Abstract
Purpose
To study in detail the expression pattern and prognostic significance of TMPRSS4 in colorectal cancer.
Methods
The expression of TMPRSS4 protein was determined using Western blot in the colorectal cancer tissues and normal tissues. Immunohistochemistry was used to detect the TMPRSS4 expression in colorectal cancer tissues, and the clinicopathologic characteristics and prognostic significance were analyzed.
Results
TMPRSS4 overexpression was associated with tumor budding, lymphovascular invasion, perineural invasion, cancerous emboli, infiltration depth, lymph node metastasis, distant metastasis, and tumor node metastasis stage (P < 0.05 for all). Interestingly, TMPRSS4 expression in the tumor budding, tumor emboli, lymph node, and liver metastatic tumor samples was higher than in the paired primary tumors. In contrast, TMPRSS4 overexpression is inversely correlated with both the overall survival and the disease-free survival of the patients with colorectal cancer (P < 0.05 for both). Also, we found that TMPRSS4 is only of significance in predicting the prognosis of stage III and IV colorectal cancer, not stage I and II.
Conclusions
TMPRSS4 was shown to be involved in the whole process of metastasis from tumor budding to lymph node and/or distant metastasis in colorectal cancer and predicted the unfavorable prognosis of stage III–IV, indicating that it is a novel target for the precise treatment of colorectal cancer with lymph node or distant organ metastasis.
Introduction
Colorectal cancer (CRC) is one of the most commonly malignant tumors worldwide, and over 1 million new cases are confirmed yearly. 1 Although great progress in therapeutic strategies, such as surgical techniques and systemic treatments, have improved the overall survival of patients with CRC when diagnosed at an early stage, 2 many patients were initially diagnosed at advanced stages with poor prognosis due to atypical early-onset symptoms. 3 It was reported that approximately 25% patients present with hepatic metastases at the initial diagnosis, 4 of which the overall median survival is 3.6 (range: 1.7–7.3) years. 5 Even more unfortunately, among these patients with hepatic metastasis, nearly 65% may suffer a relapse after hepatectomy, the majority of which will thoroughly lose the opportunity of cure. 6 Therefore, a better CRC-related biomarker for the accurate monitoring of patients' progression, their response to treatment, and prognosis is necessary.
The human type II transmembrane serine proteases (TTSPs) family has 17 members, including the 7 members of the hepsin/TMPRSS subfamily: hepsin, TMPRSS2, TMPRSS3, TMPRSS4, TMPRSS5/spinesin, mosaic serine protease large-form (MSPL), and enteropeptidase. 7 Through their dysregulated expression, most TTSPs are associated with the development and progression of tumor. TMPRSS4 is a member of the TTSPs family, which is overexpressed in lung, gastric, colon, and thyroid cancer. 8 A recent study showed that TMPRSS4 is an important mediator of invasion, metastasis, migration, adhesion, and epithelial mesenchymal transition (EMT) in human epithelial cancer cells. 9 Our group further demonstrated that TMPRSS4 induces invasion and EMT by upregulating integrin α5 and its signaling pathways. 10 It was also found that TMPRSS4 upregulates uPA gene expression through activating the JNK signaling to induce cancer cell invasion. 11 Interestingly, TMPRSS4 was demonstrated to be a poor prognostic factor for triple-negative breast, gastric, pancreatic cancer, etc. 12 To date, many studies on TMPRSS4—including our previous reports—mainly concentrated on revealing the signaling pathways. However, to our knowledge, no studies depict in detail the expression pattern of TMPRSS4 from a morphological point of view in CRC. Although higher TMPRSS4 expression was found to predict shorter overall survival (OS) and disease-free survival (DFS) in CRCs, its prognostic significance is not further revealed in different stages of CRC. 13
Herein, we firstly describe the expression pattern of TMPRSS4 in CRC tissue, and investigate the relationship between the clinicopathological characteristics of TMPRSS4 and its potential prognostic significance in detail.
Methods and materials
Cell culture
HT29 human colon cancer cell lines were obtained from the Korean Cell Bank (Seoul, Korea) and were cultured using the RPMI 1640 medium (Gibco; Thermo Fisher Scientific, Inc.) supplemented with 10% fetal bovine serum (Invitrogen, CA, USA) and 1% penicillin–streptomycin (Invitrogen) at 37°C in a humidified atmosphere of 5% CO2.
Transient transfection
HT29 human colon cancer cells were seeded in 96-well culture plates at a density of 3 × 103 cells/well and were divided into three groups: siRNA-TMPRSS4 group, empty vector group, and control group. For knockdown of TMPRSS4 studies, a pcDNA3.1(−) expression vector containing full length siRNA of the TMPRSS4 gene (Becton Dickinson and Company, New Jersey, USA) was transfected in HT29 human colon cancer cells in the siRNA-TMPRSS4 group. A pcDNA3.1(−) empty vector was transfected in HT29 human colon cancer cells of empty vector group. As a blank control, HT29 human colon cancer cells from the control group were not transfected. All groups of plasmids were then amplified in the cells.
Wound-healing migration assay
Wound-healing migration assay was performed to determine the migration of HT29 human colon cancer cells. Briefly, a “scratch” was created in the cell layers to form a wound gap using 200 μL pipette tip. HT29 human colon cancer cells were incubated and allowed to migrate for 24 h at 37°C in 5% CO2. After 24 h, the widths of the wounds were measured and imaged using a light microscope (OLYMPUS SZ61, Tokyo, Japan). The experiment was repeated three times.
Transwell invasion assay
Cell invasion was detected by the Transwell invasion assay. Briefly, the upper surface of Transwell porous membrane was coated with Matrigel substrated (Corning Inc., CA, USA). 3 × 104 cells of HT29 human colon cancer cells (3 × 104 cells/well) in 200 µL serum-free medium were seeded to the upper chambers of the Transwell. The lower chamber was filled with a standard cultivation medium. HT29 cells were allowed to invade though the porous membrane for 24 h at 37°C in 5% CO2. The invaded cells were fixed with 95% ethanol and counted in five randomly selected fields by the computer-based microscopy imaging system. The experiment was repeated three times.
Patient samples
Samples of CRC and paired normal mucosa were taken from 224 patients who underwent colorectal resection at Chonnam National University Hwasun Hospital (CNUHH) between April 2004 and November 2005. Informed consent was obtained from patients from the CNUHH-National Biobank of Korea (NBK), and the study was approved by the institutional review board of CNUHH. Immunohistochemistry (IHC) was performed to study the clinicopathological factors on the CRC specimens, which were classified according to the tumor node metastasis (TNM) classification of the American Joint Committee on Cancer (AJCC). 14 The clinical outcome was determined from the date of surgery until patients' death or 30 October 2012, which resulted in a follow-up period of from 1.9 to 99.5 months (median 64.7 months). The cases that were lost to follow-up and deaths caused by problems other than CRC were regarded as censored data in OS and DFS analysis.
Western blot analysis
Colonic tissue biopsies were homogenized in lysis buffer (1% NP-40, 50 mM Tris-Cl, 150 mM NaCl, 50 mM EDTA, 1 mM PMSF), incubated on an ice-bath for 30 min, centrifuged at 13,000 rpm for 20 min, and the supernatants were used as protein lysates. Proteins were solubilized in sodium dodecyl sulfate, separated in 12% sodium dodecyl sulfate-polyacrylamide gels using electrophoresis and transferred onto poly-vinylidene fluoride membranes. Membranes were blocked overnight at 4°C and incubated with primary antibodies of TMPRSS4 (Dilution 1:1000; Protein Tech Group, IL, USA) and GAPDH (Dilution 1:4000; Santa Cruz, CA, USA) for 3 h at room temperature. Blots were washed with TBST and probed with anti-rabbit horseradish peroxidase conjugated secondary antibody (1:4000 dilution) for 1 h and developed with ECL reagent. TMPRSS4 protein density in 12 CRC specimens and their paired normal mucosa controls were measured by multi-gauge image analysis software (Fujifilm; Tokyo, Japan).
Immunohistochemical analysis
TMPRSS4 expression in CRC and non-tumor tissues was examined by IHC. Briefly, fresh tissues were fixed in neutral buffered formalin for 24 h and infiltrated with melted Istowax. Histologic evaluation was then performed to individuate and exclude necrotic and hemorrhagic areas. The areas that represent the possible corresponding colorectal tumors were sectioned into 3 μm thickness, and the sections were deparaffinized in xylene and rehydrated with a graded series of ethanol solutions. To augment the expression of antigen in tissues, Trilogy Antigen Retrieval Reagent (Cell Marque; CA, USA) was added to the samples and the samples were boiled in a programmable high-pressure cooker. To suppress the endogenous peroxidase activity, the samples were treated with the peroxidase-blocking solution (DAKO; Glostrup, Denmark) for 20 min, and rinsed with wash buffer (DAKO). To prevent non-specific immune reactions, they were blocked with 3% normal goat serum for 10 min, and rinsed with a wash buffer. Samples were incubated with TMPRSS4 primary antibody (Dilution 1:50; Protein Tech) at 4°C overnight. After rinsing, the Polink-1 polymer HRP second antibody (GBI; Mukilteo, WA, USA) was added and reacted for 30 min. 1 × DAB + chromogen (DAKO) was added and incubated for 10 min at room temperature. The samples were counterstained with hematoxylin and sealed with Crystal Mount (Sakura; Tokyo, Japan). The negative control was prepared by the same procedure using PBS instead of the primary antibody, and the CRC tissues were used as the positive control. In all cases, cytokeratin IHC was used to evaluate tumor budding (Leica Biosystems, Newcastle, UK), and histological sections were immunostained for CD31 (dilution 1:50; DAKO).
Evaluation of immunohistochemical staining
All the slides were examined independently by two pathologists in concordance. The positive samples were further graded 1+, 2+, or 3+ scores based on their intensity of reactivity (Figure 1(a)). The highest intensity of all tissue cores of each tumor was used as its final immunohistochemical result. 15 Scores of 2+ and 3+ were respectively classified as moderate and strong TMPRSS4 expression, and a score of 1+ was weak expression. 16 Tumor budding was evaluated on the invasive margin at an area of 0.785 mm2. Lymphovascular space invasions were recorded as present when any number of tumor cells was indicated within a lymphovascular space delineated by CD31-positive endothelial cells. 17
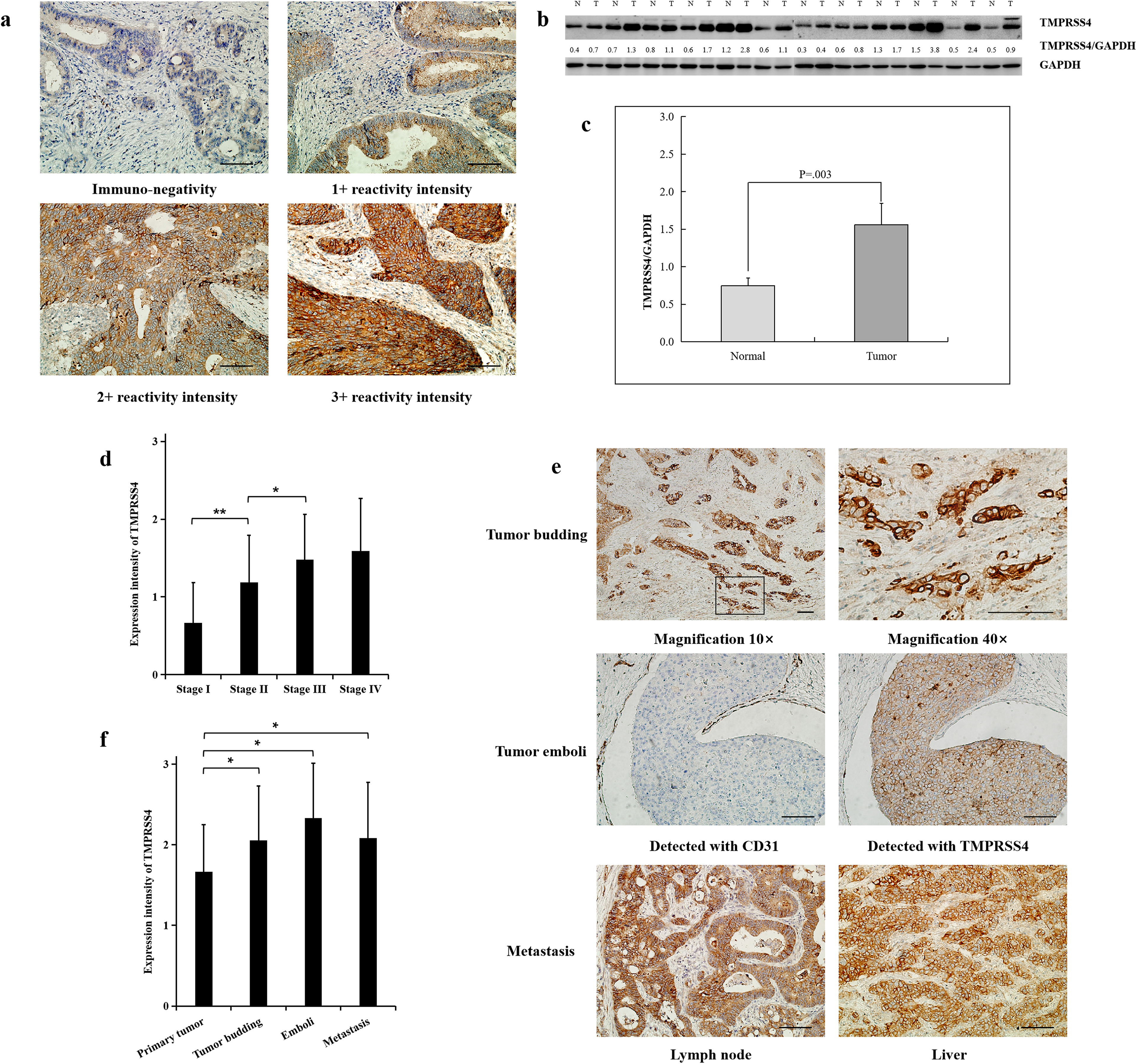
Immunohistochemical and western blot analyses of TMPRSS4 protein expression in CRC. (a) Immunohistochemical intensity classification of TMPRSS4 expression. (b and c) Western blot analysis for TMPRSS4 expression in human colorectal tissue samples from CRC patients. (b) TMPRSS4 proteins are highly expressed in tumoral tissues with specific bands of 55 KD; (c) Results are shown as mean ± SD, P < 0.05 versus the control group. (d) The expression of TMPRSS4 increased as the stage became advanced (P < 0.05); (e) Immunohistochemical analyses of TMPRSS4 protein expression in CRC-related tissues. (f) TMPRSS4 expressions in the tumor budding, cancerous emboli, metastatic lymph node and liver tumor samples (P < 0.05) showed higher reactivity than in the paired primary tumor cells. Magnification 20 × for a. scale bar =100 μm. *P < 0.05; **P < 0.001.
Statistical analysis
Differences between and among groups were evaluated using Fisher's exact test or Pearson's χ2 test for qualitative variables, and Student's t-test or analysis of variance for continuous variables. The survival curves were estimated using the Kaplan–Meier product-limit method and the significance of differences between survival curves was determined using the Log-Rank test. Univariate and multivariate analyses were performed using Cox proportional hazards regression modeling. The variables with P-values < 0.05 on the univariate analysis were entered into a multivariate analysis performed by stepwise logistic regression for evaluation the significant independent prognostic factors associated with survival. All statistical tests were two-sided, and statistical significance was defined as P < 0.05. All analyses were performed using the statistical package SPSS version 20.0 (SPSS; IL, USA).
Results
TMPRSS4 expression was increased in CRC tissues compared to the paired non-tumor tissues
IHC staining indicated that TMPRSS4 was predominantly presented in the cell membrane of CRC cells, and normal cells in the paired non-tumor CRC tissues (Figure 1(a)). CRC tissues showed significantly higher levels of the TMPRSS4 protein than their paired normal mucosa tissues (Figure 1(b)). The mean expression value of TMPRSS4 protein in CRC tissues was higher than that in the paired normal mucosa tissues (P = 0.003, t-test) (Figure 1(c)). This indicated that the increase of TMPRSS4 protein may facilitate tumorigenesis in the colon.
Expression levels of TMPRSS4 in different stages of CRC
TMPRSS4 protein was detected in 33 samples (64.7%) of 51 stage I CRC, 60 (92.3%) of 65 stage II CRC, 69 (100%) of 69 stage III CRC, and 39 (100%) of 39 stage IV CRC. Among these cases, 23 (10.3%) scored 0, 136 (60.7%) scored 1+, 56 (25.0%) scored 2+, and 9 (4.0%) scored 3+. Moderate and strong expression of TMPRSS4 protein appeared in 16 (13.8%) stage I–II and 49 (45.4%) stage III–IV tumors, while weak expression was detected in 77 (66.4%) stage I–II and 59 (54.6%) stage III–IV. Furthermore, the expression of TMPRSS4 increased as the stage became advanced, with stage IV CRCs being the highest and stage I CRCs the lowest (P < 0.05) (Figure 1(d)).
The expression pattern of TMPRSS4 in the metastatic process of CRC
The whole metastatic process of CRC may be concluded into the following four steps: tumor budding, lymphovascular invasion, lymphovascular space invasion (tumor emboli), and regional lymph node and/or distant metastasis. Therefore, in order to depict the expression pattern of TMPRSS4 in the metastatic process, we evaluated all tumor and tumor-related tissues, and only 36 patients were found with tumor budding, emboli, and metastasis. After detecting tumors from the 36 patients using IHC, we found the expression of TMPRSS4 tended to be stronger in deeply invading nests especially in the invasive margin, yet weaker in normal cells in CRC tissues. In addition, lymphovascular spaces invasion (cancerous emboli) was found when any number of tumor cells was noted within a lymphovascular space delineated by CD31-positive endothelial cells. 17 Significant correlation between TMPRSS4 expression and cancerous emboli was observed in CD31-positive endothelial cells, which was also seen in metastatic lymph node and liver tumor samples (Figure 1(e)). The statistical difference of TMPRSS4 expression was calculated and the results indicated that no marked differences of TMPRSS4 expressions were identified among the tumor budding, emboli, metastatic lymph node, and liver tumor samples, but they all showed significantly higher reactivity than in the paired primary tumor cells (P < 0.05, Figure 1(f)).
TMPRSS4 protein expression status and clinicopathological findings
TMPRSS4 protein expression was determined by IHC for 224 CRC tissues (Figure 1(a)). Scores of no staining and weak expression were regarded as “negative” for TMPRSS4 protein expression, and scores of moderate and strong expression as “positive” or “overexpression.” TMPRSS4 overexpression was statistically correlated with the presence of lymphovascular invasion (P = 0.020 < 0.05, χ2 test), perineural invasion (P < 0.001, χ2 test), invasive depth (P = 0.008, χ2 test), lymph node metastasis (P < 0.001, χ2 test), distant metastasis (P = 0.003, χ2 test), and AJCC TNM stage (P < 0.001, χ2 test). However, there was no significant correlation of TMPRSS4 expression between age, gender, tumor location, tumor size, or histological grade (P > 0.05, χ2 test) (Table 1).
Comparison of clinicopathological parameters between TMPRSS4 negative and positive CRC.
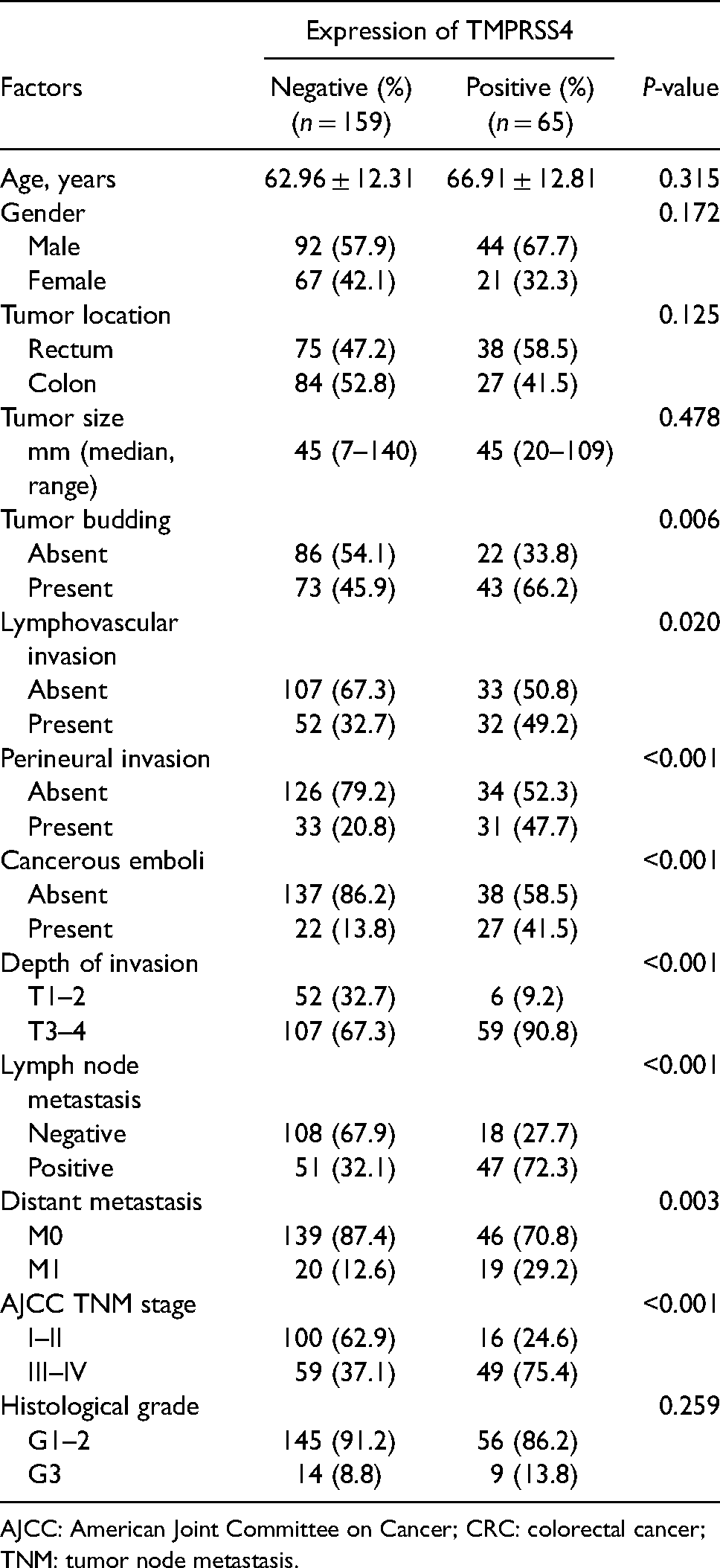
AJCC: American Joint Committee on Cancer; CRC: colorectal cancer; TNM: tumor node metastasis.
TMPRSS4 knockdown inhibits the invasion and migration of CRC cells
To study whether the invasion and migration of CRC cells are regulated by TMPRSS4, wound-healing migration and transwell assay were performed using HT29 human colon cancer cells transfected with siRNA-TMPRSS4. In the absence of siRNA-TMPRSS4, HT29 human colon cancer cells exhibited high migratory capability as indicated by the observed ability to completely close the wound gap. Subsequently, it was found that TMPRSS4 silencing effectively inhibited HT29 cell migration. In the empty vector group and control group, HT29 cells exhibited strong invasive ability and could completely penetrate through the matrigel-coated porous membranes. Therefore, the invasive activity of HT29 cells in siRNA-TMPRSS4 group after 48 h was found to be effectively inhibited by TMPRSS4 silencing (Figure 2).
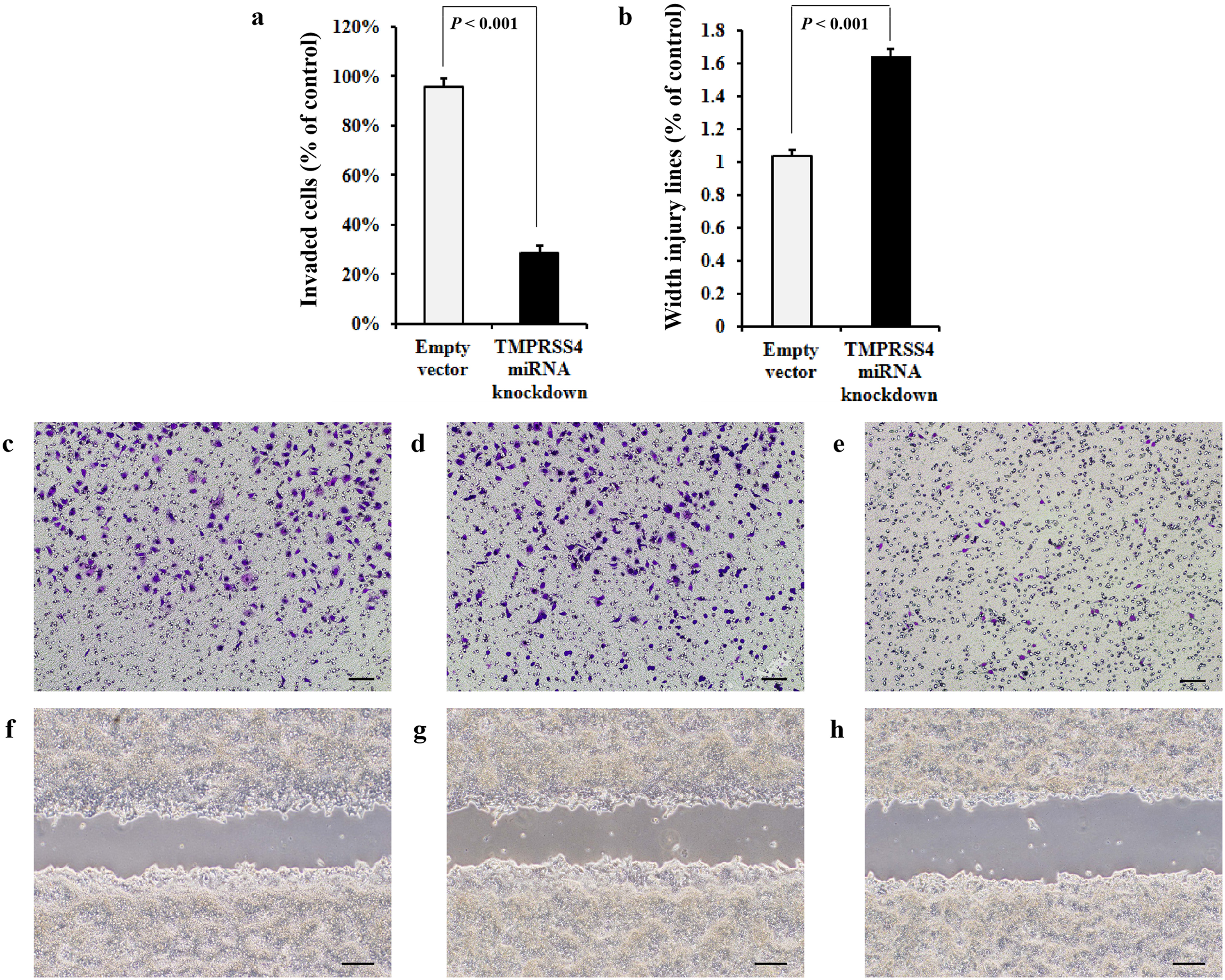
Effects of TMPRSS4 knockdown on the invasive and migratory ability of HT29 cells. (a) The number of invaded cells was significantly lower in the siRNA–TMPRSS4 group than that in the empty group. The y-axis is the number of invaded cells in the siRNA–TMPRSS4 or empty group divided by that in the control group. (b) The wound gap was significantly wider in the siRNA–TMPRSS4 group than that in the empty group. The y-axis is the width of wound in the siRNA–TMPRSS4 or empty group divided by that in the control group. (c–e) The effect of TMPRSS4 knockdown on the invasion of HT29 cells. (c) Control group. (d) Empty group. (e) siRNA–TMPRSS4 group. (f–h) The effect of TMPRSS4 knockdown on the migration of HT29 cells. (f) Control group. (g) Empty group. (h) siRNA–TMPRSS4 group.
Survival analysis
There were 83.5% of patients who had had undergone a curative resection. During the follow-up period, 66 of 224 patients (29.5%) died with CRC. Both the OS and DFS rates of patients with TMPRSS4 overexpression were significantly lower than the rate of those without TMPRSS4 overexpression (P < 0.001, and P = 0.015, respectively) (Figure 3(a) and (d)). In subgroup of TNM stage I and II, TMPRSS4 expression intensity was not significantly correlated with OS and DFS (P = 0.918 and P = 0.790, respectively) (Figure 3(b) and (e)). TMPRSS4 overexpression in advanced TNM stages III and IV of CRC tissues were significantly correlated with the OS (P = 0.006); however, this was not correlated with the DFS (P = 0.134) (Figure 3(c) and (f)).
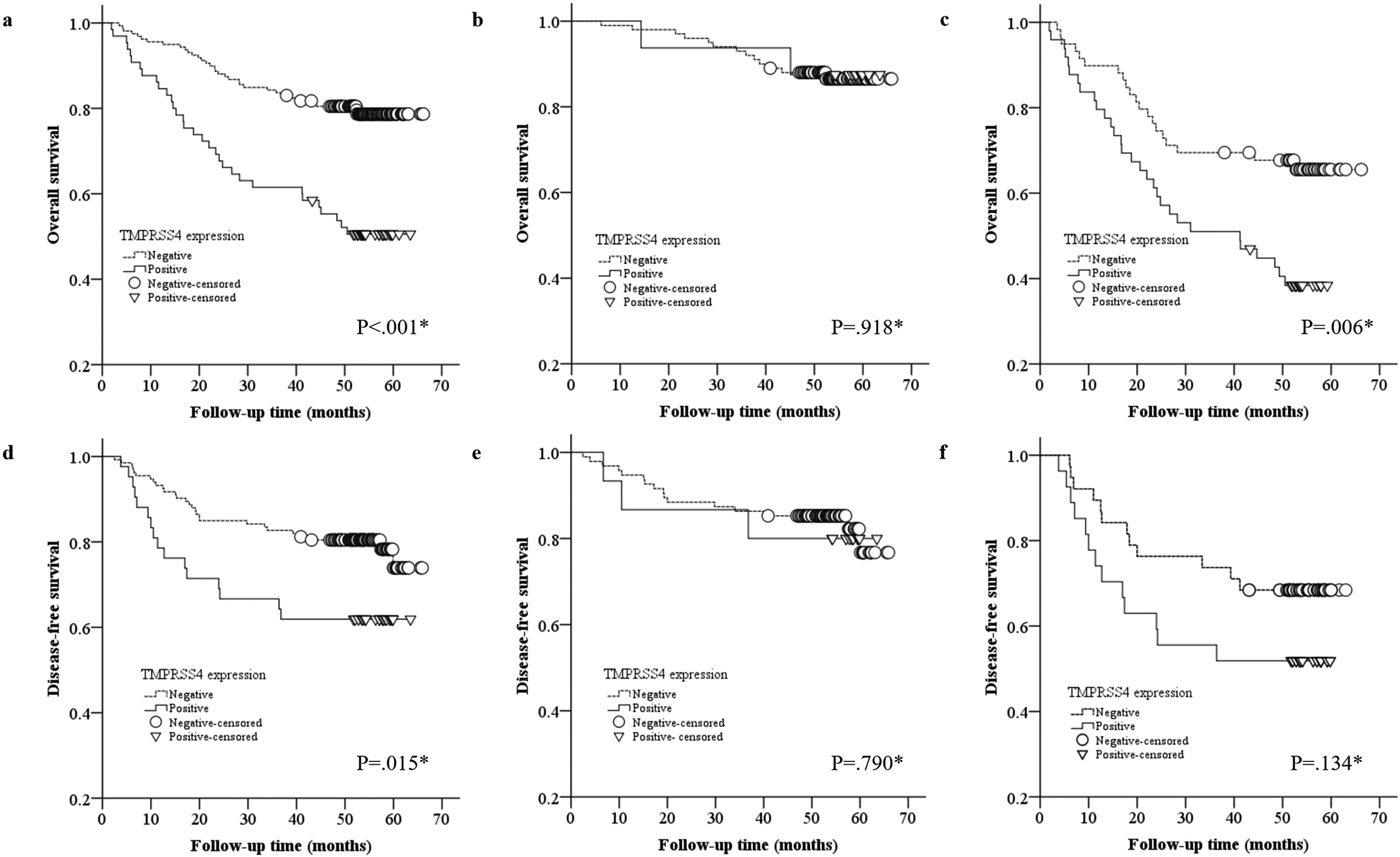
Survival curve. (a–c) Overall survival in 224 CRC cases. (a) Survival curve for all stage with CRC, P < 0.001; (b) Survival curve for stage I and II with CRC, P = 0.918; (c) Survival curve for stage III and IV with CRC, P = 0.006. (d–f) Disease-free survival in 175 CRC cases. (d) Survival curve for all stage with CRC, P = 0.015; (e) Survival curve for stage I and II with CRC, P = 0.790; (f) Survival curve for stage III with CRC, P = 0.134.
Univariate and multivariate analysis using the Cox proportional hazards model for OS revealed that tumor size, AJCC TNM stage, lymphovascular invasion, perineural invasion, histological grade, lymph node metastasis, and TMPRSS4 expression were significantly associated with prognosis (Table 2). For DFS, univariate analysis using the Cox proportional hazard showed that AJCC TNM stage, lymphovascular invasion, perineural invasion, lymph node metastasis, and TMPRSS4 overexpression were significantly associated with poor DFS. TMPRSS4 positive expression had a 2.114-fold higher risk for death than negative expression with a 95% confidence interval of 1.142–3.913 (P = 0.017) (Table 3). In a multivariate analysis, lymphovascular invasion and TMPRSS4 overexpression were independent poor prognostic factors for DFS in CRC (P < 0.001 and P = 0.016, respectively).
Cox proportional hazards analysis on patient overall survival in 224 CRCs.
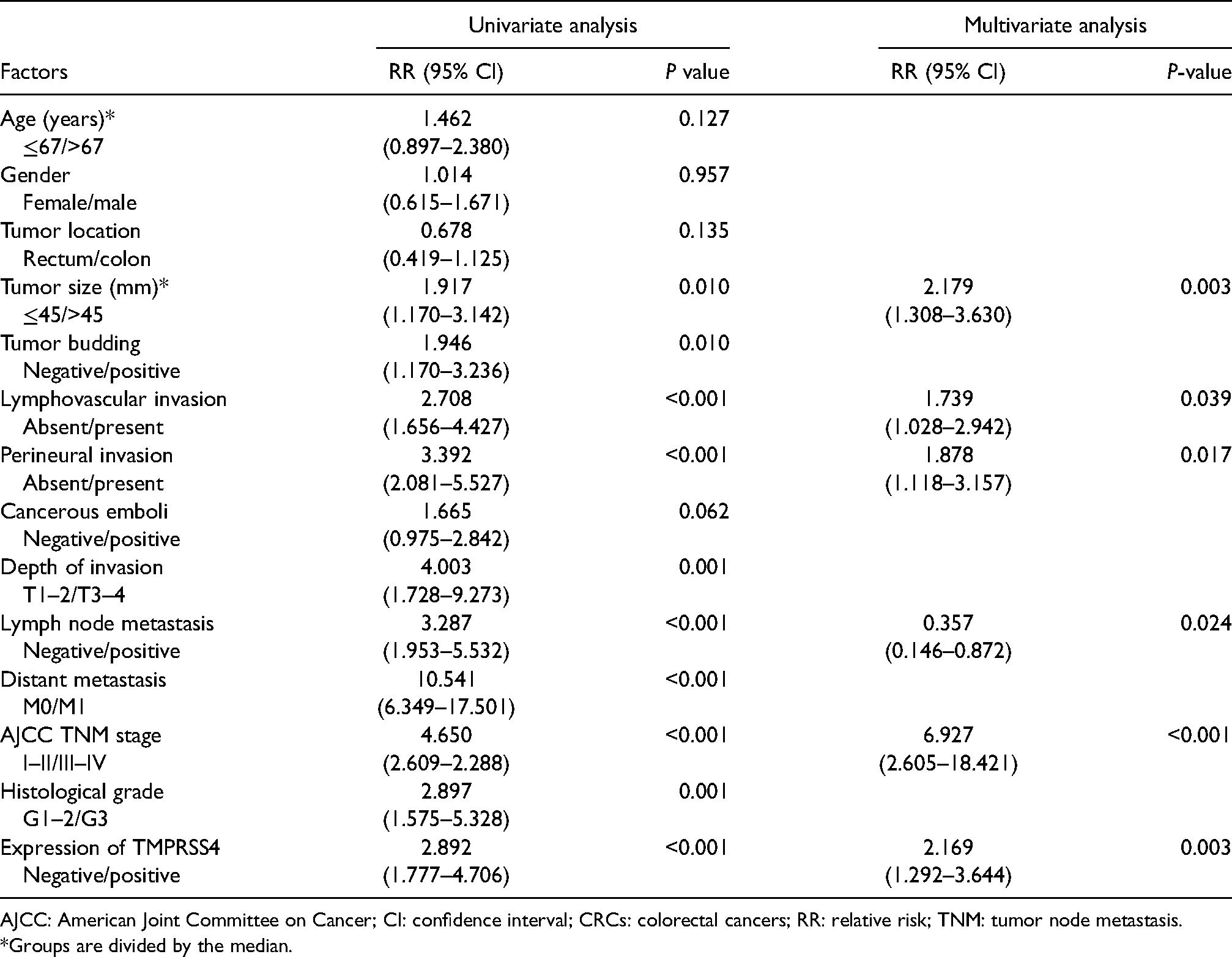
AJCC: American Joint Committee on Cancer; CI: confidence interval; CRCs: colorectal cancers; RR: relative risk; TNM: tumor node metastasis.
*Groups are divided by the median.
Cox proportional hazards analysis on patient disease-free survival in 175 CRCs.
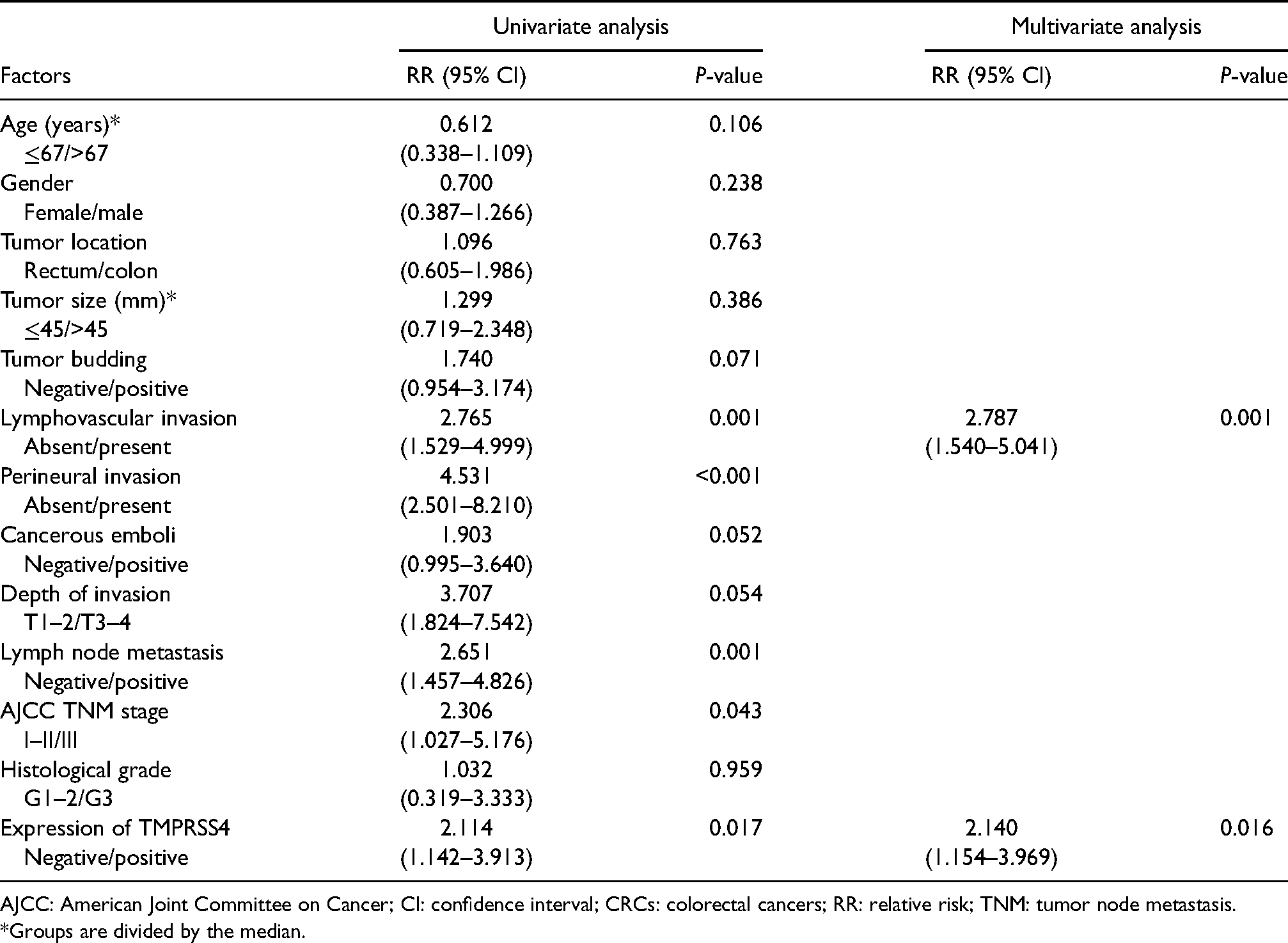
AJCC: American Joint Committee on Cancer; CI: confidence interval; CRCs: colorectal cancers; RR: relative risk; TNM: tumor node metastasis.
*Groups are divided by the median.
Discussion
Currently, the pathological stage of tumor at the initial diagnosis is the most important factor in determining the behavior, clinical course, prognosis, and treatment choice for CRC. 14 However, it has been reported that patients at the same pathological stage display different local recurrence and invasion. 18 Also, a larger number of tumors present poor behavior in spite of being regarded as low risk based on their TNM stage, 19 suggesting that a single TNM stage is not comprehensive and accurate enough in these patients. Therefore, novel biomarkers with higher specificity and efficiency on tumor behavior should be investigated for patients with CRC, such as TMPRSS4.
Previously, TMPRSS4 was found to be closely associated with many human malignancies, such as breast, 20 non-small cell lung, 21 and thyroid cancer, 22 etc. Of note, Jung et al. 9 demonstrated that TMPRSS4 was also up-regulated in CRC, and promoted invasion, migration, and metastasis of human tumor cells mainly by facilitating the EMT process. In previous studies, our group confirmed that TMPRSS4 induces invasion and EMT by upregulating integrin α5 and its signaling pathways. 10 Recently, Huang et al. 23 indicated from a quantitative perspective that there was a positive relationship between TMPRSS4 and CRC progression (TNM stage), which was also found in the present study. The significantly lower expression level of TMPRSS4 in stage I and II rather than in stage III and IV leads us to speculate that an obvious increase of TMPRSS4 expression level is a key process in the initiation of metastasis. Thus, in order to verify this speculation above, we attempted to depict the expression pattern of TMPRSS4 in the metastatic process of CRC.
In the process of tumor metastasis, cancer cells with metastatic potentiality must detach from the primary tumor, intravasate into the lymphovascular system, travel in the circulation, extravasate from the vessel, reattach to a target organ, and develop into a new focus. 24 Tumor budding—described as a morphological expression of EMT—is regarded as the first step of the metastatic process. 25 Then, penetration of tumor cells undergoing EMT through the endothelial cell layer into lymphovascular invasion is considered another key step. 26 Once tumor budding and lymphovascular invasion have occurred, tumor cells theoretically have the potential to metastasize to the regional lymph nodes and/or to more distant organs. Thus, the whole metastatic process of CRC may be covered four steps from the morphological point of view: (a) tumor budding; (b) lymphovascular invasion; (c) lymphovascular space invasion (tumor emboli); and (d) regional lymph node and/or distant metastasis. To date, many studies regarding TMPRSS4 have mainly concentrated on the revelation of the signaling pathway. However, the best of our knowledge, no studies depict in detail the expression pattern of TMPRSS4 in CRC.
In our study, the findings of tumor budding, lymphovascular invasion, tumor emboli, and lymph node and distant metastasis were strongly associated with TMPRSS4 expression. In addition, we further analyzed the correlation between immunohistochemical TMPRSS4 reactivity in the aforementioned morphological changes and the related primary tumors. There was no marked difference of TMPRSS4 expression among the tumor budding, emboli, metastatic lymph node, and liver tumor samples; however, all of them showed significantly higher expression than in the paired primary tumor cells. These findings indicated that an obvious increase of TMPRSS4 expression is necessary for the transition from primary tumor cells (with no or low metastatic potential) to metastatic cells (with high metastatic potential). In addition, the stable status of TMPRSS4 overexpression in the following metastatic process suggested that its pro-metastatic effect can be extended to last the whole metastatic course from tumor budding to lymph node and/or distant metastasis in CRC. Therefore, wound-healing migration assay and transwell assay were further conducted using HT29 cells with siRNA-TMPRSS4 to further demonstrate this hypothesis. The results suggested that TMPRSS4 knockdown significantly inhibited the invasive and migratory activity of HT29 cells. In conclusion, our study showed that TMPRSS4 plays a vital role in the metastasis of CRC, and its expression may provide some useful clues to evaluate the occurrence and progression of metastasis in CRC.
The next question investigated by us was the prognostic significance of TMPRSS4 in CRC. Previously, TMPRSS4 had been demonstrated to be associated with poor survival in many tumors, including pancreatic, 27 squamous lung, 28 liver cancer, 29 etc. Interestingly, TMPRSS4 overexpression also predicted shorter OS and DFS in CRC, 13 which was consistent with our results. Our study also demonstrated that TMPRSS4 and lymphovascular invasion were independent poor prognostic factors for OS and DFS in CRC. It is well known that metastasis is the uppermost factor for the poor prognosis in CRC. Therefore, the intimate connection between TMPRSS4 and metastasis, as revealed above, prompts us to speculate that the prognostic significance of TMPRSS4 may be noted only in patients with metastasis. Therefore, we further investigated the prognostic significance of TMPRSS4 in different stages of CRC, and interestingly, the expected results were seen. In the subgroup of TNM stages I and II, TMPRSS4 expression intensity was not significantly correlated with OS and DFS. In contrast, TMPRSS4 overexpression in advanced TNM stages III and IV of CRC tissues were significantly correlated with the OS; however, this was not correlated with the DFS. These findings indicated that TMPRSS4 is only of important significance in predicting the prognosis of stage III and IV CRC, not stage I and II, which supported our hypothesis. Since the finding that the expression levels of TMPRSS4 increased as the stage became advanced, from stage I CRCs being the lowest to stage IV CRCs the highest, had been reported by Huang et al.23 and this study, we speculated that TMPRSS4 is effective in predicting prognostic evaluation, only at a high enough expression level that can induce the initiation of metastasis in CRC.
In conclusion, our findings demonstrated within a subgroup of CRC cases that TMPRSS4 overexpression is associated with the whole process of metastasis from tumor budding to lymph node and/or distant metastasis in CRC. Moreover, TMPRSS4 overexpression was further found to only predict the unfavorable prognosis of stage III–IV CRC. Therefore, TMPRSS4 would be a useful prognostic biomarker for stage III–IV CRC and a novel therapeutic target to block and inhibit tumor metastasis.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author contributions
Xue-Feng Zhao and Yu-Shen Yang have contributed equally to this work.
author(s)
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Liaoning Provincial Natural Science Foundations of China (grant number 201602037).
Ethics approval
The study protocol was authorized by the institutional review board of CNUHH. Investigators must obtain informed consent before enrolling participants in clinical trials.
