Abstract
Background
Sex determining region Y-box 2 (SOX2) has been reported as a potential therapeutic target for cancer. However, the role of SOX2 in cervical cancer remains largely undetermined. This study was performed to evaluate the correlation of SOX2 with clinical characteristics and prognosis in cervical cancer.
Methods
Multiple databases were systematically searched for eligible publications. The combined odds ratios (ORs) or hazard ratios (HRs) with the corresponding 95% confidence intervals (CIs) were used to assess the effect sizes.
Results
A total of 17 studies with 1906 participants were identified. SOX2 expression was higher in cervical cancer than in the normal control group (OR = 10.83, 95% CI = 6.64–17.67, P < 0.001), while no significant difference was observed between cervical cancer and cervical intraepithelial neoplasia. SOX2 expression was not associated with age, tumor stage, and lymph node metastasis, but was correlated with tumor grade (grade 2–3 vs. grade 1: OR = 4.59, 95% CI = 2.76–7.62, P < 0.001) and tumor size (≥4 cm vs. ≤4 cm: OR = 1.66, 95% CI = 1.05–2.60, P = 0.028). Based on multivariate Cox analysis, SOX2 expression was not correlated with overall survival, but was closely associated with poor recurrence-free survival (HR = 5.83, 95% CI = 1.35–25.16, P = 0.018) and progress-free survival HR = 2.29, 95% CI = 1.01–5.19, P = 0.046).
Conclusion
SOX2 may serve as a novel prognostic factor and a promising molecular target for cervical cancer.
Introduction
Cervical cancer, one of the most common human cancers in women, has high incidence and mortality rates. According to the GLOBOCAN estimates, approximately 569,847 cervical cancer cases were diagnosed, and approximately 311,365 deaths were caused by this disease in 2018. 1 Despite some significant advancements in the treatment of cervical cancer, the prognosis of patients with this condition remains poor, especially in developing countries.2,3 Hence, it is crucial to identify a novel potential molecular target for treating and managing cervical cancer.
Studies have suggested the existence of cancer stem cells (CSCs) in cancer. CSCs, a small subpopulation of tumor cells, show the ability of self-renewal, uncontrolled proliferation, and differentiation.4,5 CSCs are shown to be correlated with tumor progression, prognosis, and resistance to therapy.5,6 Sex-determining region Y-box protein 2 (SOX2) is a High Mobility Group (HMG) domain transcription factor that is essential for embryonic development, stem cell fate, and pluripotent stem cell induction.7,8 SOX2 has a functional role in cellular reprogramming, and mammalian physiology, development, and pathology.9,10 As a CSC marker, SOX2 expression is correlated with a worse prognosis in advanced cancer. 7 Recently, some studies have reported the association between SOX2 expression and cervical cancer.11–13 However, the clinical and prognostic significance of SOX2 in cervical cancer remains largely unknown.
The results of SOX2 in clinical studies are still conflicting. For example, Hou et al. reported that SOX2 expression was correlated with poor overall survival (OS) in cervical cancer. 14 Nevertheless, Kim et al. showed that SOX2 expression was associated with favorable OS in cervical cancer. 15 Therefore, to determine a full insight into the role of SOX2 in cervical cancer, we first conducted a systematic meta-analysis to explore the clinical and prognostic role of SOX2 expression in this cancer.
Methods
Literature search
Our meta-analysis was performed based on the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) statement. 16 We performed a systematic search from the relevant databases (PubMed, Sciencedirect, Wang Fang, and Web of Science) before February 16, 2021. To identify eligible articles, the following key words and search terms were used: “SOX2 OR SOX-2 OR Sex determining region Y-box 2 OR SRY-Related HMG-Box Gene 2”, “cervical OR uterine cervix”, “cancer OR carcinoma OR neoplasm OR tumor OR malignancy”. The reference lists of the included articles were also scanned to identify additional studies.
Study selection
The following selection criteria in this meta-analysis were: (a) cervical cancer patients pathologically diagnosed; (b) cervical intraepithelial neoplasia (CIN) and normal tissue samples used as control groups; (c) studies that reported the expression of SOX2 using immunohistochemistry (IHC) detection from the original studies; (d) articles that reported available data for calculating odds ratio (OR) with the corresponding 95% confidence interval (CI); (e) articles that reported the prognosis of SOX2 using multivariate Cox analysis; and (f) if authors had published multiple publications using overlapping sample data from the same institution, only the study containing the most comprehensive data or the most recent study was included. Exclusion criteria were: (a) case reports, reviews, conference abstracts, letters, and comments; (b) cell or animal research; (c) studies lacking sufficient data or using univariate Cox analysis; (d) duplicate articles.
Data extraction and quality assessment
We extracted the following information from the eligible articles, including first author's surname; year of publication; country; ethnicity; sample size; mean or median age; frequency of SOX2 expression; detection method; clinical features such as tumor size, lymph node metastasis, age, tumor stage, and tumor grade; and the prognosis ((OS), recurrence-free survival (RFS), and progress-free survival (PFS)). If there were any disagreements, they were resolved by consensus from all authors. Two reviewers (DD Y and YH X) independently performed the quality evaluation for the included studies, and any discrepancies were resolved through discussion until consensus was achieved. Study quality was conducted using the Newcastle–Ottawa Quality Assessment Scale (NOS) tool. 17 The NOS maximum point was nine scores. A study with a high NOS score ranging from 6 to 9 was designated as high quality. A study with a low NOS score (scores 1–5) was deemed as low quality.
Statistical analysis
The combined ORs and 95% CIs were used to evaluate the difference of SOX2 expression between cervical cancer and CIN and normal tissue samples. We also applied the pooled ORs and 95% CIs to assess the association of SOX2 expression with clinical characteristics of cervical cancer. The pooled hazard ratios (HRs) with their 95% CIs were used to estimate the relationship between SOX2 expression and the prognosis using multivariate Cox analysis. The random-effects model was conducted in the present meta-analysis. Heterogeneity among the studies was detected based on Cochran's Q statistics test 18 Significant heterogeneity was found when a Q statistics test (P-value) was less than 0.1. When substantial heterogeneity was tested, we performed a sensitivity analysis by omitting one study or multiple studies to determine the stability of the re-calculated results. For the results with more than five studies, publication bias was detected by using Egger's test 19 This meta-analysis was carried out using Stata software, version 12.0 (Stata Corp., College Station, TX, USA).
Results
Study characteristics
The flow chart for the study selection is presented in Figure 1. Based on the inclusion criteria, 17 articles with 1906 participants (1172 cervical cancer patients and 734 control samples) were published between 2010 and 2020.8,11–15,20–30 These studies were conducted in China (n = 13), Korea (n = 1), Australia (n = 2), and The Netherlands (n = 1). Of these studies, seven studies evaluated the difference of SOX2 expression between cancer and CIN. Eight studies assessed the difference of SOX2 expression between cancer and normal tissues. A total of 13 studies with 941 patients with cervical cancer analyzed the correlation of SOX2 expression with the clinical characteristics. Three studies (n = 472) reported the prognosis of SOX2 expression using multivariate Cox analysis.14,15,24 The included studies showed that the NOS scores ranged from 4 to 8, with a mean value of 6.1. The basic characteristics of the eligible studies are summarized in Tables S1 and S2.
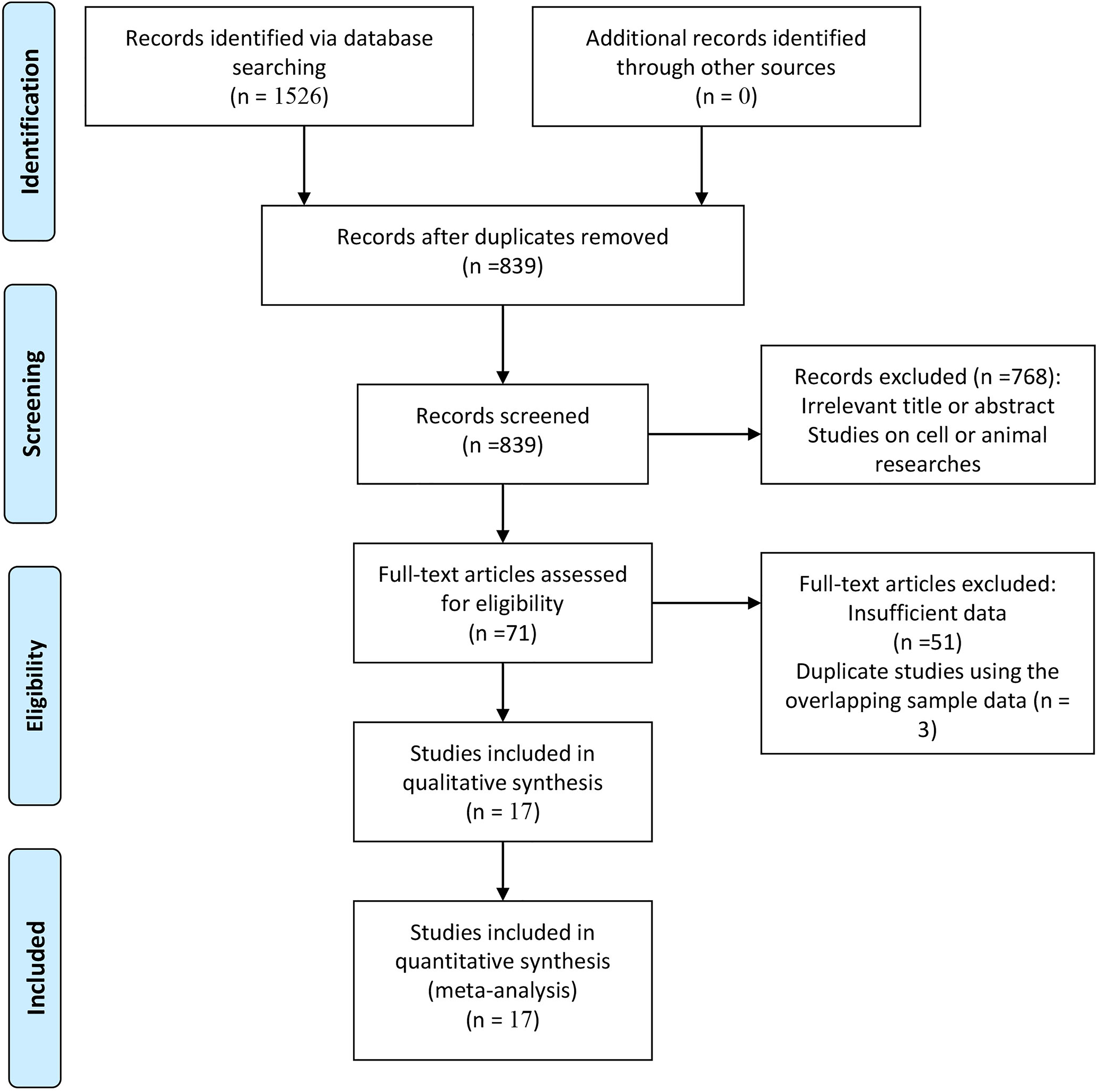
Flowchart chart of selected articles included in the current meta-analysis.
SOX2 expression in cervical cancer and the control group
Data showed no significant difference for SOX2 expression in patients with cervical cancer and CIN (382 cancer samples vs. 500 CIN samples: OR = 0.92, 95% CI = 0.35–2.38, P = 0.859) (Figure 2). SOX2 expression was significantly higher in cervical cancer than in the normal control group (OR = 10.83, 95% CI = 6.64–17.67, P < 0.001) (Figure 2), including 386 patients with cervical cancer and 234 normal tissue samples.
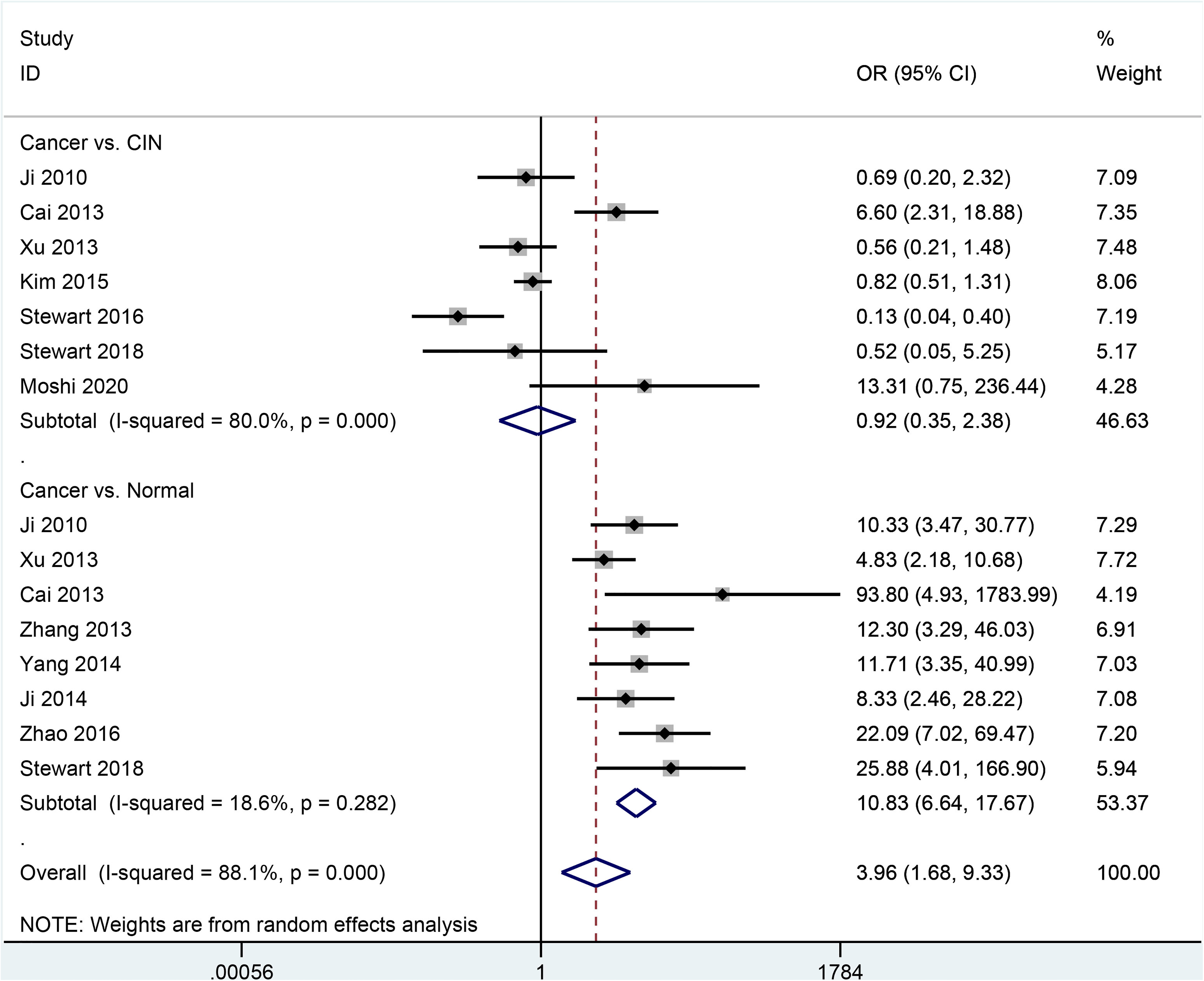
Forest plot for the difference of SOX2 expression in cervical cancer vs. cervical intraepithelial neoplasia (CIN) and normal tissue samples.
Correlation of SOX2 expression with the clinical and pathological features of cervical cancer
Data demonstrated that no association was observed between SOX2 expression and age (n = 3 studies with 227 cases; ≥50 years vs. ≤50 years: OR = 1.16, 95% CI = 0.66–2.02, P = 0.614) (Figure 3), tumor stage (n = 5 studies with 373 cases; stage 3-4 vs. stage 1-2: OR = 0.97, 95% CI = 0.49–1.95, P = 0.943) (Figure 3), and lymph node metastasis (n = 7 studies with 415 cases; positive vs. negative: OR = 0.76, 95% CI = 0.41–1.43, P = 0.402) (Figure 4).
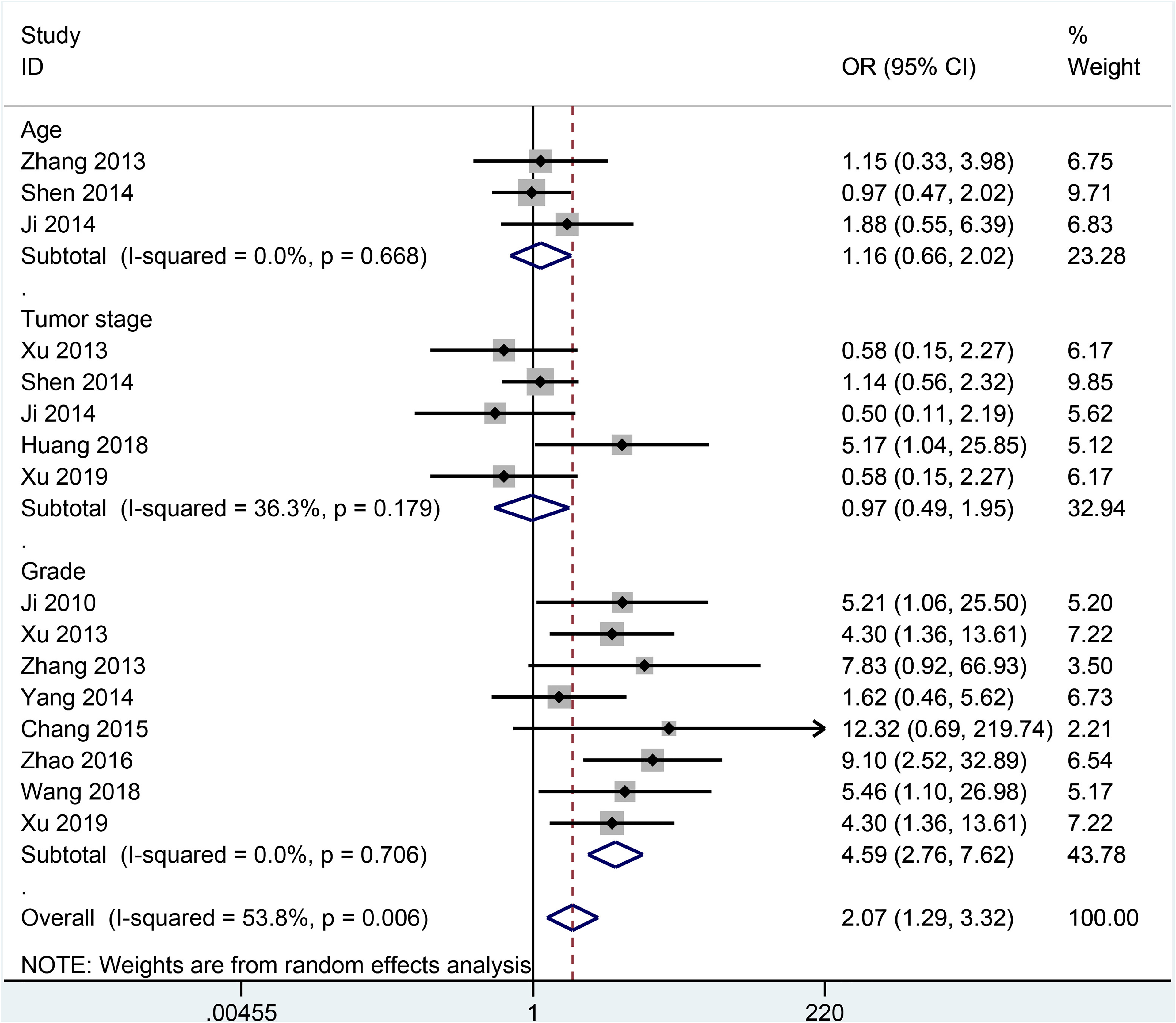
Forest plot for association between SOX2 expression and age (≥50 years vs. ≤50 years), tumor stage (3–4 vs. 1–2), and tumor grade (grade 2–3 vs. grade 1) in cervical cancer.
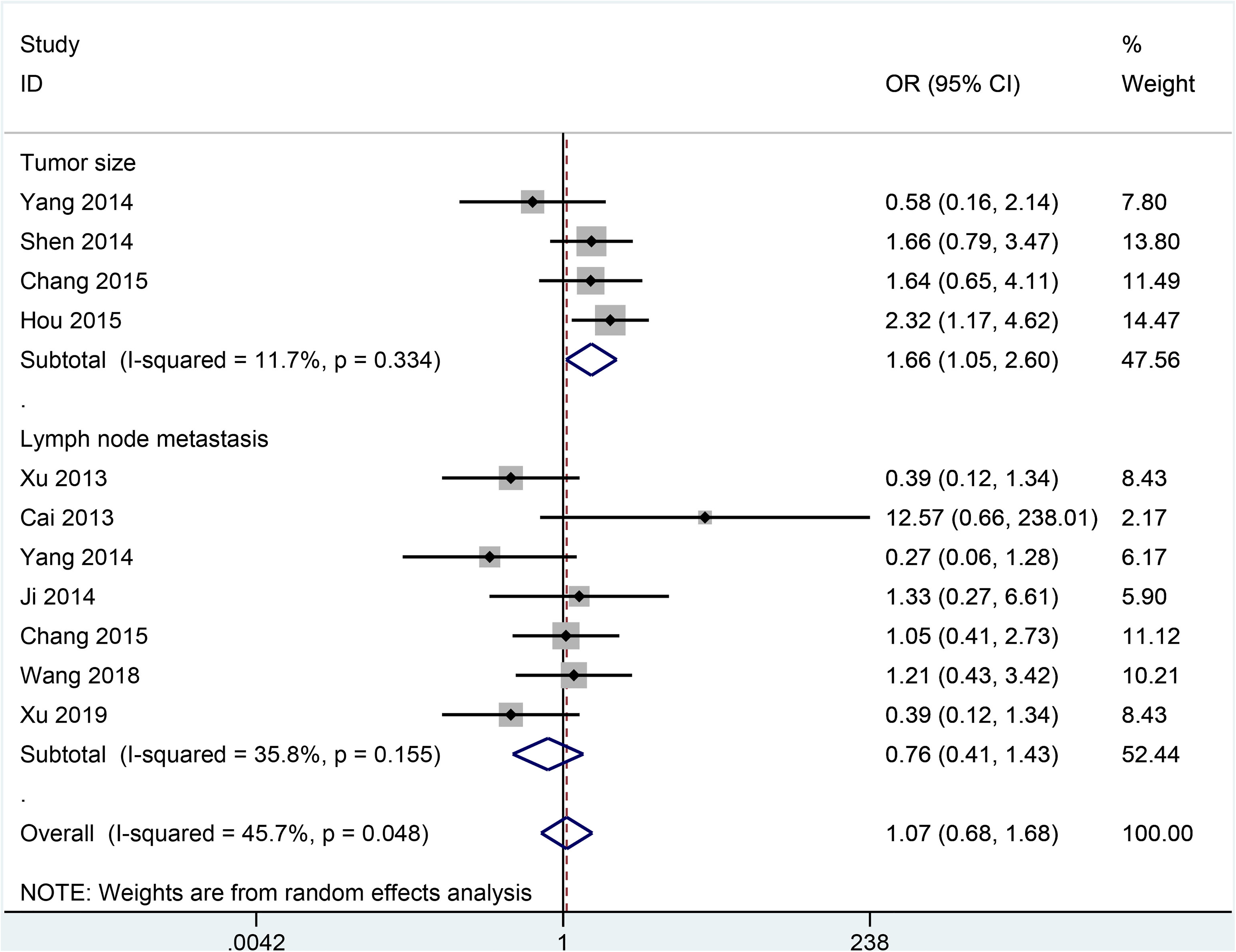
Forest plot for association between SOX2 expression and lymph node metastasis (positive vs. negative) and tumor size (≥4 cm vs. ≤4 cm) in cervical cancer.
SOX2 expression was closely correlated with tumor grade (n = 8 studies with 609 cases; grade 2–3 vs. grade 1: OR = 4.59, 95% CI = 2.76–7.62, P < 0.001) (Figure 3) and tumor size (n = 4 studies with 449 cases; ≥4 cm vs. ≤4 cm: OR = 1.66, 95% CI = 1.05–2.60, P = 0.028) (Figure 4). In addition, no significant heterogeneity was found between SOX2 and clinical features (all P values > 0.1).
Prognostic role of SOX2 in cervical cancer
Data from two studies with 340 cases showed no correlation between SOX2 expression and OS (HR = 1.26, 95% CI = 0.03–45.82, P = 0.900) (Figure 5). SOX2 expression was associated with poor RFS (n = one study with 179 cases, HR = 5.83, 95% CI = 1.35–25.16, P = 0.018) and PFS (n = one study with 132 cases, HR = 2.29, 95% CI = 1.01–5.19, P = 0.046) (Figure 5).
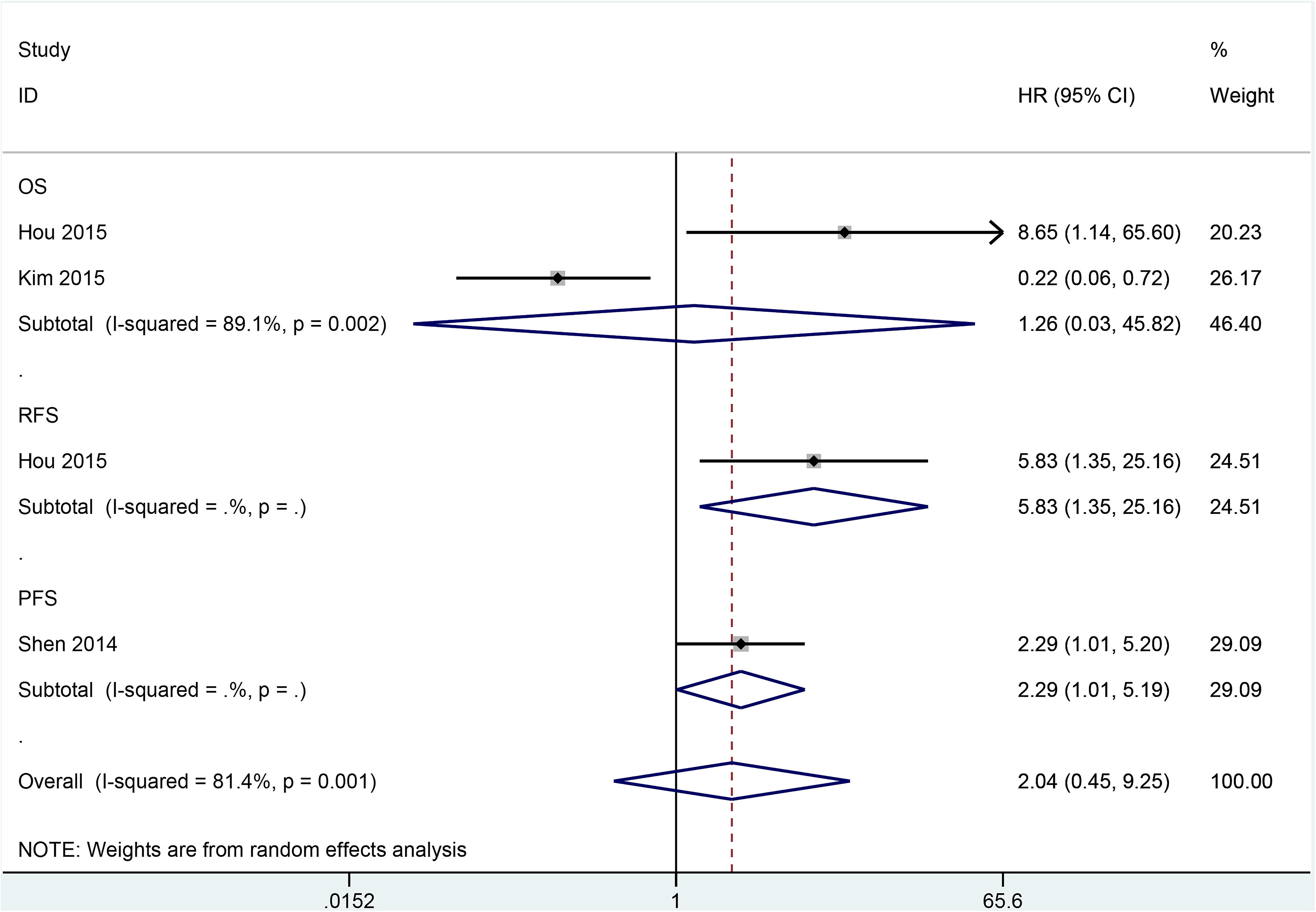
Forest plot for survival analysis of SOX2 expression in overall survival (OS), recurrence-free survival (RFS), and progress-free survival (PFS).
Heterogeneity analysis
Significant heterogeneity was observed in the comparison of cancer and CIN (P < 0.001). We conducted a sensitivity analysis to evaluate the change of the pooled results and heterogeneity. We removed the relevant studies (Cai et al. 26 and Stewart et al. 21 ); the re-calculated result was still not significant (OR = 0.78, 95% CI = 0.48–1.26, P = 0.314), with no evidence of heterogeneity (P = 0.337).
Publication bias
Publication bias was observed in the comparison of cancer and normal samples (P = 0.008), but no publication bias was found in the comparison of cancer and CIN (P = 0.798). Moreover, no publication bias was found between SOX2 and lymph node metastasis (P = 0.548) and tumor grade (P = 0.252) (Figure S1).
Discussion
Cervical cancer is the fourth most common cancer in women, with a high mortality rate. 1 The mechanism of the development of cervical cancer is complicated. 31 Increasing evidence suggests that CSCs are responsible for cervical cancer development, recurrence, and prognosis.32,33 CSCs markers have the potential to predict therapeutic therapy response. 33 SOX2 is a key embryonic stem cell gene that has a crucial role in regulating stem cell self-renewal and pluripotency.7,9 SOX2 also acts as an essential factor for cellular reprogramming and is associated with cell proliferation, cellular apoptosis, and oncogenic pathways and processes.9,34 SOX2 has been implicated in numerous cancers. For example, SOX2 expression is correlated with colorectal cancer progression, metastasis, and prognosis. 35 SOX2 expression has a significantly worse OS in head and neck cancer. 36 Some previous studies demonstrated that SOX2 was highly expressed in cervical carcinoma.11–13,20,21 However, the role of SOX2 in cervical carcinoma is still largely uncertain. The current study was performed to determine the clinical and prognostic value of SOX2 in cervical cancer.
When cervical cancer was compared to CIN and normal tissue samples, we found that SOX2 expression had a similar frequency in cervical cancer and CIN, which was in agreement with previous publications.8,11,15,20,27 Due to significant heterogeneity, we removed two studies: Stewart and Crook (2018) 20 and Yang et al. (2014). 25 The re-calculated results still showed no difference between cervical cancer and CIN, suggesting that our results were stable and credible. In comparison with normal tissues, we demonstrated that SOX2 expression was significantly higher in cervical cancer than in normal tissues, revealing that SOX2 expression may be correlated with cervical cancer tumorigenesis.
We further evaluated the association of SOX2 with clinical and pathologic characteristics via pooling available studies in colorectal cancer. No relationship was observed between SOX2 expression and age, tumor stage, and lymph node metastasis. These findings were consistent with the previous studies on age,23,24 tumor stage,8,12,23,24 and lymph node metastasis.8,12,22,23,25,26 We found that SOX2 expression was correlated with advanced tumor grade and tumor size. These findings were consistent with previous studies showing a significant association between SOX2 and tumor grade8,12,27 and tumor size. 14 These results revealed that SOX2 might be correlated with cervical cancer progression. We also investigated whether SOX2 might become a potential prognostic biomarker. Based on multivariate Cox survival analysis, we observed that SOX2 expression was not correlated with OS, but was associated with worse RFS (HR = 5.83, P = 0.018) 14 and PFS (HR = 2.29, P = 0.046), 24 which suggested that SOX2 may predict poor prognosis of cervical cancer in RFS and PFS. However, the results should be carefully considered with caution due to the small sample sizes. Further clinical studies are essential to confirm the predictive role of SOX2 in RFS and PFS in the future.
The present meta-analysis had some limitations. First, most studies consisted of Asians; other ethnic groups, such as Europeans and Africans, were insufficient. More high-quality studies from the American and African continents are needed to explore the clinical role of SOX2 in cervical cancer. Second, heterogeneity was observed in cervical cancer versus CIN. Although we performed a sensitivity analysis to analyze the potential heterogeneity, the detailed reasons were not very clear for heterogeneity. The different cut-off values of SOX2 expression and different IHC staining or antibody source were used from the original articles, which may cause the potential heterogeneity. Third, a slight publication bias was observed in the comparison of cancer and normal samples. Possible reasons were: the current meta-analysis was limited to papers published in English and Chinese. Studies with positive results could be more easily published than studies with negative results. Some studies with negative results may not be reported. This may result in publication bias. Fourth, the mean NOS scores of 6.1 were observed, suggesting that the quality of the included studies was not very high. This may be inevitable in the meta-analysis. Additional well-designed and high-quality studies with larger sample sizes are necessary to validate our results.
In conclusion, our results provided more evidence that SOX2 expression was higher in cervical cancer than in normal samples. SOX2 expression was associated with tumor grade, tumor size, and poor RFS and PFS. SOX2 could be a valuable prognosticator for predicting survival in cervical cancer. Targeting SOX2 has the potential to serve as a useful target. In the future, more high-quality clinical studies with larger sample sizes are required to confirm our findings.
Supplemental Material
sj-jpg-1-jbm-10.1177_17246008211042899 - Supplemental material for SOX2 as a prognostic marker and a potential molecular target in cervical cancer: A meta-analysis
Supplemental material, sj-jpg-1-jbm-10.1177_17246008211042899 for SOX2 as a prognostic marker and a potential molecular target in cervical cancer: A meta-analysis by Dandan Yuan, Jian Wang, Mingyu Yan and Yaohui Xu in The International Journal of Biological Markers
Supplemental Material
sj-docx-2-jbm-10.1177_17246008211042899 - Supplemental material for SOX2 as a prognostic marker and a potential molecular target in cervical cancer: A meta-analysis
Supplemental material, sj-docx-2-jbm-10.1177_17246008211042899 for SOX2 as a prognostic marker and a potential molecular target in cervical cancer: A meta-analysis by Dandan Yuan, Jian Wang, Mingyu Yan and Yaohui Xu in The International Journal of Biological Markers
Supplemental Material
sj-docx-3-jbm-10.1177_17246008211042899 - Supplemental material for SOX2 as a prognostic marker and a potential molecular target in cervical cancer: A meta-analysis
Supplemental material, sj-docx-3-jbm-10.1177_17246008211042899 for SOX2 as a prognostic marker and a potential molecular target in cervical cancer: A meta-analysis by Dandan Yuan, Jian Wang, Mingyu Yan and Yaohui Xu in The International Journal of Biological Markers
Footnotes
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors’ contributions
DD Y provided the study conception and design. YH X contributed to the drafting of the article and final approval of the submitted version. All authors provided the analyses and interpretation of the data and completion of figures and tables. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
