Abstract
Objective
Clinical and laboratory guidelines recommend thyroglobulin antibodies (TgAbs) measurement with every thyroglobulin (Tg) measurement for the follow-up of differentiated thyroid cancer (DTC) patients. However, no evidence exists on the need for perpetual TgAbs testing in patients who are TgAb-negative at baseline. Our study was carried out to evaluate the prevalence, the dynamic changes, and the clinical significance of TgAbs that appeared de novo during the follow-up of DTC patients who were TgAb-negative at baseline.
Methods
The data of DTC patients with negative pre-ablation TgAbs were reviewed retrospectively. The main characteristics of patients with both transient and sustained de novo TgAbs appearance were analyzed. DTC patients with persistently negative TgAbs served as controls.
Results
Among 119 patients with pre-ablation negative TgAbs, 14 cases (11.7%) with de novo TgAbs appearance (10 and 4 patients with a transient and sustained de novo TgAbs appearance, respectively) were detected. No differences in disease-free survival were observed in patients with de novo TgAbs appearance compared to controls. The TgAbs peak value was higher in patients with sustained de novo appearance compared to patients with transient de novo. Two of 14 patients with de novo TgAbs developed structural disease with concurrently detectable Tg in both cases.
Conclusions
Transient de novo TgAbs appearance is not infrequent during DTC patients’ follow-up, and it has no apparent clinical impact. Sustained de novo TgAbs appearance is rare and may predict structural recurrences; however, similar disease-free survival was observed in patients with sustained de novo TgAbs and TgAb-negative DTC patients.
Keywords
Introduction
Differentiated thyroid cancer (DTC) accounts for 3.1% of all malignancies in United States, and registered about 53.990 new cases in 2018. 1 While the percentage of patients surviving 5 years after diagnosis of DTC is estimated at 98.1%, persistence and recurrence rates of disease may reach 30%, representing a significant issue of DTC management. 1 , 2
Serum thyroglobulin (Tg) and neck ultrasound (US) are currently the basic tools used for follow-up of patients with DTC. 2 Notably, positive Tg autoantibodies (TgAbs) are detected in 15%–30% of DTC patients (compared to ∼ 10% in the general population) 3 , 4 and may interfere with Tg measurement, leading to underestimated or falsely undetectable Tg concentrations in widely adopted immunometric assays (IMAs). 5 , 6 In such circumstances, serial TgAb measurements (i.e. TgAbs trend) play a useful role as a surrogate DTC tumor marker, both in patients with or without histologically confirmed chronic lymphocytic thyroiditis. 2 , 3 , 7 , 8 Indeed, a consistent reduction in serum TgAbs levels is an indication that the patient is likely free of disease. 2 , 3 In contrast, a consistent rise of serum TgAbs levels should raise the suspicion of recurrence. 2 , 3 Unchanged serum TgAbs levels must be regarded as indeterminate, although the risk of persistent disease or recurrence is higher in these patients than in those with decreasing TgAbs levels. 3 , 9 , 10
Accordingly, the American Thyroid Association (ATA) guidelines recommend that in the postoperative evaluation of DTC, the quantitative assessment of TgAbs always has to be carried out with every measurement of Tg. 2 Notably, even though most TgAbs assays on the market are claimed to have been standardized against the 1st International Reference Preparation 65/93 standard, wide interassay variability between different TgAbs assays is observed. 3 , 7 , 11 , 12 Then, for longitudinal consistency of clinical care, consecutive measurements of TgAbs levels should be performed in the same laboratory using the same assay every time. 3 Should a change of assays occur, the TgAb measurement should be performed simultaneously at least once with both assays in each individual patient in order to establish an individual baseline level with the new assay. 3 However, it must be said that in clinical practice establishing an individual baseline level with the new assay is not always feasible. 11
Finally, although there is some evidence of cases in which an elevation of TgAbs was the only biochemical sign of recurrence, such cases mostly concern patients who were TgAbs-positive at diagnosis.13–15 Therefore, no evidence exists on the need for perpetual TgAbs testing in patients who were never TgAbs- positive. In patients with negative TgAbs at baseline, de novo TgAbs appearance is commonly encountered in the first few months after thyroidectomy, radioiodine (RAI) therapy, fine-needle aspiration cytology (FNAC) or after immunotherapies. 3 , 9 In these scenarios, TgAbs are typically transiently positive and disappear over time. 3 , 7 , 9 Accordingly, a minimum waiting period of 6 months seems to be appropriate before considering an increase in TgAbs to indicate potentially progressive disease. 3 , 7 , 9 Conversely, progressive and sustained de novo TgAbs appearance is reported in some clinical cases as a sign of structural disease with concurrent undetectable or low Tg levels.15–19
Therefore, our study was carried out to evaluate the frequency of de novo TgAbs appearance in DTC patients with negative antibodies at baseline, the dynamic of TgAbs changes over time, and the relationship between DTC recurrences and concurrent Tg levels.
Methods
Institutional management and follow-up protocol
At the Clinic for Nuclear Medicine and Competence Center for Thyroid Diseases, Imaging Institute of Southern Switzerland in Bellinzona and Lugano (Switzerland), DTC patients—except those with microcarcinomas—underwent near-total thyroidectomy with central or lateral neck dissection (depending on risk and intraoperative findings) and subsequent RAI therapy. The latter was performed 4–6 weeks after surgery without starting levothyroxine replacement (i.e. a certified thyrotropin (TSH) value > 30 mIU/L was required), whereas recombinant human TSH (rhTSH, Thyrogen, Genzyme) was used in selected cases on the decision of the attending physicians. Three to 7 days after RAI administration, a post-treatment whole-body scan (RxWBS) was performed. Based on preoperative assessment, intraoperative findings, pathology reports, and RxWBS results, patients were classified at high, intermediate, and low risk according to the ATA risk stratification system. 2 In low- to intermediate-risk patients, TSH levels were maintained below 0.5 mIU/L until a cure was demonstrated, and between 0.5 and 2.0 mIU/L later. 20 The initial response to primary therapy was evaluated 6 months after RAI by neck US and basal Tg testing. Additional rhTSH-stimulated diagnostic whole-body scan (WBS) and Tg testing were performed in high-risk patients or when at RxWBS RAI uptake outside of the thyroid bed and/or positive preablation TgAbs were found. Overall, further follow-up visits were scheduled every 6–12 months as deemed appropriate by the attending physician. Follow-up visits consisted of physical examination, serum TSH, Tg, and TgAb measurement (on thyroxine). The primary treatment was considered to be completed (i.e. excellent response to treatment) if there was no evidence of disease (NED). Otherwise, further visits and examinations (including 18F-FDG positron emission tomography/computed tomography (PET/CT), CT of the chest and abdomen, FNAC) and, if needed, additional treatments (i.e. surgery, radioiodine) were performed according to our multidisciplinary thyroid board decisions.
Study design
This was a retrospective study reviewed and approved by the Institutional Review Board of Ente Ospedaliero Cantonale and the Ethics Committee of Canton Ticino, Bellinzona (Switzerland). Patients gave blanket consent to use their data for future studies. The entire study was conducted in accordance with the Helsinki Declaration. DTC patients consecutively seen at the Clinic for Nuclear Medicine and Competence Center for Thyroid Diseases, Imaging Institute of Southern Switzerland in Bellinzona and Lugano (Switzerland), from June 2008 until November 2018 were initially considered for inclusion if they met the following criteria: (a) they underwent total thyroidectomy (with or without central or lateral neck dissection) followed by RAI therapy (primary therapy); (b) they had negative serum TgAbs before RAI therapy (i.e. negative preablation TgAbs); (c) they carried out postoperative follow-up visits with available and complete follow-up data (clinical, imaging and laboratory); and (d) isolated TgAbs positivity was confirmed by a repeat analysis within a short interval of 4 months. DTC patients with TgAb-positive microcarcinomas were excluded. The medical records were retrieved from our database and reviewed to derive a specific set of data (i.e. date of DTC diagnosis, cancer histotype, risk category, number and dose of RAI treatments, TSH levels, and disease status at the time of Tg and TgAbs sample collection). Notably, TgAb measurements performed within 6 months after surgery, RAI treatments, and FNAC were excluded to minimize the effects of these settings on TgAb levels. 3 , 7 Definition of disease status was based on the longitudinal review of the clinical, imaging, and biochemical data (including Tg, TgAbs, and TSH) and any cytology/histology reports available. Disease status was assessed in conference by two experienced clinical thyroidologists (L.G., P.T.) for each visit. Patients were classified as having no evidence of structural disease (NESD) if there was no clinical, imaging, or cytological/histological evidence of disease at the time of the follow-up visit. Otherwise, patients were classified as having evidence of structural disease (ESD) and were further divided into patients with loco-regional (loc-ESD) or distant ESD (dist-ESD). When ESD was detected, further follow-up time was not considered for the study purpose.
Laboratory assessment
Our institutional laboratory provided all serum measurements. TSH was measured with chemiluminescence immunoassays (HYPERsensitive hTSH) on a UniCel DxI 800 automated platform (Beckman Coulter SA, Nyon, Switzerland); this HYPERsensitive hTSH assay is based on the Third International Standard (World Health Organization) for human TSH (IRP 81/565) having analytical sensitivity of 0.003 mIU/L, functional sensitivity of 0.0 mIU/L, and reference ranges between 0.40 and 4.00 mIU/L. Serum Tg and TgAbs were measured on the Kryptor® compact Plus instrument (BRAHMS Thermo Fisher Scientific, Henningsdorf, Germany). This method uses two fluorophores, Lumi4 and Cyanin5.5, which are detected separately or combined in the same antigen-antibody complex—a method known as time resolved amplified cryptate emission (TRACE). Serum hTg Kryptor® assay is calibrated against the BCR®457 international reference standard. The functional sensitivity (corresponding to inter-assay imprecision of 20% assessed in the appropriate matrix (human serum) over 6–12 months (to reflect the typical DTC interval) including at least two batches of reagents and/or calibrators) was estimated at 0.15 μg/L (Instructions For Use, ThermoFisher). The Kryptor® TgAbs assay is standardized against the 1st International Reference Preparation 65/93, and in-house limit of quantification (LoQ) was estimated at 33 IU/mL. Then serum with TgAb levels ≤33 IU/mL were considered negative (i.e. free from TgAbs interference in Tg measurement).
Definition of “de novo” TgAbs appearance
In DTC patients with negative TgAb levels detected at baseline, the elevation of serum TgAbs above 33 IU/in at least one scheduled follow-up visit was defined as de novo TgAbs appearance. In isolated TgAbs positivities confirmed by repeat analysis, the higher values were reported. Transient and sustained de novo TgAbs appearances were defined as elevations that disappeared over the follow-up visits and elevations that continued over every follow-up visit (with stable or even rising trends), respectively. TgAb peak levels (the highest positive levels achieved) were expressed as median and interquartile range (IQR). Time to TgAbs appearance indicated the time interval from preablation TgAbs assessment to the first positive TgAbs result. Time for TgAbs disappearance referred to transient de novo appearance cases, and meant the time taken to become negative (from the first new appearance).
Statistical analysis
Continuous variables were expressed as median and IQR and analyzed by non-parametric test (Mann–Whitney test). Categorical variables were presented as number (percentage) and compared by chi-square or Fisher testing. The Kaplan–Meier method was used to estimate the disease-free survival (DFS) of the two groups with persistently negative TgAbs or de novo TgAbs appearance. Survival curves compared by log-rank test and hazard ratio (HR, with a 95% confidence interval (CI)) were obtained. P < 0.05 was considered statistically significant. Statistical analysis was performed by GraphPad Prism, version 7 (GraphPad Software Inc., San Diego, CA, USA).
Results
Patient characteristics
We retrospectively collected the complete follow-up data of 145 consecutive patients with DTC diagnosed at the Competence Center for Thyroid Diseases, Ente Ospedaliero Cantonale, Bellinzona (Switzerland) between 2008 and 2018. After exclusion of 26 DTC patients with preablation positive TgAbs, our cohort study consisted of 119 patients with preablation negative TgAbs. The main features of the cohort study are shown in supplementary Table 1. Median age at diagnosis of DTC was 52 years (range, 44–62 years) with female/male ratio ∼2:1. Papillary thyroid carcinoma (PTC) was the main histological subtype—almost four times more common than FTC—and the percentage of patients who were classified as low, intermediate, and high risk was 51.3, 45.4, and 3.3, respectively. However, we found only eight cases of disease recurrence (with six cases of dist-ESD); 93.3% of patients did not develop recurrence and no disease-related death was registered among the 119 DTC patients.
During a median follow-up of 36 months (IQR 12–72 months), we detected 14 cases of de novo TgAbs appearance (11.7% of the cohort study) while the majority (105 patients) persistently remained TgAbs-negative. As shown in Table 2, the 14 patients were followed-up for a median of 78 months (range, 57–99 months). At TgAbs appearance, serum Tg was undetectable in 11 patients, while detectable values were present in 3 patients.
Main characteristics of patients with de novo TgAbs appearance (a) and comparison of transient versus sustained de novo TgAbs appearance behavior (b).
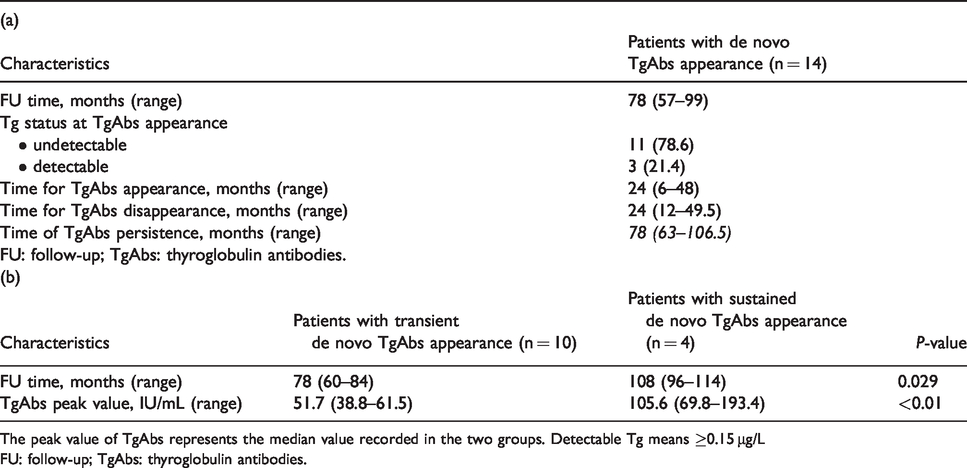
The peak value of TgAbs represents the median value recorded in the two groups. Detectable Tg means ≥0.15 μg/L
FU: follow-up; TgAbs: thyroglobulin antibodies.
According to dynamic changes, 10 patients showed a transient de novo TgAbs appearance (having a median follow-up time of 78 months), whereas sustained de novo TgAbs positivity was maintained by 4 patients during follow-up (median follow-up of 108 months, P = 0.029). Moreover, TgAbs peak value was higher among patients with sustained de novo TgAbs compared to patients with transient de novo TgAbs (105.6 IU/mL vs. 51.7 IU/mL, P < 0.01). Both median time for TgAbs appearance and TgAbs disappearance (i.e. transient de novo TgAbs appearance) was 24 months. However, for patients with sustained de novo TgAbs behavior, the median time of TgAbs persistence was 78 months. Figure 1 illustrates the dynamic changes of TgAb levels in patients with sustained and transient de novo TgAbs appearance.
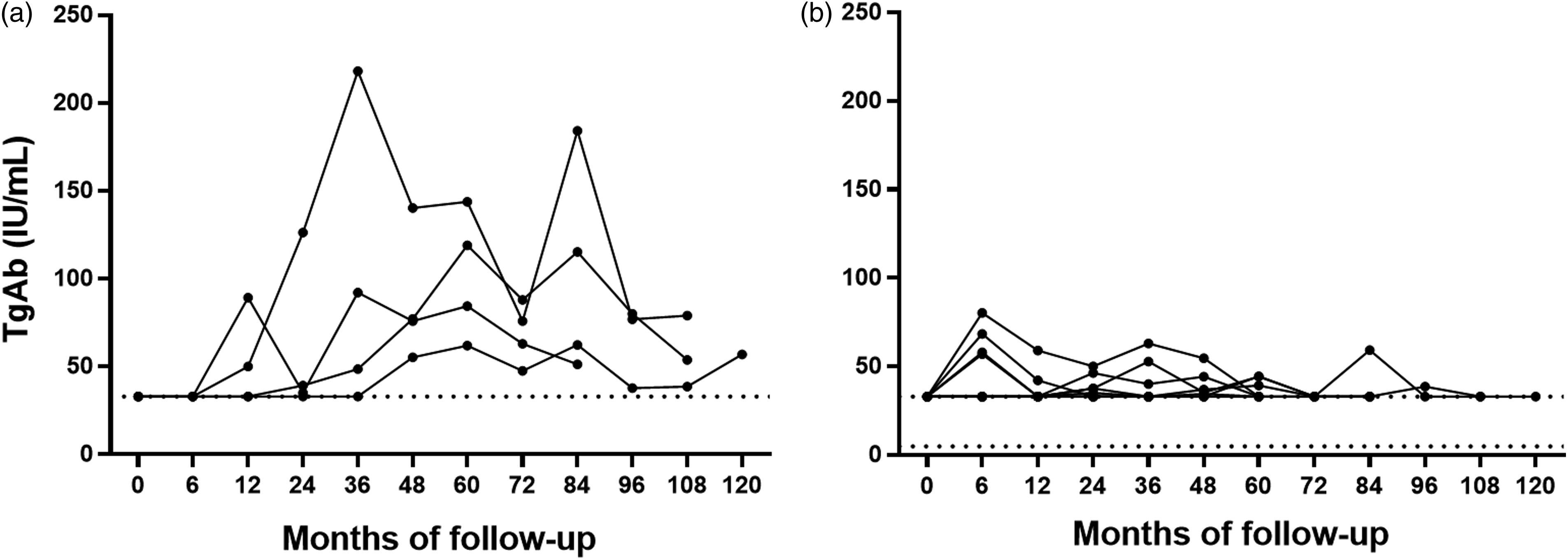
Trend lines of TgAbs in patients with sustained (a) and transient (b) de novo TgAbs appearance.
As summarized in Table 3, no demographic, clinical, and histopathological differences were observed between persistently TgAb-negative patients and those with de novo TgAbs appearance (except for median follow-up time, which was longer for patients who developed de novo TgAbs (78 vs. 36 months, P = 0.0007)). Likewise, the number of settings potentially linked to de novo TgAbs appearance (i.e. number of RAI treatments, neck surgical reinterventions, FNAC, and the presence of thyroid remnant at RxWBS) was not statistically different among the two groups. Also, no differences in the overall prevalence of both NESD and ESD (i.e. recurrence, as loc-ESD and dist-ESD) were found between patients with persistently negative TgAbs and patients with de novo TgAbs appearance (P = 0.21).
Comparison of the main characteristics of the cohort study according to the development of new positivity of TgAbs during follow-up time.
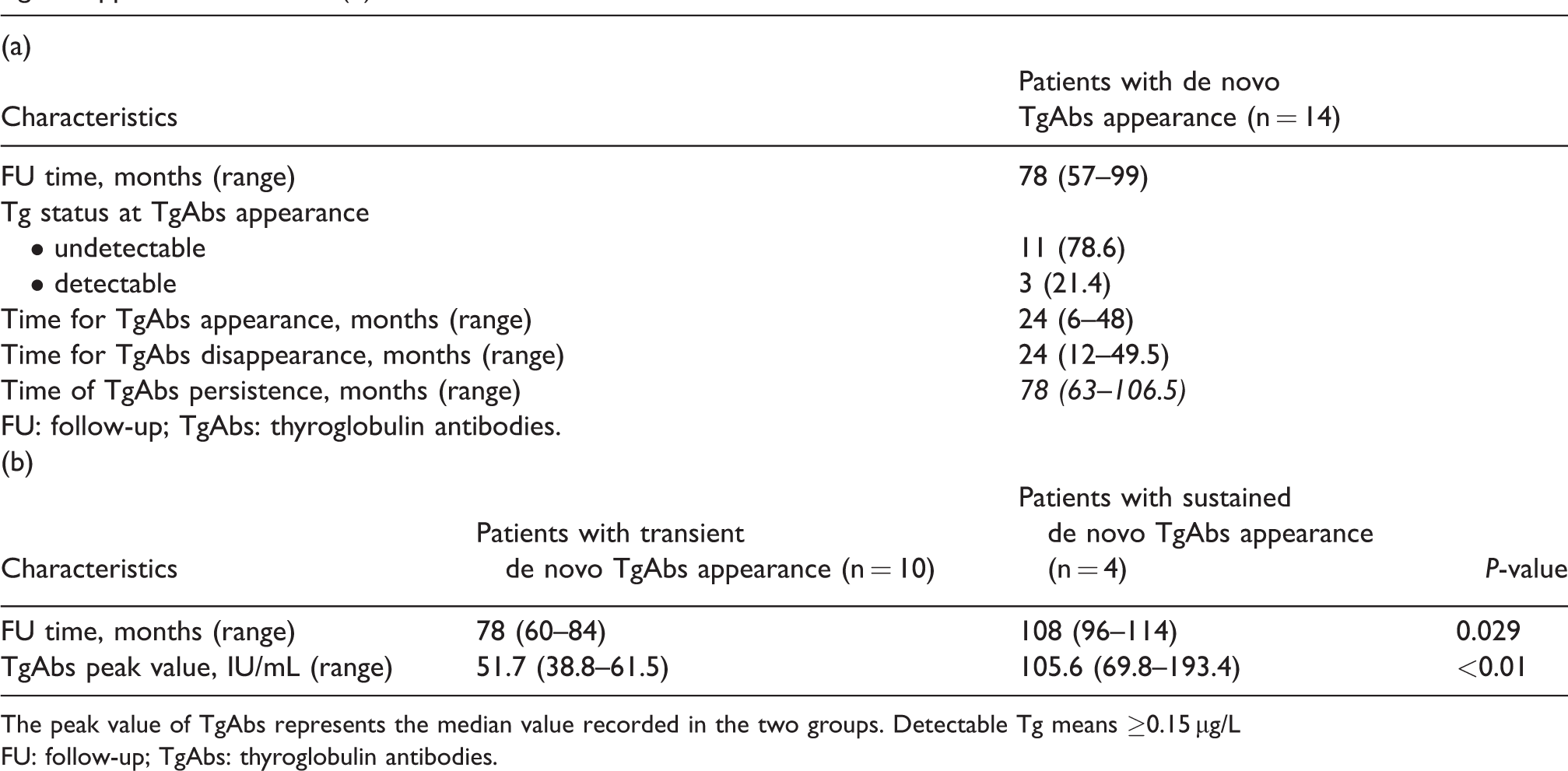
Thyroglobulin and TSH refer to overall basal values during the FU.
ATA: American Thyroid Association; ESD: evidence of structural disease; dist-ESD: distant ESD; loc-ESD: local ESD; FNAC: fine-needle aspiration cytology; FTC: follicular thyroid carcinoma; FU: follow-up; NESD: no evidence of structural disease; PTC: papillary thyroid carcinoma; RAI: radioiodine; RxWBS: post-treatment whole body scan; TgAbs: thyroglobulin antibodies.
Figure 2 shows that de novo TgAbs appearance was not associated with shorter DFS, compared to persistently negative TgAbs status (P = 0.67, HR 0.92, 95% CI 0.17, 4.97). In detail (Figure 3), we found 2 cases of ESD (dist-ESD) among the 14 patients with de novo TgAbs appearance (1 with transient and 1 with sustained de novo TgAbs appearance). In both cases, serum Tg at the time of the de novo TgAbs positivity and at the time of recurrence detection was already detectable. A descriptive case presentations is summarized in supplementary Table 4 (parts (a) and (b)).
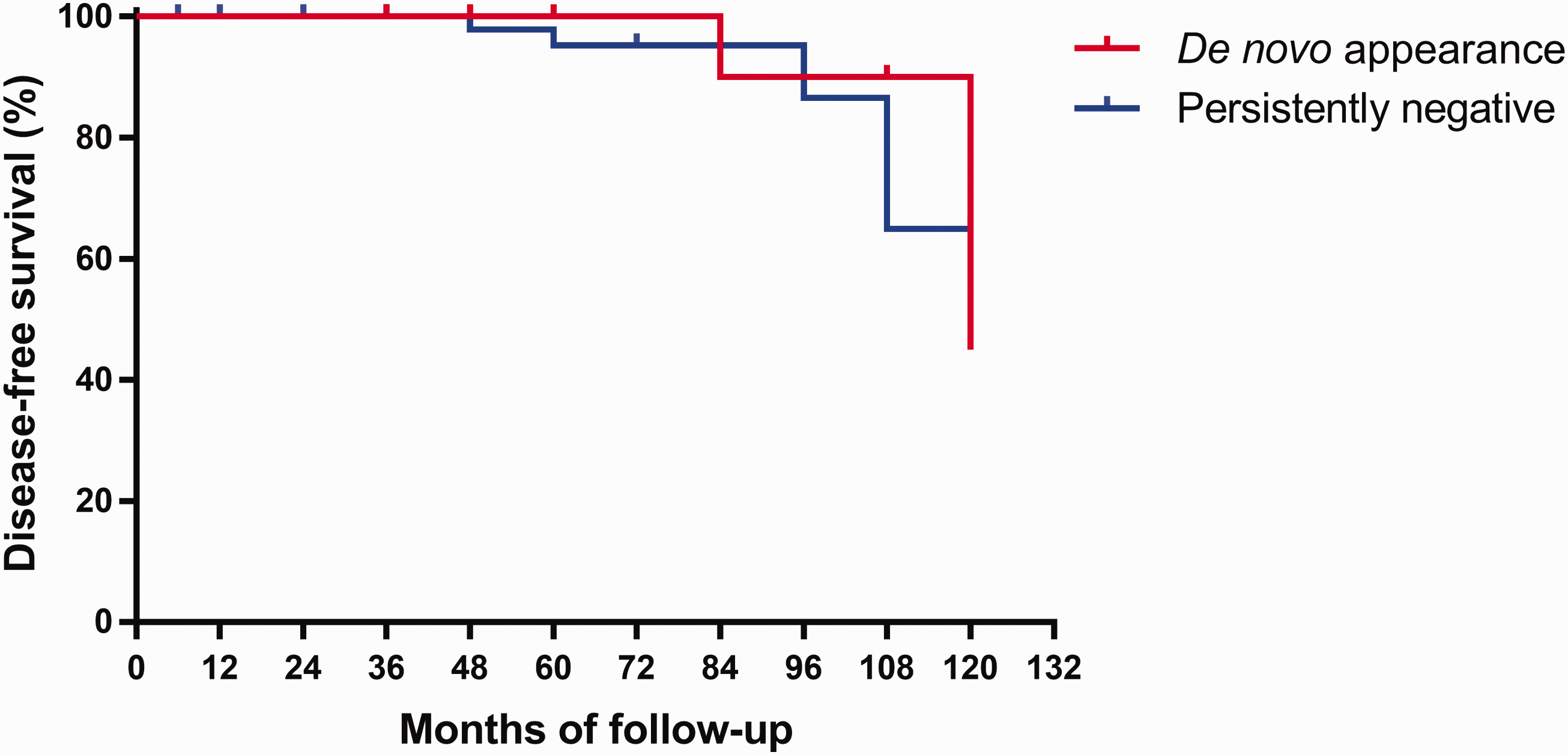
Kaplan–Meier curve of disease-free survival according to de novo TgAbs appearance or persistently negative TgAbs during follow-up time.
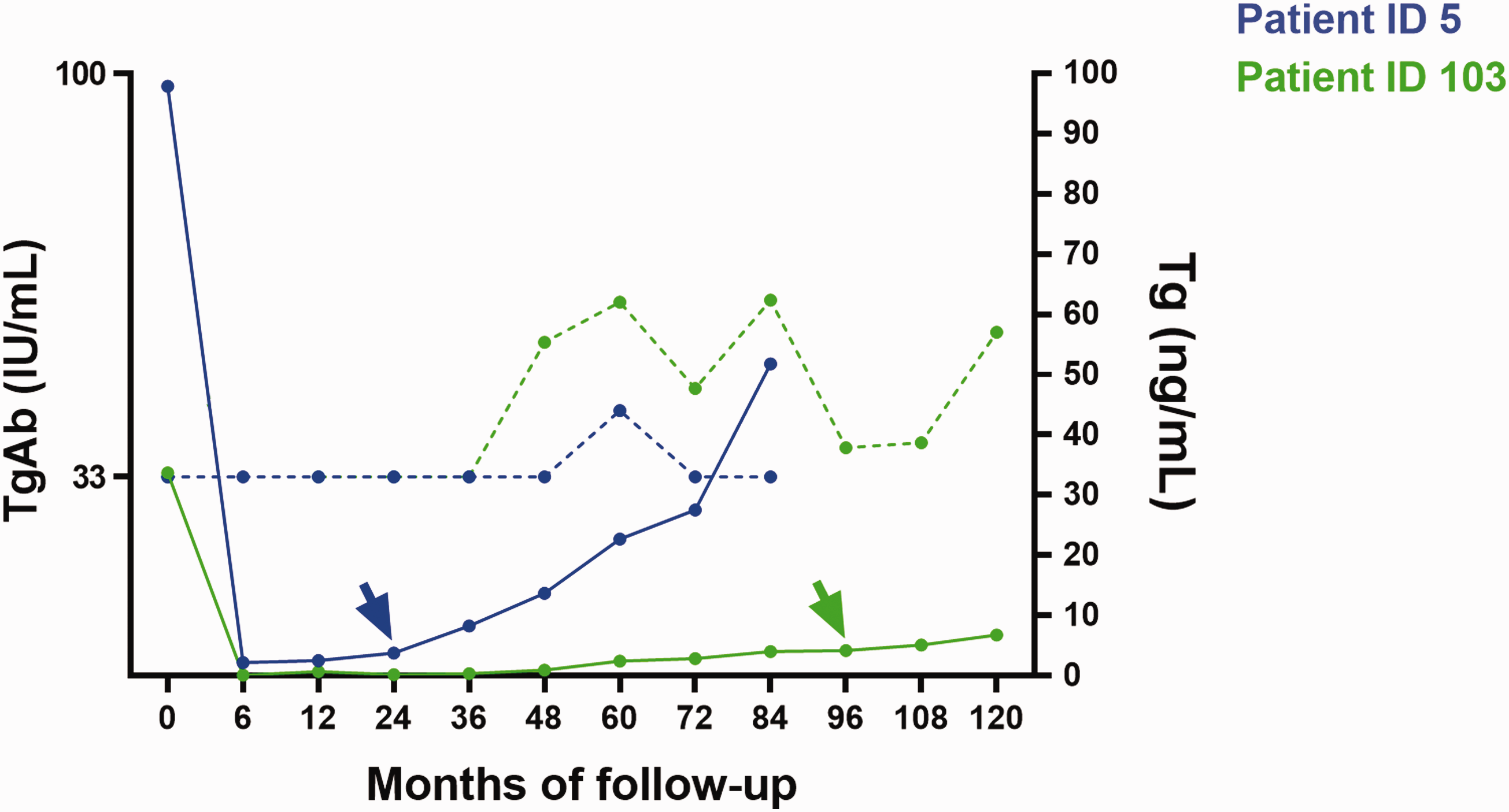
Trend lines of TgAbs and Tg values of the two patients (IDs 5, 103) with de novo TgAbs appearance who developed distant-ESD during follow-up. Continuous line represents Tg values; dashed line represents TgAb values; arrows indicate the time of ESD detection.
Discussion
The “new appearance” of TgAbs in a previously TgAb-negative DTC patient was associated with an increased risk of disease recurrence. 2 However, transient TgAbs appearance is mostly related to acute damage of thyroid tissue and should not be regarded as a herald of structural disease. 3 , 7 , 9 In contrast, immune system may react against malignant thyroid tissue with a sustained de novo TgAbs formation signaling evidence of structural disease,15–19, 21 as reported in anecdotal cases summarized in Table 5.
De novo TgAbs events to date reported in the literature.
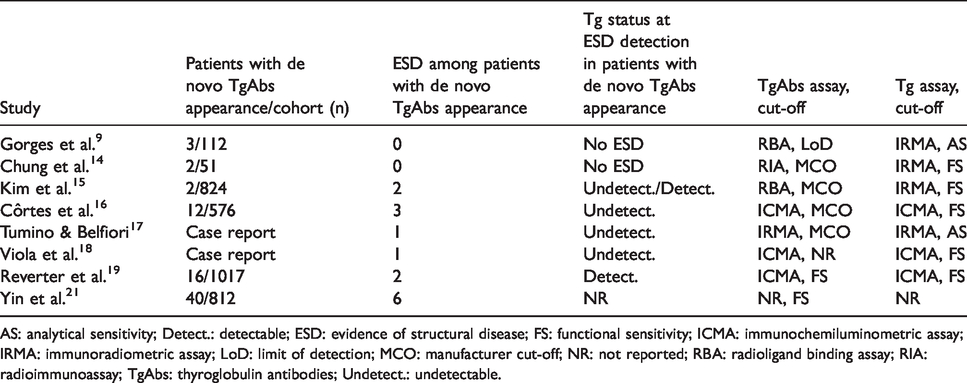
AS: analytical sensitivity; Detect.: detectable; ESD: evidence of structural disease; FS: functional sensitivity; ICMA: immunochemiluminometric assay; IRMA: immunoradiometric assay; LoD: limit of detection; MCO: manufacturer cut-off; NR: not reported; RBA: radioligand binding assay; RIA: radioimmunoassay; TgAbs: thyroglobulin antibodies; Undetect.: undetectable.
To the best of our knowledge, this is the second specifically designed study that has analyzed the prevalence of de novo TgAbs appearance and its clinical implications (the paper by Yin et al. 21 was published during the revisions of our study). Compared to the study by Yin et al., 21 we found a similar prevalence of de novo TgAbs (3.3% in our study considering only the sustained de novo TgAbs vs. 5%). We came to the same conclusion that de novo TgAbs development does not seem to be significantly associated with DTC structural recurrence, with respect to persistently TgAb-negative patients. 21
Our cohort of 119 TgAb-negative DTC patients before ablation were followed-up for a median period of 3 years. Among all cases, 12% developed de novo TgAbs appearance. This subgroup was followed-up for a median time of 6.5 years, and in most cases TgAbs developed ∼ 2 years after RAI ablation. In ∼ 70% of cases TgAbs spontaneously disappeared within ∼ 2 years, whereas TgAbs remained detectable for ≥6 years in remaining cases. Sustained de novo TgAbs appearance was characterized by higher peak values as compared with transient de novo appearing TgAbs. Since TgAb measurements were within 6 months after surgery, RAI, and FNAC were not extracted (to minimize the effects of these settings on TgAbs levels 3 , 7 ), transient de novo TgAb positivities likely reflect spontaneous TgAb fluctuations and interassay variability (i.e. IDs 5, 29, 30, 31, 41, 101). 7 , 16 Although at least 6 months from RAI had elapsed before the TgAb measurements were performed, RAI treatment could have contributed to transient de novo TgAb positivity in three cases (i.e. IDs 2, 27, 95). We did not investigate immunotherapies or vaccines3 in the cohort, and we cannot exclude either of them as the cause of de novo TgAb appearances in some of our patients. One hypothetical reason of the (re)appearance of TgAbs could be the persistence of thyroid autoimmunity after therapy for DTC. In other words, thyroglobulin antibody-positive DTC patients could first lose serum positivity after therapy for cancer, then they could develop new TgAbs in the presence of Tg stimulus. Thus, one could speculate that in a patient (ID 64), serum TgAbs existed before surgery and transitorily (re)appeared after primary therapy in the presence of Tg stimulus. Nevertheless, even if the pre-surgery autoimmune profile was lacking, for this patient (ID 64) RxWBS showed no thyroid remnants and an adequate time from RAI elapsed before the measurement of TgAbs. In this setting, the assessment of thyroid peroxidase antibodies (TPOAbs) could be informative and added to TgAbs to explore the existence of thyroid autoimmunity. However, TPOAbs are not among the current tools for DTC follow-up. 2
One among four patients with sustained de novo TgAbs appearance (ID 103) had ESD. Notably, serum Tg was already detectable (0.98 μg/L) at the time of de novo TgAbs appearance (48-month follow-up visit), and continued to increase until lung metastases were detected at 18F-FDG PET/CT (96-month follow-up visit, Tg 4.23 μg/L). The remaining patients (IDs 12, 58, 82) had stable TgAbs trends with concurrent undetectable Tg values and remained disease-free during follow-up. In the latter three patients, repeat scans could eventually remove the doubts about structural disease.
In summary, our results showed that the appearance of de novo TgAbs is common among DTC patients. Transient de novo TgAbs appearance accounts for most cases and has no apparent clinical value; in contrast, sustained de novo TgAbs appearance is rare and not related to a reduction in DFS compared with persistently TgAb-negative DTC patients. Notably, a concurrent undetectable highly sensitive Tg measurement likely excluded ESD, while detectable Tg levels were noted in one patient carrying ESD (among patients with sustained de novo TgAbs). This is well in line with recent data; the adoption of highly sensitive Tg assays can help to reliably monitor TgAb-positive DTC patients and to predict their outcome. 22 , 23
A major strength of the current study is that for all patients the consecutive Tg and TgAb measurements were performed in the same laboratory using the same assay at each follow-up visit as recommended for longitudinal consistency of clinical care. 3 Additionally, dynamic changes of TgAbs and their clinical meaning were evaluated over a long follow-up period (median 6.5 years). However, there were some limitations. First, a selection bias cannot be excluded considering the retrospective analysis of data. However, DTC patients were consecutively diagnosed and monitored by the same physicians (L.G., P.T.) making a significant impact on our results unlikely. Second, a relatively small number of DTC patients were reviewed to investigate de novo TgAbs appearance, but this was also related to our highly selective inclusion criteria. Third, the consequence of the small number of DTC patients with de novo TgAbs, and the distinction between transient and sustained de novo TgAbs is not definite since it may be due to the length of the follow-up. Fourth, because of the well-known interassay variability between different Tg and, especially, TgAbs assays, 3 , 12 our results primarily apply to this specific methodological context.
In conclusion, considering the limitations of the current study along with the presence in the literature of anecdotal cases of de novo TgAbs associated with poor prognosis and undetectable Tg values,15–18 it would be prudent to continue measuring both variables (Tg and TgAbs) in each individual sample during the follow-up of patients with differentiated thyroid cancer, as recommended by clinical position papers and guidelines. 2 , 3 However, our preliminary findings would suggest that there is no need to adopt an aggressive diagnostic strategy when encountering de novo TgAbs along with undetectable high-sensitivity thyroglobulin, except when the de novo TgAbs trend is progressively increasing.15–18
Supplemental Material
sj-pdf-1-jbm-10.1177_1724600820931517 - Supplemental material for Significance of “de novo” appearance of thyroglobulin antibodies in patients with differentiated thyroid cancer
Supplemental material, sj-pdf-1-jbm-10.1177_1724600820931517 for Significance of “de novo” appearance of thyroglobulin antibodies in patients with differentiated thyroid cancer by Lorenzo Scappaticcio, Pierpaolo Trimboli, Frederik A. Verburg and Luca Giovanella in The International Journal of Biological Markers
Footnotes
Acknowledgments
L.S. contributed to this paper as the recipient of an internal grant (“programma STAR”) by the University of Federico II (Naples, Italy) and as a Fellow in the Clinic for Nuclear Medicine and Competence Centre for Thyroid Disease, Ente Ospedaliero Cantonale (Bellinzona, Switzerland).
Author Contributions
Conceptualization: L.G.; data curation: L.G., L.S.; formal analysis: L.G., L.S., P.T.; investigation: L.G., L.S.; methodology: L.S., P.T.; project administration: L.G.; resources: L.G.; supervision: L.G., F.A.V.; writing—original draft: L.S.; and writing—review and editing: L.G., L.S., P.T., and F.A.V.
Declaration of conflicting interest
L.G. is a member of Roche Diagnostics Advisory Board and has received research grants from Roche Diagnostics and speaker honoraria from Roche Diagnostics, BRAHMS GmbH, EISAI, Siemens Healthcare, and Sanofi-Genzyme. F.A.V. is a consultant to EISAI, Jubilant Draximage and Genzyme, and has received speaker honoraria from Genzyme. L.S. and P.T. have nothing to disclose related to this work.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
