Abstract
Background:
Albumin to fibrinogen ratio (AFR) play a crucial role in the progression and prognosis of many malignant tumors. This study aimed to comprehensively assess the diagnostic value of AFR as single markers or in combination with squamous cell carcinoma antigen (SCC-Ag), cancer antigen 125 (CA-125) in cervical cancer.
Methods:
A total of 323 cervical cancer inpatients, 143 patients with cervical intraepithelial neoplasia (CIN) and 317 healthy controls were analyzed. Differences in laboratory parameters and clinicopathological features were calculated using the Mann–Whitney U or Kruskal–Wallis H test. The receiver operating characteristic (ROC) curve was used to evaluate the predicted value of AFR, alone or combined with SCC-Ag, CA-125 for the diagnosis of cervical cancer.
Results:
The levels of AFR in patients with cervical cancer were significantly lower than those in the CIN patients and the control subjects. AFR were not only negatively correlated with the tumor stage, but also related to histology typing, lymph node metastasis, distant metastasis, depth of stromal infiltration, tumor size, and tumor stage; however, it was not associated with the blood group. AFR combined with SCC-Ag possessed a larger area under the curve (AUC; AUCAFR+SCC-Ag = 0.924, 95% confidence interval (CI) 0.900, 0.944) than AFR (P < 0.001), SCC-Ag (P < 0.001), or CA-125 (P < 0.001) did alone.
Conclusions:
The pretreatment levels of AFR, alone or combined with SCC-Ag, CA-125 could improve the diagnostic efficiency of cervical cancer.
Introduction
Cervical cancer is the fourth most common cause of female cancer-associated morbidity and mortality worldwide, with around 0.5 million new cases and 0.3 million deaths in 2018. 1 Almost one-half of the new cases and over one-half of the cancer deaths occurred annually in Asia, especially in underdeveloped countries. 2 So far, screening is the most effective way of preventing and diagnosing cervical cancer 3 ; however, cytology screening is not optimal due to shortcomings that are mainly related to sample collection, low sensitivity, and the time required for analysis. In addition, although persistent human papillomavirus (HPV) infection is the pathogenetic risk factor for cervical cancer, HPV testing only detects transient infection, which may lead to unnecessary treatment or excessive medication. 4 Moreover, the lower screening coverage rates and poor sanitation in developing countries are other causes of poor performance. 5 Therefore, a simple, economical, and effective biomarker is required to precisely predict cervical cancer.
Inflammation is a main crucial hallmark of malignancies and is involved in various tumors, including cervical cancer.6,7 Research on inflammation and inflammatory mediators has revealed that inflammation markers can trigger a premetastatic niche to escape the immunological surveillance and to play decisive roles at every step of tumorigenesis and development. 8 These inflammatory molecules—such as neutrophils, 9 platelets, 10 lymphocytes, 11 fibrinogen (Fib), 12 albumin (Alb), 13 and combinations of several single markers in the forms of ratios,14,15 which are usually derived from circulating immune cells and acute-phase reactive proteins—have been reported as valuable predictors in a variety of human tumors. Currently, many studies have applied these routine parameters for the diagnosis and prognosis of malignant tumors.
Fib and Alb have both been recognized as key components in systemic inflammation. Fib is not only one of the vital acute-phase proteins in the systemic inflammation response but also a crucial element in the coagulation cascade. A high level of circulating Fib is generally present in solid malignancies, and the plasma Fib levels is commonly related to advanced tumor stage, deep stromal invasion, large tumor size, and tumor recurrence.16-18 Meanwhile, research on the role of Alb used for the early diagnosis and prognosis of malignant tumors is increasing, and hypoalbuminemia has been reported as part of a prognostic model regarding cervical cancer survival. 19 The albumin to fibrinogen ratio (AFR), as a novel inflammation-based marker that combines Fib and Alb was recently proposed and is currently being reported on. The diagnostic and prognostic power of the ratio of AFR was exhibited in several types of human cancers 20 other than cervical cancer. However, few studies have been conducted on the diagnostic values of AFR as single markers or in combination with SCC-Ag, CA-125 for cervical cancer. Hence, we investigated the diagnostic efficacy of these prospective parameters for cervical cancer in this study.
Materials and methods
Eligible patients
All participants in this study were enrolled form the First Affiliated Hospital of Guangxi Medical University between 2012 and 2018. A total of 323 cervical cancer cases, 143 cervical intraepithelial neoplasia (CIN) patients, and 317 healthy volunteers were analyzed in the present study. The control group was healthy individuals who matched with the cervical cancer patients in age, and were selected from individuals who had come to the Physical Examination Center of the hospital for a health examination. The healthy controls had no history of cancer and no clinical evidence of cervical disease. The CIN patients were confirmed by colposcopy-directed biopsy and underwent conization. Women with a diagnosis of gynecological malignant tumor, previous laser therapy, and vaginal surgery were excluded from the study. The inclusion criteria for the cervical cancer patients were: (a) newly diagnosed with cervical cancer as treated by surgery and verified by histopathology; (b) clinical staging of cervical cancer according to the International Federation of Gynecology and Obstetrics (FIGO) staging criteria (FIGO 2018); (c) not receiving any anticoagulant drugs before enrollment; and (d) no hematological diseases, autoimmune, infectious disease or concomitant malignant tumors. According to clinical characteristics and imaging results, there were 18 patients with stage IA, 194 patients with stage IB, and 121 patients with stage IIA. This study was approved by the ethics committee of the First Affiliated Hospital of Guangxi Medical University. Informed consent was obtained from all enrolled subjects.
Laboratory measurements
All the data, except for fibrinogen concentrations in healthy controls, were obtained from the hospital’s electronic medical record for each eligible participant. Peripheral blood samples of 2 mL were phlebotomized in the early morning before pretreatment. After 10 min of centrifugation at 4000 rpm/min, serum and plasma were collected for analysis within 4 hours. The concentrations of the coagulation profile were all tested using a Sysmex CA7000 automatic coagulation analyzer and related reagents were used. Whole peripheral blood cell parameters were measured by a Beckman-Coulter LH 780 hematology analyzer (Beckman Coulter, Brea, CA). The serum albumin concentration was detected with a Hitachi 7600 automatic biochemical analyzer (Tokyo, Japan) using the bromocresol green method produced by Shanghai Zhicheng Bio-Technology Co., Ltd. The values of serum CA-125 and SCC-Ag were detected with a Roche E6000 analyzer (Roche Diagnostics, Basel, Switzerland).
Statistical analysis
The distribution of continuous variables was performed using the Kolmogorov–Smirnov test. The mean and SD were applied for normally distributed data, and non-normally distributed data were expressed as the median and quartile. Clinical/laboratory parameters and relevant indicators were assessed using the Mann–Whitney U test or Kruskal–Wallis H test. Statistical analysis was carried out using SPSS 16.0 software. GraphPad Prism 5 was used for analyzing the correlation between laboratory data and FIGO stages in cervical cancer patients and for presenting a scatter diagram of relevant parameters. The diagnostic values of the candidate markers were estimated with the receiver operating characteristic (ROC) curve and area under the curve (AUC) using MedCalc statistical software (version 18.1.1). A two-tailed P-value < 0.05 was considered statistically significant.
Results
Study population
The laboratory results and demographic characteristics of patients with cervical cancer, CIN, and healthy controls are summarized in Table 1. These parameters were not in accordance with normal distribution. The median age (median; quartile) of the cervical cancer patients, the CIN cases, and the control individuals were 46 (41–51), 45 (38–51), and 45 (35–55) years, respectively. The measured results for white blood cells (WBC), platelets (PLT), Fib, serum squamous cell carcinoma antigen (SCC-Ag), and cancer antigen 125 (CA-125) were higher in cervical cancer patients compared with those in the control and CIN groups. Conversely, the levels of median Alb, hemoglobin (Hb), and AFR were significantly lower in the cervical cancer patients than in the controls and the CIN patients. SCC-Ag levels were only obtained from 296 patients with cervical cancer. According to the FIGO stage, 194 patients had stage I, 95 had stage II, 17 had stage III, and 17 had stage IV. The numbers of cases in CIN I, II, and III were 15, 123, and 5, respectively.
Laboratory parameters among cervical cancer patients, patients with cervical intraepithelial neoplasia, and healthy controls.
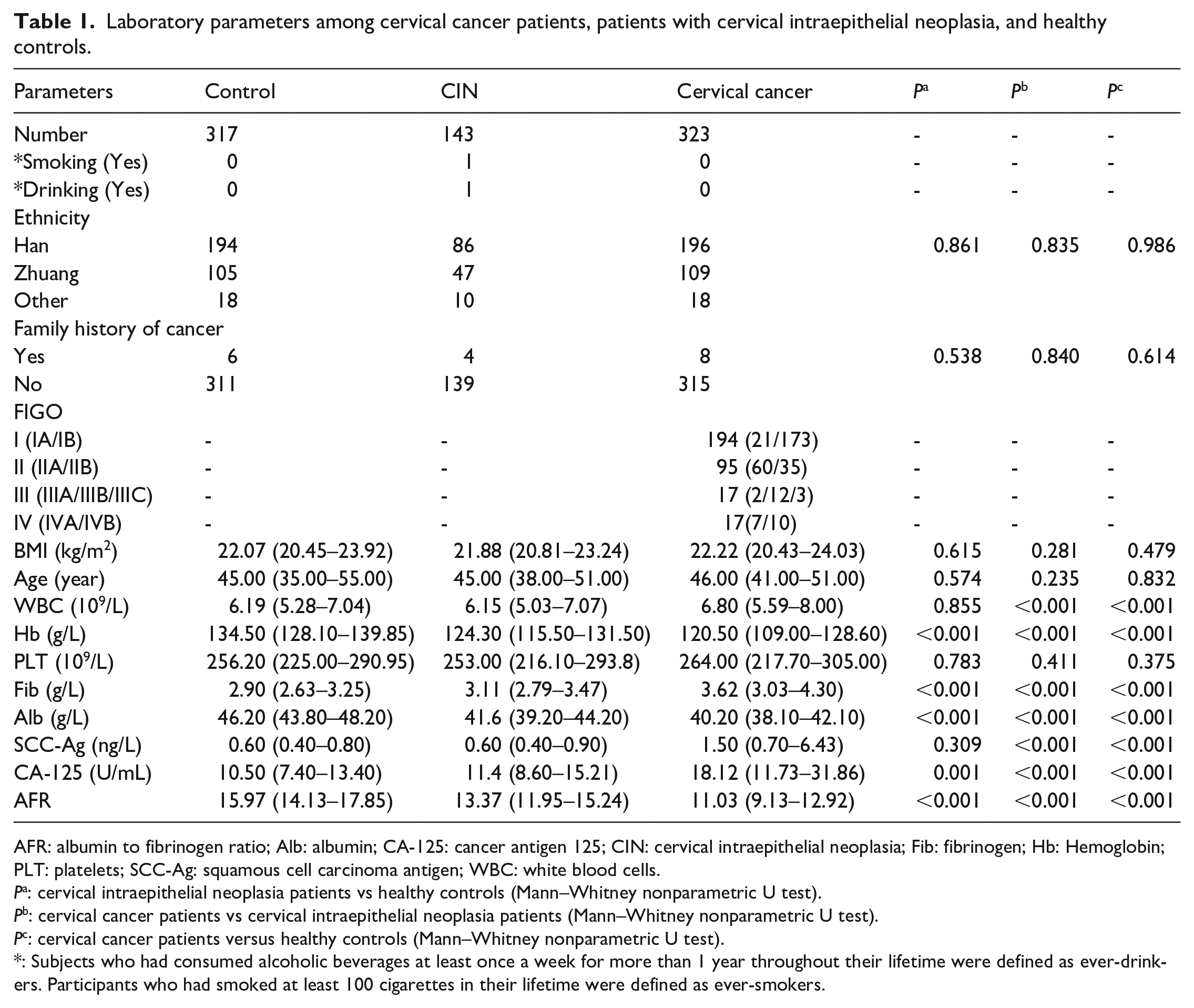
AFR: albumin to fibrinogen ratio; Alb: albumin; CA-125: cancer antigen 125; CIN: cervical intraepithelial neoplasia; Fib: fibrinogen; Hb: Hemoglobin; PLT: platelets; SCC-Ag: squamous cell carcinoma antigen; WBC: white blood cells.
Pa: cervical intraepithelial neoplasia patients vs healthy controls (Mann–Whitney nonparametric U test).
Pb: cervical cancer patients vs cervical intraepithelial neoplasia patients (Mann–Whitney nonparametric U test).
Pc: cervical cancer patients versus healthy controls (Mann–Whitney nonparametric U test).
: Subjects who had consumed alcoholic beverages at least once a week for more than 1 year throughout their lifetime were defined as ever-drinkers. Participants who had smoked at least 100 cigarettes in their lifetime were defined as ever-smokers.
Correlations between AFR, SCC-Ag, CA-125, and clinicopathological characteristics in patients with cervical cancer
The values of SCC-Ag, CA-125, and AFR in the cervical cancer patients were both closely related to the clinical features, including histology typing, lymph node metastasis, depth of stromal infiltration, distant metastasis, tumor size, and International Federation of Gynecology and Obstetrics (FIGO) stage, but they were not associated with blood group (Table 2).
Correlation between clinicopathological features and SCC-Ag, CA-125, and AFR in cervical cancer.
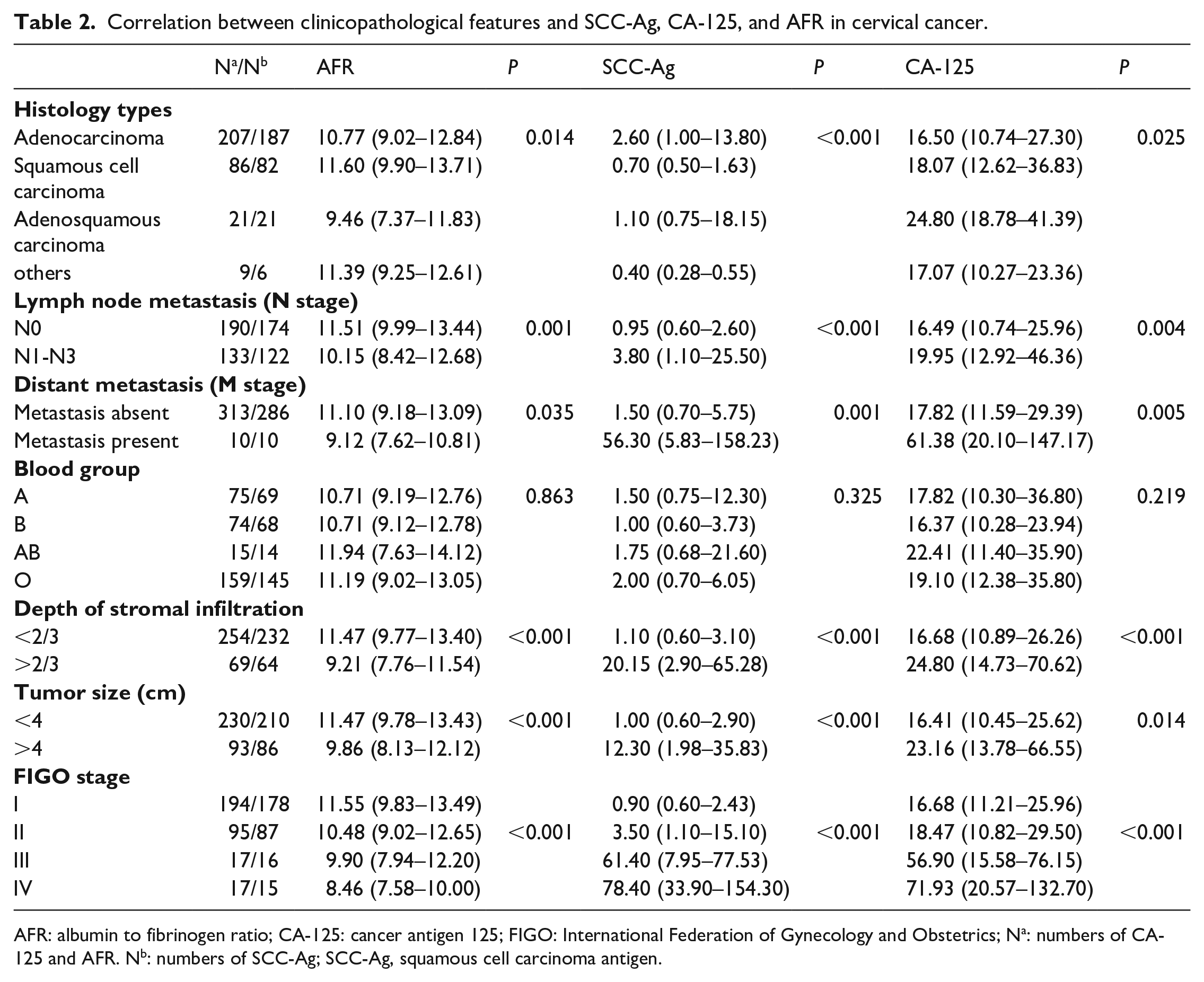
AFR: albumin to fibrinogen ratio; CA-125: cancer antigen 125; FIGO: International Federation of Gynecology and Obstetrics; Na: numbers of CA-125 and AFR. Nb: numbers of SCC-Ag; SCC-Ag, squamous cell carcinoma antigen.
As shown in Figure 1, the results demonstrated that Fib (P < 0.001), SCC-Ag (P < 0.001), PLT (P = 0.006), and CA-125 (P < 0.001) were positively and consistently correlated with an advanced cervical cancer stage. Nevertheless, the Hb levels and AFR continuously and sharply decreased with tumor progression (all P < 0.001). Compared with those of controls, the median differences in AFR were −27.68%, −34.38%, −38.01%, and −47.03%, respectively. However, serum concentrations of Alb suddenly declined by stage IV after a steady increase from stage I to III, and there was no significant difference between the cancer stages. No significant interaction between WBC and tumor stage was found.
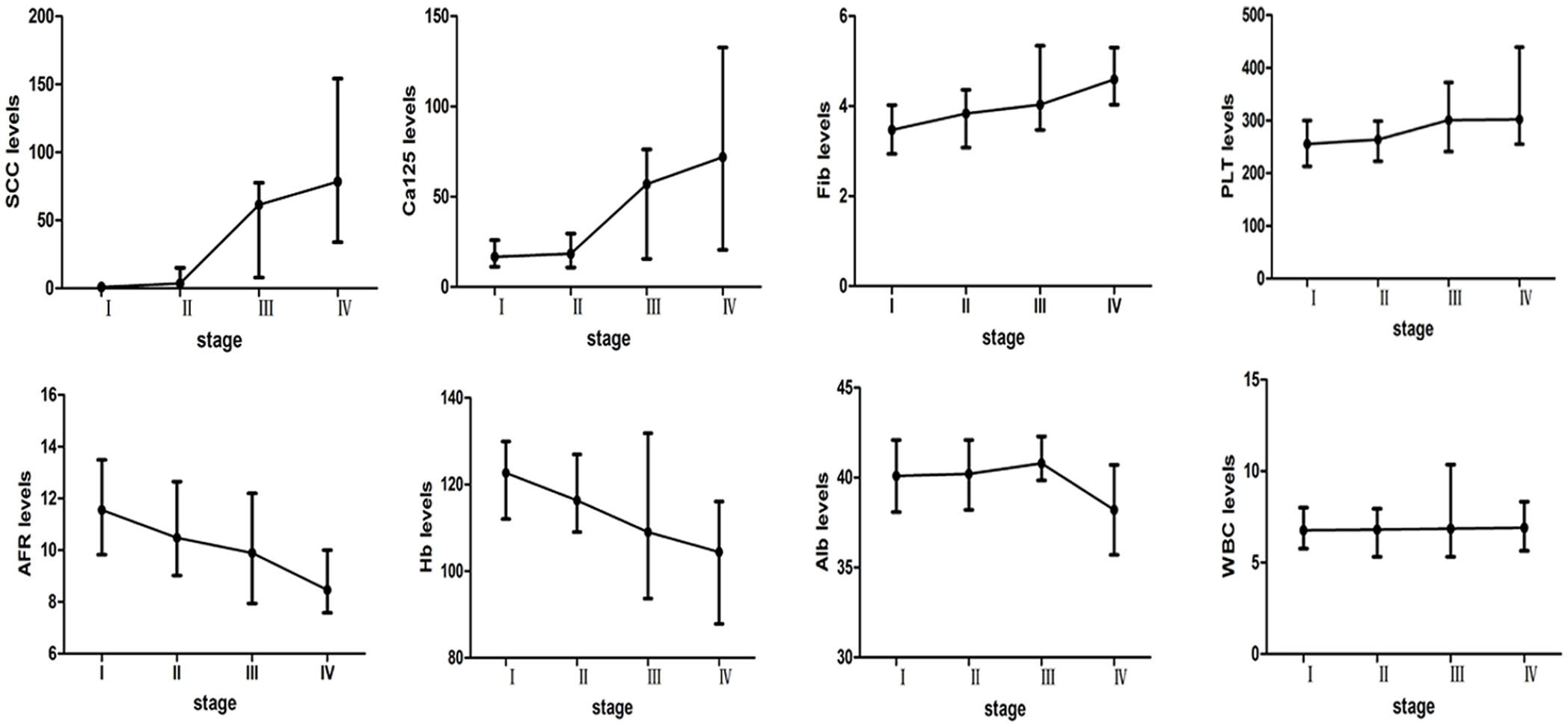
Association of laboratory parameters with FIGO stages in cervical cancer patients.
The treatment protocols for cervical cancer patients may vary depending on the FIGO stage. Patients with a stage below IIB tend to be treated with surgery, whereas patients at stage IIB and above are advised to undergo adjuvant radio-chemotherapy. The diagnostic significance of pretreatment values of SCC-Ag, CA-125, and AFR for tumor staging were performed by ROC analysis. The diagnostic efficiency of AFR (AUC = 0.863) for patients with tumor stage below IIB were superior to SCC-Ag (AUC = 0.759) and CA-125 (AUC = 0.759). However, the results revealed that AFR had poorer predictive power than SCC-Ag for patients with tumor stage IIB, suggesting that AFR was the best among these biomarkers for detecting early cervical cancer (Supplementary Table 1).
Diagnostic efficacy of SCC-Ag, CA-125, and AFR alone or in combination
The results of the ROC curve analysis are presented in Table 3 and Figure 2. In the single index analysis, AFR had the highest sensitivity (74.61%) and AUC (0.879) in differentiating the cervical cancer group from the control group, whereas CA-125 had the highest specificity (93.06%). The optimal cutoff values of AFR, SCC-Ag, and CA-125 were 12.89 (positive likelihood ratio (PLR) = 8.16, negative likelihood ratio (NLR) = 0.28), positive predictive value (PPV) = 89.3%, negative predictive value (NPV) = 77.8%), 1.40 (PLR = 32.77, NLR = 0.49, PPV = 96.8%, NPV = 68.6%), and 16.30 (PLR = 8.03, NLR = 0.48, PPV = 89.1%, NPV = 67.4%), respectively. The AUC and Youden value of the combination of SCC-Ag and AFR (0.924, 0.712) was larger than the values of the other combinations and SCC-Ag, CA-125, and AFR used alone.
Diagnostic efficacy of SCC-Ag, CA-125, and AFR alone or in combination.
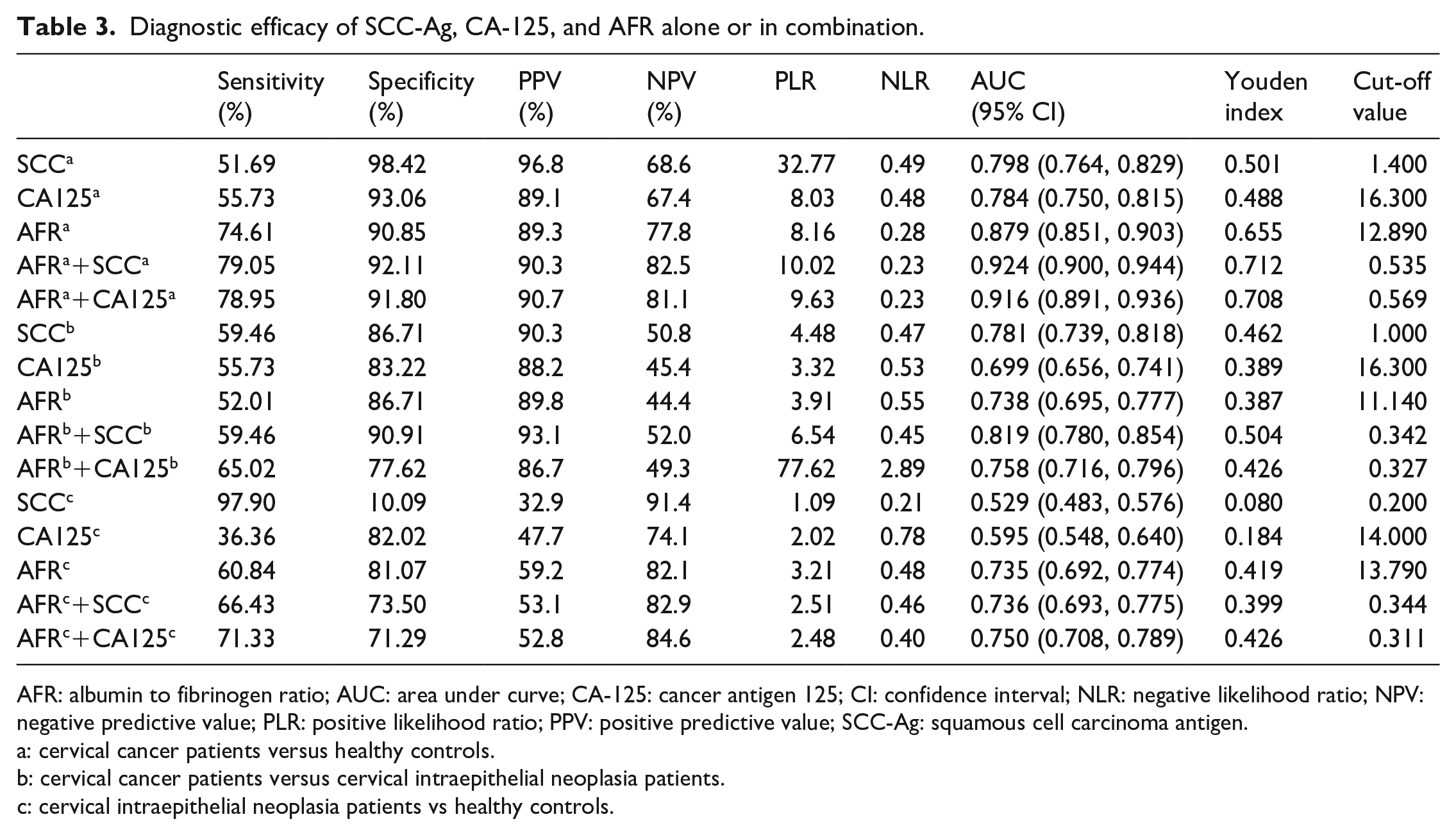
AFR: albumin to fibrinogen ratio; AUC: area under curve; CA-125: cancer antigen 125; CI: confidence interval; NLR: negative likelihood ratio; NPV: negative predictive value; PLR: positive likelihood ratio; PPV: positive predictive value; SCC-Ag: squamous cell carcinoma antigen.
: cervical cancer patients versus healthy controls.
: cervical cancer patients versus cervical intraepithelial neoplasia patients.
: cervical intraepithelial neoplasia patients vs healthy controls.
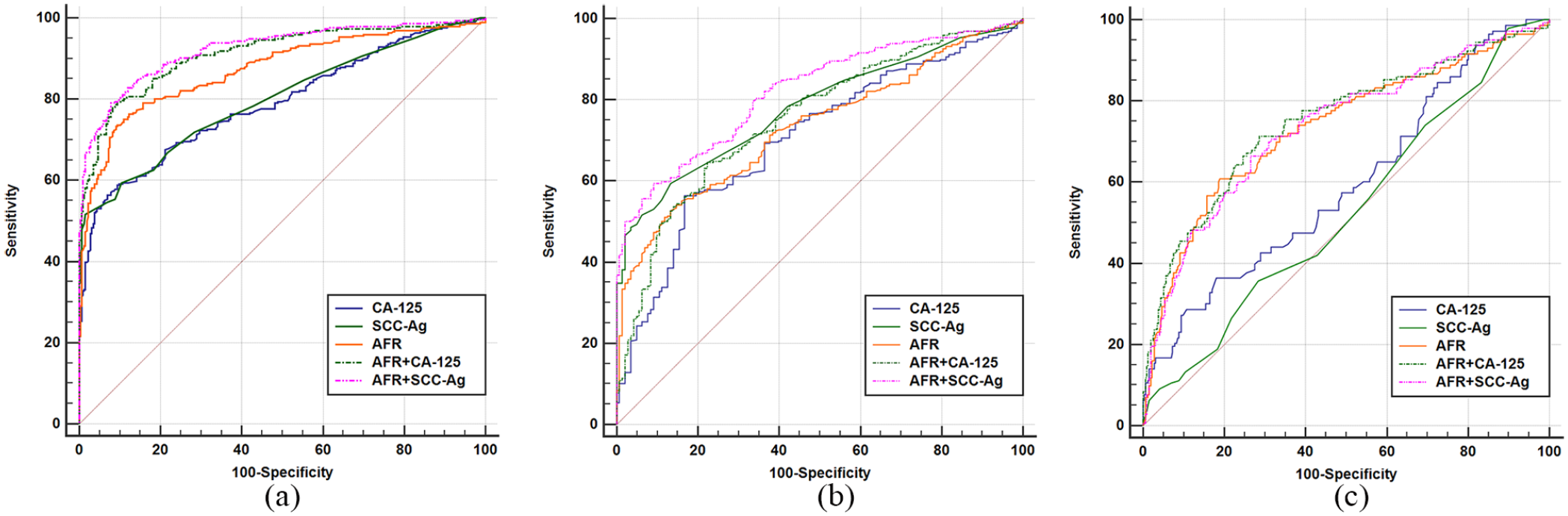
Diagnostic efficacy of SCC-Ag, CA-125, and AFR alone or in combination. (a) Cervical cancer patients versus healthy controls. (b) Cervical cancer patients versus cervical intraepithelial neoplasia patients. (c) Cervical intraepithelial neoplasia patients versus healthy controls.
In the cervical cancer patients and CIN cases, the predictive value of SCC-Ag was moderately better than AFR and CA-125, while the AUC was 0.781, 0.738, and 0.699, respectively. However, AFR in conjunction with SCC-Ag enhanced the diagnostic efficacy compared to SCC-Ag, CA-125, and AFR alone.
The AUC value of AFR (0.735) was superior to SCC-Ag (0.529) and CA-125 (0.595) in discriminating CIN patients from healthy controls. The sensitivities of AFR, SCC-Ag, and CA-125 were 60.84%, 97.90%, and 36.36%, respectively, while the specificity was 81.07%, 10.09%, and 82.02%, respectively.
Discussion
Early screening and definitive diagnosis are vital for controlling the development of cervical cancer. Research has demonstrated that no effective tumor marker is commonly applied in the early diagnosis, prognostic evaluation, treatment monitoring, and follow-up of patients with cervical cancer. 21 So far, the detection and diagnosis of cervical cancer mainly rely on cytology and biopsy, although these approaches are characterized by low sensitivity, and they are not routine in physical examinations. There is an urgent need to develop specific tumor markers to aid the diagnosis of cervical cancer, which will further reduce the economic burden on patients.
As a valuable indicator, AFR or fibrinogen to albumin ratio (FAR), was applied to assess the systemic inflammatory response (SIR) and/or nutrition status in many cancers and other serious diseases.13,18,22,23 The diagnostic and clinicopathological value of AFR has been reported in numerous cancers. Li et al. 24 reported that AFR was a prospective biomarker to predict tumor size and clinical outcome in non-small cell lung cancer. The FAR was significantly correlated with tumor length, pT status and pN status in another esophageal cancer study. 23 Moreover, a mate-analysis uncovered that high FAR was significantly correlated with deeper infiltration, positive lymph node metastasis, distant metastasis, and advanced clinical stage, whereas no intergroup difference was observed between the FAR and histological grade. 20 Similarly, our findings demonstrated that patients with low AFR had a significantly higher tumor stage than those with high AFR, as well as an association with other clinicopathological features (except blood group) in this study.
The mechanism of AFR that could help to monitor the tumor characteristics of cervical cancer is unclear. The following causes and evidence may account for our findings: as an acute-phase protein in inflammatory environments, Fib promotes the synthesis of proinflammatory cytokines and fibroblast growth factors to induce the malignant proliferation of tumor cells and accumulates vascular endothelium to enhance tumor angiogenesis.25,26 The results of the previous studies indicated that the pretreatment concentration of Fib was significantly higher in patients with cancer compared with controls, and the plasma levels of Fib consistently and steadily increased with the advanced cancer stage, deep stromal invasion, large tumor size, and tumor recurrence in patients with early stage cervical cancer.27,28 Also, multivariate logistic regression analysis revealed that high plasma levels of fibrinogen represented a risk factor for pelvic lymph node metastasis in patients with early stage squamous cell carcinoma of the uterine cervix. 29 Similarly, we discovered that the concentration of Fib was elevated with tumor stage in patients with cervical cancer. Furthermore, as fibrinogen receptors, platelets induce fibrinogen adherence to malignant cells, leading to the formation of micro-emboli in targeted organs, which increase with tumor progression and metastasis. 30 Interestingly, this research also proved that the number of platelets in peripheral blood moderately increased with tumor stage, which is consistent with previous studies. On the other hand, Alb also plays a vital role in the systemic inflammatory response. In one study, Alb levels were inhibited by a series of cancer-related cytokines and were negatively correlated with tumor-related inflammatory responses. 31 Furthermore, clinical studies have shown that a low concentration of Alb is associated with the prognosis of various types of tumors; that is, cancers with unfavorable prognosis had lower serum Alb levels.32,33 Hence, the AFR was made up to further amplify the opposing effects for improving its predictive value, which was proved in our and previous research.13,20
Previous literature reported data on the value of serum tumor-associated antigens in the diagnosis management of cervical cancer, showing that the elevation rates of pretreatment CA125 levels and serum SCC-Ag levels are 28%–88% and 20%–75%, respectively. 21 In the present study, the pretreatment SCC-Ag and CA125 levels positively correlated with tumor stage, whereas the SCC-Ag data were not available for some patients, and the sensitivities of CA125 and SCC-Ag were only 55.73% and 51.69%, which is in accordance with previous results. The relatively low sensitivity and high cost of these serum tumor markers drives the discovery of better predictive markers. Our results demonstrated that the AUC and sensitivity of AFR was larger in discriminating cervical cancer patients from healthy controls compared with SCC-Ag or CA-125 used alone. AFR was superior to SCC-Ag or CA-125 alone for distinguishing CIN patients from healthy controls. Also, the diagnostic efficacy of AFR (AUC = 0.863) in FIGO stage below IIB cervical cancer was better than SCC-Ag and CA-125, indicating that AFR was a superior biomarker to others for detecting early cervical cancer. The predicted value of SCC-Ag combined with AFR produced superior accuracy in distinguishing cervical cancer patients from controls or CIN cases, exhibiting a larger AUC than SCC-Ag, CA-125, or AFR used alone. These parameters are all conveniently based on information available from routine blood and laboratory tests, highlighting the clinical value of AFR for diagnostic decision making. Hence, AFR, alone or in combination with SCC-Ag, CA-125, may represent a promising diagnosis biomarker for the progression of cervical cancer.
Although the present study first clarifies the diagnostic value of AFR in cervical cancer, several limitations are inevitable and should be carefully considered. First, this study only analyzed subjects from the same hospital, and the relevant data on patients with cervicitis were not available, and more than 85% (123/143) of patients with CIN II was not suitable for graded analysis. Second, the uncommon distribution of the CIN lesions and the small number of patients with advanced cervical cancer and distant metastasis may create bias in evaluating the predictive value of these markers. Third, relevant personal information, including hygiene habits and viral infections, was not collected, which may have affected the final results. The present conclusions need to be validated by a larger cohort size and multi-center clinical prospective study.
In summary, this analysis evaluated the diagnostic significance of the markers of AFR, SCC-Ag, and CA-125 for cervical cancer, representing the first such study. The results showed that AFR was significantly associated with histological type, lymph node metastasis, distant metastasis, depth of stromal infiltration, tumor size, and FIGO stage in cervical cancer. AFR, alone or in combination with SCC-Ag, CA-125, may be helpful for predicting the development and clinical characteristics of cervical cancer.
Supplemental Material
Supplementary_Table_1 – Supplemental material for Diagnostic Value of Albumin to Fibrinogen Ratio in Cervical Cancer
Supplemental material, Supplementary_Table_1 for Diagnostic Value of Albumin to Fibrinogen Ratio in Cervical Cancer by Li Huang, Zhuning Mo, Linyan Zhang, Shanzi Qin, Simeng Qin and Shan Li in The International Journal of Biological Markers
Footnotes
Author contributions
Li Huang and Zhuning Mo contributed equally to this work and should be considered as co-first authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Self-financing Scientific Research Subject of Guangxi Health Department (Z20170315) and the Youth Science Foundation of The People’s Hospital of Guangxi Zhuang Autonomous Region (QN2018-7).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
