Abstract
Background
This study aimed to investigate the diagnostic value of prealbumin-to-fibrinogen ratio (PFR) and albumin-to-fibrinogen ratio (AFR) alone or in combination in Helicobacter pylori-negative gastric cancer (Hp-NGC) patients.
Methods
This study included 171 healthy controls, 180 Hp-NGC patients, and 215 Helicobacter pylori-negative chronic gastritis (HpN) patients. We compared the differences of various indicators and pathological characteristics between groups with Mann–Whitney U test and Chi-square test. The diagnostic value of PFR and AFR alone or in combination for Hp-NGC patients was assessed by the receiver operating characteristic (ROC) curve.
Results
PFR and AFR were related to the progression and clinicopathological characteristics of Hp-NGC. As the disease progressed, PFR and AFR values gradually decreased and were negatively related to the tumor size and depth of invasion. In addition, the area under the curves (AUCs) that resulted from combining PFR and AFR to distinguish Hp-NGC patients from healthy controls and HpN patients were 0.908 and 0.654, respectively. When combined with PFR and AFR in the differential diagnosis of tumors with a maximum diameter ≥ 5 cm and the T3 + T4 stage, the AUCs were 0.949 and 0.922; the sensitivity was 86.32% and 80.74%; and the specificity was 94.74% and 92.98%, respectively.
Conclusions
PFR and AFR may be used as diagnostic biomarkers for Hp-NGC. The combination of PFR and AFR was more valuable than each indicator alone in the diagnosis of Hp-NGC.
Keywords
Introduction
Gastric cancer (GC) is a malignant tumor that ranks fifth among the most common cancers and third among the most common causes of cancer death in the world. 1 Despite a decline in its incidence, the prognosis of GC remains poor. 2 GC is caused by many factors, including diet, genetic mutations, and Helicobacter pylori (H. pylori) infection.3,4 Among these factors, persistent H. pylori infection can cause chronic gastritis, which may eventually lead to GC. 5 At present, serum carcinoembryonic antigen (CEA), CA724, and CA199 are still important markers for early GC screening, 6 but a small number of patients with GC are negative for H. pylori, which is called Hp-NGC. Although these markers have certain diagnostic value for GC, they are not recommended for early diagnosis of Hp-NGC due to its unique clinical and pathological features. 7 In addition, due to the extremely low prevalence of Hp-NGC, there are relatively few clinical studies on markers for diagnosing Hp-NGC.7,8 Although methods such as endoscopy and pathological biopsy can accurately diagnose Hp-NGC, they are invasive and unsuitable for early screening. 5 At present, many potential markers have been proposed for the diagnosis of GC, such as long non-coding RNAs cancer upregulated drug resistant, long stress-induced noncoding transcript 5, phosphatase and tensin homolog pseudogene 1, and microRNA. 9 However, these markers are expensive to detect and are not suitable for routine testing. In addition, no useful non-invasive GC biomarkers have been identified. 10 Therefore, new, convenient, and economical biomarkers are needed to predict early GC, especially in Hp-NGC patients.
Inflammation is closely connected with the progression and prognosis of tumors, and abnormal blood coagulation and nutritional status may also affect the occurrence and development of malignant tumors. 11 Common inflammatory markers include prealbumin (PA), albumin (ALB), Fibrinogen (Fib), and new markers that are composed of the ratio of these indicators, which are usually abnormal in patients with GC. 12 ALB and PA, which are produced by the liver, are often used as biomarkers to assess inflammation and nutritional status. 13 In addition, a decrease in the serum ALB level indicates malnutrition and decreased immunity and accelerates the infection and progression of tumors. 14 However, the half-life of PA is shorter than that of ALB and is also more sensitive to malnutrition. It is a new and feasible indicator of a poor prognosis for patients with GC.15–17 Several other studies have also confirmed that ALB and PA are potential prognostic factors in patients with GC. 13 Fib is an acute-phase reactive protein that can regulate the proliferation, metastasis, and signal transduction of tumor cells. 18 Palaj et al. found that high levels of Fib are related to tumor development, metastasis, and survival in GC patients. 19 Therefore, we speculated that PFR and AFR may be related to inflammation and tumor progression and are expected to become new biomarkers to predict and diagnose patients with Hp-NGC.
Existing studies have assessed the prognostic value of PFR and AFR in GC and other cancers; as a result, there is a lack of research on the predictive value of PFR and AFR in patients with Hp-NGC. Therefore, we used PFR and AFR both alone and in combination as diagnostic markers to evaluate patients with Hp-NGC and analyzed their relationship with clinicopathological characteristics.
Materials and methods
Patients
This study was a retrospective analysis that included 171 healthy controls, 215 HpN, and 180 Hp-NGC patients from the First Affiliated Hospital at Guangxi Medical University between January 2013 and October 2020. The inclusion criteria for Hp-negative patients were as follows: (a) newly diagnosed with gastric adenocarcinoma via clinical pathology; (b) a diagnosis of chronic gastritis; (c) a negative for H. pylori by endoscopy, pathological biopsy, urea breath test (UBT), or serum H. pylori antibody; (d) no history of radiotherapy, chemotherapy or anti-inflammatory treatment; (e) no other malignant tumors or related infectious diseases (such as autoimmune diseases, infectious diseases, hepatitis, cirrhosis, etc.); (f) no cardio cerebrovascular disease; and (g) complete clinical and pathological data. In addition, we also included 171 healthy people with no history of cancer and gastrointestinal diseases as healthy controls. Our study was approved by the Ethics Committee of the First Affiliated Hospital of Guangxi Medical University and was in line with the Declaration of Helsinki. All participants gave informed consent.
Clinical data collection and calculation
All relevant data were obtained from patient medical records, including gender, age, PA, Fib, white blood cells (WBC), neutrophils (NEU), hemoglobin (Hb), platelets, ALB, CEA, and clinicopathological characteristics. A Beckman-Coulter LH 780 (Beckman Coulter, Brea, CA) and Roche E6000 Analyzer (Roche Basel, Switzerland) were used to carry out routine blood and CEA analyses, respectively. We also used a Hitachi 7600 automatic biochemical analyzer (Tokyo, Japan) to determine PA and ALB. Plasma Fib was detected by Sysmex CA7000 automatic coagulometer. PFR and AFR values were generated from formula calculations: PFR = PA/Fib and AFR = ALB/Fib, respectively. We obtained the optimal cutoff values for PFR and AFR using MedCalc software and divided them into a high-value group and a low-value group.
Statistical analysis
The data in this study were analyzed using the SPSS software (version 25.0). All data did not follow a normal distribution; therefore, they were represented using the median and interquartile ranges. In addition, we used a Chi-square test and a Mann–Whitney U test to analyze the differences in the laboratory markers and pathological characteristics between the groups. MedCalc software (version 18.1.1) was used to determine the area under the curve (AUC) and receiver operating characteristic. For all the analysis results, P < 0.05 indicated statistical significance.
In this study, the sample sizes of the healthy control group, HpN group, and Hp-NGC group were 171, 215, and 180, respectively. The current sample size had more than 99% power to detect differences in PFR and AFR between different groups (α<0.05). The sample size of 180 patients had 98.05% power to detect the difference in PFR and AFR between subgroups of tumor size or tumor invasion depth (α<0.05).
Results
Baseline data of patients
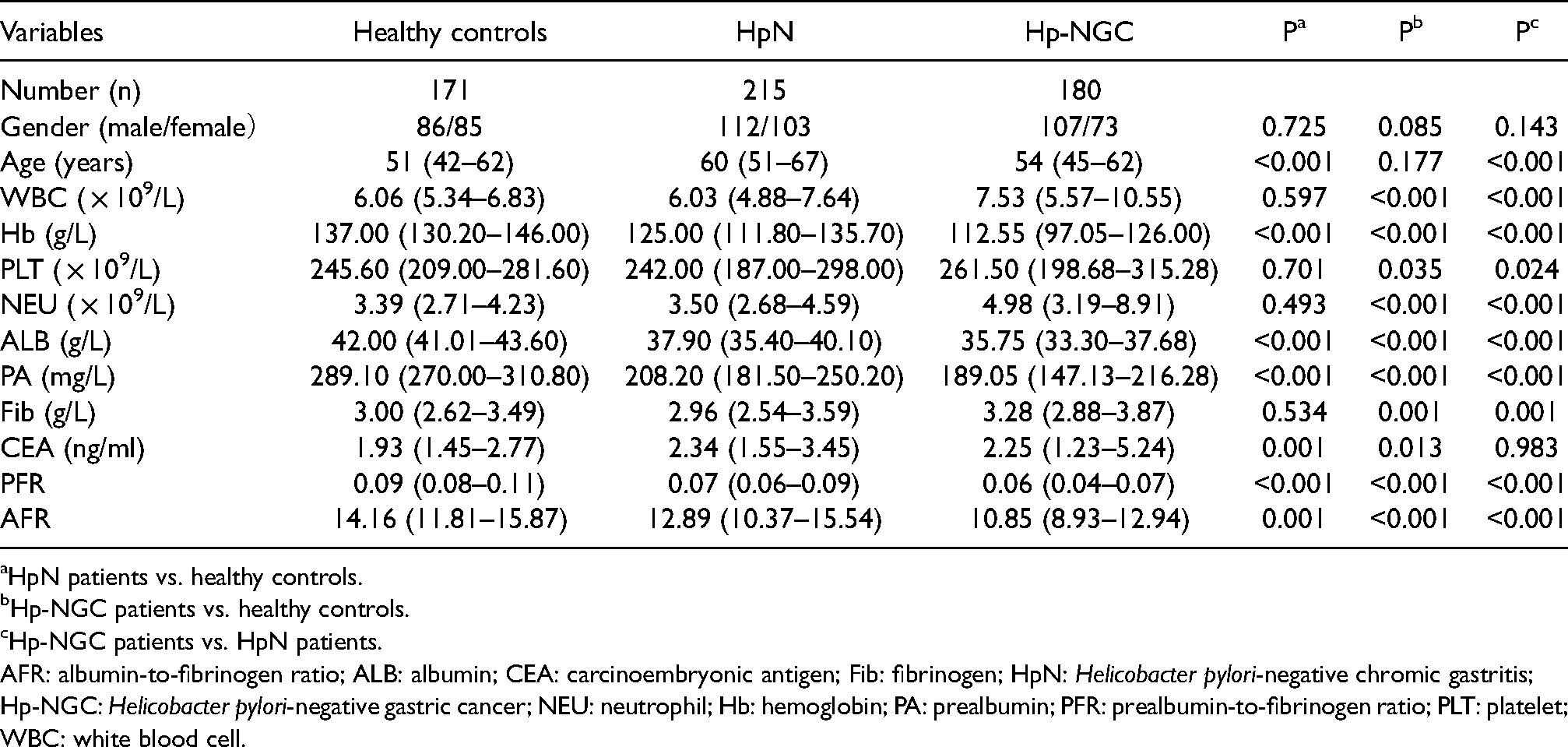
The baseline data of the study population are shown in Table 1 and Figure 1. In terms of gender, there was no statistical significance among the three groups, but Hb, ALB, PA, PFR, and AFR were all statistically significant. PFR and AFR values decreased gradually as the disease progressed. In addition, Hb, ALB, PA, PFR, and AFR values of Hp-NGC patients were lower than those of HpN patients and healthy controls, while WBC and NEU findings in Hp-NGC patients were higher than those of the other groups.
Baseline characteristics of Hp-negative patients and healthy controls.
HpN patients vs. healthy controls.
Hp-NGC patients vs. healthy controls.
Hp-NGC patients vs. HpN patients.
AFR: albumin-to-fibrinogen ratio; ALB: albumin; CEA: carcinoembryonic antigen; Fib: fibrinogen; HpN: Helicobacter pylori-negative chromic gastritis; Hp-NGC: Helicobacter pylori-negative gastric cancer; NEU: neutrophil; Hb: hemoglobin; PA: prealbumin; PFR: prealbumin-to-fibrinogen ratio; PLT: platelet; WBC: white blood cell.
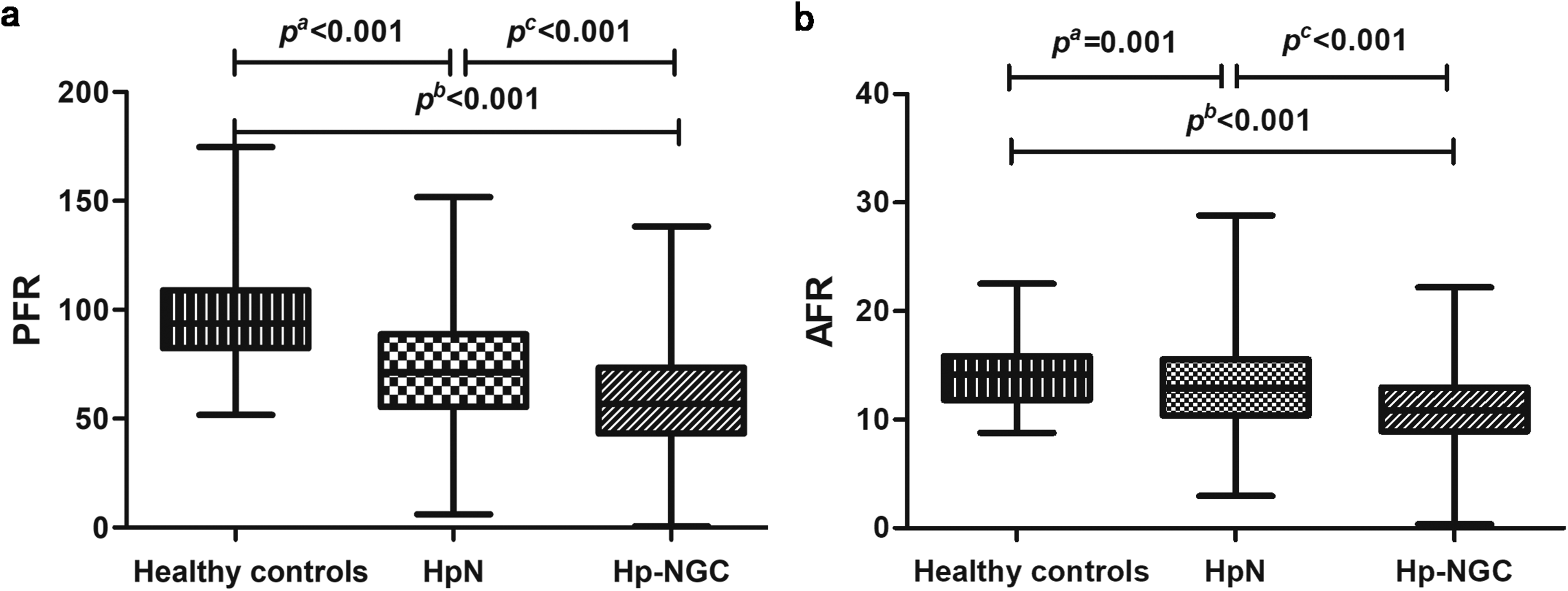
A comparison of PFR and AFR values in Hp-NGC patients and all other groups. (a) The difference in the PFR in Hp-NGC patients and all other groups. (b) The difference in the AFR in Hp-NGC patients and all other groups.
Correlation between PFR, AFR, and clinicopathological characteristics in Hp-NGC
According to Table 2, PFR and AFR were both associated with tumor size and depth of invasion (P < 0.05) but not with age, smoking, drinking, breakthrough of a serous membrane, or ulcer invasion (P > 0.05). PFR was also not associated with gender, lymph node metastasis, or tumor stage (P > 0.05) but was associated with distant metastasis (P < 0.05). In contrast, AFR was related to gender, lymph node metastasis, and tumor stage (P < 0.05) but was not associated with distant metastasis (P > 0.05).
The relationship between PFR, AFR, and clinicopathological characteristics of Hp-NGC patients.
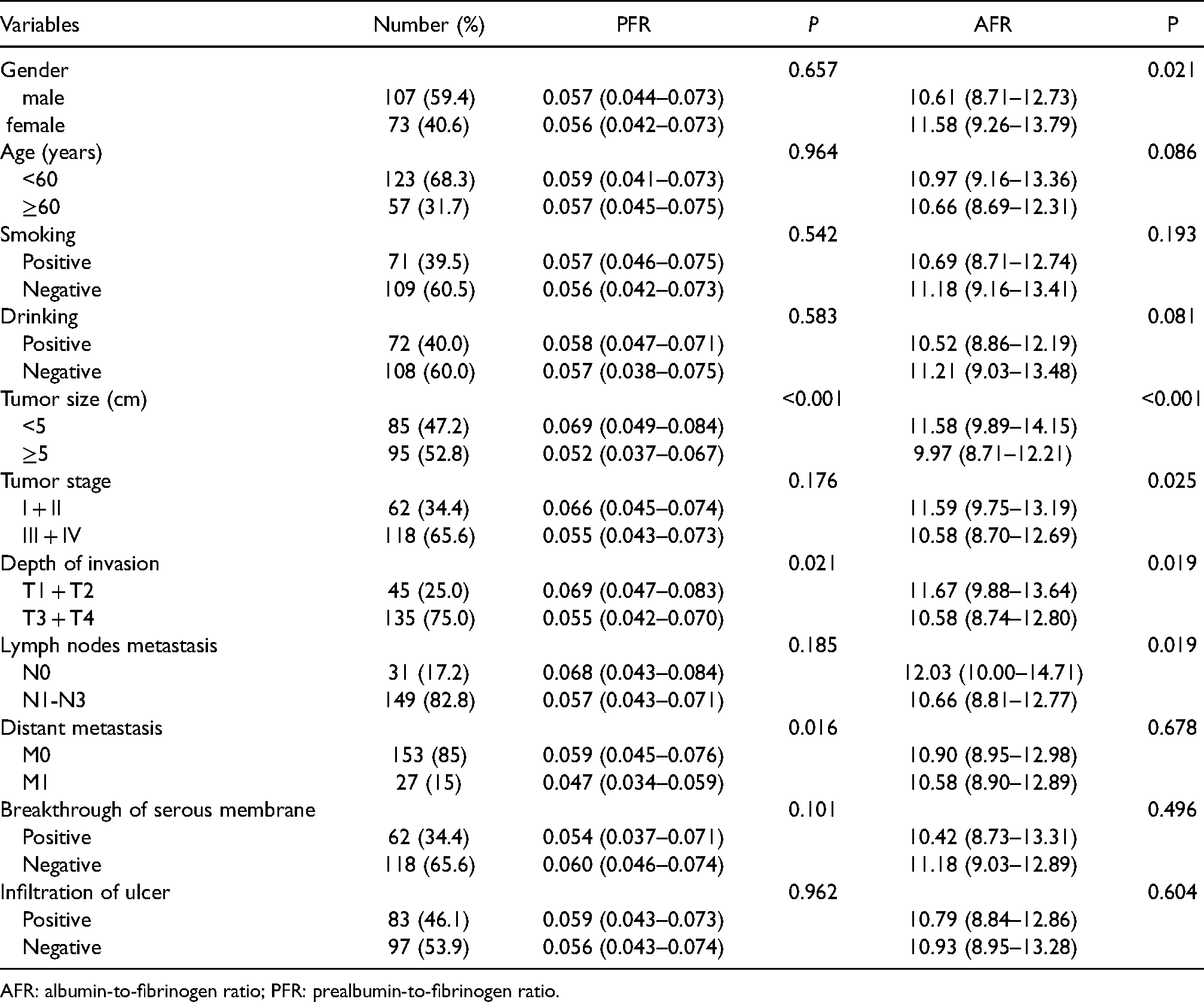
AFR: albumin-to-fibrinogen ratio; PFR: prealbumin-to-fibrinogen ratio.
Correlation between PFR and AFR stratification and clinicopathological characteristics
As shown in Table S1, there were 79 (60.3%) males and 52 (39.7%) females in the low-PFR group and 28 (57.1%) males and 21 (42.9%) females in the high-PFR group. There were 66 (66%) males and 34 (34%) females in the low-AFR group and 41 (51.2%) males and 39 (48.8%) females in the high-AFR group. The low-PFR group had a larger maximum tumor diameter (P < 0.001), a deeper invasion depth (P = 0.026), and more lymph node metastasis (P = 0.043) and distant metastasis (P = 0.041) than the high-PFR group. However, gender and tumor stage were not statistically significant between the high-PFR group and the low-PFR group (P > 0.05). The low-AFR group had more male patients (P = 0.045), a larger maximum tumor diameter (P = 0.006), a later tumor stage (P = 0.019), a deeper depth of invasion (P = 0.006), and more lymph node metastasis (P = 0.038) than the high-AFR group, but distant metastasis was not statistically significant between the high-AFR group and the low-AFR group (P > 0.05). Finally, there was no significant difference in age, smoking, drinking, breakthrough of a serous membrane, or ulcer infiltration between the low and the high groups of either PFR or AFR (P > 0.05).
The diagnostic value of PFR and AFR between Hp-NGC patients and other participants
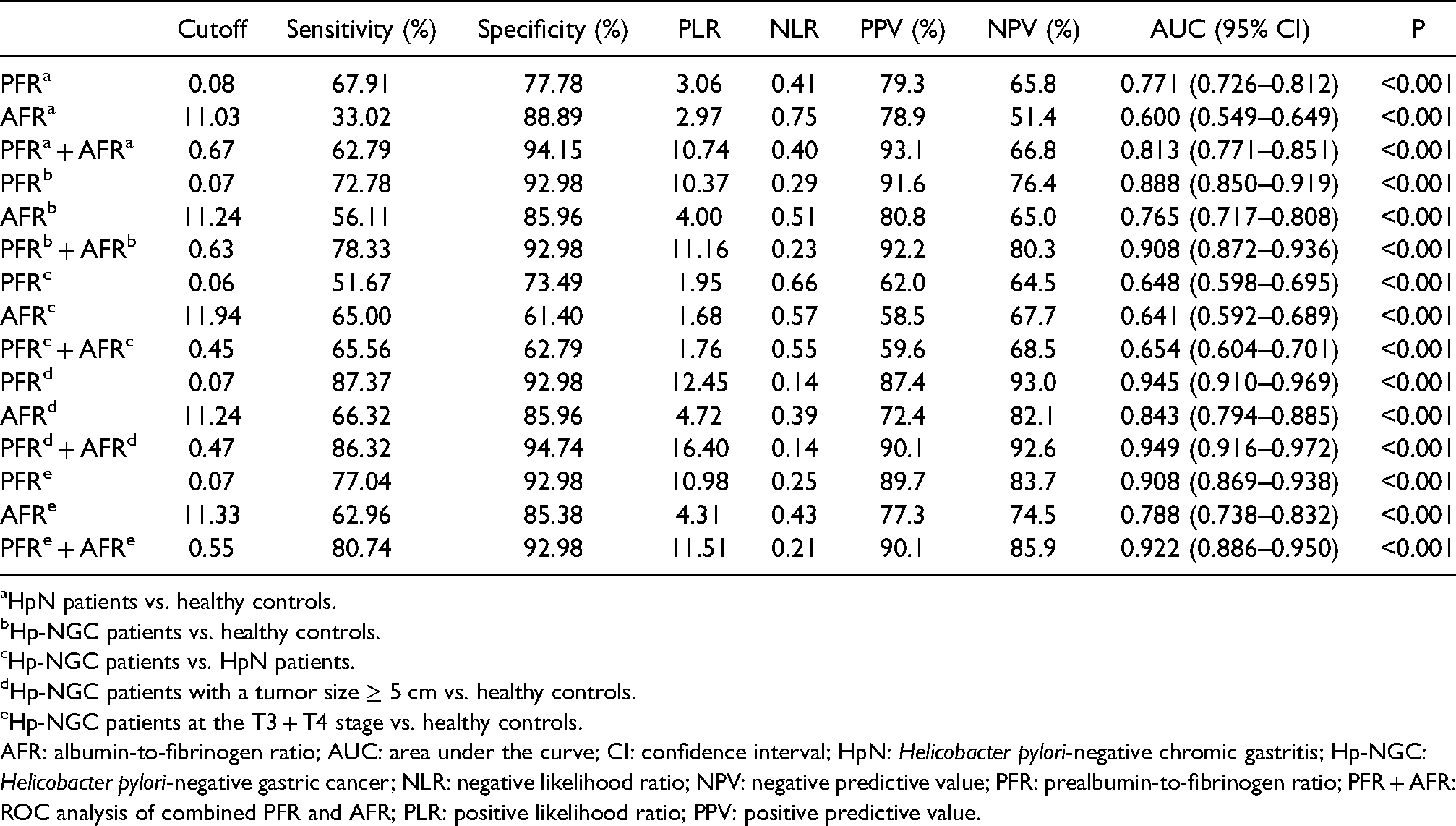
The diagnostic value is shown in Table 3 and Figure 2. First, the AUC values of PFR and AFR for differentiating Hp-NGC patients from the healthy control group alone were 0.888 and 0.765, respectively, while the combination of PFR and AFR improved the diagnostic value (AUC = 0.908) due to its sensitivity and specificity values of 78.33% and 92.98%, respectively. Second, when PFR and AFR were combined to diagnose Hp-NGC patients with a tumor size ≥ 5 cm and healthy controls and to diagnose T3 + T4 stage and healthy controls, the AUC values were 0.949 and 0.922; the sensitivity results were 86.32% and 80.74%; and the specificity values were 94.74% and 92.98%, respectively. Finally, after combining the two markers to differentiate HpN patients from healthy controls and to differentiate HpN patients from Hp-NGC patients, the AUC values were 0.813 and 0.654, the sensitivities were 62.79% and 65.56%, and the specificities were 94.15% and 62.79%, respectively. In all models, it was suggested that the combination of PFR and AFR had a better diagnostic value than any single marker alone.
The diagnostic value of PFR and AFR as single or combined indicators for distinguishing Hp-NGC patients from other patients.
HpN patients vs. healthy controls.
Hp-NGC patients vs. healthy controls.
Hp-NGC patients vs. HpN patients.
Hp-NGC patients with a tumor size ≥ 5 cm vs. healthy controls.
Hp-NGC patients at the T3 + T4 stage vs. healthy controls.
AFR: albumin-to-fibrinogen ratio; AUC: area under the curve; CI: confidence interval; HpN: Helicobacter pylori-negative chromic gastritis; Hp-NGC: Helicobacter pylori-negative gastric cancer; NLR: negative likelihood ratio; NPV: negative predictive value; PFR: prealbumin-to-fibrinogen ratio; PFR + AFR: ROC analysis of combined PFR and AFR; PLR: positive likelihood ratio; PPV: positive predictive value.
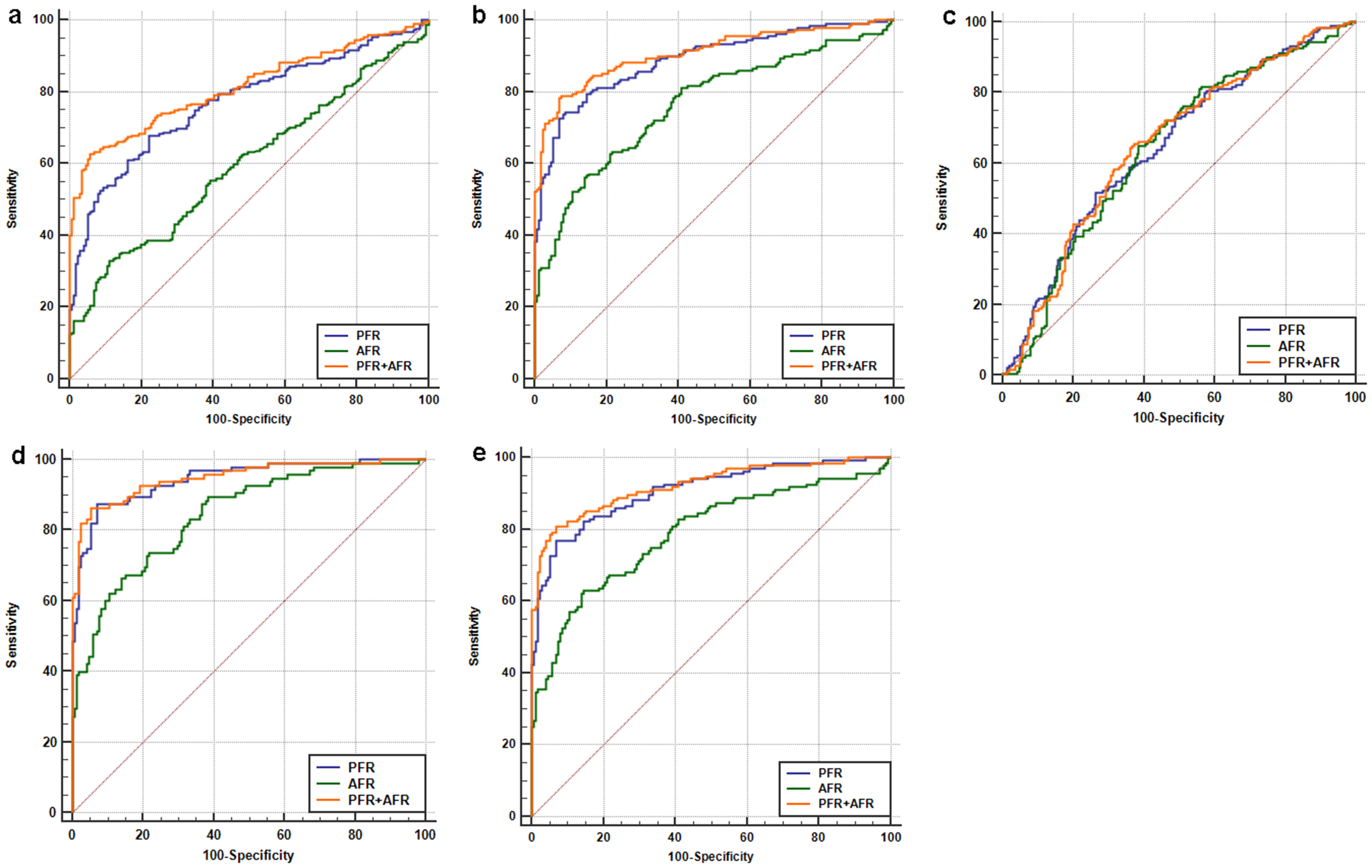
The diagnostic value of PFR and AFR in Hp-NGC patients and all other groups. (a) HpN patients vs. healthy controls. (b) Hp-NGC patients vs. healthy controls. (c) Hp-NGC patients vs. HpN patients. (d) Hp-NGC patients with a tumor size ≥ 5 cm vs. healthy controls. (e) Hp-NGC patients at the T3+T4 stage vs. healthy controls
Discussion
In recent years, early GC has not been easily detected, and most cases are diagnosed at an advanced stage. Although the incidence has gradually decreased following developments in surgery and adjuvant therapy, the prognosis remains poor. 20 There are many diagnostic methods for GC, but effective methods are usually invasive, such as gastroscopy and biopsy. As a common tumor marker of gastrointestinal malignant tumors, CEA is of great significance in the diagnosis of GC. 21 However, the diagnostic efficiency of CEA is not high (AUC = 0.644). 22 Therefore, it is particularly important to identify economical, efficient, and non-invasive diagnostic biomarkers. At present, many prognostic biomarkers are used to evaluate a variety of malignant tumors, such as AFR, fibrinogen-to-prealbumin (FPR), neutrophil-to-lymphocyte ratio, and platelet-to-lymphocyte ratio.23–26 However, the diagnostic value of these markers for Hp-NGC remains unclear. Therefore, this study explored the clinical value of PFR and AFR in the diagnosis of Hp-NGC.
Some studies have shown that Fib is a pro-inflammatory protein that is related to the clinicopathological characteristics and prognosis of many tumors, including gastrointestinal cancer, lung cancer, and prostate cancer. 27 Hyperfibrinogenemia has been significantly related to tumor enlargement, an advanced cancer stage, and a poor prognosis in GC patients. 28 Huang et al. reported that the Fib level in cancer patients was significantly increased compared with the control group. 29 This finding was in line with our results. In addition, a decrease in ALB and PA may lead to malnutrition and immune response disorders, which also may affect the prognosis of GC.30,31 Gupta and Huang et al. confirmed that ALB and PA levels in tumor patients were significantly lower compared with the healthy control group.32,33 Therefore, PFR and AFR, which are composed of PA, ALB, and Fib, may be closely associated with the progression of cancer. PFR was also closely related in acute pancreatitis and cancer 16 and could be used to evaluate the severity of acute pancreatitis and the possibility of complications. 34 In another study, FPR could be used as a useful biomarker for the diagnosis of colorectal cancer and was associated with tumor stage. 35 At the same time, other studies have confirmed AFR as a new tumor prognostic biomarker, 36 and had a relationship with the tumor size, its depth of invasion, and potential lymph node metastasis. 37 Huang et al. showed that AFR was related to the clinicopathological features of tumors in their study of cervical cancer; and AFR was significantly lower in cervical cancer patients. 29 Based on these studies, PFR and AFR values gradually decrease as the disease progresses, which was also in line with our results. We found that PFR and AFR were related to Hp-NGC and that they decreased with increases in the tumor size and depth of invasion. As a result, PFR and AFR could be used as indicators of tumor aggressiveness.
This study evaluated the correlation between PFR and AFR and Hp-NGC. The results showed that PFR and AFR gradually decreased as the disease progressed. We also analyzed the relationship between PFR and AFR and clinicopathological characteristics and found that they were related to the tumor size and depth of invasion. A further stratified analysis showed that the larger the maximum tumor diameter, the deeper the invasion depth and the lower the PFR and AFR levels. Such patients were more prone to neurovascular invasion, suggesting that lower levels of PFR and AFR in patients with GC were associated with a higher postoperative recurrence rate and a worse prognosis. Therefore, it is necessary to strengthen postoperative chemotherapy and perform frequent follow-up examinations.
When analyzing the diagnostic value of PFR and AFR in Hp-NGC patients and other populations, the diagnostic efficiency of combining PFR and AFR in differentiating Hp-NGC from healthy controls was higher than that of any single indicator (AUC = 0.908, sensitivity = 78.33%, specificity = 92.98%). In addition, the AUCs of the combined PFR and AFR in the diagnosis of HpN patients and healthy controls and of HpN patients and Hp-NGC patients were 0.813 and 0.654, respectively, indicating that PFR and AFR can also be used as moderate predictors for the differential diagnosis of HpN and Hp-NGC. However, the AUCs of HpN patients and healthy controls and of Hp-NGC patients and healthy control models were lower than the AUCs of Hp-NGC patients and healthy control models; therefore, Hp-NGC patient and healthy control models were still the best diagnostic models to use to diagnose Hp-NGC. Based on these results, the combined application of PFR and AFR had higher diagnostic efficiency for Hp-NGC than either method used alone.
There were some limitations to our study. First, the sample size was small, and all participants came from the same hospital, which might have caused errors in the results. Second, the study was a single-center retrospective study, and the results might not be comprehensive enough. Finally, prospective multi-center studies should be conducted in the future to verify these results more comprehensively.
Conclusions
Low PFR and AFR values were related to the tumor size and depth of invasion. PFR and AFR may be used as diagnostic biomarkers for Hp-NGC. The combination of PFR and AFR had a higher diagnostic efficiency than a single diagnosis of Hp-NGC. It might be an economical, convenient, and promising biomarker.
Supplemental Material
sj-docx-1-jbm-10.1177_17246008211072875 - Supplemental material for Diagnostic value of combined prealbumin-to-fibrinogen and albumin-to-fibrinogen ratios in Hp-negative gastric cancer
Supplemental material, sj-docx-1-jbm-10.1177_17246008211072875 for Diagnostic value of combined prealbumin-to-fibrinogen and albumin-to-fibrinogen ratios in Hp-negative gastric cancer by Linyan Zhang, Simeng Qin, Liuyi Lu, Li Huang and Shan Li in The International Journal of Biological Markers
Footnotes
Author contributions
Shan Li conceived the study design, Linyan Zhang wrote the manuscript, Simeng Qin was involved in data acquisition, Liuyi Lu and Li Huang performed the analyses. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant number 81460431).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
