Abstract
Background:
This work was designed to explore the roles of PIM-1 in the development of cervical cancer.
Methods:
There were 90 paired cervical tumor samples and the non-tumor adjacent tissue. The levels of PIM-1 in different samples were examined using quantitative reverse transcriptase-polymerase chain reaction (qRT-PCR) methods. The potential diagnostic value of PIM-1 was analyzed by the receiver operating characteristic (ROC) curve; furthermore, the expression of EGFR in tumor samples was detected, and Pearson’s correlation analysis was performed to analyze the relationship between the expression of PIM-1 and EGFR. Finally, cervical cancer cell line Hela cells were cultured and treated by PIM-1 siRNA, and MTT assay and Pi/Annexin V assay were performed to explore the effects of PIM-1 siRNA on the growth and apoptosis ability of the Hela cells.
Results:
PIM-1 was significantly up-regulated in cervical cancer tissue compared to adjacent tissue, and the expression of PIM-1 in patients with cervical cancer is positively associated with the size and metastasis of the tumor. ROC analysis showed PIM-1 is a sensitive biomarker for the diagnosis of cervical cancer. Furthermore, EGFR was over-expressed in cervical cancer tumor tissues, and the levels of PIM-1 and EGFR in cervical cancer tissue were positively correlated. Finally, PIM-1 siRNA dramatically inhibited the viability and promoted the apoptosis of the Hela cells.
Conclusion:
Our findings prove that PIM-1 may function as an oncogene in cervical cancer and can regulate the EGFR signaling in cervical cancer.
Keywords
Introduction
Cervical cancer (CC) is now a common female cancer and one of the leading causes of death among women.1-3 Previous studies indicated that CC usually derives from a sequential accumulation of molecular and genetic alterations within colon epithelial cells, leading to uncontrolled cell growth. 4 , 5 However, the pathogenesis of CC remains unclear. The current treatment of CC includes chemotherapy, radiotherapy, or surgery, 6 , 7 unfortunately, due to the strong metastatic activity of this cancer. However, none of these treatments has achieved the desired therapeutic effects, and the prognosis of patients with late-stage of CC remains poor. 2 , 8 Therefore, searching for new molecule targets for the diagnosis and treatment of CC is in great demand.
The PIM kinase family is comprised of three proteins, including PIM-1, PIM -2, and PIM-3. 9 It has been reported that different transcription factors can recruit PIM-1 to the chromatin and consequentially activate the transcription of the target genes. 10 PIM-1 was shown to regulate different cellular activities; for example, PIM-1 has been reported to inhibit cell apoptosis and promote cell proliferation. 11 , 12
Previous studies indicated that PIM-1 functions as an oncogene in several human cancers including breast, gastric, and prostate 11 , 13 , 14 ; however, reports on the functions of PIM-1 in CC is relatively rare and whether PIM-1 functions as an oncogene in CC is still unclear. Therefore, we designed the current study to explore the clinical significance of PIM-1 in CC. The levels of PIM-1 in tumor tissues of patients with CC and the adjacent tissue will be compared, and the potential diagnostic value will be analyzed; also, the roles of PIM-1 on the growth as well as apoptosis of the CC cells will also be evaluated.
Material and methods
Patients and clinical information
The present study included 90 cervical tumor samples as well as the paired non-tumor adjacent normal tissues at Women & Children Health Institute Futian Shenzhen, between May 2018 and September 2019 from patients who have been histologically diagnosed with CC and received surgery in our hospital. All samples were stored in liquid nitrogen immediately after surgery. Patients who received preoperative radiotherapy or chemotherapy were excluded from this study. Follow-up data were obtained by reviewing outpatient charts or through correspondence with the patients. The study was approved by Women & Children Health Institute Futian Shenzhen’s ethic committee, and informed consent was obtained from each of the participants.
Quantitative Reverse Transcriptase-Polymerase Chain Reaction
The expression levels of PIM-1 and EGFR in CC tumor samples and the adjacent non-tumor tissues were examined by real-time quantitative PCR methods as previously described 11 . In brief, TRIzol was used to extract the total RNAs (Invitrogen), and SYBR ExScript RT-PCR kit (TaKaRa, Osaka, Japan) was used for the polymerase chain reaction (PCR) reaction. The ABI 7900 System was used to amplify the genes. The primers used in the present work have been designed by Sangon Biotech (Shanghai, China). GAPDH was used as the internal control to quantify the levels of PIM-1 and EGFR by 2−ΔΔCt method.
Cell culture and transfection
To determine the effect of PIM-1 siRNA on the proliferation and apoptosis of the CC cells, human CC cell line Hela cells (Shanghai Institute of Cell Biology, Chinese Academy of Sciences, Shanghai, China) were transfected by PIM-1 siRNA or the PIM-1 siRNA negative control with Lipofectamine 3000 (Invitrogen). The PIM-1 siRNA was purchased from GenePharma (Shanghai, China).
MTT cell viability analysis
The effects of PIM-1 siRNA on the viability of the Hela cells was determined by the MTT assay with the commercially available kit (Beyotime, Shanghai, China) following the protocol provided by the manufacturer.
Flow cytometry apoptosis assay
To determine the effects of PIM-1 siRNA on the apoptosis of the Hela cells, the Hela cells were double-stained by PI and annexin V (PI/annexin V kit was purchased from Beyotime) after 48 h of transfection, and cell apoptosis was measured by the FACS Calibar flow cytometer (Becton Dickinson, Franklin Lakes, NJ, USA).
Western blot
The expressions of PIM-1 and EGFR in Hela cells were determined by the western blot method. Briefly, 48 h after transfection, the Hela cells were lysed by radioimmunoprecipitation assay (Beyotime). Next, the proteins in each sample were separated using 10% gels via SDS-PAGE. The proteins were then transferred to PVDF (Millipore, Burlington, MA, USA) membranes, blocked by 5% skim milk and treated by the anti-PIM-1 (ab98004, 1:1000), anti-EGFR (ab52894, 1:1000), or anti-GAPDH (ab8245, 1:2000) antibodies (all purchased from Abcam, Cambridge, MA, USA). The membranes were then washed and treated by HRP-conjugated secondary antibodies, and incubated by BeyoECL Plus (Beyotime, Shanghai, China) for colorization. The images were captured by Tanon 6200 image system (Tanon, Shanghai, China). GAPDH was used for normalization.
Statistical analysis
The data were analyzed by SPSS 19.0 and GraphPad prism 7.0. The data between the two groups were analyzed using the Student t-test, and the data among the three groups were analyzed by one-way analysis of variance (ANOVA). Pearson’s coefficient was used for the correlation analysis. P<0.05 was set as a significant difference.
Results
Over-expression of PIM-1 in CC
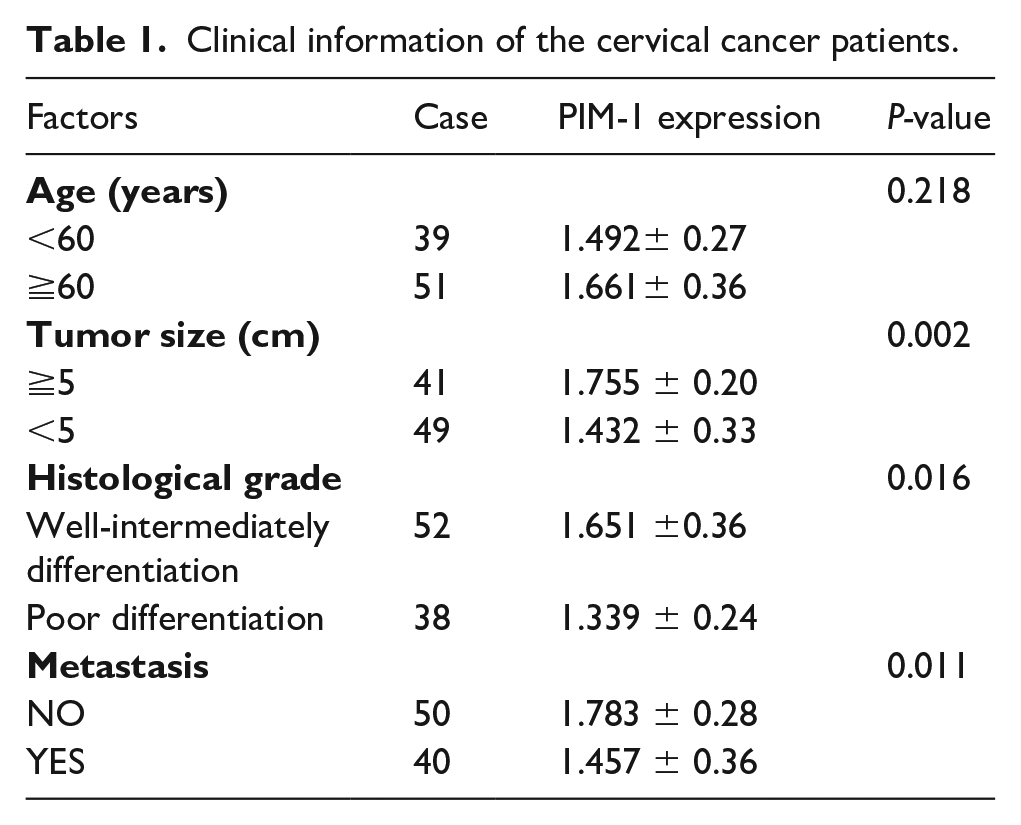
We examined the expression of PIM-1 in tissue samples of patients with CC and the adjacent tissue by quantitative reverse transcriptase (qRT)-PCR methods (Figure 1(a)). We observed that the expression of PIM-1 in CC tissue was significantly up-regulated compared with the adjacent non-tumor tissues (P<0.001); furthermore, as shown in Table 1, increased expression of PIM-1 in CC patients may indicate increased tumor size (P<0.001) and increased risk with metastasis (P<0.05) of the patients.
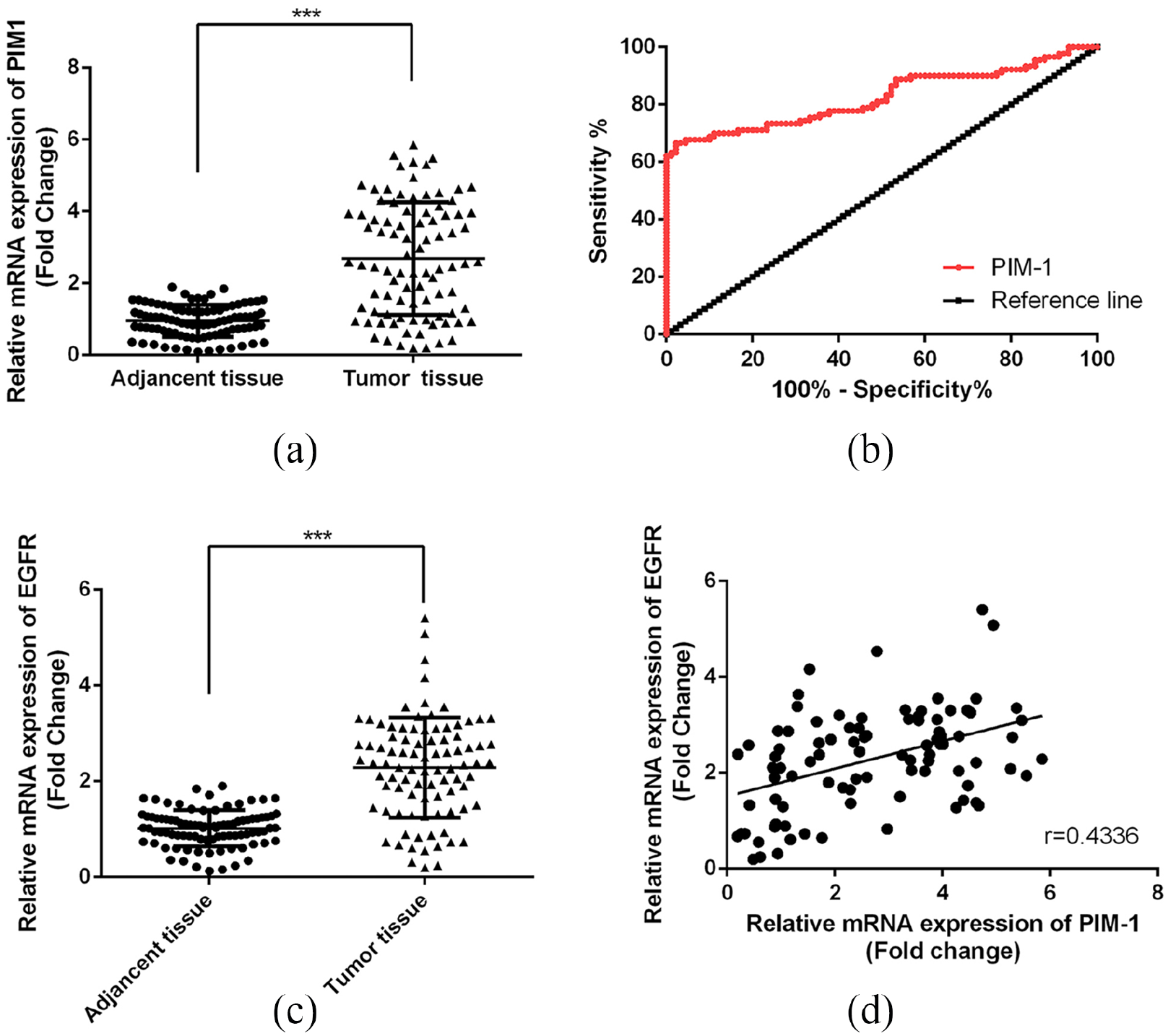
Up-regulation of PIM-1 in CC. (a) Comparison of the mRNA levels of PIM-1 in CC tumor tissue and the adjacent tissue. (b) Results of ROC analysis. ***P<0.01 versus control. (c) Comparison of the mRNA levels of EGFR in CC tumor tissue and the adjacent tissue. (d) Results of correlation analysis. ***P<0.001 versus control.
Clinical information of the cervical cancer patients.
PIM-1 may serve as a diagnostic marker for CC
Next, to determine the diagnostic value of PIM-1 for CC, we performed receiver operating characteristic (ROC) curve analysis. As Figure 1(b) shows, the area under the curve (AUC) for PIM-1 to distinguish the CC tumor and the normal tissue was 0.8252 (cut-off value 1.288; 95% CI 0.7614, 0.8890), indicating that PIM-1 could be used for the diagnosis of CC tumors with high sensitivity and specificity (Figure 1(b)).
The levels of PIM-1 were positively correlated with the level of EGFR in patients with CC
Pearson’s coefficient was used to analyze the correlation between the expression of PIM-1 and EGFR in tumor samples of the CC patients. As shown in Figure 1, EGFR was over-expressed in CC tissue (Figure 1(c)), and we found that the levels of PIM-1 and EGFR were positively correlated (Figure 1(d); r= 0.4336; P<0.001).
Knockdown of PIM-1 decreases the viability and promotes the apoptosis of the Hela cells via decreasing the expression of EGFR
Finally, we treated CC cell line Hela cells with the PIM-1 siRNA to determine the roles of PIM-1 on the viability as well as apoptosis of the cells. We designed two PIM-1 siRNAs, and compared them with untransfected cells. Both siRNA-1 and siRNA-2 lead to a dramatic decrease in the mRNA (Figure 2(a); P<0.01) and protein (Figure 2(b) and (c); P<0.01) expression levels of PIM-1 in CC cells. siRNA-1 showed better inhibitory effects and was used in the following experiments (Cell proliferation, apoptosis and WB assays). The results of MTT assay indicated that PIM-1 siRNA-transfected cells showed a decreased ability of proliferation at 12, 24, and 48 h (Figure 2(d); P<0.05). Moreover, the apoptosis of Hela cells with different treatments were determined by flow cytometry methods. We found that PIM-1 siRNA markedly promoted cell apoptosis in vitro (Figure 3; P<0.01). Finally, the expression of EGFR in different groups was examined by the western blot method, and as Figure 4 shows, PIM-1 siRNA induced the dramatic down-regulation of EGFR in Hela cells in vitro (Figure 4; P<0.01).
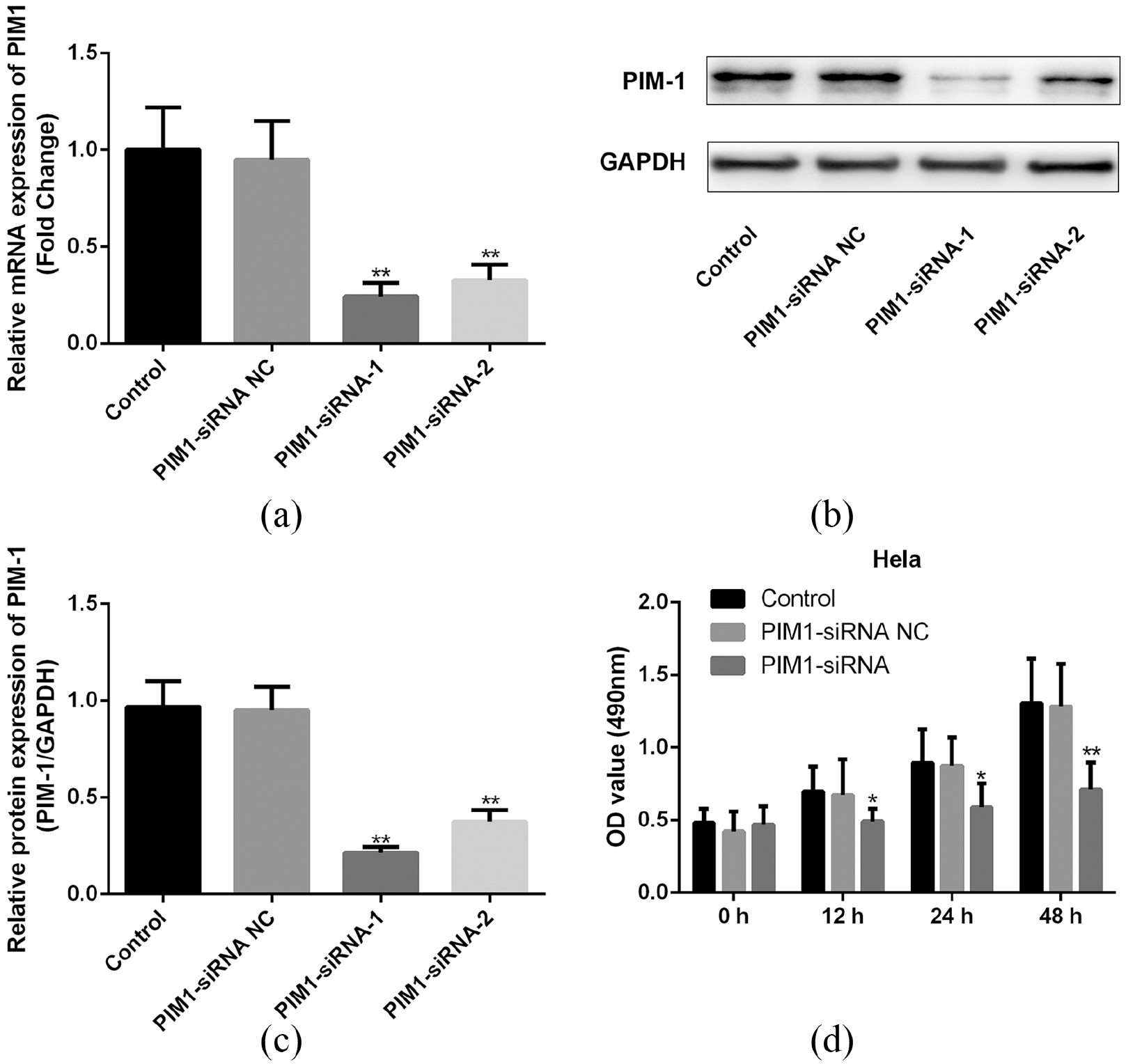
Effect of PIM-1 on the proliferation of CC cells in vitro. (a) Effect of PIM-1 siRNA on the mRNA expression of PIM-1 in Hela cells. (b) Effect of PIM-1 siRNA on the protein expression of PIM-1 in Hela cells. (c) Quantified results of (b). (d) Results of MTT assay. *P<0.05, **P<0.01 versus control.
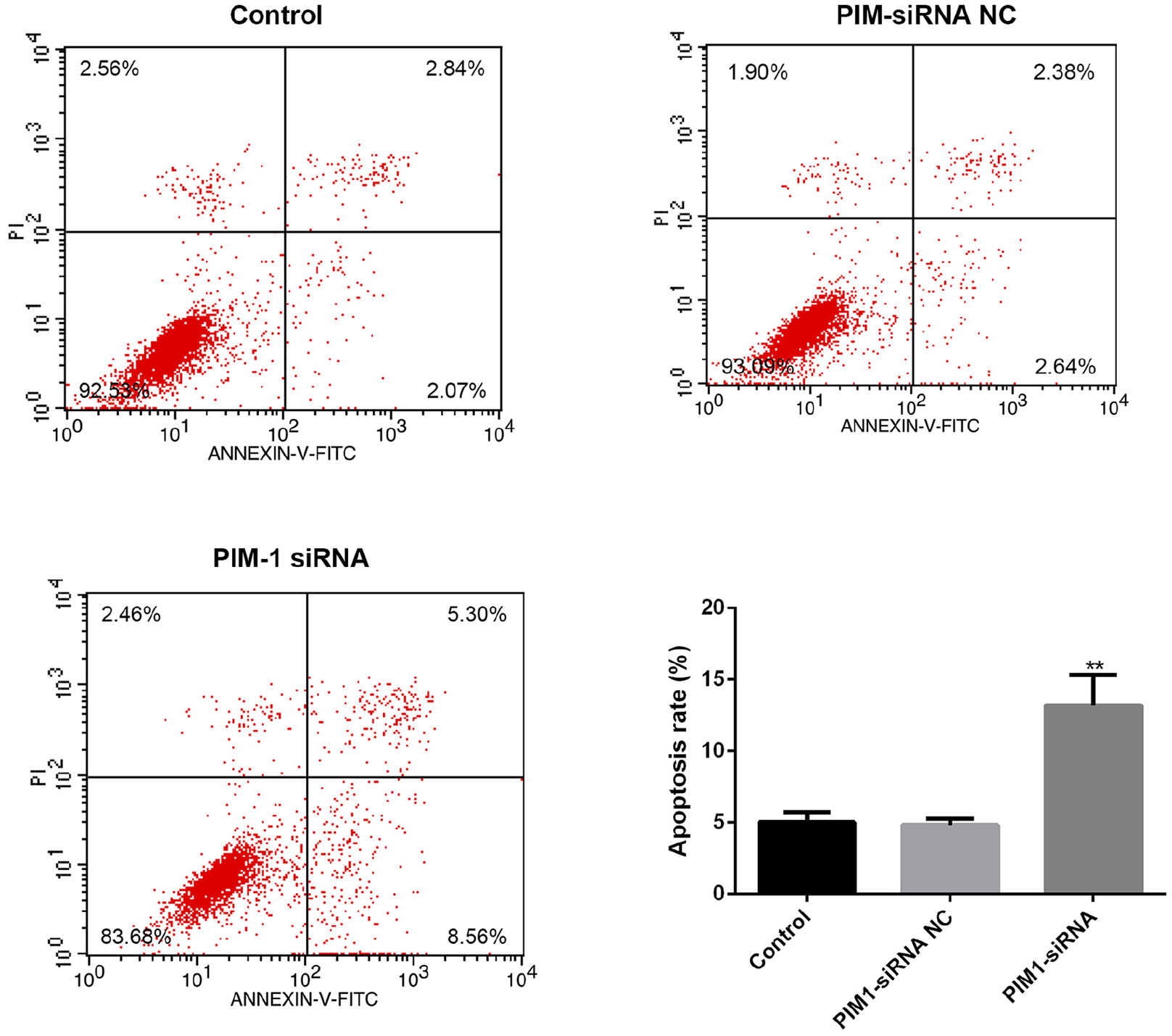
Effect of PIM-1 on the apoptosis of Hela cells in vitro. **P<0.01 versus control.
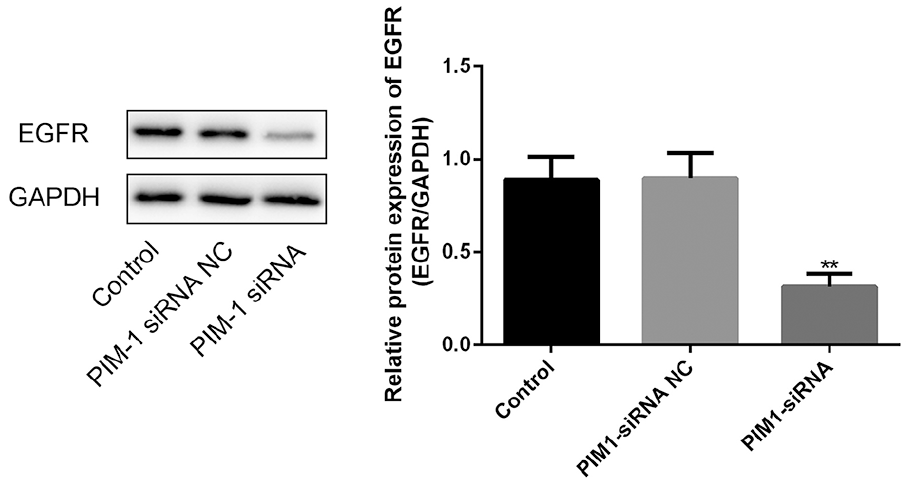
Effect of PIM-1 on the expression of EGFR in Hela cells in vitro. **P<0.01.
Discussion
In this study, we explored the clinical significance of PIM-1 in CC. Our results prove that PIM-1 may function as an oncogene in CC, and PIM-1 may exert its carcinogenic effects through activating the EGFR signaling via increasing the proliferation and decreasing the apoptosis of CC cells.
The PIM kinases were first reported as most significantly up-regulated genes during the process of embryonic development 9 , 15 ; however, in recent years, PIMs were also found to be over-expressed in several cancers. The role of PIM-1 as an oncogene has been investigated in different studies. 11 , 14 , 16 , 17 For example, PIM-1 was significantly up-regulated in gastric cancer tissue, and was regulated by microRNA-101-3p to affect the growth and migration of the gastric cancer cells. 11 Moreover, PIM-1 was over-expressed in salivary gland adenoid cystic carcinoma and exerted carcinogenic behaviors on the cancer cells 16 ; furthermore, PIM-1 was also abnormally increased in nasopharyngeal carcinoma, and the knockdown of PIM-1 could inhibit the proliferation and migration of the cancer cells. 17 However, little was known about the function of PIM-1 in CC. In the present work, we first reported that PIM-1 was markedly over-expressed in CC tumor samples, and increased levels of PIM-1 in CC patients were positively associated with increased tumor size and increased chance of metastasis. Interestingly, the results of the ROC analysis indicated that the levels of PIM-1 can distinguish the CC tumor samples and the non-tumor adjacent tissues, suggesting that PIM-1 may serve as a potential diagnostic marker for CC. Taken together, our data suggest that PIM-1 may function as an oncogene in CC, and the levels of PIM-1 were positively correlated with the severity and progress of CC.
The over-expression or mutation of EGFR was reported in different types of tumors, including CC, 7 , 8 , 18 and currently an EGFR inhibitor is widely used in the clinical field for the treatment of CC. 7 , 18 In contrast, the correlation between PIM kinases and EGFR has been reported previously. 14 , 15 Siu et al. 14 suggested that PIM-1 can regulate the EGFR signaling in prostate cancer cells; Broutian et al. 15 reported that PIM-1 affected the EGFR signaling in head and neck squamous cell carcinoma. However, whether PIM-1 can regulate the EGFR signaling in CC still needs to be investigated. Consistent with the previous observation, we observed that EGFR was significantly increased in CC tissue; we found that the levels of PIM-1 and EGFR were positively correlated. Therefore, the above results confirmed that the aberrant expression of PIM-1 may lead to the abnormality in the EGFR signaling and consequentially contribute to the development of CC.
PIM-1 is known to play critical roles in regulating the viability of different cells.14,17,19-23 For example, prostate cancer cells, 14 nasopharyngeal carcinoma cells, 17 acute myelocytic leukemia cells, 19 vascular smooth muscle cells, 20 B-cells, 21 pulmonary artery smooth muscle cells, 22 and T lymphocytes. 23 Therefore, we hypothesized that PIM-1 may also regulate the viability and apoptosis of CC cells. We observed that the knockdown of PIM-1 siRNAs in Hela cells inhibits the viability and increases apoptosis of the cells. Interestingly, it was found that PIM-1 siRNA could decrease the expression of EGFR in Hela cells, which was consistent with our observation in clinical samples. Taken together, the above data suggest that PIM-1 may affect the viability and apoptosis of Hela cells by suppressing EGFR signaling in vitro.
In summary, we reported for the first time that PIM-1 was over-expressed in CC; we also found that PIM-1 may affect the viability as well as the apoptosis of Hela cells through regulating EGFR signaling. Our data have provided novel evidence for the potential of PIM-1 for the early diagnosis and treatment of CC.
Footnotes
Acknowledgements
This work is supported by grants from the Nonprofit Health Research of Futian District, Shenzhen, in 2016, No. FTWS20160044.
Author contributions
Hongwen Yang and Kui He contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
