Abstract
Keywords
Introduction
There were about 567,233 new cases of thyroid cancer in 2018 worldwide, with 41,071 deaths. 1 Papillary thyroid cancer (PTC) is a carcinoma that arises from thyroid follicular cells, is classified as differentiated thyroid cancer, and accounts for about 80% of all thyroid cancers. 2 PTC can be sporadic or genetic and can be subtyped as well-differentiated or poorly differentiated, with each subtype encompassing multiple variants (some of which are more aggressive than others). 2 Risk factors for PTC include female sex, prior exposure to radiation in childhood or adolescence, history of iodine deficiency, excess intake of iodine, female sex, and family history of thyroid cancer.3,4 The management of PTC is comprehensive and includes surgery, chemotherapy, radiation therapy, and targeted therapy.5–7 The prognosis is usually good, with a 15-year overall survival of 91% and cancer-specific survival of 99%.
Physical and ultrasound examinations for cervical adenopathy are part of the routine staging for potential PTC.2,5,8 Cervical lymph node (LN) metastases are associated with reduced survival in patients of all ages and perhaps more so for patients >45 to 50 years.9,10 The 2015 American Thyroid Association guidelines highlight that LN metastases increase the risk of PTC recurrence when there are >5 positive LNs, clinically palpable LNs, and LNs >3 cm.5 Therefore, detecting lateral LN metastasis can influence the management of patients with thyroid cancer. Still, some metastases are occult in the early stage of the disease, and predicting their presence is important for proper patient management.5–7
A fine-needle aspiration (FNA) should be performed in the presence of abnormal or suspicious cervical LN.5,8 Some studies reported thyroglobulin (Tg) detection in FNA of cervical LN (LN-FNA-Tg) to be diagnostic of thyroid carcinoma metastasis.5,11–13 Still, some issues remain, including a lack of standardized methods, the wide variability of cutoff values, and interpreting these cutoff values in the clinical context.14,15 Nevertheless, the studies showed that LN-FNA-Tg has a high sensitivity for cervical LN metastasis but poor specificity.5,13–15 In addition, a retained thyroid gland can decrease the diagnostic accuracy of LN-FNA-Tg16–19 and anti-Tg antibodies (TgAb) can also influence the results by underestimating the association.20,21 Still, it should be noted that cytological evaluation remains the gold standard, and only when indeterminate cases or acellular aspirates are encountered, can LN-FNA-Tg be a confirmatory test.
Therefore, this study aimed to analyze whether LN-FNA-Tg and LN-FNA-TgAb can predict LN metastasis and obtain the cutoff value for the study population. The results could help guide the physicians to make an accurate evaluation of the LN status in PTC.
Materials and Methods
Study Design and Participants
The patients admitted to the Department of General Surgery, Endocrinology, and Otolaryngology of our hospital from January 2020 to March 2021 were prospectively enrolled. This study was approved by the Medical Ethics Committee of our hospital (20190019). All enrolled patients signed the informed consent for the study.
The adult patients (≥18 years of age) who were pathologically diagnosed with PTC who underwent thyroid cancer surgery and lateral cervical LN dissection in our hospital and the patients who underwent thyroid cancer surgery in other hospitals and then underwent lateral cervical LN dissection in our hospital were enrolled. Most patients were enrolled before surgery, and a very small percentage of patients were enrolled after LN abnormalities were detected during postoperative review in our hospital. Patients who underwent index thyroidectomy in other hospitals were included as long as the investigation for the LNs was made at the authors’ hospital. The exclusion criteria were (1) postoperative pathology showed heterogeneous tumor comprising PTC with follicular carcinoma, poorly differentiated carcinoma, medullary carcinoma, undifferentiated carcinoma, etc; (2) history of cervical LN tuberculosis or other cervical tumors; (3) coagulation dysfunction; or (4) history of radiotherapy, chemotherapy, 131I ablation, and other tumor-related treatments before surgery.
Ultrasound-Guided FNA
The ultrasound features of suspect enlarged LNs were (1) morphological changes (round, quasi-round, or irregular [longitudinal/transverse diameter ratio <1.5]), (2) centripetal thickening of LN cortex or decreased echo of cortex and medulla, (3) hyperechoic masses or mutual fusion were found in LNs, (4) abundant peripheral and internal blood supply or disordered blood flow signals, (5) absence of hilar structure, (6) minimum diameter ≥6 mm, (7) multiple fine calcifications or cluster calcification, and (8) partial liquefaction of LNs. The suspect metastatic LNs of PTC in the lateral neck were detected under the guidance of color ultrasound (GE LOGIQ E9, GE Healthcare; Aloka α5, Hitachi; Kaili S60/China Kaili Medical Co., Ltd), and FNA was performed (puncture needles of 21-25 G with 5 mL syringes). A cytological examination was performed on all FNAs. During the FNA, each LN was aspirated at least 4 times to absorb sufficient puncture material. For liquefied cystic LNs, the original puncture fluid was used. For unliquefied solid LNs, the needle and syringe were cleaned with 1.0 mL normal saline to make an eluent of about 1.0 mL volume, which was stored in an eppendorf tube to detect Tg and TgAb. At the same time, 2 mL of venous blood was taken to detect Tg and TgAb. The locations of all LNs were marked on the skin, and the anatomical region of cervical LNs (I-VII areas) was recorded. The size was measured and compared with that of the intraoperative LNs for confirmation. The punctured LNs were separately submitted for pathological examination.
All FNAs were performed by the same chief physician experienced in color ultrasound and cytology puncture. All LN surgeries were performed by the same chief surgeon with rich experience in thyroid cancer surgery and neck LN dissection. Pathological examinations were all performed by the same associate chief pathologist. All patients were diagnosed according to the histological results. The participants were grouped according to the LN results.
Biochemistry
The LN-FNA eluate and blood Tg and TgAb were detected by electrochemiluminescence immunoassay. The detection concentration range of TgAb and Tg was 10 to 4000 IU/mL and 0.04 to 500 ng/mL, respectively. Specimens exceeding the range were diluted with normal saline 30 to 50 or 100 times. Tg detection reagents were calibrated with the CRM-457 standard (Roche Diagnostics) to reduce bias between tests. The functional sensitivity of the Tg detection reagent was at least 1 ng/mL to ensure that a very small amount of Tg could be detected. All patients should be tested in the same laboratory using the same methods.
Statistical Analysis
The Shapiro-Wilk method was used to test the normality of continuous data. Measurement data conforming to the normal distribution were presented as mean ± standard deviation, and Student's t-test was used for statistical analysis. Nonnormally distributed continuous data were presented as median (25th percentile, 75th percentile), and the Mann-Whitney U test was used for statistical analysis. Categorical data were expressed as n (%) and analyzed using the chi-square test or Fisher's exact probability method. Statistical analysis was performed using SPSS 22.0 (IBM, Armonk, NY, USA). The receiver operating characteristic (ROC) curve was used to test the diagnostic efficiency. The point with the highest Youden’s index was selected as the critical value for diagnosis. Sensitivity, specificity, positive predictive value, negative predictive value, accuracy, and area under the curve (AUC) were calculated. The ROC analysis was performed using MedCalc 19.5.6 (MedCalc Software bvba, Ostend, Belgium). We have deidentified all patient details.
Ethics Statement and Consent to Participate
This study was approved by the Medical Ethics Committee of Panzhihua Central Hospital (20190019) and all methods were carried out in accordance with relevant guidelines and regulations. All enrolled patients signed the informed consent for the study.
Results
Characteristics of the Participants
Table 1 presents the participants’ characteristics. There were 29 participants in the LN metastasis group and 42 in the nonmetastasis group. Compared with the nonmetastasis group, the participants in the metastasis group were younger (median: 39 vs 44.5 years, P = .048), had a lower frequency of hypoechoic LN (50.0% vs 73.2%, P = .003), a higher frequency of cystic solid LN (21.0% vs 4.1%, P = .001), and larger LNs (median: 9 vs 7 mm, P < .001).
Characteristics of the Patients.

Abbreviations: LN-FNA-Tg, thyroglobulin washout of fine-needle aspiration of a lymph node; LN-FNA-TgAb, antithyroglobulin antibody washout of fine-needle aspiration of a lymph node; Tg, thyroglobulin; TgAb, antithyroglobulin antibody.
*The P-value was obtained by Fisher's exact probability method.
Diagnostic Evaluation of LN-FNA-Tg and LN-FNA-TgAb
Compared with the nonmetastasis group, the participants in the metastasis group had higher LN-FNA-Tg (median: 1897 vs 7.74 ng/mL, P < .001), higher LN-FNA-TgAb (median: 15.65 vs 8.21 IU/mL, P < .001), and higher serum Tg (median: 25.4 vs 18.81 ng/mL); there were no differences in serum TgAb (median: 26.6 vs 28.6 IU/mL, P = .477; Table 1).
The best accuracy (87.5%) was observed with LN-FNA-Tg of >227.1 ng/mL, resulting in an AUC of 0.927, 84.5% sensitivity, and 89.5% specificity (Table 2). LN-FNA-TgAb >10.85 IS/mL had an accuracy of 79.6%, sensitivity 64.8%, and specificity 89.5%. Serum Tg and TgAb had the lowest accuracy, with 64.2% and 57.4%, respectively, with a sensitivity of 53.5% and 67.6%, and specificity of 71.4% and 50.5% (Table 2 and Figure 1).

ROC curve to determine the cutoff value of LN-FNA-Tg/LN-FNA-TgAb/serum Tg/serum TgAb.
Diagnostic Value Evaluation.

Abbreviations: AUC, area under the curve; LN-FNA-Tg, thyroglobulin washout of fine-needle aspiration of a lymph node; LN-FNA-TgAb, antithyroglobulin antibody washout of fine-needle aspiration of a lymph node; Tg, thyroglobulin; TgAb, antithyroglobulin antibody.
Subgroup Analysis
Table 3 presents the characteristics of the participants according to the first-operation (index thyroidectomy performed at the authors’hospital) versus postoperative suspicion (index surgery at another hospital and postoperative LN investigation at the authors’ hospital). Compared with the postoperative group, the LNs in the first-operation subgroup had a higher frequency of envelope invasion (70.7% vs 35.7%, P = .017), but the LNs were smaller (median: 7 vs 9 mm, P = .010). The serum TgAB levels were higher in the first-operation group (median: 27 vs 18.2 IU/mL, P = .001), while there were no differences in LN-FNA-Tg, LN-FNA-TgAb, and serum Tg levels (all P > .05).
Subgroup Analyses.
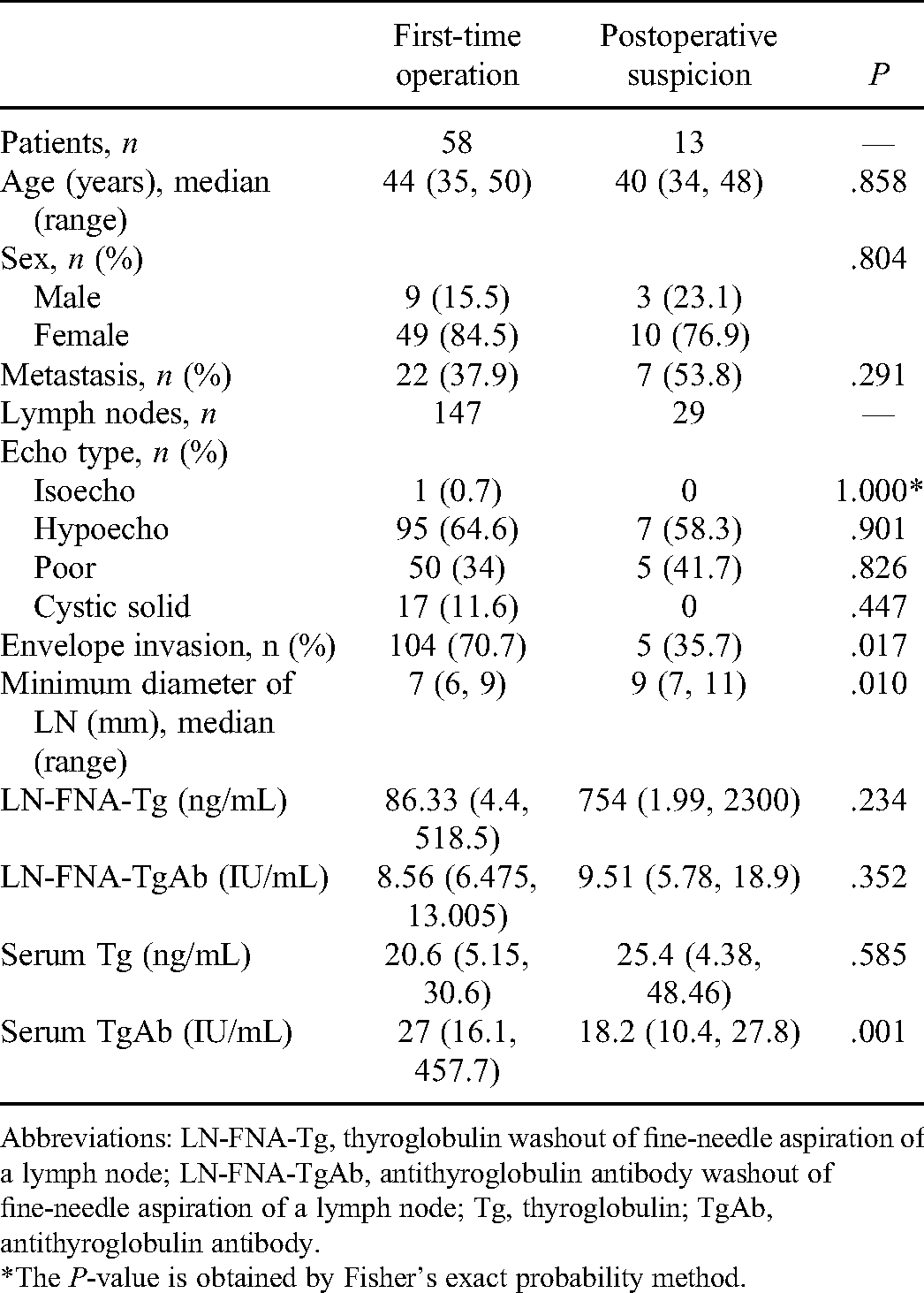
Abbreviations: LN-FNA-Tg, thyroglobulin washout of fine-needle aspiration of a lymph node; LN-FNA-TgAb, antithyroglobulin antibody washout of fine-needle aspiration of a lymph node; Tg, thyroglobulin; TgAb, antithyroglobulin antibody.
*The P-value is obtained by Fisher's exact probability method.
In the first-operation group, LN-FNA-Tg >227.1 ng/mL and LN-FNA-TgAb >10.85 IU/mL had the highest accuracy (85.7% and 80.3%, respectively), with an AUC of 0.902 and 0.763, sensitivity 80.8% and 63.5%, and specificity 88.4% and 89.5%. On the other hand, serum Tg and TgAb had low accuracy (Table 4 and Figure 2).

ROC curve: the cutoff value of Tg and TgAb in the first-time operation subgroup.
Diagnostic Value Evaluation in the First-Time Operation Subgroup.

Abbreviations: AUC, area under the curve; LN-FNA-Tg, thyroglobulin washout of fine-needle aspiration of a lymph node; LN-FNA-TgAb, antithyroglobulin antibody washout of fine-needle aspiration of a lymph node; Tg, thyroglobulin; TgAb, antithyroglobulin antibody.
In the postoperative group, LN-FNA-Tg >6.8 ng/mL had an AUC of 1.00, an accuracy of 100%, sensitivity of 100%, and specificity of 100%. LN-FNA-TgAb >8.91 IU/mL had an accuracy of 79.3%, with an AUC of 0.774, sensitivity 79.0%, and specificity 80.0%. Again, serum Tg and TgAb had low accuracy (Table 5 and Figure 3).

ROC curve: the cutoff value of Tg/TgAb in the postoperative suspicion subgroup.
Diagnostic Value Evaluation in the Postoperative Suspicion Subgroup.

Abbreviations: AUC, area under the curve; LN-FNA-Tg, thyroglobulin washout of fine-needle aspiration of a lymph node; LN-FNA-TgAb, antithyroglobulin antibody washout of fine-needle aspiration of a lymph node; Tg, thyroglobulin; TgAb, antithyroglobulin antibody.
Discussion
Most of the current research results showed that the FNA aspirate examination had high sensitivity in diagnosing lateral cervical LN metastasis.5,13–15 Still, the specificity is generally poor, and there are many influencing factors. Therefore, this study aimed to analyze whether LN-FNA-Tg and LN-FNA-TgAb can predict LN metastasis. The results indicate that LN-FNA-Tg has high accuracy, sensitivity, and specificity for detecting cervical LN in patients with PTC.
The current guidelines support the use of LN-FNA-Tg to diagnose metastatic cervical LN, especially in patients who underwent total thyroidectomy or in the absence of TgAb.5,13 Still, the diagnostic performance of LN-FNA-Tg in the general population of patients with PTC remains controversial.5,13–15 Of note, the use of LN-FNA-Tg is highly dependent upon the detection of suspicious LN at an ultrasound, which in itself has a sensitivity of 65.0% to 90.3% and specificity of 80.9% to 82.0%22,23 and is highly dependent upon the experience of the radiologists. Therefore, a prerequisite for the diagnostic value of LN-FNA-Tg is the quality of the ultrasound examination, highlighting the need for a careful examination of the neck. Furthermore, the cytological examination of the LN-FNA is associated with a 6% to 50% false-negative rate, especially in small LNs and cystic LNs. 24 Previous studies showed that LN-FNA-Tg measurement is more accurate than cytological examination,11,25–30 but one study reported that LN-FNA-Tg and cytological examination had a similar value, 31 again highlighting the role of the radiologist and pathologist. Still, LN-FNA-Tg could be of particular value when the cytological examination is inconclusive.11,32,33
In the present study, LN-FNA-Tg showed 84.5% sensitivity and 89.5% specificity, which is slightly lower than a recent study 31 but using a different cutoff level. Indeed, the most optimal cutoff level for LN-FNA-Tg is a matter of debate. A meta-analysis suggested that the cutoff value of LN-FNA-Tg should be within 0.2 to 50 ng/mL, 26 which is a wide range, with the lowest value (0.2 ng/mL) below the detection level of many clinical assays (1 ng/mL). In that previous meta-analysis, the vast heterogeneity among the patient populations probably explains this wide range. In the present study, the cutoff level with the highest Youden’s index was 227.1 ng/mL, which is much higher than the range reported in the meta-analysis 26 and a previous study (2.1 ng/mL, but that previous study excluded extreme values). 31 Still, this high cutoff value resulted in an AUC of 0.927 and 87.5% accuracy.
Some studies reported that the presence of the thyroid gland could influence the reliability of the LN-FNA-Tg measurement because of peripheral blood contamination of the specimen, 28 but there are discrepancies among the studies.12,16–19,25,29,34–36 Still, an author suggested that blood contamination should be negligible in properly performed FNA. 12 In the present study, the predictive value of LN-FNA-Tg was similar between patients operated on for the first time and those with suspicious LNs after surgery. This is supported by previous studies that showed that the presence of the thyroid gland does not compromise LN-FNA-Tg measurement.31,34–36
Some studies suggested that TgAb could influence the value of LN-FNA-Tg for metastasis.20,21 Still, two studies showed that the value of LN-FNA-Tg was not affected by the LN-FNA-TgAb.11,31 In addition, it was suggested that serum TgAb did not influence LN-FNA-Tg because of no direct contact between serum and the LN content.16,37,38 In the present study, LN-FNA-TgAb also had a predictive value for cervical LN metastasis, but its value was lower than LN-FNA-Tg.
Serum Tg data are already available from the literature.1–6 In recent years, many studies have found that tumor size, characteristics of LN metastasis, degree of vascular invasion, and molecular pathological characteristics are important factors in predicting differentiated thyroid carcinoma recurrence.7–10 Therefore, the 2015 guidelines revised and supplemented the factors that affect the risk stratification of recurrence. 11 The number of metastatic LNs, their diameter, and extranodal invasion were included in the recurrence risk stratification system. In the present study, we wanted to clarify the view that LN metastasis in the lateral neck is associated with the ultrasonographic characteristics of the thyroid, including tumor size, LN metastasis, degree of vascular invasion, and whether there is a correlation between the integrity of the thyroid capsule. In particular, determining the number of LN metastasis cannot be determined only by the level of serum TgAb and Tg. Therefore, we believe that the determination of Tg/TgAb in the FNA eluent of each LN is a feasible method to determine whether the LN is involved.
This study has limitations. The sample size was small and from a single institution. Only suspicious LNs were sampled, and the contribution of benign LNs to the diagnostic accuracy of LN-FNA-Tg might be underestimated. No power calculation was done for the estimation of the sample size. Previous studies examined the diagnostic or predictive value for LN-FNA-Tg, and each study reported a different cutoff point based on their specific patient sample,5, 13–15 as did the present study. The cutoff point for LN-FNA-Tg appears to vary among studies and, for now, lacks standardization and generalizability. Multicenter studies should be conducted to determine the diagnostic value of LN-FNA-Tg for cervical LN metastasis and a cutoff point that could be used in a more general manner.
This study has some strengths. It was a prospective study that ensured that all examinations and surgery were performed by the same physicians. In addition, the LNs sampled with FNA were marked and sent separately for the pathological examination. The novelty of the present study is that the diagnostic ability of LN-FNA-Tg does not appear to be affected by whether thyroidectomy has been performed or not. In addition, most previous studies examined serum TgAb levels, while the present study examined LN-FNA-Tg.
Conclusion
LN-FNA-Tg has high accuracy, sensitivity, and specificity for detecting cervical LN in patients with PTC. The presence or absence of the thyroid gland does not significantly affect the accuracy of LN-FNA-Tg.
Footnotes
Abbreviations
Acknowledgments
Authors' Contributions
XW contributed to conceptualization, methodology, formal analysis, writing original draft preparation, writing review and editing, project administration, and funding acquisition. YL was involved in the investigation and data curation. KL, YY, and PL were involved in the investigation. JL was involved in visualization and SK in data curation. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study was funded by the second batch of health research topics popularization project (project number 19PJ225) of Sichuan Province 2019.
