Abstract
BACKGROUND:
Systemic inflammatory biomarkers reflect level of inflammatory response, which have been suggested as prognostic factors in cancer patients.
OBJECTIVE:
To estimate the prognostic value of inflammatory biomarkers in 149 patients with head and neck soft tissue sarcoma (HNSTS).
METHODS:
Pre-treatment neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), lymphocyte-to-monocyte ratio (LMR), systemic immune-inflammation index (SII), neutrophil-platelet score (NPS) and Aarhus Composite Biomarker Score (ACBS) were analyzed for association with overall survival (OS) and progression-free survival (PFS). Nomograms were consisted of independent predictors for OS and PFS, and evaluated by calibration curve and concordance index (C-index).
RESULTS:
In multivariate analysis, LMR (HR
CONCLUSIONS:
Pre-treatment LMR, ACBS and AJCC stage were effective predictor of OS. The NLR and AJCC stage could independently predict PFS. The nomogram might act as a promising prognostic model for OS and PFS in HNSTS.
Introduction
Soft tissue sarcoma (STS) is a rare tumor, accounting for approximately 1% of all cancers, with annual incidence around 5 among 100,000 persons [1]. STS comprises over 50 histological types of heterogeneous malignancies arising from various kind of tissue, including smooth muscle, adipose tissue and so on [2]. Head and neck soft tissue sarcoma (HNSTS) is the rarest site, which takes up about 5–15% of sarcoma and 1% of head and neck tumor [3]. However, HNSTS is rare but aggressive, with poor five-year overall survival rate (31–80%) and high rates of local recurrence (22–53%) and distant metastases (10–40%) [4]. Conventional prognostic factors such as tumor size and histologic grade still have imperfections of inaccuracy and inconvenience in clinical practice [5]. Therefore, it is important to estimate novel prognostic markers that identify high-risk HNSTS patients and optimize therapeutic strategy to improve survival outcomes.
Increasing evidences showed that cancer-related inflammation has an important role on tumor microenvironment, which promotes tumor proliferation, angiogenesis and metastasis via different inflammatory cells and cytokines [6]. Therefore, as blood indicators of inflammatory response, the systemic inflammatory biomarkers such as neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR) and lymphocyte to monocyte ratio (LMR) were suggested as effective prognostic factors [7, 8]. In previous studies of STS, pre-treatment NLR, PLR, LMR and Aarhus Composite Biomarker Score (ACBS) were practical in predicting overall survival or disease progression [9, 10, 11, 12]. However, to the best of our knowledge, the prognostic value of these inflammation biomarkers has not been separately analyzed for patients with HNSTS. Moreover, the nomogram incorporating inflammatory biomarkers and clinical characteristics could provide an intuitive graphical representation of statistic model, which was considered as a practical method to predict clinical outcome in esophageal cancer, breast cancer and so on [14, 15]. Also, the application of nomograms based on inflammatory biomarkers for HNSTS has not been reported.
Therefore, the purpose of present study was to evaluate the prognostic value of pre-treatment inflammatory biomarkers including NLR, PLR, LMR, systemic immune-inflammation index (SII), neutrophil-platelet score (NPS) and ACBS, then to establish inflammatory biomarkers-based nomograms to reveal the association between inflammatory biomarkers and survival results in patients with HNSTS.
Materials and methods
Patients selection
We searched the electronic medical records of the *BLIND* Hospital, and reviewed all patients with STS between January 2006 and December 2016. The inclusion criteria: 1) Patients with primary STS of head and neck, who were pathological diagnosed and had surgery. 2) Patients had complete laboratory data of inflammatory biomarkers and medical records before initial treatments (surgical resection, neoadjuvant chemotherapy or radiotherapy). 3) Patients received regular and complete follow-up, and the corresponding clinical outcomes were definite. The exclusion criteria: 1) Patients had primary STS of other site, or 2) Lacked pathological reports, pre-treatment blood tests and follow-up data. 3) Patients with some diseases, such as respiratory infection, atherosclerosis and hematologic disease, or medication history that could affect inflammatory markers level were also excluded. This study was approved by the institution Ethics Commission of *BLIND* Hospital of *BLIND* University. The need for consent to participate was waived by Ethics Commission.
Data collection
The peripheral blood tests were performed within 1 week before first treatment, which contained platelet counts (P), neutrophil counts (N), lymphocyte counts (L), levels of hemoglobin, monocytes, albumin and C-reactive protein (CRP). SII was calculated by this formula: SII
All patients received routine follow-up every three months during the first three years, every six months in fourth to fifth years, and then once a year until December 2018 or their death [17]. Times for overall survival (OS) were calculated from the period from initial histological diagnosis to latest clinical observation or death. The progression-free survival (PFS) was defined as the date of histological diagnosis until first evidence of local relapse, lymph node or distant metastases, death or the latest follow-up.
Statistical analysis
According to the results of OS, we performed receiver operating curve (ROC) analysis to obtain area under the curve (AUC) of NLR, PLR, LMR and SII. Then we obtained optimal cut-off values via calculating best Youden index. The differences of clinicopathological characteristics between high and low inflammatory biomarkers were compared by the chi-square test. Crude survival analyses were showed in Kaplan-Meier curves and compared by the log-rank test. These factors also were assessed by univariate Cox proportional hazard model, and significant markers would be included in multivariate Cox analysis using the likelihood ratio test. Association between prognostic factors and survival outcome were calculated as the hazard ratio (HR) and 95% confidence interval (CI). The significant
Nomograms were established using “rms” package on R software 3.5.3. The significant markers in multivariate Cox analysis were included in nomogram, then the predicted probabilities of 3-year, 5-year and 8-year OS and PFS were presented. We performed bootstrapping method with 1000 resamples to generate calibration curves that verified the deviation between predicted survival and actual observation, with the Hosmer-Lemeshow test. The discriminative abilities of nomograms and traditional markers were compared using Harrell’s concordance index (C-index) and ROC analysis.
Results
Clinical characteristics of patients
There were 149 Asian patients diagnosed with HNSTS in present study, including 90 males and 59 females. All patients were Chinese, which were mostly Han Chinese (
The best cut-off value was 2.2 for NLR (AUC
Clinicopathological characteristics of patients
Clinicopathological characteristics of patients
Abbreviations: NLR neutrophil-lymphocyte ratio, PLR platelet-lymphocyte ratio, LMR lymphocyte-monocyte ratio, SII systemic immune-inflammation index, NPS neutrophil-platelet score, ACBS Aarhus Composite Biomarker Score.
The median duration of follow-up was 44 months (range, 5–183 months). Totally 113 patients (75.8%) were alive at the latest follow-up with 3-year OS rate of 80.6%, 5-year OS rate of 73.9% and 8-year OS rate of 64.1%. Sixty-nine patients (46.3%) were observed to have disease progression, which included 42 cases (28.2%) with recurrence in situ, 16 patients (10.7%) with distant metastases (mostly in lung and bone) and 9 cases (6.0%) of cervical lymph node metastases. The probabilities of 3-year, 5-year and 8-year PFS were 65.2%, 53.1% and 45.8%, respectively.
As shown in Table 2, NLR (
Univariate Cox analysis of prognostic factors affecting survival
Univariate Cox analysis of prognostic factors affecting survival
Abbreviations: NLR neutrophil-lymphocyte ratio, PLR platelet-lymphocyte ratio, LMR lymphocyte-monocyte ratio, SII systemic immune-inflammation index, NPS neutrophil-platelet score, ACBS Aarhus Composite Biomarker Score.
Kaplan-Meier curves of overall survival stratified by NLR (A), PLR (B) and LMR (C). Kaplan-Meier curves of progression-free survival stratified by NLR (D), PLR (E) and LMR (F).
Multivariate Cox analysis of prognostic factors affecting survival
Abbreviations: NLR neutrophil-lymphocyte ratio, PLR platelet-lymphocyte ratio, LMR lymphocyte-monocyte ratio, SII systemic immune-inflammation index, ACBS Aarhus Composite Biomarker Score.
Nomograms for predicting the probability of 3-year, 5-year and 8-year OS (A) and PFS (B). Calibration curves of nomograms for predicting 3-year and 5-year OS (C) and PFS (D).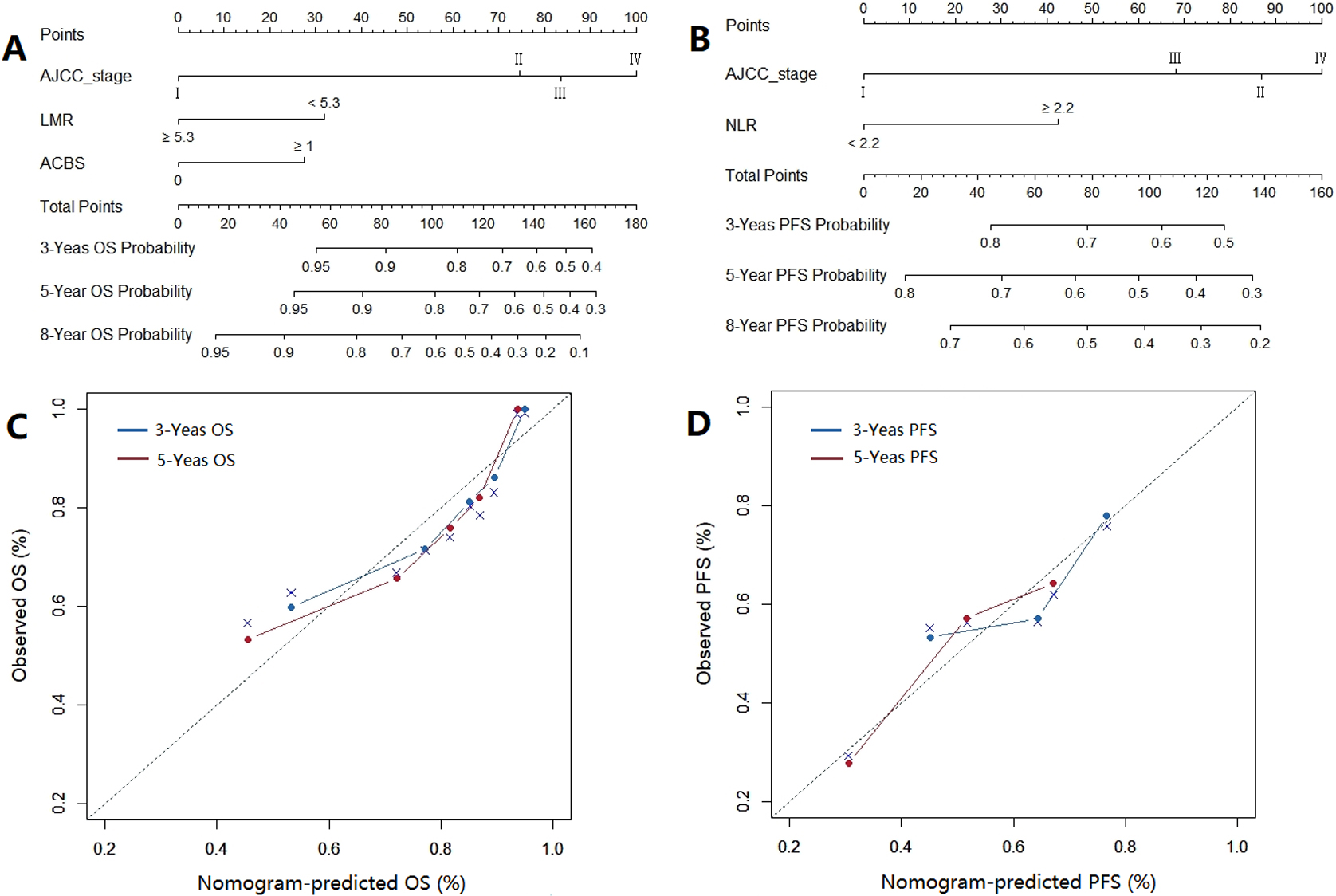
In multivariate analysis (Table 3), the lower LMR (HR
As independent predictors, AJCC stage, LMR and ACBS were included in nomogram for OS (Fig. 2A). The AJCC stage and LMR were used to establish nomogram for PFS that predicting probability of survival events by calculating total points (Fig. 2B). The calibration curves for 3-year and 5-year OS and PFS reflected good agreement between prediction and observed survival (Fig. 2C and D). The P value of Hosmer-Lemeshow test was 0.746 for OS and 0.262 for PFS. The C-index of nomogram for OS and PFS was 0.748 (95% CI
ROC curves comparing the discriminative ability of nomograms, clinicopathological features and inflammatory biomarkers for OS (A) and PFS (B).
First reported by Rudolf Virchow, the cancer-related inflammation is considered to contribute to tumor development and progression via inducing angiogenesis, cell proliferation and tissue infiltration [18]. Systemic inflammatory biomarkers reflected level of inflammatory and immune response, thus were suggested as effective predictors of survival and cancer progression [19]. In present research, we analyzed the prognostic value of six inflammatory biomarkers for first time in HNSTS. The results showed that pre-treatment LMR and ACBS were independently associated with OS, and NLR was an unfavorable independent predictor of PFS. Nomograms cooperating staging system and inflammatory biomarkers also were convenient and practical in predicting OS and PFS in patients with HNSTS.
The allogeneic phenotype of tumor cell stimulates the body to produce inflammatory cells, which leads to the collapse of tumor tissues and results in broad and lasting non-specific inflammatory response [6]. The inflammatory response may lead to enhancement of leukocytes, neutrophils and platelets as well as reduction in lymphocytes [20]. Increased tumor infiltrating neutrophils (TINs) could recruit tumor cell proliferation and promote angiogenesis by secreting various cytokines such as TNF-
Platelets were also reported to involve the inflammation and support cancer progression. For instance, platelets can promote angiogenesis by proangiogenic factors, stimulate tumor cells proliferation via TGF-
Systemic immune-inflammation index (SII) is an integrative indicator based on levels of lymphocytes, neutrophils and platelets. Previous studies have suggested SII as a superior predictor of long-term survival outcome compared to NLR and PLR in colorectal cancer [35], lung cancer [36] and so on. It also reported that SII was significantly correlated with the number of circulating tumor cells, which potential promoted the metastases and progression of gastric cancer [37]. However, for all we know, the SII for STS has not been estimated before, and the prognostic role of SII remained unclear in patients with STS. Although the SII was not an independent predictor for OS and PFS in this study, the significant results in univariate analyses indicated the potential prognostic ability of SII, which required further investigation.
Neutrophil-platelet score (NPS) could reflect the levels of neutrophils and platelets, then was suggested as effective predictor of patient’s survival and disease development in many cancers [38]. Evaluation of NPS has not been reported in STS, HNSTS patients with NPS score
The nomogram is a convenient and practical identification method to predict clinical events and prognosis of patients with visualized possible risk factors. It was worth that we established nomograms combined by traditional staging system and inflammatory biomarkers in HNSTS, which was only reported in osteosarcoma and spinal Ewing’s sarcoma expect for STS [19, 40]. The results of C-index, ROC analysis and calibration curve showed improved accuracy of nomograms than AJCC stage and inflammatory biomarkers for OS and PFS. The nomogram-based model could be a novel potential tool to predict prognosis and therapeutic results of HNSTS patients in clinical work.
The present study had several limitations. Firstly, there were significant correlations between inflammatory biomarkers, tumor stage and treatments, which could be potential con-founders of survival analysis. In addition, due to the rarity of STS, especially of head and neck region, the relatively small sample could lead to selection bias. For instance, the median age at diagnosis was lower than that of previous epidemiologic studies [41, 42]. Also, the inevitable heterogeneity of histological types of STS may affect the reliability and generalizability of results. Furthermore, this study was limited by retrospective design in single center, thus it was better to verify the prognostic role inflammatory biomarkers in multi-center prospective research.
Conclusions
For patients with head and neck soft tissue sarcoma, pre-treatment LMR, ACBS and AJCC stage were independent prognostic factors of OS. The elevated NLR and advanced AJCC stage were independently associated with worse PFS. The prognostic nomograms integrating NLR, LMR, ACBS and AJCC staging system might be applied to predict clinical outcome and optimize therapeutic strategy.
Footnotes
Conflict of interest
The authors declare that they have no conflict of interests.
Supplementary Figure
The flowchart of patient selection.
Kaplan-Meier curves of overall survival stratified by SII (A), NPS (B) and ACBS (C). Kaplan-Meier curves of progression-free survival stratified by SII (D), NPS (E) and ACBS (F).
