Abstract
Background:
BRCA1 and BRCA2 have been well studied for their roles in tumorigeneis, plus cancer diagnosis and treatment, but their prognostic value in colon cancer, especially for early-stage cancer, has not been fully illuminated. This study examined the expression levels of BRCA1 and BRCA2 proteins in sporadic colon cancer cases and investigated their value in prognosis.
Methods:
The expression levels of BRCA1 and BRCA2 in 275 colon cancer patients who underwent radical surgeries were assayed by immunohistochemical staining in dissected tumor samples. Also, its correlation with clinicopathological characteristics, disease-free survival, and overall survival was investigated.
Results:
Tumors with low expression levels of BRCA1, BRCA2, and both were 19.6%, 17.8%, and 6.5%, respectively. The levels of BRCA1/2 expression were not associated with clinicopathological parameters (gender, age, histological differentiation, and tumor node metastasis stage). Patients with low-levels of BRCA1 protein in their tumors demonstrated a lower chance of 5-year disease-free survival (55.6% vs. 69.7%, P=0.046), which was more obvious in the patients with stage I–II tumors without chemotherapy (52.6% vs. 82.6%, P=0.006). Neither BRCA1 nor BRCA2 affected overall survival in this cohort. Multivariate analysis revealed that pathologic stage and the level of BRCA1 protein were independent factors of long-term disease-free survival.
Conclusion:
This study highlights BRCA1 as an independent prognosticator of early-stage colon cancer.
Keywords
Introduction
Colon cancer (CC) is the third most common cancer type worldwide and the second most lethal cancer in western countries. 1 Although various molecular markers have been developed for a more accurate prediction of clinical outcome, biomarkers are needed to differentiate patients with high risks of recurrences after surgery from patients with low risks, in order to provide more personalized health management for early-stage cancer (stage I–II).
BRCA1 and BRCA2 are two important tumor suppressor genes that are functionally relevant in maintaining genomic stability, and their germline mutations contribute to 40%–60% of inherited breast cancer and ovarian cancer.2-5 In addition, mutations of these two genes also exist in other sporadic cancer, including CC.6, 7 Loss of heterozygosity (LOH) is the most common form of BRCA1/2 mutations in tumor, leading to loss of function of the two genes. 8 Most studies focused on mutations of BRCA1/2 rather than their expression levels in CC and suggest that LOH of BRCA1 exists in 40% of sporadic CC.6, 7 However, LOH could not accurately reflect the level of protein expression in tumor tissues, due to other factors regulating gene expression, in addition to LOH. Grabsch et al. 9 reported a low level of BRCA1 protein was associated with worse overall survival (OS) in advanced colorectal cancer. Here, we aimed to examine the levels of BRCA1/2 protein expression in tumor tissues and study their clinical significance in early-stage CC.
Materials and methods
Patients
A total of 275 patients with primary colon adenocarcinoma who underwent radical surgery with or without adjuvant chemotherapy between January 2007 and February 2012 were enrolled based on the availability of resected tissue. Patients with familial adenomatous polyposis and clinical criteria for hereditary non-polyposis colorectal cancer were excluded from this study. All including patients signed the informed consent before treatment, and the study was approved by the Peking University Cancer Hospital Ethics Committee. Adjuvant chemotherapy with FOLFOX or XELOX regimen was proposed to patients with stage III CC in the absence of any clinical contraindications, and to those with stage II CC who had high risks of recurrence, such as perforated cancer, pT4N0 with vascular emboli, and/or intestinal obstruction. 10 A total of 43 out of 123 patients with stage II tumor received adjuvant chemotherapy. Patients were followed up every 3 months during the first 3 years post-surgery, then every 6 months for an additional 2 years, and annually after 5 years following surgery. Tumor progression was assessed by physical examination with measurement of serum carcinoembryonic antigen (CEA) levels, chest radiograph, and computed tomography. Patients who became lost to follow-up were excluded.
Immunohistochemistry and tissue microarray
The expression of BRCA1 and BRCA2 were assessed by immunohistochemical staining of tumor and adjacent colorectal tissue. Immunohistochemistry (IHC) was performed on formalin-fixed, paraffin-embedded tissues. The tissue microarrays with a 3-mm punch taken from each paraffin-embedded block were used as described before. 11 The slides containing tumor and adjacent colon tissues were incubated with antibodies specific for BRCA1 (CAS# ab16780) or BRCA2 (CAS# ab53887). The pathological annotation process included an evaluation of the immunoreactivity intensity (negative, 0; weak, 1; moderate, 2; strong, 3) and the fraction (%) of positive cells (<5%, 0; 5%–25%, 1; 25%–50%, 2; 50%–75%, 3; >75%, 4). The final score was calculated as intensity multiplied by fraction. All samples were categorized as “high-level expression” or “low-level expression” based on a cut-off score of 6. The staining of the normal colonic epithelium and lymphocytes served as the negative controls. All slides were examined by two experienced pathologists unaware of the clinical information and therapeutic outcome. For the discrepant results, both pathologists re-evaluated the slides together to reach a final consensus. Mismatch repair (MMR) deficiency was assessed by evaluating MLH1, MSH2, MSH6, and PMS2 using IHC, as previously reported. 12
Statistical analysis
Data were collected and analyzed using SPSS 19.0 package (SPSS Inc., Chicago, IL, USA). The relationship between BRCA1/2 protein expression and clinicopathological features of the patients was evaluated with a chi-square test. DFS and OS were evaluated using the Kaplan–Meier method, and differences between curves were tested with a log-rank test. The multivariate Cox proportional hazards regression model (Enter method) was used to identify independent risk factors affecting survival time. All tests were 2-sided and a P< 0.05 was considered statistically significant.
Results
The expression of BRCA1/2 in CC tumor
Clinicopathological characteristics of the included patients are listed in Table 1. The positive staining of BRCA1 and BRCA2 were observed in the nucleus of cells, as shown in Figure 1. The high-level expression rate of BRCA1, BRCA2, and both were 80.4% (221/275), 82.2% (226/275), and 69.1% (190/275), respectively. There was no correlation between expression levels of BRCA1/2 and the clinicopathological parameters such as gender, age, tumor location, histological differentiation, pathologic stage, vascular invasion, and serum CEA; while all MMR-deficient cases (n=21) were high-level BRCA1 expressed in this cohort (Table 2).
Clinicopathological characteristics of the patients in this study.
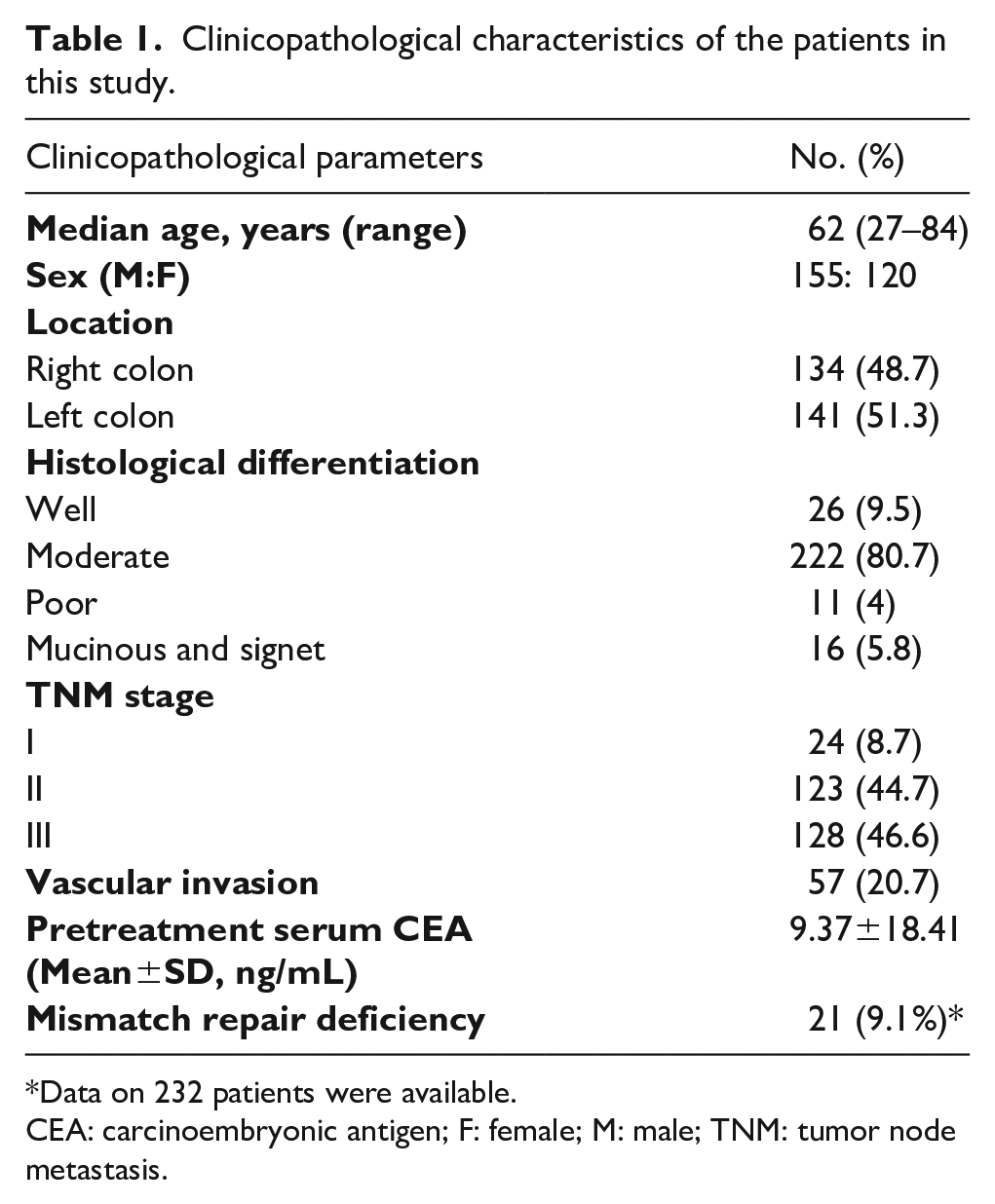
Data on 232 patients were available.
CEA: carcinoembryonic antigen; F: female; M: male; TNM: tumor node metastasis.
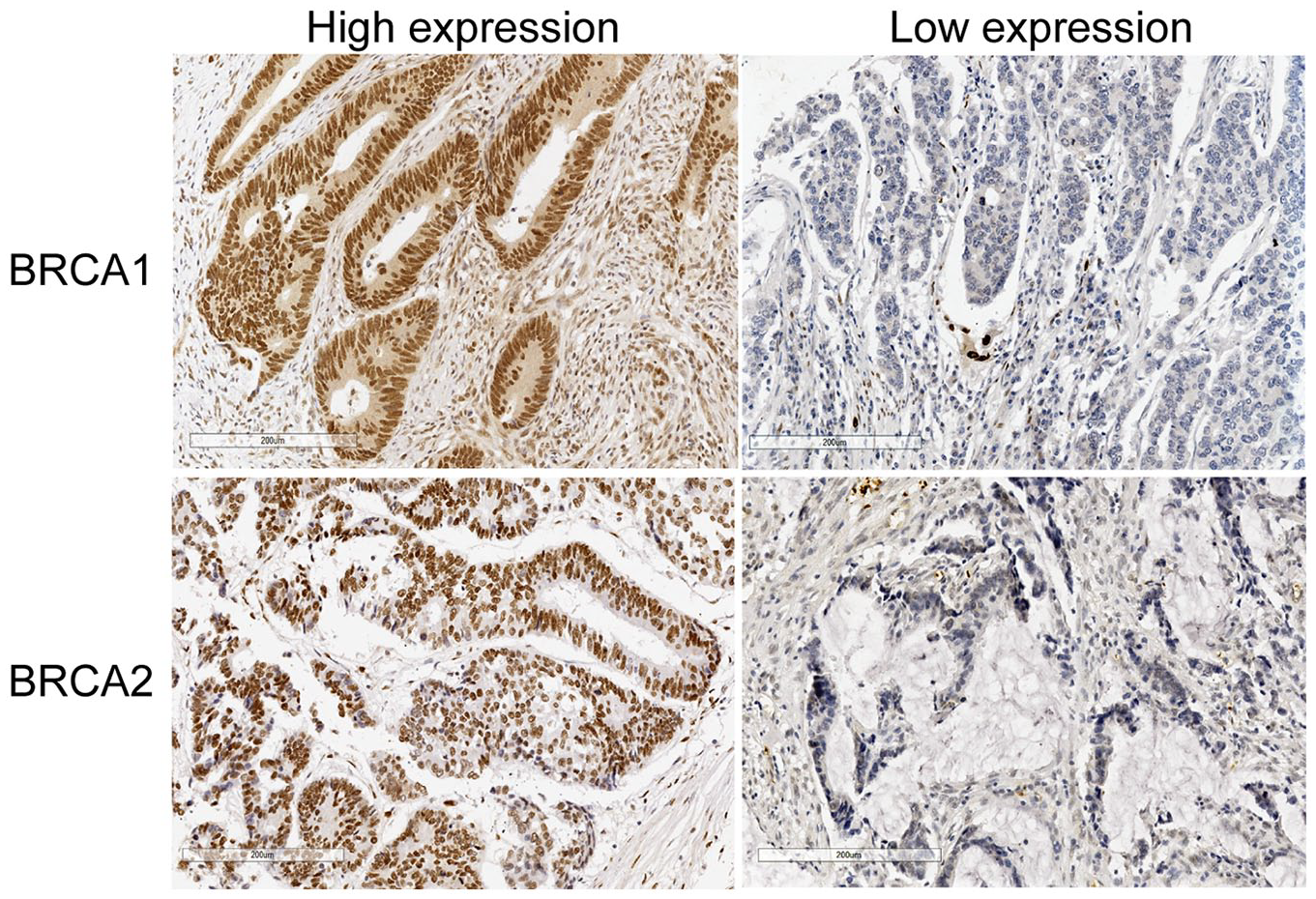
The expression of BRCA1 and BRCA2 in CC tissue. Assay: immunohistochemistry staining (IHC).
The association between the expression of BRCA1/2 and clinical parameters.
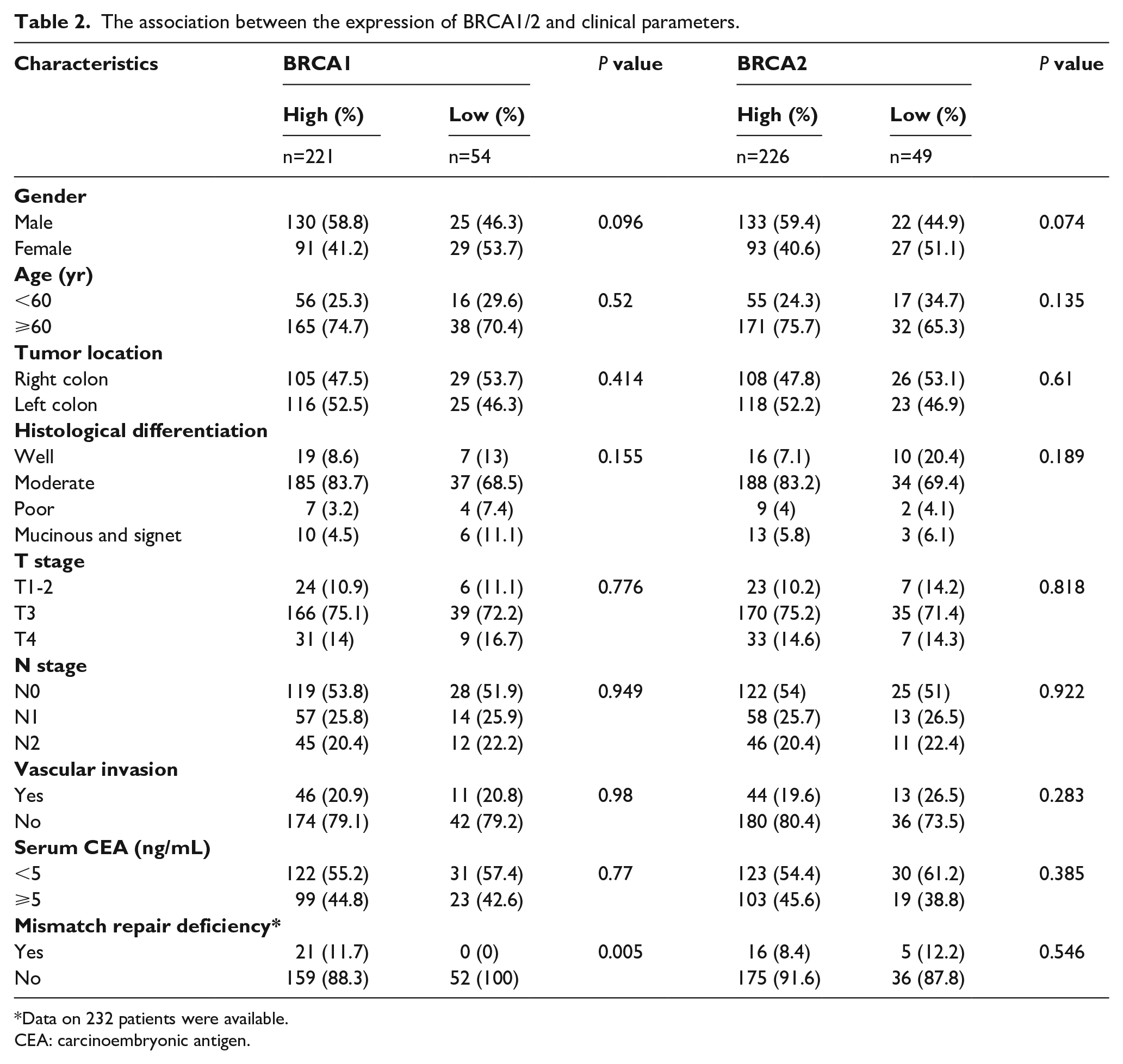
Data on 232 patients were available.
CEA: carcinoembryonic antigen.
Prognosis analysis for BRCA1/2 expression
With respect to clinical outcome, the 5-year DFS rate and 5-year OS rate for all patients were 66.9% and 73.8%, respectively, with 91 patients developing recurrence; and 72 deaths. The 5-year DFS rate in the BRCA1 low-level expression group was significantly lower than that in the BRCA1 high-level expression group (55.6% vs. 69.7%, P=0.045; Figure 2(a)), whereas no difference was observed for BRCA2 (67.3% in low-level expression group vs. 66.8% in high-level expression group, P=0.96; Figure 2(b)). For patients with stage I–II cancer who did not undergo adjuvant chemotherapy, patients with BRCA1 low-level expression presented a worse DFS compared to those with BRCA1 high-level expression (52.6% vs. 82.6%, P=0.006, Figure 2(c)). For the patients with stage I–II tumor who received chemotherapy, no statistical difference was observed in DFS between the low-level and high-level BRCA1expression groups (57.1% vs. 61.5%, P=0.566, Figure 2(d)). Neither the BRCA1 nor the BRCA2 expression levels affected OS (Figure 2(e) and (f)).
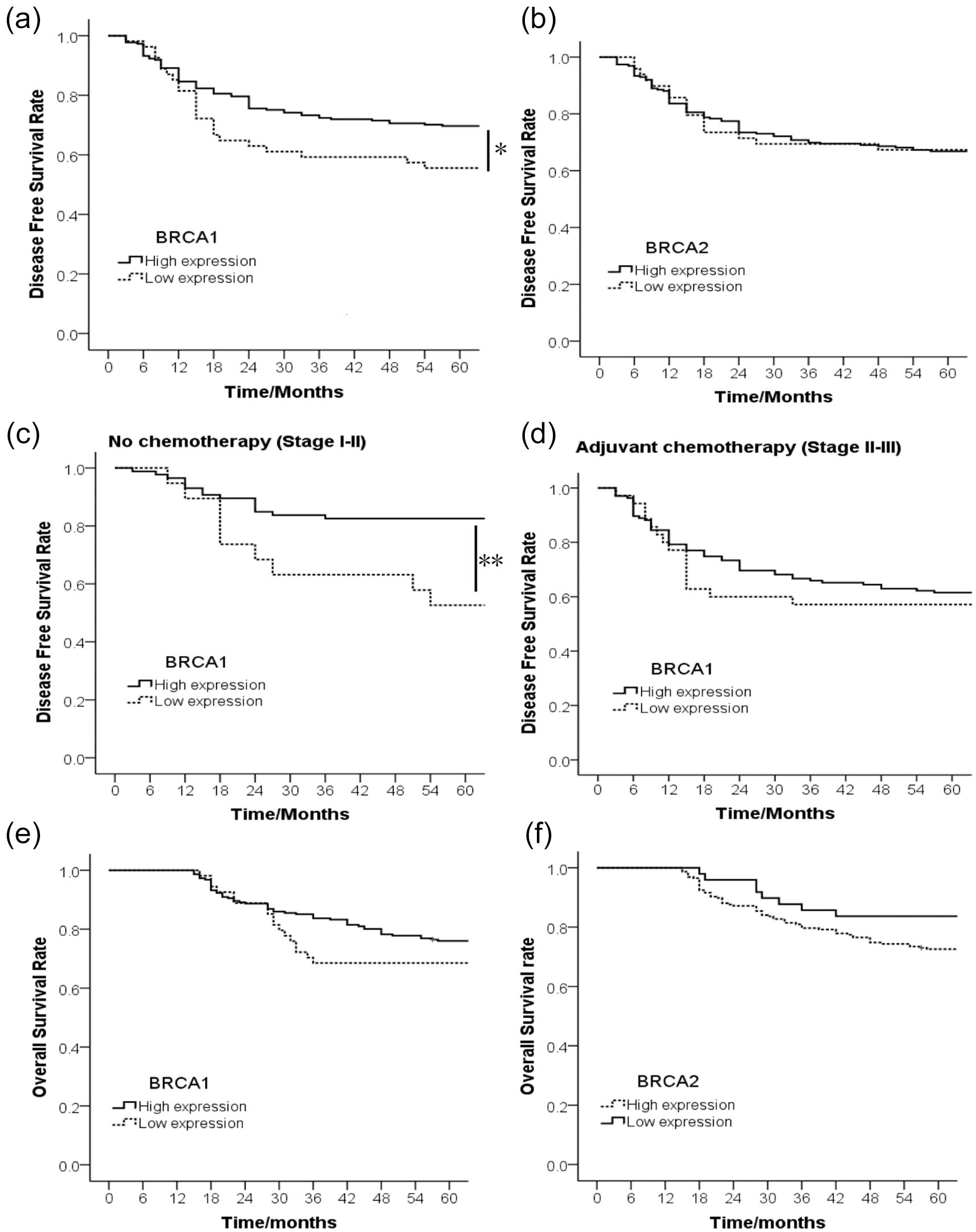
(a) and (b) The 5-year disease-free survival of the patients with different expression status of BRCA1 (a) and BRCA2 (b). (c) and (d) The association between 5-year disease-free survival and BRCA1 expression status in the patients without chemotherapy (c) or with chemotherapy (d). (e) and (f) The 5-year overall survival of the patients with different expression status of BRCA1 (e) and BRCA2 (f). *P<0.05, **P<0.01.
Cox proportional hazards regression model of disease-free survival
Clinicopathological parameters including gender, age, tumor location, histological differentiation, pathological T stage, N stage, vascular invasion, serum CEA, and MMR status as well as expression levels of BRCA1 and BRCA2 were analyzed using the Cox proportional hazards regression model. Multivariate analysis showed that the pathological T stage, N stage, and expression levels of BRCA1 were left in the model and were therefore the independent prognostic factors (Table 3).
Multivariate analysis of disease-free survival by Cox proportional hazards regression (Enter method).
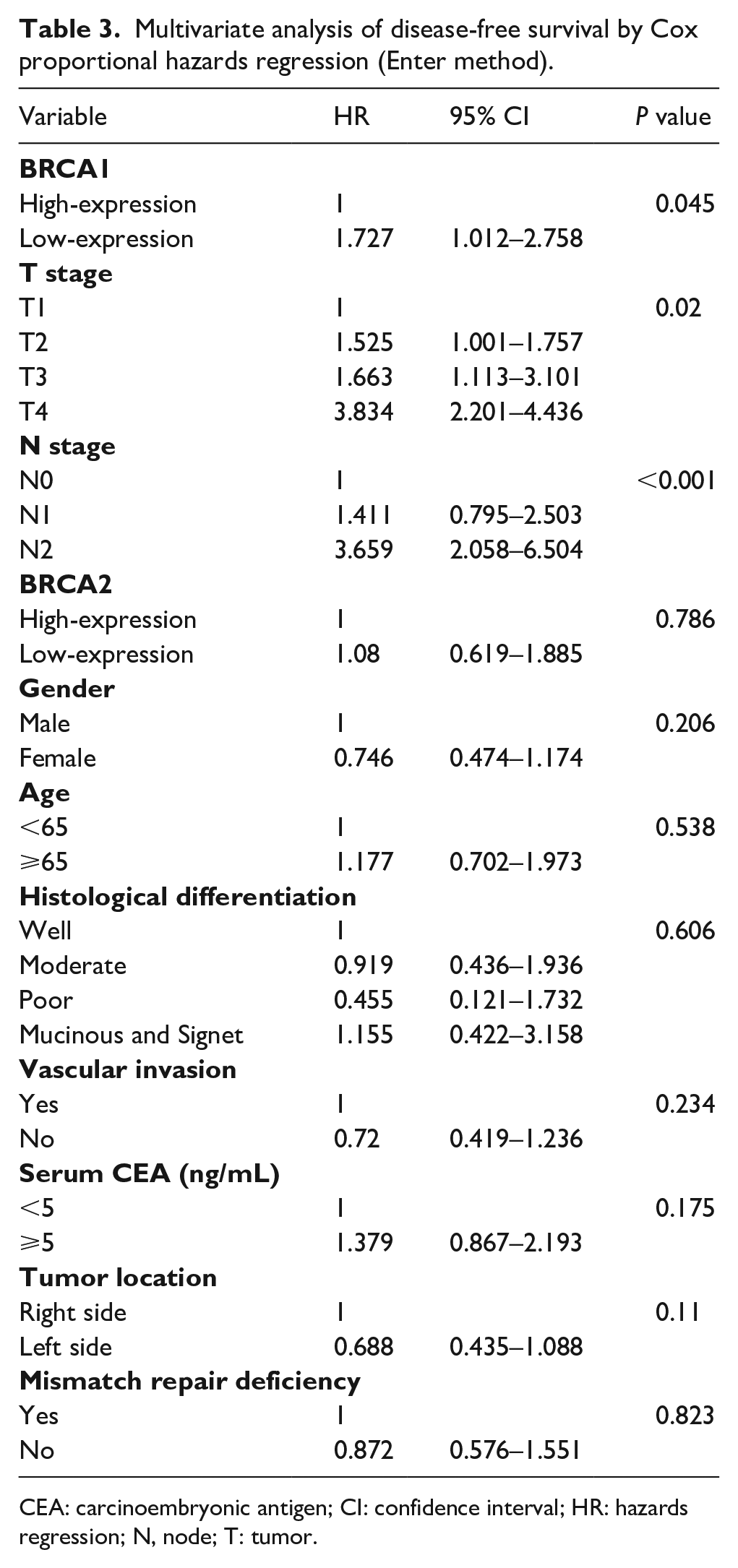
CEA: carcinoembryonic antigen; CI: confidence interval; HR: hazards regression; N, node; T: tumor.
Discussion
As the cancer therapy has stepped into a precision medicine era, the significance of reliable biomarkers cannot be emphasized enough. Biomarkers not only provide abundant prognostic information, but also allow physicians to adopt personalized therapies for their patients. 13 An effective prognostic marker is especially critical for early stage (I-II) CC, since the majority of these patients are not given adjuvant chemotherapy after surgery under the current consensus. 14 Individualized treatment based on prognostic markers is critical to minimize the risk of tumor progression for those patients with high risks.
BRCA1 and BRCA2 are two well-established tumor suppresser genes in maintaining genomic stability, exerting their complicated functions in DNA damage repair, transcription regulation, and signal transduction.15-17 Loss of function in BRCA1/2 leads to genomic instability and attenuated homozygous recombination in DNA replication, promoting tumorigenesis in breast and ovary.8, 18 Furthermore, the drug toxicity and response to DNA repair inhibitors are different in the BRCA1/2-mutated patients. 19
Whether germline mutation of BRCA1/2 increases the risk of colorectal cancer is still controversial5, 20; BRCA1/2 mutant colorectal tumors are known to be different from BRCA1/2 wild-type tumors by demonstrating a higher percentage of mucinous adenocarcinoma and left-sided tumors.6, 21 LOH of BRCA1 was shown to have a poor DFS after surgery. 7 Compared to gene mutation or LOH tests, IHC is more accessible in hospitals.9, 22 In 2006, Grabsch et al. 9 reported that a low level of BRCA1 expression by IHC correlated with a worse OS in patients with colorectal cancer. In their study, they included 342 patients with either CC or rectal cancer—38 of whom received chemotherapy or radiotherapy. In contrast to their conclusion, our results demonstrated that (a) expression levels of BRCA1/2 had no association with OS; and (b) low levels of the BRCA1 protein predicted a worse DFS in stage I-Ⅱ tumor without receiving chemotherapy, but not in the advanced disease group undergoing chemotherapy. One possible reason contributing to the difference may come from the rapid development of therapy in the past decade, especially the advance in surgical techniques and chemotherapeutic drugs, leading to a dramatic improvement of clinical outcome.23, 24 The understanding and clinical indication of adjuvant therapy also changed significantly. In Grabsch et al.’s report, 9 only less than one-third of the patients with lymph node metastasis were given adjuvant therapy, which affected the prognostic outcome. Another possibility is the difference in patient enrollment criteria as the majority of previous studies included both CC and rectal cancer cases. Only in recent years has it been understood that the two cancer types are different in terms of gene expression profile, biological behavior, clinical treatment, and prognosis.25, 26 A heterozygous mixture of rectal cancer cases was excluded from the current study; therefore, results from this study provide a potential prognostic marker for early stage CC under the current therapeutic modality.
A limitation of this study is that it did not involve more biomarkers, such as K-RAS, N-RAS, and BRAF. The mutations of the above genes not only predicted worse prognosis in CC, but also were relevant to the therapeutic outcome. 27 Thus, further prospective studies are needed to compare the prognostic importance of BRCA1 with other biomarkers.
Footnotes
Acknowledgements
We appreciate the help from Mrs. Fangping Zhao for her professional suggestions and language revisions.
Author contributions
Changzheng Du and Yifan Peng designed the study, undertook the research and wrote the paper. Yiping He provided professional consultation of the research and helpful suggestions on writing. Guoan Chen and Hao Chen provided technical support. Changzheng Du and Yifan Peng contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by university grant no.Y01416218 from Southern University of Science and Technology.
