Abstract
Background:
Colorectal cancer is one of the most common cancers worldwide in terms of both incidence and mortality. The associations of expressions of tissue and plasma miR-29b were not detected in this study.
Methods:
There are 400 healthy age- and gender-matched controls enrolled in this study in a rate of 1:2. The receiver operating characteristic curve analysis was undertaken using the expression level for miR-29b in the colorectal cancer specimens from patients with cancer and healthy controls to assess the diagnostic accuracy of both tissue and plasma miR-29b levels.
Results:
It was found that the expression of plasma miR-29b is associated with the tissue miR-29b. Advanced study showed that aberrant miR-29b expression in both cancer tissues and plasma is associated with the clinicopathological data of patients with colorectal cancer. Tissue miR-29b showed an AUC of 0.883, with a sensitivity of 81.6% and a specificity of 84.9%. However, the AUC for plasma miR-29b was 0.743, with a sensitivity of 61.4% and a specificity of 72.5%. The analyses of the biological effects of miR-29b for colorectal cancer showed that miR-29b could inhibit the cell viability and migration.
Conclusion:
In summary, our data suggest that both the tissue and the plasma miR-29b levels have some value as a diagnostic tool for colorectal cancer. Advanced biological effects were conducted to detect the potential effect on the cell viability and migration. Future investigations including larger patient populations and patients with early-stage colorectal cancer are needed to confirm the potential diagnostic value of miRNA-29b in colorectal cancer.
Introduction
Colorectal cancer (CRC) is one of the most common cancers worldwide in terms of both incidence and mortality. 1 In the United States, nearly 6% of individuals will develop this malignancy during their lifetime and one-half of those will die from it. 2 The risk of developing CRC is influenced by both environmental and genetic factors. Worldwide, the CRC incidence rose in parallel with economic development, with the majority of cases occurring in industrialized countries since it has been strongly associated with a western lifestyle. 3 Recently, progress has been made through epidemiological studies investigating environmental risk factors for CRC. A large number of factors have been reported to be associated with an increased risk of CRC development such as obesity, high red meat consumption, cigarette smoking, and alcohol abuse. 4 Although primary prevention strategies focusing on modifying environmental and lifestyle risk factors are options, the best strategy for reducing CRC mortality is based on early diagnosis. 5,6
Noncoding RNAs (ncRNAs) are divided into 2 main categories based on transcript size: small ncRNAs (microRNAs [miRNAs]) and long noncoding RNAs. Micro RNAs are small (∼22 nucleotides) endogenous ncRNAs that regulate gene expression at the posttranscriptional level through RNA interference. 4,7,8 Micro RNAs are actively involved in many biological processes and have an impact on many diseases including cancer. Differential expression of miRNAs has been demonstrated in many types of cancer. 9 These miRNAs carry both diagnostic and prognostic information and have been shown to alter cancer cell phenotype by targeting biological pathways critical to tumorigenesis. MiR-29b, a member of the miR-29 family, was found to be highly associated with the development and progression of CRC. 10,11 For example, recent studies have suggested that members of miR-29 family may play an important role in cancer by regulating cell proliferation, differentiation, apoptosis, migration, and invasion. Downregulation of miR-29 family members has been observed in leukemia, melanoma, liver, cervical, and lung cancer. 12,13 Aberrant expression of miRNAs in CRC has been reported recently, suggesting a potential for their use as biomarkers for disease diagnosis. Imaging techniques are widely used for the diagnosis of CRC, and images of the tumor tissue may confirm the diagnosis of CRC via ophthalmoscopy, ultrasonography, computed tomography, and magnetic resonance imaging. 14 An increasing number of studies demonstrate that miRNA expression profiles may be specific to certain types of cancer, and tumor-derived miRNAs may be stably detected in the plasma or serum. These findings highlight the potential of circulating miRNAs as biomarkers for the diagnosis of cancer. A low level of miR-29b contributes to the vascular endothelial growth factor proteins in CRC cells, which are considered as one of the key genes of CRC.
However, the associations of expressions of tissue and plasma miR-29b were not clear by now. Taking into consideration the results of that microarray assay and the fact that miR-29b has been investigated in several types of cancer as a biomarker, we hypothesized that miR-29b may serve as noninvasive circulating biomarkers for the diagnosis of CRC. However, the associations of tissue and plasma miR-29b levels were also tested in this study. Besides, the effect of miR-29b on the proliferation and migration of CRC cell line was also detected in this study.
Materials and Methods
Patients and Samples
Our study was approved by the Ethics Review Board of Shandong Provincial Hospital Affiliated to Shandong University, China. Informed consent was obtained from all patients. All the samples were collected once informed consent was obtained from the patients or the legal guardian. There are 400 healthy age- and gender-matched controls enrolled in this study in a rate of 1:2. We studied CRC specimens (tumor and adjacent nontumor colon tissues) from 200 patients who had undergone resection at the Shandong Provincial Hospital between March 2013 and June 2014. We gathered all samples in the same manner, and they were snap frozen immediately in liquid nitrogen and stored at −80°C until RNA extraction could be performed. Each participant donated 5-mL blood sample for plasma miRNA analysis. For patients with CRC, blood samples were taken before and after definitive surgical intervention and/or adjuvant therapy. For healthy controls, blood samples were taken at the time when they were included in this study.
RNA Extraction
Total RNA in tissue was extracted from cancer and control tissues using the mirVana miRNA isolation kit (Applied Biosystems, Foster City, California). Total RNA in plasma was extracted from 400 μL of plasma with a mirVana PARIS kit (Ambion, Austin, Texas) and eluted into 100 μL of preheated (95°C) elution solution according to the manufacturer’s instructions. The qualification and quantification of RNA were verified by measuring ultraviolet absorbance (A260 and A280) on the NanoDrop Spectrophotometer 2000 (Thermo Scientific, Wilmington, Delaware). The A260–A280 ratio should be close to 2.0 (ratios between 1.8 and 2.1 are acceptable).
Cell Culture and Transfection
Colorectal cancer cell line HT-29 and the normal colon cell line SW480 were maintained as adherent cultures in Dulbecco Modified Eagle Medium (GIBCO) supplemented with 10% fetal bovine serum (FBS), 100 U/mL penicillin, and 100 mg/mL streptomycin and were incubated at 37°C in a humidified chamber supplemented with 5%
Analysis of Cell Viability
The in vitro cell viability of HT-29 cells posttransfection was measured using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium Bromide (MTT) method. In brief, in the indicated time points, spent cell cultural medium was replaced with 0.1 mL fresh medium containing 0.5 mg/mL MTT. Cells were then incubated at 37°C for 1 hour and resolved by 0.1 mL dimethyl sulfoxide (Sigma). The absorbance was measured at 570 nm.
Cell Migration Assays
Migration assay was conducted with Transwell inserts with 8.0 mm pore size membrane (24-well format; Corning, New York). To measure the invasion ability of CRC cell line HT-29, the previously mentioned inserts were precoated with Matrigel matrix (BD Science, Sparks, Maryland). The cells (1 × 105) were resuspended in serum-free medium and seeded to the upper chamber. The lower chambers were filled with complete culture medium containing 10% FBS. After incubation at 37°C for 24 hours, the migrated cells present on the lower side of the membrane were fixed, stained, and counted. Each experiment was performed in triplicate.
Quantitative Real-Time Polymerase Chain Reaction
Quantitative real-time polymerase chain reaction (qRT-PCR) was carried out on the 7900HT thermocycler (Applied Biosystems). Each sample was tested in triplicate using the TaqMan microRNA assay (TaqMan Universal PCR Master Mix II, No AmpErase UNG; Applied Biosystems) along with the TaqMan probes and primers (Applied Biosystems). Fifteen microliters of polymerase chain reaction (PCR) master mix of the PCR reactions started at 95°C for 10 minutes, followed by 50 cycles of 95°C for 15 seconds and 60°C for 60 seconds. The reaction includes 50 nmol/L stem-loop RT primer, 1× RT buffer, 0.25 mmol/L each of deoxynucleotides, and 3.33 U/μL MultiScribe reverse transcriptase in a total volume of 15 μL. The 15 μL reactions were mixed well and incubated for 30 minutes at 16ºC, 30 minutes at 42ºC, 5 minutes at 85ºC, and then held at 4ºC. All reverse transcriptions and no template controls were run at the same time following the RT steps. The average quantification cycle (Ct) and standard deviation were calculated with the SDS software, along with the coefficient of variation (CV) of each sample. Samples with CV > 0.05 were tested again. In our miRNA test, all the samples had a CV < 0.01. Samples with Ct < 15 or Ct > 35 were excluded. Real-time PCR data were analyzed using the SDS software v2.3 (settings: automatic baseline; threshold: 0.2), and relative miR-29b levels were calculated with the RQ Manager v1.2.1 (Applied Biosystems). In addition, the raw Ct data obtained from each sample were normalized to the mean expression of spike-in U6 with the 2−ΔΔCt transition: ΔCt = Ct(miR-29b) − Ct (U6). In the next steps, the ΔΔCt value and the normalized miRNA expression were calculated: ΔΔCt = ΔCt(cancer sample) − ΔCt(control sample). Normalized miRNA expression in the sample = 2−ΔΔCt. The investigators were blinded to the results of clinical and pathological diagnosis. The primers of both miR-29b and U6 were reported in Table 1.
Primers used in qPCR.
Abbreviation: qPCR, quantitative polymerase chain reaction.
Statistical Analysis
For all analyses, TNM staging was categorized as stage I versus stage II/III/IV. The receiver operating characteristic curve (ROC) analysis was undertaken using the expression level for miR-29b in the CRC specimens from patients with cancer and healthy controls to assess the diagnostic accuracy of both tissue and plasma miR-29b levels. Using this approach, the area under the ROC (AUC) identified optimal sensitivity and specificity levels at which to distinguish normal individuals from patients with cancer from CRC, and corresponding thresholds were calculated for the miR-29b. Parametric tests (the t test between 2 groups and the one-way analysis of variance for 3 or more groups) were used to analyze the relationship between the miR-29b expression level and various clinic–pathologic characteristics. Nonparametric tests (the Wilcoxon–Mann–Whitney test between 2 groups and the Kruskal–Wallis test for 3 or more groups) were used to analyze the relationship between the miR-29b expression level and various clinic–pathologic characteristics. Spearman correlation analysis was performed to determine the correlation between expression of miR-29b and CRC stages. Statistical analysis was performed using Statistical Program for Social Sciences (SPSS) software 19.0 (SPSS Inc, Chicago, Illinois). All P values shown were 2 sided, and a P value of <.05 was considered statistically significance.
Results
MiR-29b Expression Levels Were Downregulated in Tissue and Plasma in CRC Cases
The expression of miR-29b was downregulated in CRC tissues and plasma. First, to examine whether the miR-29b is differentially expressed in human CRC tissues, its expression level was determined using TaqMan real-time PCR in 200 pairs of CRC cancer tissues and pair-matched adjacent noncancerous colon tissues. Our results demonstrated that the expression level of miR-29b was significantly reduced in CRC tissues in comparison with the adjacent noncancerous colon tissues. Besides, it was also found that the plasma miR-29b levels in cases with CRC were also reduced compared with the 400 paired controls (Figure 1). We found that neither age nor gender was different between the 200 cases and 400 matched controls (Table 2).

The relative expression of miR-29b in the tissue and plasma in the colorectal cancer (CRC) versus controls.
Clinicopathological features of 200 Patients With CRC and 400 Controls.

Abbreviation: CRC, colorectal cancer.
Considering that both the miR-29b levels in tissue and plasma were downregulated in cases with CRC, we also conducted the Spearman correlation between tissue and plasma miR-29b levels. It was found that the expression of miR-29b in tissue was associated with level in plasma (P = .001). The liner association was displayed in Figure 2.

The association between the levels of miR-29b in the tissue and plasma.
Associations of Aberrant miR-29b Expression With Clinicopathological Data of Patients With CRC
In general, it was found that miR-29b (tissue—case vs control: 4.21 ± 1.79 vs 11.21 ± 3.18, P = .001; plasma—case vs control: 2.31 ± 0.97 vs 7.81 ± 1.34, P = .001) was downregulated in both the tissue and plasma samples in cases with CRC. We then investigated the associations between the altered miR-29b expressions with the clinicopathological characteristics of the patients with CRC. We found that neither tissue nor plasma miR-29b expressions were associated with age and gender (P > .05). In advanced studies, the expressions of miR-29b were lower in the tissue and plasma with larger tumor size group (over 3 cm). Advanced study showed that low expression of tissue and plasma was strongly associated with CRC differentiation status (P = .004 and P = .012). Besides, the expression of miR-29b in tissue and plasma was associated with the lymph node metastasis (P < .001 and P < .001, respectively). Besides, we also found that both tissue and plasma miR-29b levels were associated with the stage classifications (P < .001 and P = .001, respectively). All the detailed data presented in Table 3.
Associations of Aberrant miR-29b Expression in Tissue and Plasma With Clinicopathological Data of Patients With CRC.

Abbreviation: CRC, colorectal cancer.
The Expression of Tissue and Plasma miR-155 After the Surgery
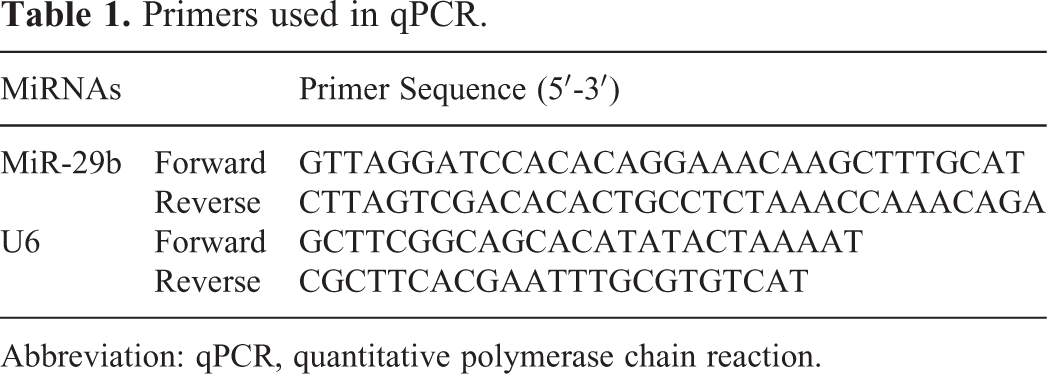
A total of 200 cases with CRC were obtained to detect the change in the plasma miR-29b. Compared with the preoperative expression, plasma miR-29b was demonstrated to be significantly increased. As showed in Figure 3, the expression of tissue miR-29b changed from 2.31 to 7.84 (P = .002). In the male group, the expression of plasma miR-29b was 1.94 ± 0.743, while the postoperative level is 8.3 ± 0.92 (P = .001). The level of miR-29b in the female group changed from 2.91 ± 1.43 to 7.50 ± 1.32, and a significant difference was detected (P = .016).
The preoperation and postoperation level of miR-29b in the colorectal cancer (CRC) cases.
Diagnostic Performance of Tissue and Plasma miR-29b for CRC
The diagnostic performances of tissue and plasma miR-29b for the CRC were detected. Cutoff points were determined using the highest sum of sensitivity and specificity. The cutoff points for tissue and plasma miR-29b were 5.46 and 2.86, respectively. Tissue miR-29b showed an area under the curve (AUC) of 0.883, with a sensitivity of 81.6% and a specificity of 84.9%. However, the AUC for plasma miR-29b was 0.743, with a sensitivity of 61.4% and a specificity of 72.5% (Figure 4).

Diagnostic performance of tissue and plasma miR-29b for colorectal cancer (CRC).
The Effects of miR-29b Overexpression on Cell Viability
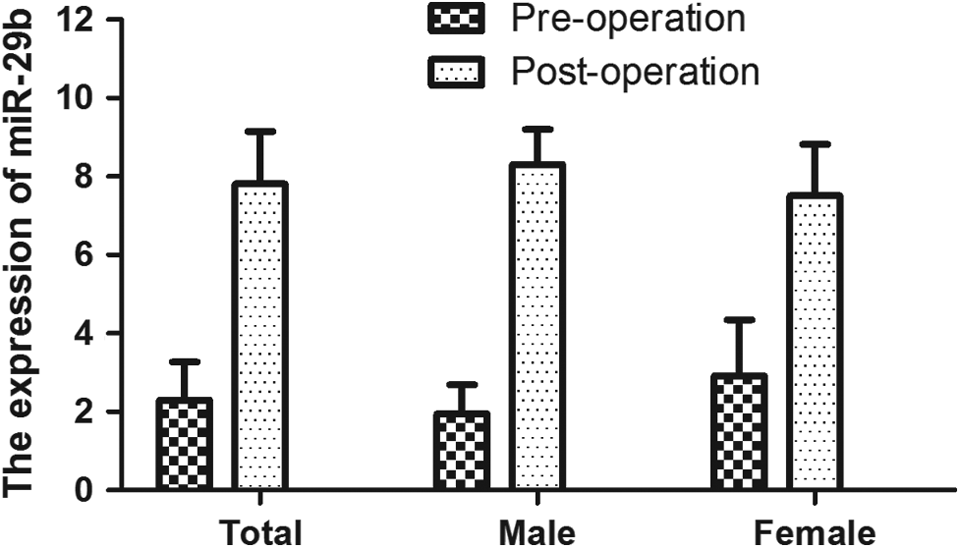
In order to assess the effects of miR-29b on CRC cell line HT-29 growth, the miR-29b precursor was transfected into HT-29 cells, and cell growth at various posttransfection time points was examined. MiR-29b was found to downregulate compared with the normal colon tissue cell (Figure 5A) and significantly inhibited proliferation in cells 3 days posttransfection with miR-29b precursor (data showed in Figure 5B).
The effects of miR-29b overexpression on cell viability. A, MiR-29b was found to downregulate compared with the normal colon tissue cell. B, MiR-29b significantly inhibited proliferation in cells 3 days posttransfection with miR-29b precursor.
The Effects of miR-29b Overexpression on Migration
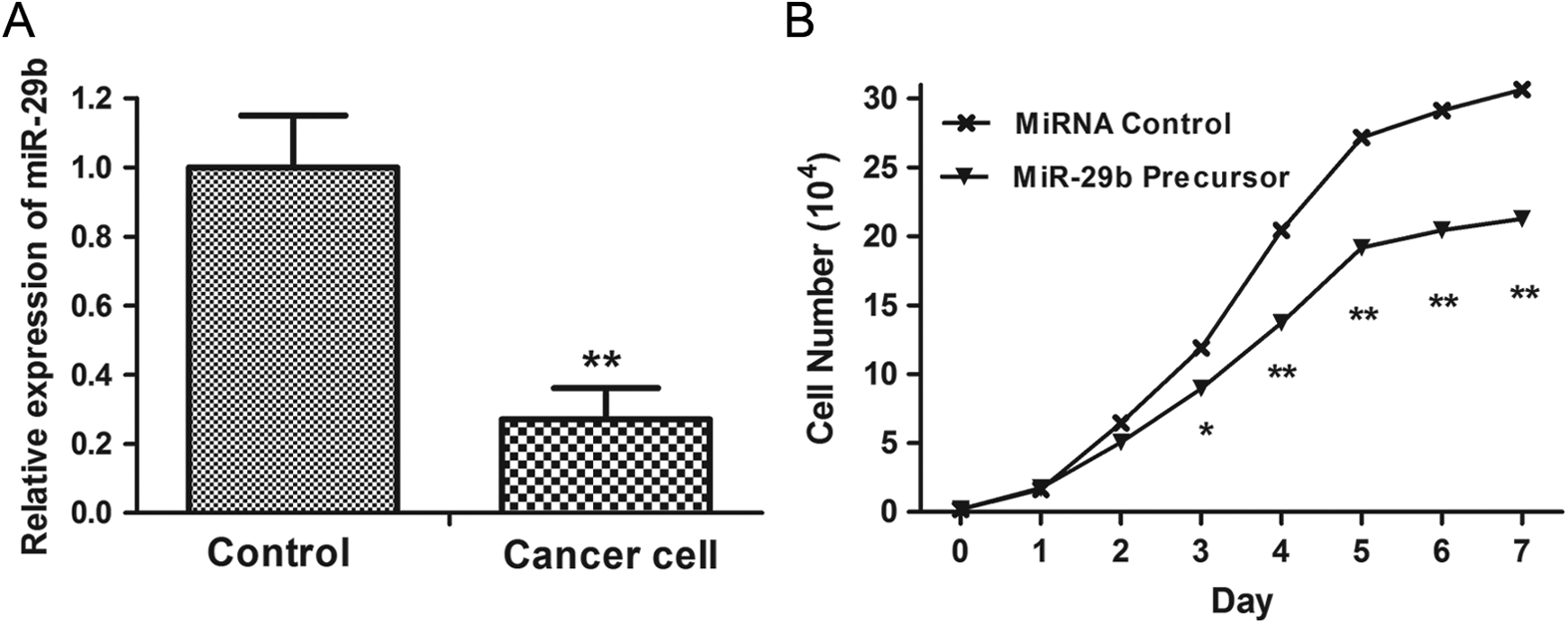
In addition to their role in the different expression of miR-29b on the lymph node metastasis in the cases with CRC, however, the role of miR-29b in regulating cell migration in CRC cells is currently unknown. To address this question, we investigated whether upregulation of miR-29b impacts CRC cell mobility. As shown in Figure 6, upregulation of miR-29b significantly inhibited the migration ability of CRC cells compared with the control miRNA.
The effects of miR-29b overexpression on migration. Upregulation of miR-29b significantly inhibited the migration ability of colorectal cancer (CRC) cells compared with the control miRNA.
Discussion
Early CRC diagnosis remains a major challenge but holds promise of resulting in a more favorable disease outcome. 15 In light of the fact that growing evidence indicating that miRNAs are novel classes of cancer-relevant molecules that regulate gene expression at the posttranscriptional level, the application of miR-29b for the CRC is a promising approach. In this study, we detected the effect of both the tissue and plasma miR-29b as the biomarkers for the CRC. We found that the expression of plasma miR-29b is associated with the tissue miR-29b. Advanced study showed that aberrant miR-29b expression in both cancer tissues and plasma is associated with the clinicopathological data of patients with CRC. Tissue miR-29b showed an AUC of 0.883, with a sensitivity of 81.6% and a specificity of 84.9%. However, the AUC for plasma miR-29b was 0.743, with a sensitivity of 61.4% and a specificity of 72.5%. The analyses of the biological effects of miR-29b for CRC showed that miR-29b could inhibit the cell viability and migration.
Expression of miRNA-29b is important in the development of kinds of cancers. 16 Recent studies have demonstrated that miR-29b expression is decreased during the development of CRC, and miRNA-29b can act as a tumor suppressor. 17 Consistent with these findings, we observed a significant decrease in both tissue and plasma miRNA-29b levels in patients with CRC compared with the controls. In the most recent studies, the associations between cases with miR-29b and CRC were detected. To present proof-of-principle application for using miRNAs as diagnostic markers for colon cancer, Ahmed et al carried out global microarray expression studies on stool samples obtained from 15 individuals (3 controls and 3 each with TNM stage 0-1, stage 2, stage 3, and stage 4 colon cancer) to select for a panel of miRNA genes for subsequent focused semi-qPCR analysis studies. 18 In their results, miR-29b is one of the 8 miRNAs that had decreased expression in the stool of patients with CRC, which was also more pronounced from early to later TNM stages. 19
In the most recent studies, the applications of circulating miRNAs for the diagnosis and prognosis of kinds of diseases were detected in different studies. Kiko et al reported that the qRT-PCR results showed that plasma miR-34a and miR-146a levels and cerebrospinal fluid (CSF) miR-34a, miR-125b, and miR-146a levels in patients with Alzheimer disease were significantly lower than that in controls. On the other hand, CSF miR-29a and miR-29b levels were significantly higher than that in the controls. 20 In a previous study, it was identified that the expression values of miR-29b were correlated with gene expression data from the same samples to assess their biologic impact on chronic lymphocytic leukemia. In this study, we also found that IGHV-unmutated high expression of ZAP-70 protein and low expression of the miR-223, miR-29c, miR-29b, and miR-181 family were strongly associated with disease progression in chronic lymphocytic leukemia cases harboring 17p deletion, whereas in those harboring trisomy 12, only high expression of the miR-181a, among the analyzed parameters, suggested more aggressive disease. However, the association between tissue and plasma miR-29b in cases with CRC was quite poorly understood. 21 In this study, we conducted the association between the levels of miR-29b in cancer tissues and plasmas. Through anglicizing the data form 200 cases, it was found that the expression of miR-29b in tissue was associated with plasma level (P = .001). Although the strong association between tissue and plasma miR-29b was detected, the ROC analyses showed that the tissue miR-29b showed better sensibility and specificity. While considering the atraumatic acquisition of plasma from the cases, it provides a promising application of plasma miR-29b for the diagnosis of the CRC.
The effect of miR-29b on the CRC progression was not that clear by now. In this study, we conducted an in vitro study to detect the effect of miR-29b on the CRC cell line. Through the relevant study, we found that overexpression of miR-29b could inhibit the cell viability and migration. In a previous study, Poudyal et al confirmed that HAG induces miR-29b expression to target matrix metalloproteinase-2 (MMP-2), thereby suppressing the migration of colon cancer cells. Results are consistent with this hypothesis. The study supports the understanding that targeting MMP-2 by miR-29b is a mechanism by which HAG suppresses the migration of colon cancer cells. 10 Considering the important role of miR-29b in the CRC development, miR-29b targeted the 3′ untranslated region of BCL9L and decreased its expression with a consequent decrease in nuclear translocation of β-catenin. Ectopic expression of miR-29b inhibited anchorage independent cell growth, promoted reversal of epithelial to mesenchymal transition, and reduced the ability of conditioned medium from SW480 cells to induce in vitro tube formation in endothelial cells. These results have unraveled a novel role of miR-29b in Wnt signaling in human CRC cells with implications in the treatment of CRC. 9
Conclusion
In conclusion, our data suggest that both the tissue and the plasma miR-29b levels have some value as a diagnostic tool for CRC. It is reported that plasma miRNA-29b levels were correlated well with miRNA levels in tumor tissue, so miRNA-29b may be able to serve as a noninvasive diagnostic marker for CRC. In the present study, we included a total of 200 patients and 400 age- and gender-matched controls. Advanced biological effects were conducted to detect the potential effect on the cell viability and migration. Future investigations including larger patient populations and patients with early-stage CRC are needed to confirm the potential diagnostic value of miRNA-29b in CRC.
Footnotes
Authors’ Note
L.L.P., G.Y., Z.L., G.X.B., L.J.L., and J.C.Q. participated in the design of the study, writing, statistical analyses, and clinical sample and data collection. L.L.P. and J.C.Q. carried out the miRNA analyses. L.L.P., G.Y., C.Y.Z., W.J.S., Z.L., G.X.B., L.J.L., and J.C.Q. interpreted data and participated in revising the manuscript. Z.L., G.X.B., L.J.L., and J.C.Q. contributed to conducting the study, clinical sample, data collection, statistical analyses, and editing the manuscript. All authors read and approved the final manuscript. L.L., G.Y., and C.Y. contributed equally to this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
