100% of patients with malignant tumors on laboratory investigations, including those without thrombotic manifestations [25]. Numerous possibilities of utilizing platelet kinetics in the diagnostic and prognostic evaluation of hepatocellular carcinoma (HCC) have already been observed [3].
VWF provides a bridge between collagen and plat-elets inducing rolling of platelets that results into platelet activation accompanied by shape change and exposure of phosphatidyl-serine on the outer membrane, and release of platelet VWF and other growth factors [26]. Clinical studies have reported an increased concentration of plasma VWF antigen in a variety of malignancies including colorectal carcinoma [27, 33], urinary bladder cancer [36], ovarian carcinoma [8], and lymphoblastic leukemia [12]. Likewise, local and plasma VWF concentrations were found elevated in HCC as compared to chronic hepatitis-B patients [18].
In this study, we investigated the kinetics of IP and platelet-derived (serum) VWF-Ag after PH in patients with HCC and followed them for 2 years. Since the properties of IP VWF-Ag differ from the plasma VWF-Ag, in this study, we have focused on the IP and serum VWF-Ag dynamics and their association with LR and post-resection HCC recurrence.
Patients and methods
Prospective study cohorts
Forty patients with primary HCC (all with Child-Pugh class A subgroup) who went on to have liver resection were enrolled in the study from May 2013 to June 2015. This study is registered in UMIN Clinical Trial Registry (UMIN000026380). The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki (6th revision, 2008) as reflected in a priori approval by the institution’s human research committee. Patients were recruited from 3 different institutions; Kagoshima university hospital, Kirishima medical center and Kagoshima medical center, national hospital organization. All three institutional ethics committee approved analyses of blood samples and patients’ data; signed, informed consent was obtained from all patients. All methods were performed in accordance with relevant guidelines and regulations.
All patients underwent a thorough laboratory evaluation, including serum
Definition of Postoperative liver dysfunction
Postoperative liver dysfunction (LD) was assessed based on the “50–50 criteria” criteria by Balzan et al. [4]. This study focused on the later phase (delayed) hepatic regeneration and not merely liver failure, thus a serum bilirubin concentration
Follow up
After the first-month follow-up, patients were followed with ultrasonography (USG) every 3 months after resection, and if USG suggested any evidence of tumor, subsequent contrast-enhanced CT or magnetic resonance imaging (MRI) of the abdomen and non-contrast CT of the chest were performed. The diagnosis of intrahepatic recurrence was made on the basis of imaging alone if the tumor exhibited the typical enhancement characteristics. A 2-year follow-up was described as the end point for early HCC recurrence in this study.
Sample preparation
Serum and plasma
Venous blood was collected preoperatively (PRE OP) and after four weeks post-resection (POST OP).
Whole blood was collected in a serum separating tube and 3 citrate tubes, containing 0.5 ml of sodium citrate (for plasma and platelet preparations), an EDTA-2k tube (Venoject II, Terumo Corp., Tokyo, Japan) was used to collect blood for cell count. Serum tube was incubated at room temperature for 30 minutes to allow clotting. Serum and plasma tubes were centrifuged at 1710
Platelet isolation
Citrate tubes containing venous blood were centrifuged at 90
Quantification of cytokines
Serum and platelet extracts were analyzed together by commercially available enzyme-linked immunosorbent assay (ELISA) tests for human Von Willebrand factor (Abcam, Waterloo, Australia, ab108918) according to the manufacturer’s guidelines.
Calculation of IP VWF concentration
Platelet content of VWF-Ag per 10 [6] platelets was calculated using the previously described equation [2]. Briefly, 220
(220
Clinico-pathological data at the time of resection of primary HCC and after 2-years follow-up
Clinico-pathological data at the time of resection of primary HCC and after 2-years follow-up
Footnotes
Acknowledgments
Dr. Bibek Aryal is a fellow of Japan society for the promotion of science (JSPS). This study is supported by JSPS KAKENHI Grant Numbers: JP18F16420, JP16H05229 and JP26293130. UMIN clinical Trial Registry (Registration identification number: UMIN000019145).
Conflict of interest
No conflict of interest to disclose.
Supplementary data
Preoperative VWF-Ag concentration in patients with and without HCC recurrence in: serum (a) and IP (b). IP VWF concentration expressed per 10