Abstract
The presence of hypoxia is a typical feature of solid tumors and has been identified in many neoplasms, favouring the survival of malignant cells in a hostile environment and the expression of an aggressive phenotype. Malignant brain tumors have large proportions of hypoxic tissue, thus contributing to resistance to radiation and chemotherapy. Positron emission tomography (PET) is an attractive technique to gain a non-invasive assessment of tumor hypoxia within the whole tumor, with 18F-fluoromisonidazole (18F-FMISO) and 18F-flouroazomycin arabinoside (18F-FAZA) being the most promising radiotracers. In this short review, we aim to discuss the available clinical studies focused on the use of 18F-FAZA PET/computed tomography in patients affected by high-grade glioma.
Background
Hypoxia and tumor aggressiveness
The presence of hypoxia is a typical feature of solid tumors and has been identified in many neoplasms. This biological phenomena is related to changes in gene expression and genetic instability, as a result of its resistance to apoptosis and decreased DNA repair. 1
Hypoxia favors the survival of malignant cells in a hostile environment and the expression of an aggressive phenotype; in fact, the oxygenation status of a tumor is involved in the regulation of gene expression for malignant progression as well, leading to an increase of local aggressiveness and rate of metastases through the secretion of hypoxic stress proteins and loss of apoptotic potential. 2
Hypoxia is one of the main causes of resistance to radiotherapy; in fact, the reduced presence of oxygen decreases the free formation of radicals, which radiotherapy relies on to cause DNA damage to tumor cells. 2 The reduced sensitivity to ionizing radiation also interferes with many chemotherapy regimens by hampering efficient drug delivery to the tumor. Additionally, it is well recognized that hypoxic tumors present chemotherapeutic resistance due to the reduced drug penetration (caused by the irregular vascularization), extracellular acidification, and genomic instability and resistance to apoptosis. This evidence underlines how hypoxia is a major adverse prognostic factor for tumor progression and resistance to anticancer treatment. 3
Malignant brain tumors have large proportions of hypoxic tissue, thus contributing to resistance to radiation and chemotherapy. High-grade gliomas (III–IV) in particular are highly vascularized with an inefficient vascular microcirculation compared to that of normal brain. 4
These high-grade brain tumors are also characterized by histological features of intratumor necrosis and vascular endothelial proliferation; these aspects are known to distinguish low-grade from high-grade gliomas, and it has been demonstrated that the extent of necrosis inversely correlates with patients’ outcome and survival. 5
Intratumor heterogeneity along with the lack of an efficient and universal technique able to identify intratumor hypoxic regions represent a major challenge for clinical practice in oncology. Moreover, studies correlating the impact of hypoxia on patients’ outcome are currently lacking.
The identification of tumor hypoxia would improve tumor characterization, thus reflecting on patients’ prognosis, treatment response, and patients’ selection by discriminating those patients eligble to “hypoxia-directed therapies” from those requiring more intensive treatment apporaches.
Tumor hypoxia assessment
Various strategies have been proposed to detect the presence of tumor hypoxia. Although the clinical relevance of tumor hypoxia could be demonstrated with direct oxygenation measurements, this methodology is limited by its invasiveness and feasibility to only superficially assessable tumors. In addition, electrode measurements reflect the oxygenation status solely in a particular portion of the tumor and, thus, do not represent the tumor tissue as a whole. 6
Tumor hypoxia could be assessed also by using exogenous (i.e. pimonidazole) or endogenous biomarkers. Among the latter ones, hypoxia-inducible factor-1a, carbonic anhydrase IX, vascular endothelial growth factor, and lucose transporter-1 are the most widely applied.
However, equivocal results regarding the correlation between differential expression of the abovementioned hypoxia-related biomarker and patients’ outcome have been reported. These discrepancies might be due to the differential expression of these biomarkers within a given tumor, as they are conditioned by specific microenvironments.
Therefore, in vivo brain hypoxia imaging, which might be able to non-invasively provide a reliable assessment of the whole tumor hypoxic status, might represent a valid alternative and be a promising tool to provide an accurate tumor characterization prior treatment, to plan effective and tailored treatments, and to ultimately predict the outcome in patients affected by high-grade glioma.
Currently, dynamic susceptibility contrast and dynamic contrast-enhanced magnetic resonance imaging (MRI) have emerged as powerful tools for the diagnosis of various central nervous system diseases, and in particular in grading gliomas; these techniques can provide a quantittive assessment of the microcirculation of tumors through tissue blood flow, vascularity, and parenchymal contrast uptake measurements. In addition, diffusivity measures derived from MRI (diffusion weighted imaging (DWI)/diffusion tensor imaging (DTI)) represent an accurate measure of tumor cellularity and peritumoral infiltration. 7 Positron emission tomography (PET) is an attractive technique to gain a non-invasive assessment of tumor hypoxia within the whole tumor. The optimal tracer to be used for hypoxia imaging should be retained only under hypoxic circumstances, and should be only minimally influenced by other factors, such as cerebral blood flow or pH. 8
Several hypoxia specific PET tracers have been proposed for tumor hypoxia imaging. Considering al the radiotracers that have been tested so far, Fluorine-18-Fluoromisonidazole (18F-FMISO) and 18F-flouroazomycin arabinoside (18F-FAZA) are the most promising. 8 Both tracers are part of the 2-nitroimidazoles group and they are both radiolabeled with Fluorine-18. Their lipophilicity facilitates their entrance into cells, after which these radiotracers are reversibly reduced to reactive oxygen radicals in a hypoxic environment, and at low values of oxygen these radicals remains trapped within the cells. 9
Compared to 18F-FMISO, 18F-FAZA is more hydrophilic and it does not accumulate in blood–brain barrier (BBB) preserved tumors, although both 18F-FMISO and 18F-FAZA can accumulate in tumors where the BBB is unpaired. An advantage of the hydrophilic nature of 18F-FAZA is its rapid clearance from the urinary tract, while the lipophilic 18F-FMISO is excreted via the hepatobiliary system. 10
The relatively short biological half-life of 18F-FMISO hampers the possibility of performing late imaging, with image quality at several hours after the injection being reduced because of poor contrast between hypoxic and not-hypoxic areas. Moreover, 18F-FMISO also presents non-specific accumulation in normoxic tissue. 11
Conversely, 18F-FAZA, which is a second-generation nitroimidazole, is a valid and promising alternative to 18F-FMISO for its longer biological half-life and for its higher contrast to non-target tissue. This radiotracer has faster blood clearance and therefore an improved tumor-to-background ratio, resulting in a better image quality also at later imaging acquisition. 12
In this short review we aim to discuss the available clinical studies focused on the use of 18F-FAZA PET/computed tomography (CT) in patients affected by high-grade glioma.
Methods
A literature search from the PubMed and Scopus databases has been performed with the purpose of identifying reports on the use of the 18F-FAZA hypoxia PET tracer in the clinical setting of high-grade glioma. The following research filters were applied: (a) time frame: last 10 years; (b) language: English. The searching keywords were “PET” and “glioma” and “FAZA” and they led to nine papers. After filtering only for studies on human subjects, three papers were considered.
Results and discussion
In the papers considered for the analysis, nine patients affected by high-grade glioma were studied with 18F-FAZA PET/CT overall.
Postema and colleagues 13 were the first group reporting their clinical experience on 18F-FAZA in high-grade gliomas. In their study, including 50 oncological patients, 7/50 were affected by glioblastoma. The authors reported high tumor-to-blood ratios, mainly due to the lack of uptake of 18F-FAZA in the normal brain tissue. This radiotracer provided good quality images at 2–3 hours post-injection, which did not require later scanning. A disrupted BBB surely plays a role in 18F-FAZA uptake in gliomas. However, the radiotracer retention is strictly dependent from the reduction process that happens only under hypoxic conditions and explains the absent uptake of 18F-FAZA in normal brain tissue. Therefore, Postema et al. 13 confirmed that 18F-FAZA was highly representative of tumor hypoxia within glioblastoma.
Recently, our group reported the potentiality of 18F-FAZA PET/CT in guiding tailored radiotherapy treatment in high-grade glioma. In particular, the possibility of providing additional boost on more hypoxic regions by using 18F-FAZA PET images in a simulated radiotherapy scheme was described. Additionally, this molecular imaging technique has been used to assess tumor response after radiotherapy with promising implications for clinical practice. 14
Our group also reported—for the first time—the ability of 18F-FAZA in guiding stereotactic biopsy on the more hypoxic regions in order to identify the tumor areas with the highest aggressive potential. In order to reach this goal, MRI and 18F-FAZA PET/CT images were co-registered to identify the areas of both high-perfusion MRI markers and high 18F-FAZA uptake, and therefore guide the bioptic sampling on the most representative tumor region (Figure 1). 15
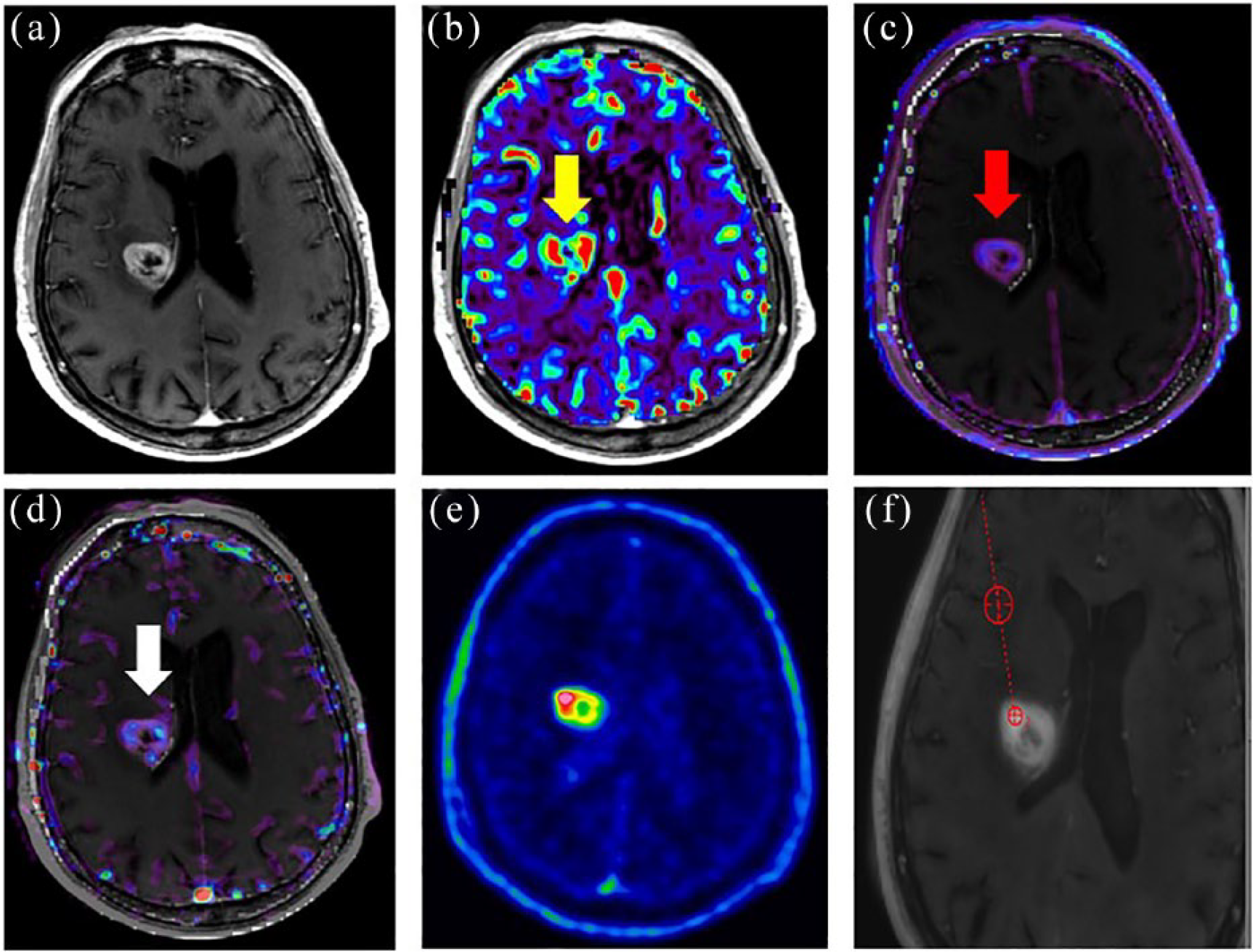
Role of 18F-FAZA PET/CT in guiding stereotactic biopsy in a 77-year-old patient affected by high-grade glioma. Gadolinium-enhanced T1 images (a) showed an enhancing, edematous lesion in the right basal ganglia and corona radiata with mass effect on the right ventricle. The lesion presented high values of relative cerebral blood volume ((b): yellow arrow), transfer constant ((c): red arrow) and fractional plasma volume ((d): white arrow). The brain lesion showed 18F-FAZA uptake with a central photopenic area, possibily due to necrotic tissue, on PET images (e). MRI and 18F-FAZA PET/CT images were then co-registered and an area of both high-perfusion MRI markers and high 18F-FAZA uptake was selected for biopsy after appropriate image fusion (f) to select the most representative tumor region to be sampled. 15
The treatment of choice in high-grade glioma is represented by radical radiotherapy and concomitant chemotherapy with temozolomide, which seems to increase the benefit of radiotherapy especially in glioblastoma. 16 Despite this progress, the results of the best association of therapies including radical surgery, adjuvant radiotherapy, concomitant and adjuvant temozolomide, are not encouraging. The Brain Tumor Cooperative Group NIH Trial 87-01, the Radiation Therapy Oncology Group 93-05 protocol, and the RTOG 0023 phase II trial have not demonstrated an increase in survival adding an interstitial or stereotactic radiotherapy boost to escalate the dose to the tumor. In addition, the combined radio-chemotherapy treatment increases the likelihood of radiation necrosis compared with radiotherapy alone, and the differential diagnosis is sometimes difficult, but necessary, to decide whether or not to continue adjuvant chemotherapy or to switch to another treatment.
As evidenced from the papers analyzed in this review, 18F-FAZA seems to be particularly promising for clinical purposes, with encouraging results regarding the possibility of guiding treatments by adapting treatment in the course of radiotherapy, predicting treatment success in combined radiochemotherapy schemes, or identifying possible tumor regions highly inclined to recurrence.17-19
This radiotracer might also have a role in both treatment planning and monitoring. The possibility to perform hypoxia imaging-guided biopsies could properly represent the vascular heterogeneity within the tumor, being a strong support for planning treatment and predicting patient outcome.
Future pespectives and conclusion
Few papers have investigated the potentiality of 18F-FAZA PET in brain tumors so far, despite the high relevance and implications of the use of this radiotracer in several steps of the management of patients affected by high-grade glioma.
The use of PET, with the possibility to assess different aspects of tumor metabolism by using different radiotracers, together with the ability of MRI with advanced techniques (DWI/DTI) in providing tumor morpho-functional features, highlights the potential of the new PET/MRI hybrid scanners in the clinical setting of brain tumors, thus improving diagnostic accuracy and, in turn, patient management.
One of the main fields of application of this technique is surely for those patients with high-grade glioma after concomitant radiochemotherapy for whom routine follow-up imaging is recommended for distinguishing between pseudo-progression and true tumor progression. These very common clinical scenarios have to be recognized, so that efficient alternative treatment may be promptly applied; in this view, PET/MRI hybrid imaging with the use of PET hypoxia tracers could speed up decision making compared to sequential imaging strategies.
In conclusion, the identificaiton of imaging hypoxia biomarkers is essential for the clinical development of hypoxia-targeted treatments, although a standardization and validation process is still necessary in order to establish a definitive clinical role.
The knowledge of tumor heterogeneity, and therefore the possibility to evaluate treatment response and prognosis, might improve the process towards hypoxia-personalized treatments.
Footnotes
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics
This review article does not report new data obtained by the authors on human or animal subjects besides those previously reported in the literature, for which the ethical issues have already been fully clarified.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present work was supported by the Italian Association for Cancer Research (grant IG 2014 Id.1524; EudraCT: 2015-000679-28).
